
Cholesterol Management
Cholesterol Management
Last Section Update: 07/2025
Contributor(s): Colleen Mazin, MS/MPH; Maureen Williams, ND; Debra Gordon, MS
1 Overview
Summary and Quick Facts for Cholesterol Management
- Emerging research into underappreciated aspects of cholesterol biochemistry has revealed that levels of cholesterol account for only a portion of the cardiovascular risk profile, while the properties of the molecules responsible for transporting cholesterol through the blood, called lipoproteins, offer important insights into the development of atherosclerosis.
- Although the use of pharmaceutical treatment has saved lives, Life Extension® has long recognized that optimal cardiovascular protection involves a multifactorial strategy that includes at least 17 different factors responsible for vascular disease.
- For optimal health effects and vascular support, innovative strategies for decreasing vascular risk should be incorporated thorough cholesterol and lipoprotein testing, as well as strategic nutrient and pharmaceutical intervention.
How Does Cholesterol Contribute to Heart Disease?
Cholesterol is a steroid molecule that is a large component of cellular membranes and a precursor to steroid hormones and vitamin D, among other functions. Cholesterol is transported throughout the body as lipid-protein complexes called lipoproteins. Low-density lipoproteins (LDLs) and high-density lipoproteins (HDLs) are generally measured when testing blood cholesterol levels.
LDL (commonly referred to as “the bad cholesterol”) can transport cholesterol to arterial walls, contributing to the development of atherosclerosis (hardening and narrowing of arteries) and cardiovascular disease. Less commonly known is that size, density, and metabolic processes (eg, oxidation and glycation) that alter lipoproteins play a large role in how damaging LDL can be to the endothelial cells that line arteries. HDL (often referred to as “the good cholesterol”) transports cholesterol away from tissues to the liver for reprocessing or disposal.
Natural interventions such as pantethine and artichoke may help with managing cholesterol levels and maintaining a healthy heart.
Note: High cholesterol is not the only risk factor for cardiovascular events. People interested in reducing their cardiovascular risks should follow the cholesterol-lowering strategies outlined in this protocol and read the Life Extension Magazine article titled “How to Circumvent 17 Independent Heart Attack Risk Factors.”
What are Conventional Medical Treatments for Managing Cholesterol?
- HMG-CoA reductase inhibitors (statins) to reduce cellular production of cholesterol
- Medications to lower cholesterol absorption from the intestine (eg bile acid sequestrants like colesevelam)
- PCSK9 inhibitors, which allow LDL to bond to its receptors and be removed from the blood
- For people who require additional LDL lowering, the new drug bempedoic acid may be recommended in addition to statin therapy.
What Dietary and Lifestyle Changes Can Help Manage Cholesterol?
- Dietary modifications to reduce intake of saturated and trans fats, sodium, added sugar, and cholesterol, while increasing fruits and vegetables, whole grains, nuts and legumes, fish and poultry, and oils low in saturated fat (like olive oil).
- Caloric restriction may help some individuals.
- Regular exercise—at least 150 minutes of moderate-intensity activity or 75 minutes of high-intensity activity each week
What Natural Interventions Can Help Manage Cholesterol?
- Artichoke. Artichoke may help the body get rid of excess cholesterol. Several clinical trials have shown artichoke reduces LDL cholesterol.
- Pantethine. Pantethine, a derivative of vitamin B5, and its metabolites appear to act on the body’s fat and cholesterol metabolism pathways. Clinical studies have demonstrated pantethine’s ability to reduce LDL and triglycerides in people with high cholesterol.
- Indian gooseberry (amla). Amla, traditionally used in Ayurvedic medicine, favorably influences blood lipids by lowering total cholesterol and triglycerides—one head-to-head study even showed it was comparable to simvastatin.
- Garlic. Garlic’s benefits on blood lipid profiles have been confirmed in multiple reviews and over 30 clinical trials. Garlic can lower total cholesterol and LDL, as well as triglycerides. Garlic also has favorable effects on blood pressure for added cardiovascular benefits.
- Plant sterols. Plant sterols are steroid molecules found in plants that may compete with cholesterol for absorption in the intestines, reducing LDL levels. Numerous clinical studies have repeatedly shown plant sterols to be effective for reducing LDL.
- Soluble fibers. The cholesterol-lowering effects of soluble fibers, like beta-glucan, pectin, and those from oats and barley, have been substantiated in hundreds of clinical trials. Intake of soluble fiber is linked with lower incidence of cardiovascular disease as well.
- Coenzyme Q10. Coenzyme Q10 (CoQ10) is well known for its impact on cardiovascular health. Statins, first-line drugs for treatment of high cholesterol, suppress CoQ10 levels. It is advisable for those taking statins to also supplement with CoQ10. Additionally, CoQ10 may reduce total cholesterol and increase HDL.
- Pomegranate. Pomegranate contains high amounts of polyphenols, especially punicalagins. Several placebo-controlled clinical trials have shown pomegranate can lower total cholesterol and LDL levels in people with high cholesterol and may also improve other parameters indicative of atherosclerosis.
- Other natural ingredients, such as Gynostemma pentaphyllum, hesperidin, guggul/gum guggul, carotenoids, fish oil, niacin, red yeast rice, and more, may be beneficial as well.
2 Introduction
We now know more about cholesterol and its role in cardiovascular disease than ever before. Ample clinical trial data has established unequivocally that lowering low-density lipoprotein (LDL) ("bad cholesterol") with statins and other drugs in appropriately selected patients delivers meaningful cardiovascular risk reduction.1 Furthermore, the advent of PCSK9-inhibiting drugs in the mid-2010s paved the way for further LDL lowering and risk reduction in people whose cholesterol is not adequately controlled by statins, although cost remains a barrier.2,3
That said, keeping cholesterol levels in check is still only one piece of the cardiovascular health puzzle. As described in Life Extension's Atherosclerosis and Cardiovascular Disease protocol, many other risk factors should be appropriately addressed for optimal cardiovascular health. Nevertheless, keeping cholesterol levels in a healthy range remains a central pillar of cardiovascular risk reduction.
Cholesterol and related blood lipids are far more complex than the traditional cholesterol blood test would lead us to believe. Most people are familiar with the traditional lipid risk factors: LDL, high-density lipoprotein (HDL), total cholesterol, and triglyceride levels. What few people outside the cardiovascular research community appreciate is that a more detailed look, that includes non-traditional blood lipids, can offer important insights into an individual’s cardiovascular risk.
The protein apolipoprotein B (ApoB), a structural component of all non-HDL cholesterol particles including LDL- and very low-density lipoprotein (VLDL)-cholesterol, has emerged as perhaps the most valuable marker of cardiovascular risk. ApoB is an even stronger predictor of risk than LDL alone.4,5,236,237,238 A 2018 expert panel on cholesterol management, representing numerous cardiology and physician societies, including the American College of Cardiology and American Heart Association, referred to ApoB as “a stronger indicator of atherogenicity than LDL-cholesterol alone,” and recommended measurement of ApoB for cardiovascular risk assessment, especially in those with elevated triglycerides.5 In 2019 a lipid management guideline from the European Society of Cardiology and European Atherosclerosis Society made similar recommendations.239 ApoB also improves risk prediction in individuals with metabolic syndrome and diabetes.238
A prospective cohort analysis published in JAMA Cardiology in 2021, which included a large population study (UK Biobank) and two large international clinical trials, found ApoB was the best predictor of heart attack risk when compared with triglycerides, LDL-cholesterol, or non-HDL cholesterol. They further suggested ApoB may be the main driver of atherosclerosis.240 A commentary on this study that appeared in the same journal concluded that not only is ApoB superior for risk prediction compared with traditional lipid risk factors, it also better predicted benefit from lipid-lowering treatment.236 An observational study in over 13,000 statin-treated Danish patients, published in 2021, also found ApoB was more accurate than LDL-cholesterol for predicting risk of myocardial infarction and is a better predictor of all-cause mortality in statin-treated patients.237
Also, the size and density of cholesterol-transporting proteins, called lipoproteins, are important factors that influence cardiovascular risk. Large, buoyant LDL particles are less atherogenic than smaller, more dense LDL particles. Similarly, large, buoyant HDL particles offer greater vascular protection than smaller, denser HDL. The development of advanced lipid testing strategies that take the importance of lipoprotein particle size into consideration, such as the NMR LipoProfile, allows for a deeper assessment of cardiovascular risk than a conventional lipid profile.6
Furthermore, metabolic processes, such as oxidation and glycation, modify the functionality of lipoproteins, transforming them from cholesterol transport vehicles into highly reactive compounds capable of damaging the delicate endothelial cells that line our arterial walls. This endothelial damage both initiates and promotes atherogenesis. Some natural interventions and lifestyle changes can target the formation of these modified lipoproteins and help avert vascular damage and dysfunction.
A comprehensive strategy for decreasing vascular risk should incorporate dietary and lifestyle changes along with thorough cholesterol and lipoprotein testing and strategic nutrient and pharmaceutical interventions.
3 The Blood Lipids: Cholesterol and Triglycerides
Cholesterol
Figure 1: Steroid Biosynthesis Pathways7
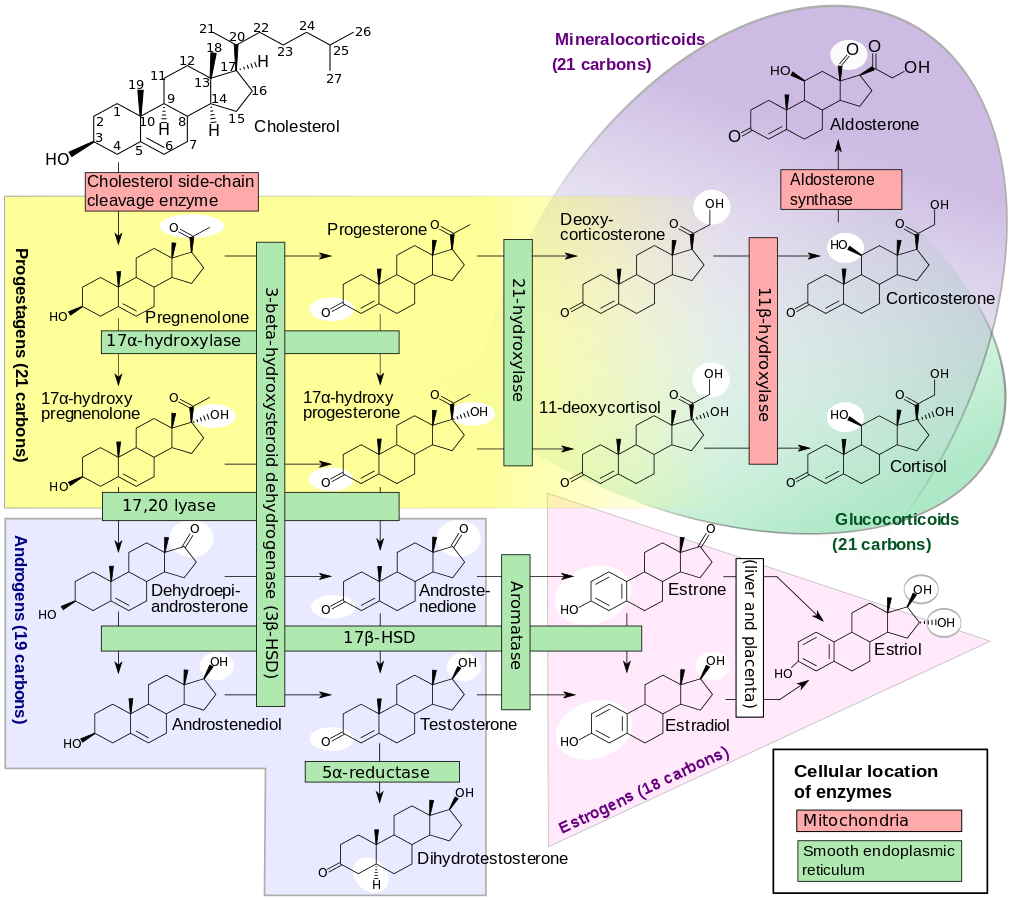
Cholesterol is a wax-like steroid molecule that plays a critical role in metabolism. It is a major component of cellular membranes, where its concentration varies depending on cell type. For example, the lipid portion of the membrane of liver cells consists of about 17% cholesterol, while that of red blood cells consists of about 23%.8
The cholesterol in cell membranes serves two primary functions. First, it modulates the fluidity of membranes, allowing them to maintain their function over a wide range of temperatures. Second, it prevents leakage of ions by acting as a cellular insulator.8 (Ions are molecules used by cells to interact with their environment.) This effect is critical for the proper function of neuronal cells because the cholesterol-rich myelin sheath insulates neurons and allows them to transmit electrical impulses rapidly over long distances.
Cholesterol has other important roles in human metabolism. Cholesterol serves as a precursor to steroid hormones, which include the sex hormones (androgens and estrogens); mineral-corticoids,9 which control the balance of water and mineral excretion in the kidneys; and glucocorticoids, which control protein and carbohydrate metabolism, immune suppression, and inflammatory processes (Figure 1).7 Cholesterol is also the precursor to vitamin D. Finally, cholesterol provides the framework for the synthesis of bile acids, which emulsify dietary fats for absorption.
Triglycerides
Triglycerides are storage lipids that have a critical role in metabolism and energy utilization. They are molecular complexes of glycerol (glycerin) and three fatty acids.
While glucose is the preferred energy source for most cells, it is a bulky molecule that contains little energy for the amount of space it occupies. Glucose is primarily stored in the liver and muscles as glycogen. Fatty acids, on the other hand, when packaged as triglycerides, are denser sources of energy than carbohydrates, which make them superior for long-term energy storage (the average human can only store enough glucose in the liver for about 12 to 24 hours worth of energy without food, but can store enough fat to power the body for substantially longer).10
Lipoproteins: Blood Lipid Transporters
Lipids (cholesterol and fatty acids) are insoluble in plasma, and so must be transported throughout the body as lipid-protein complexes called lipoproteins. These have cholesterol esters and triglycerides in their core, with a surface composed of phospholipids, free cholesterol, and apolipoproteins on their surface.238
These more water-soluble lipoproteins can be delivered to tissues throughout the body, including into arterial walls where one of these proteins, ApoB, plays a critical role in atherosclerotic plaque formation.241 Lipoproteins can also carry fat-soluble nutrients, like coenzyme Q10 (CoQ10), vitamin E, and carotenoids, which protect the transported lipids from oxidative damage. Vitamin E and CoQ10 also help prevent the oxidative modification of LDL particles, which in turn protects the blood vessel lining from damage. This will be discussed in greater detail later in this protocol.
There are four main classes of lipoproteins, each with a different, important function11:
- Chylomicrons are produced in the small intestines and deliver energy-rich dietary fats to muscles (for energy) or fat cells (for storage). They also deliver dietary cholesterol from the intestines to the liver.
- Very low-density lipoproteins (VLDLs) take triglycerides, phospholipids, and cholesterol from the liver and transport them to fat cells.
- Low-density lipoproteins (LDLs) carry cholesterol from the liver to cells that require it. In aging people, LDL often transports cholesterol to the linings of their arteries where it may not be needed.
- High-density lipoproteins (HDLs) transport excess cholesterol (from cells, or other lipoproteins like chylomicrons or VLDLs) back to the liver, where it can be re-processed and/or excreted from the body as bile salts. HDL removes excess cholesterol from the arterial wall.
Among its myriad of functions, the liver has a central role in the distribution of cellular fuel throughout the body. Following a meal, and after its own requirements for glucose have been satisfied, the liver converts excess glucose and fatty acids into triglycerides for storage and packages them into VLDL particles for transit to fat cells. VLDLs travel from the liver to fat cells, where they transfer triglycerides/fatty acids to the cell for storage. VLDLs carry between 10% and 15% of the total cholesterol normally found in the blood.12
As VLDLs release their triglycerides to fat cells, their cholesterol content becomes proportionally higher (which also causes the VLDL particle to become smaller and denser). The loss of triglycerides causes the VLDL to transition to an LDL. The LDL particle, which averages about 45% cholesterol, is the primary particle for the transport of cholesterol from the liver to other cells of the body; about 60‒70% of serum cholesterol is carried by LDL.12
Each non-HDL cholesterol particle contains exactly one Apo-B molecule, making measurement of Apo-B levels a highly accurate indicator of the number of these atherogenic particles.238 Although Apo-B particles play a role in clearing LDL-cholesterol, they tend to get trapped in arterial walls, leading to or worsening atherosclerotic disease.241,242 ApoB has been found to be a superior predictor of cardiovascular and other risk as compared to a measurement of the cholesterol content of those particles.13
LDL particles are brought into cells via LDL receptors, and the more these receptors are active, the more LDL particles are removed from the blood resulting in a lower blood concentration of LDL.14 The enzyme PCSK9 (proprotein convertase subtilisin/kexin type 9) degrades the LDL receptor. PCSK9-inhibitor drugs that are antibodies against this enzyme have been developed, and they markedly lower LDL cholesterol.
Because of the correlation between elevated blood levels of cholesterol carried in LDL and the risk of heart disease, LDL is commonly referred to as the "bad cholesterol." LDL is, however, more than just cholesterol, and its contribution to disease risk involves more than just the cholesterol it carries.
Not all LDL particles are created equal. In fact, LDL subfractions are divided into several classes based on size (diameter) and density, and are generally represented from largest to smallest in numerical order beginning with 1. The lower numbered classes are larger and more buoyant (less dense); size gradually decreases and density increases as the numbers progress. Smaller, denser LDLs are significantly more atherogenic for two reasons: they are much more susceptible to oxidation,15-17 and they pass from the bloodstream into the blood vessel wall much more efficiently than large, buoyant LDL particles.18 A more comprehensive lipid test, such as the NMR LipoProfile,6 allows for assessment of the size and density of LDL particles, a feature that increases the prognostic value and sets these advanced tests apart from conventional lipid tests. If an individual is found to have a greater number of small dense LDLs, they are said to express LDL pattern B and are at greater risk for heart disease than an individual with more large buoyant LDL particles, which is referred to as pattern A.
Lipoprotein(a) , also called Lp(a), is a subclass of lipoprotein particles composed of LDL-like particles bound to another particle, called apolipoprotein(a). Lp(a) is a known marker of cardiovascular risk; that is, elevated levels correlate with greater risk for cardiovascular disease. Lp(a) levels are mostly determined by genetics (as opposed to diet and lifestyle as with other blood lipid markers). Generally, Lp(a) levels above 50 mg/dL (~125 nmol/L) are considered to indicate high cardiovascular risk, whereas levels below 30 mg/dL (~72 nmol/L) are associated with low risk.19
Lp(a) levels should be interpreted in the context of other cardiovascular and lipid risk markers, and family history of cardiovascular disease is an indication for measurement of Lp(a). As of mid-2019, no reliable data from randomized controlled trials have shown that targeting Lp(a) reduction with medication is an effective risk-reduction strategy. As of this writing, the only intervention available that appears promising for lowering Lp(a) is lipoprotein apheresis.20 Lipoprotein apheresis involves the removal of lipoproteins from the blood via a blood filtration process used only in people with very elevated blood lipids despite maximal lifestyle and drug therapy. Thus, Lp(a) is primarily useful as a marker for identifying people who might benefit from adopting a more intensive overall cardiovascular risk reduction strategy.
Nevertheless, some intriguing interventions that target Lp(a), such as antisense oligonucleotides, are currently under development and may represent a novel intervention if research progresses as hoped, but further studies are needed.5,21-23
HDLs are small, dense lipoprotein particles assembled in the liver. They carry about 20‒30% of total serum cholesterol.12 Cholesterol carried in the HDL particle is called "good cholesterol," in reference to the protective effect HDL particles can have on cardiovascular disease risk. HDL particles can pick up cholesterol from other tissues and transport it back to the liver for re-processing and/or disposal as bile salts. HDL can also transport cholesterol to the testes, ovaries, and adrenals to serve as precursors to steroid hormones. HDLs are identified by their apolipoproteins ApoA-I and ApoA-II, which allow the particles to interact with cell surface receptors and other enzymes.
The movement of cholesterol from tissues to the liver for clearance, mediated by HDLs, is called reverse cholesterol transport. If the reverse cholesterol transport process is not functioning efficiently, lipids can build up in tissues such as the arterial wall. Thus, reverse cholesterol transport is critical for avoiding atherosclerosis.
4 Blood Lipids and Disease Risk
The initial association between cholesterol and cardiovascular disease was born out of the detection of lipid and cholesterol deposits in atherosclerotic lesions during the progression of atherosclerosis.27 Subsequently, studies clarified the role of LDLs in cardiovascular disease development, particularly the role of oxidized LDL (ox-LDL; LDL particles that contain oxidized fatty acids) in infiltrating and damaging arterial walls, and leading to development of lesions and arterial plaques.28,29
Upon exposure of the fatty acid components of LDL particles to free radicals, they become oxidized and structural and functional changes occur to the entire LDL particle. The ox-LDL particle can damage the delicate endothelial lining of the inside of blood vessels.30 Once the ox-LDL particle has disrupted the integrity of the endothelial barrier, additional LDL particles flood into the arterial wall (intima). As oxidized LDL accumulates behind the endothelium, immune cells begin to infiltrate the site to attempt to deal with the oxidized LDL particles. Monocytes differentiate into macrophages, which engulf ox-LDL then express many adhesion molecules that limit their movement. This process results in accumulation of these macrophages rich in adhesion molecules (called foam cells), which contributes to subendothelial plaque burden. Immune cells that have infiltrated the subendothelium also release cytokines, which results in recruitment of more immune cells and LDL particles to the site. This accumulative cycle results in the formation of atherosclerotic plaque deposits, which cause the arterial wall to protrude and disrupt blood flow, a process referred to as stenosis.
Another modification to LDL thought to play a role in the development of atherosclerosis is glycation. LDL can become glycated when a sugar molecule modifies its structure. The reaction is not driven by enzymes and is dependent on the concentration of sugar in the blood, so it is easy to understand why individuals with diabetes have a greater degree of LDL glycation. LDL glycation can play a direct role in the development of atherosclerosis and also makes the LDL molecule more prone to oxidation.31,32
The recognition that modification of LDL is an initiator of endothelial damage allows for a clearer understanding of LDL’s role in the grand scheme of heart disease. Though an elevated number of native LDL particles does not directly endanger endothelial cells, it does mean that there are more LDL particles available to become oxidized (or otherwise modified), which then become more likely to damage endothelial cells.
Lowering serum cholesterol to a more healthful range—total cholesterol about 160–180 mg/dL and LDL cholesterol ideally 40— 80 mg/dL)— is one of the most frequently used strategies for reducing heart disease risk in persons without coronary heart disease (CHD).33 For young adults (age 20‒39), a 2018 expert panel recommended cholesterol-lowering therapy for those with LDL cholesterol over 160 mg/dL and a history of early-onset atherosclerosis in a close family member. For older adults, cholesterol-lowering therapy should be considered if one has diabetes, an LDL concentration ≥ 190 mg/dL, or increased risk as determined by a risk algorithm.1
Measuring Blood Lipids
The determination of relative levels of blood lipids and their lipoprotein carriers is an important step for assessing cardiovascular disease risk, as well as determining appropriate measures for attenuating this risk. Most physicians conduct a routine, fasting blood chemistry panel during a patient's annual physical. This test includes the traditional lipid panel or lipid profile, which measures total cholesterol, HDL, and triglycerides from a fasting blood sample; LDL cholesterol levels are calculated from this data.37 An extended lipid profile may also include tests for non-HDL and VLDL.38
The recognition of some limitations of conventional lipid profile testing has led to the development of advanced lipid testing, which may have an improved prognostic power over conventional lipid panels.
One such advanced lipid analysis technique is nuclear magnetic resonance (NMR) spectroscopy,39 which can directly quantitate LDL particle number. An NMR lipid analysis includes a standard lipid profile, (LDL-C, HDL-C, triglycerides, and total cholesterol) plus the following important measurements:
- LDL particle number
- Number of small LDL particles
- HDL particle number
- LDL particle size
- An analysis of insulin sensitivity based on lipid particle chemistry
Other parameters that can offer insight into cardiovascular risk associated with blood lipids include:
- Lipoprotein (a)
- Lp-PLA2 (which measures activity of an enzyme that can contribute to vascular plaque rupture)
However, as stated elsewhere in this protocol, blood lipids represent only some of the factors that can contribute to cardiovascular risk. Life Extension’s Atherosclerosis and Cardiovascular Disease protocol covers many other factors that can increase risk and which can be measured through blood testing.
What are the Best Lab Tests for Heart Health?
5 Many Factors Contribute to Vascular Disease
A 2010 analysis of the decline in CHD death rates from 1980 to 2000 highlighted the need to address multiple risk factors to protect against heart disease mortality. In this study, cholesterol reduction accounted for only a 34% decrease in mortality in individuals with heart disease. By comparison, the same paper estimated that reductions in systolic blood pressure were responsible for 53%, while smoking cessation accounted for 13%, and 5% was attributable to increased physical activity.40
In the Copenhagen Heart Study, which tracked 12,000 participants for 21 years, high cholesterol was the 6th most relevant risk factor for developing CHD in both men and women; diabetes, hypertension, smoking, physical inactivity, and no daily alcohol intake (light alcohol consumption is heart-healthy) presented larger risks for the disease.41 The controversial JUPITER trial, which examined prevention of CHD by statin drugs in persons with very low LDL cholesterol (but elevated highly-sensitive C-reactive protein [hs-CRP]) supported the conclusion that non-LDL cholesterol risk factors (such as inflammation) represent enough risk for CHD to warrant treatment, even if lipids are within low-risk ranges.42
In order to reduce risk, there must be a systematic approach and understanding of the multiple factors of cardiovascular risk and atherosclerosis. Optimal cholesterol management is important for risk reduction, but so is managing the several other established cardiovascular disease risk factors. Accordingly, efforts to lower cholesterol to mitigate cardiovascular risk should be paired with measures to reduce other risk factors such as inflammation, oxidation, hypertension, excess plasma glucose, excess body weight, excess fibrinogen, excess homocysteine, low vitamin K, insufficient vitamin D, hormone imbalance, etc.43 More information is available in Life Extension's Atherosclerosis and Cardiovascular Disease protocol.
6 Managing Blood Lipids and Lipoproteins
Deciding whether and how to treat elevated blood lipids hinges on a variety of factors, including overall cardiovascular risk and likelihood of adherence to dietary and lifestyle modification. People whose lipids are mildly elevated but are otherwise healthy may do well with dietary changes and initiation of an exercise regimen. On the other hand, people with high risk of cardiovascular disease (such as a history of cardiovascular events) may need to start on medication(s) in addition to changing their diet and exercising.
Dietary and Lifestyle Changes
Dietary modifications aim to reduce the intake and uptake of unhealthy fats such as saturated and trans fats and cholesterol from the diet. The inclusion of specific dietary compounds with cholesterol-lowering50 or cardioprotective properties may also reduce cardiovascular disease risk by several different mechanisms.
Diet is the most important aspect of a cholesterol management program. The American Heart Association and other experts recommend a diet that emphasizes51,52:
- Fruits and vegetables
- Whole grains
- Nuts and legumes
- Fish and skinless poultry, with limited amounts of red meat
- Non-tropical vegetable oils, such as olive, sunflower, safflower, canola, and other oils low in saturated fat. Note that the American Heart Association does not recommend deep frying foods regardless of the type of oil used. They also do not recommend coconut oil, which is high in saturated fat.
- Avoiding hydrogenated oils to reduce dietary trans fat
- Reducing added sugar and sodium
- Limiting daily alcohol consumption to no more than one drink for women and two drinks for men
In controlled clinical trials that replaced dietary saturated fat with polyunsaturated vegetable oils such as those listed above, cardiovascular disease was reduced by about 30%, which is roughly the degree of protection conferred by statin drugs. Substituting saturated fat for polyunsaturated vegetable oil lowers LDL cholesterol, which may in part explain the benefits observed. On the other hand, oils high in saturated fat, like coconut oil (which is approximately 90% saturated fat), can increase LDL cholesterol. In a systematic review and meta-analysis of 16 clinical trials published in the journal "Circulation," coconut oil consumption was shown to increase LDL cholesterol levels by 10.47 mg/dL compared with non-tropical vegetable oils. The authors concluded that this increase in LDL may lead to an increased risk of major vascular events, and "coconut oil should not be viewed as healthy oil for cardiovascular disease risk reduction and limiting coconut oil consumption because of its high saturated fat content is warranted."224 Observational trials have also found that higher intake of monounsaturated and polyunsaturated fats is associated with lower rates of death from cardiovascular disease or any cause. It should be noted that replacing saturated fat with refined carbohydrates and sugars has not been shown to reduce cardiovascular disease.53
Specific dietary approaches that are generally heart healthy are the Dietary Approaches to Stop Hypertension (DASH) diet and Mediterranean diet.
Caloric Restriction
Caloric restriction is the reduction of dietary calories (by up to 40%) while still maintaining good nutrition.54 Restricting energy intake slows down the body’s growth processes, causing it instead to focus on protective repair mechanisms; the overall effect is an improvement in several measures of health. Observational studies have tracked the effects of caloric restriction on lean, healthy individuals, and demonstrated that moderate restriction (22‒30% decreases in caloric intake from normal levels) improves heart function and reduces markers of inflammation (C-reactive protein, tumor necrosis factor [TNF]), risk factors for cardiovascular disease (LDL cholesterol, triglycerides, blood pressure), and diabetes risk factors (fasting blood glucose, insulin levels).55-58 Preliminary results of the Comprehensive Assessment of Long-Term Effects of Reducing Intake of Energy (CALERIE) study, a long-term multicenter trial on the effects of calorie-restricted diets in healthy overweight volunteers,59 showed moderate calorie restriction can reduce several cardiovascular risk factors (LDL cholesterol, triglycerides, blood pressure, and C-reactive protein).60
More information is available in Life Extension's Caloric Restriction protocol.
Exercise
Exercise is a fundamental component of any lipid management strategy. The American Heart Association and American College of Cardiology recommend adults engage in at least 150 minutes of moderate-intensity activity or 75 minutes of high-intensity activity each week for the prevention of cardiovascular disease.61 Exercise helps raise HDL levels and boost the efficiency of reverse cholesterol transport, the process by which cholesterol is carried from the blood vessels back to the liver for excretion.62 A small study published in 2019 found exercise had a greater effect on cholesterol synthesis than calorie restriction.63 Exercise reduces LDL significantly when combined with a heart-healthy diet—much more so than a healthy diet without exercise.64
A meta-analysis of 37 studies found that exercise usually resulted in moderate-to-strong improvements in levels of total cholesterol, LDLs, triglycerides, and HDLs. This same analysis also found that exercise consistently improved glucose and insulin levels, which are often elevated in people with an unhealthy lipid profile.65 Both aerobic and anaerobic exercise have beneficial effects on blood lipids, so an exercise regimen that combines strength training (eg, weightlifting) and endurance training (eg, running, swimming) is suggested.66
Medications to Manage Blood Lipids
Reduction of total cholesterol and LDL cholesterol (and/or triglycerides) by drugs usually involves inhibiting cholesterol production in the body or preventing the absorption/reabsorption of cholesterol from the gut. When the availability of cholesterol to cells is reduced, they are forced to pull cholesterol from the blood (which is contained in LDL particles). This has the net effect of lowering LDL cholesterol. Therapies that increase the breakdown of fatty acids in the liver or lower the amount of VLDL in the blood (like fibrate drugs or high-dose niacin)67 also result in lower serum cholesterol levels. Often, complementary strategies (eg, statin to lower cholesterol production plus a bile acid sequestrant to lower cholesterol absorption) are combined to meet cholesterol-lowering goals.
Statins
Decreasing cellular cholesterol production is the most common strategy for reducing cardiovascular disease risk, with HMG-CoA reductase inhibitors (statins) being the most commonly prescribed cholesterol-lowering treatments. Statins inhibit the activity of the enzyme HMG-CoA reductase, a key enzyme in cholesterol synthesis. Since cholesterol levels in cells are tightly controlled (cholesterol is critical to many cellular functions), the shutdown of cellular cholesterol synthesis causes the cell to respond by increasing the activity of the LDL receptor on the cell surface, which has the net effect of pulling LDL particles out of the bloodstream and into the cell. Statins may also reduce CHD risk by other mechanisms, such as reducing inflammation.68
Statins may induce serious side effects in some individuals, the most common being muscle pain or weakness (myopathy). The prevalence of myopathy is fairly low in clinical trials (1.5‒3.0%), but can be as high as 33% in community-based studies and may rise dramatically in statin users who are active (up to 75% in statin-treated athletes).69,70 The mechanisms leading to statin-induced muscle damage are not entirely understood; however, a protein called glucocorticoid-induced leucine zipper (GILZ) may be involved. GILZ plays a variety of roles in the body, including immunomodulation, adipocyte differentiation, and skeletal muscle differentiation. Statins can cause downregulation of the Akt signaling pathway, which is involved in cell survival, and eventually lead to the induction of GILZ. In turn, this promotes apoptosis (cell death) in muscle cells. A better understanding of these mechanisms is necessary to develop ways of preventing myopathy in statin users.225 Occasionally, statins may cause an elevation of the liver enzymes aspartate aminotransferase (AST) and alanine aminotransferase (ALT). These enzymes can be monitored by doing a routine chemistry panel blood test. Additionally, by inhibiting HMG-CoA reductase (an enzyme not only required for the production of cholesterol, but other metabolites as well), statins may reduce levels of the critically important antioxidant molecule CoQ10.
Targeting Cholesterol Absorption
Lowering cholesterol absorption from the intestines reduces LDL cholesterol in a different fashion; by preventing uptake of intestinal cholesterol, cells respond by making more LDL receptor, which pulls LDL particles out of the bloodstream. Ezetimibe (Zetia) and bile acid sequestrants (colesevelam [Welchol], cholestyramine [Prevalite, Questran], colestipol [Colestid]) are two classes of prescription treatment that work in this fashion. Ezetimibe acts on the cells lining the intestines (enterocytes) to reduce their ability to take up cholesterol from the intestines. Ezetimibe, when added to a statin, has been shown to reduce cardiovascular events in high-risk patients and has been recommended as an alternative treatment in those who cannot take statins.71 Bile acid sequestrants bind to bile acids in the intestine, which reduces their ability to emulsify fats and cholesterol. This has the net effect of preventing intestinal cholesterol absorption. Bile acid sequestrants may also increase HDL production in the liver, which is usually inhibited by the reabsorption of bile acids.72
PCSK9 Inhibitors
The newest medications on the scene are agents that directly inhibit the enzyme PCSK9, effectively removing an inhibitor in the LDL receptor pathway. This means that more LDL receptors are free to grab LDL particles and remove them from the blood, thus potently lowering blood LDL concentrations. While highly effective in reducing LDL, the cost of PCSK9 inhibitors severely limits their use, but they are expected to become more affordable with time.73
Bempedoic Acid
On February 21st, 2020, the Food and Drug Administration (FDA) approved bempedoic acid (Nexletol), an oral, non-statin medication taken once daily as an adjuvant to statin therapy to further lower LDL cholesterol levels. This drug is recommended for use in those with a genetic predisposition to very high cholesterol levels and those at risk of atherosclerotic cardiovascular disease.226 Several clinical trials have demonstrated the efficacy of bempedoic acid.
In a recent randomized, double-blind, placebo-controlled trial of 779 people with atherosclerotic cardiovascular disease or persistently elevated LDL (ie, LDL > 70 mg/dL despite lipid-lowering therapy), subjects received either 180 mg of bempedoic acid or placebo once daily for 52 weeks. Patients in the treatment group had significantly lower LDL levels as compared with the placebo group, especially over the first 12 weeks of the trial.227 In another randomized placebo-controlled trial, patients with high cholesterol took 80 mg of atorvastatin (Lipitor) (a common lipid-lowering medication that blocks cholesterol production) for four weeks and then added either 180 mg of bempedoic acid or placebo to their treatment regimen for four additional weeks. The treatment group had lower LDL cholesterol values compared with the placebo group.228
In a double-blind, randomized, placebo-controlled trial, 177 people with elevated LDL and normal or elevated triglyceride levels received 40, 80, or 120 mg of bempedoic acid once daily for 12 weeks. Treatment with bempedoic acid reduced LDL levels in all groups in a dose-dependent manner, regardless of triglyceride level.229
In another randomized, double-blind, placebo-controlled trial of 300 people with high cholesterol levels and at high risk of cardiovascular disease, subjects were randomized to receive either 180 mg of bempedoic acid, 10 mg of ezetimibe (which prevents the intestinal absorption of cholesterol), a combination of the two medications (180 mg of bempedoic acid and 10 mg of ezetimibe), or placebo in addition to stable statin therapy for 12 weeks. The combination of bempedoic acid and ezetimibe lowered LDL and C-reactive protein levels significantly more than placebo or monotherapy.230 These data suggest bempedoic acid is safe and effective and can be combined with other drug therapies to support healthy LDL cholesterol levels and promote cardiovascular health.231
In a randomized controlled trial involving 2,230 people with atherosclerotic cardiovascular disease or familial hypercholesterolemia (LDL > 70 mg/dL on statin therapy, with or without other lipid-lowering medications), individuals received either 180 mg of bempedoic acid or placebo daily for 52 weeks. Bempedoic acid added to statin therapy reduced LDL, non-HDL, and total cholesterol, apolipoprotein B, and C-reactive protein levels significantly from baseline to week 12, as compared with placebo. Smaller improvements were seen through week 52. While no significant differences were observed between groups in terms of adverse cardiovascular events and overall mortality, there was a greater number of cancer deaths reported in the treatment group. Blinded trial investigators concluded that these deaths most likely represented pre-existing cancers and that bempedoic acid did not lead to a higher incidence of adverse events.232 Long-term follow-up studies will help clarify whether this potential safety signal merits further investigation.
Bempedoic acid’s mechanism of action is still being uncovered. Recent animal research suggests this drug inhibits ATP citrate lyase (ACL), an enzyme involved in cholesterol and fatty acid synthesis.233,234 In humans, ACL inhibition affects only cholesterol synthesis. Further research is required to determine if bempedoic acid impacts cardiometabolic risk factors, such as hyperglycemia and insulin resistance.233
7 Nutrients
Artichoke
Artichoke has traditional usage as a liver protectant and choleretic (compound that stimulates bile flow). In stimulating bile flow, artichoke may aid the body in the disposal of excess cholesterol. In vitro studies suggest its mechanisms of action may be linked to reduction of LDL oxidation, or the ability of one of its constituents, luteolin, to indirectly inhibit HMG-CoA reductase.74
Several randomized controlled trials support the ability of artichoke extract to lower total- and/or LDL cholesterol. In one trial, artichoke extract (1,800 mg/day) for six weeks reduced total cholesterol (-9.9%) and LDL cholesterol (-16.6%) in 71 hypercholesterolemic patients, with no differences in HDL cholesterol or triglycerides.75 In another trial in hypercholesterolemic patients, 1,280 mg artichoke extract per day for 12 weeks reduced total cholesterol by 6.1% when compared with a control group. Changes in LDL cholesterol, HDL cholesterol, and triglycerides were insignificant.76 Some studies indicate artichoke supplementation can raise HDL levels.77,78 Artichoke extract also improved parameters of endothelial function in a small trial.79
Pantethine
Pantethine, a derivative of pantothenic acid (vitamin B5), can serve as a source of the vitamin. Pantethine and its metabolites appear to act on the body’s fat and cholesterol metabolism pathways. One notable function of vitamin B5 is its conversion into coenzyme A, a necessary factor in the metabolism of fatty acids into cellular energy. The pantethine derivative cysteamine may also function to reduce the activity of liver enzymes that produce cholesterol and triglycerides.80 Studies of pantethine consumption have demonstrated significant reductions in total cholesterol and LDL cholesterol (up to 13.5%), triglycerides, and elevation of HDL cholesterol in people with high cholesterol81,82 and diabetic individuals83 when taken at 900‒1,200 mg/day, although significant effects on triglycerides have been observed at dosages as low as 600 mg/day.84 A well-controlled clinical trial demonstrated that, when added to a diet low in saturated fat and cholesterol, pantethine provided an additional reduction in total cholesterol and LDL.85
Indian Gooseberry (Amla)
Indian gooseberry or amla, a common name for the plant Phyllanthus emblica (Emblica officinalis), has been used traditionally in Indian Ayurvedic medicine for a variety of conditions.86 In animal models, Indian gooseberry has demonstrated powerful antioxidant and anti-inflammatory effects.87 For instance, in an animal model of metabolic syndrome induced by a high-fructose diet, administration of amla extract controlled rising cholesterol and triglyceride levels and significantly reduced the expression of some inflammation-related genes typically elevated in metabolic syndrome.88
Several clinical trials have demonstrated that Indian gooseberry extract favorably influences blood lipids and cholesterol. Healthy and diabetic volunteers given 2 or 3 grams per day of Indian gooseberry for three weeks experienced significant decreases in total cholesterol and triglycerides.87 A head-to-head comparison between amla and simvastatin (Zocor) in 60 hyperlipidemic individuals demonstrated similar effects on lipid profile.89 In individuals with type 2 diabetes, doses of either 250 or 500 mg of an amla extract standardized to 60% low molecular weight hydrolyzable tannins, taken twice daily, significantly lowered total cholesterol, LDL and VLDL cholesterol, triglycerides, and significantly raised HDL cholesterol levels compared to both baseline and placebo.90 In a 12-week trial in 15 overweight or obese adults who were 36 years old on average, 500 mg twice daily of this same high-tannin extract had a significant beneficial impact on markers of cardiovascular risk. Compared with baseline, the amla extract reduced calculated LDL cholesterol, total cholesterol/HDL ratio, and hs-CRP levels.91
Garlic
Garlic supplements are available in a variety of formulations and preparations. Some common variations include aged garlic extract (often as branded Kyolic); high-allicin, non-aged garlic extract; black garlic extract; and garlic powder in tablets or capsules. The cardiovascular benefits of various garlic preparations have been extensively substantiated in clinical trials.253-258
A meta-analysis conducted in 2023 examined 19 randomized controlled trials including 999 participants and found that various garlic preparations significantly reduced triglycerides, total cholesterol, and LDL cholesterol compared with placebo. Studies ranged from six to 24 weeks, and the doses of garlic powder ranged from 188 to 2,400 mg daily.255
Black garlic (ie, garlic that has undergone an aging process under high heat and humidity) has been studied for its beneficial effects on cholesterol and other blood lipids. Black garlic provides a higher amount of S-allyl-cysteine (SAC) than raw garlic, and SAC is thought to contribute to the health benefits of aged and black garlic.259 A double-blind placebo-controlled trial randomized 56 healthy individuals aged 25–65 years with borderline cardiovascular disease (CVD) risk factors to receive placebo or 500 mg aged black garlic (providing 2.5 mg SAC) for 12 weeks. At the end of the intervention period, those given aged black garlic had statistically significant and clinically meaningful improvements in total cholesterol, LDL cholesterol, HDL cholesterol, and triglyceride levels compared with placebo.260
Gynostemma pentaphyllum (G. pentaphyllum)
G. pentaphyllum is used in Asian medicine to treat several chronic conditions, including diabetes and inflammatory disorders. Its effects are due in part to its ability to activate a critical enzyme called adenosine monophosphate-activated protein kinase (AMPK).97,98 This enzyme, which affects glucose metabolism and fat storage, has been called a “metabolic master switch” because it controls numerous metabolic pathways.99,100
Activation of AMPK stimulates glucose uptake in muscles and beta oxidation, in which fatty acids are broken down, while reducing the production of fat and cholesterol in the liver.97 It can also prevent damage to blood vessel lining (endothelial) cells caused by oxidized LDL.101
G. pentaphyllum stimulates AMPK activation and affects cholesterol levels in the blood and liver. A study in obese mice showed eight weeks of supplementation with G. pentaphyllum led to weight loss and improvements in glucose metabolism and cholesterol levels. Mice treated with 150 mg/kg (about 900 mg for an adult human) or 300 mg/kg (about 1,800 mg for an adult human) of the extract had total cholesterol reductions of 14.2% and 7.1%, respectively, compared with the control group.97 In a 12-week placebo-controlled trial of G. pentaphyllum in healthy obese men and women, abdominal fat, body weight, overall body fat and percent body fat were all significantly reduced compared with placebo.100
Hesperidin
Hesperidin and related flavonoids are found in a variety of plants, but especially in citrus fruits, particularly their peels.102,103 Digestion of hesperidin produces a compound called hesperetin along with other metabolites. These compounds are powerful free radical scavengers and have demonstrated anti-inflammatory, insulin-sensitizing, and lipid-lowering activity.104,105 Findings from animal and in vitro research suggest hesperidin’s positive effects on blood glucose and lipid levels may be related in part to activation of the AMPK pathway.106-108 Accumulating evidence suggest hesperidin may help prevent and treat a number of chronic diseases associated with aging.104
Hesperidin may protect against diabetes and its complications, partly through activation of the AMPK signaling pathway. Coincidentally, metformin, a leading diabetes medication, also activates the AMPK pathway. In a six-week randomized controlled trial on 24 diabetic participants, supplementation with 500 mg hesperidin daily improved glycemic control, increased total antioxidant capacity, and reduced oxidative stress and DNA injury.109 Using urinary hesperetin as a marker of dietary hesperidin, another group of researchers found those with the highest level of hesperidin intake had 32% lower risk of developing diabetes over 4.6 years compared to those with the lowest intake level.110
In a randomized controlled trial, 24 adults with metabolic syndrome were treated with 500 mg hesperidin per day or placebo for three weeks. After a washout period, the trial was repeated with hesperidin and placebo assignments reversed. Hesperidin treatment improved endothelial function, suggesting this may be one important mechanism behind its benefit to the cardiovascular system. Hesperidin supplementation also led to a 33% reduction in median levels of the inflammatory marker hs-CRP,111 as well as significant decreases in levels of total cholesterol, ApoB, and markers of vascular inflammation relative to placebo.107 In another randomized controlled trial in overweight adults with evidence of vascular dysfunction, 450 mg per day of a hesperidin supplement for six weeks resulted in lower blood pressure and a decrease in markers of vascular inflammation.112 In yet another randomized controlled trial including 75 heart attack patients who received 600 mg hesperidin or placebo daily for four weeks, those taking hesperidin had significant improvements in HDL cholesterol and markers of vascular inflammation and fatty acid and glucose metabolism.113
Plant Sterols
Plant sterols are steroid compounds found in plants that function similarly to cholesterol in animals (ie, as components of plant cell membranes, and precursors to plant hormones).114 Like cholesterol, they can exist as free molecules or sterol esters. Esters of sterols have a higher activity and better fat solubility, which allows for lower effective dosages (2‒3 grams/day as opposed to 5‒10 grams/day for unesterified sterols).115 Sterols themselves are poorly absorbed from the diet, but because of their chemical similarity to cholesterol, are thought to compete with cholesterol for absorption in the intestines, which has the net effect of reducing LDL levels.116 Sterols may also reduce cholesterol production in the liver, reduce the synthesis of VLDLs, increase LDL particle size, and increase LDL uptake from the blood.117,118 HDL is generally not affected by sterol intake.119
There have been numerous studies of the effects of sterol esters on reducing mean total cholesterol and LDL cholesterol in healthy, hypercholesterolemic, and diabetic individuals. An analysis of 57 trials involving over 3,600 individuals reported an average LDL cholesterol reduction of 9.9% at a mean intake of 2.4 grams sterol esters/day.120 This benefit extends to sterol supplements delivered in tablet form.114 Sufficient evidence for the cholesterol-lowering effects of sterols prompted the U.S. Food and Drug Administration (FDA) to permit the health claim that sterol esters may be associated with a reduced risk of CHD, when taken at sufficient levels in the context of a healthy diet.121 The National Cholesterol Education Program122 and American Heart Association123 both support the use of sterols in their dietary recommendations. A comprehensive review by a lipid expert panel suggests plant sterols alone or in combination with other dietary supplements and/or ezetimibe could be considered as alternatives or add-on therapy to statins.124
Green Coffee Extract
Extract from unroasted coffee beans, or green coffee extract, has been used to promote cardiovascular and metabolic health. Green coffee contains high levels of polyphenols called chlorogenic acids (CGAs), which have been suggested as important health-promoting compounds in coffee. However, conventional coffee roasting methods typically reduce polyphenol levels.243 Preclinical studies indicate CGA may help regulate cholesterol levels by contributing to the increased expression of cholesterol 7 alpha-hydroxylase (CYP7A1), the enzyme responsible for catalyzing the first step of converting cholesterol to bile acids.244 Chlorogenic acid was also shown, in a mouse model, to decrease plasma levels of total and LDL cholesterol and promote cholesterol efflux from macrophages, a process that helps prevent the buildup of atherosclerotic plaque. It also decreased triglycerides and some inflammatory biomarkers.245
Numerous clinical studies, both in healthy populations and in those with cardiometabolic diseases, have demonstrated the benefits of green coffee extract on lipid levels, as well as other cardiometabolic markers.246 In a meta-analysis of 14 randomized controlled trials including 766 participants, green coffee extract at dosages ranging from 90 to 6,000 mg/day (providing 13.5 to 1,200 mg/day CGA) for up to 16 weeks lowered serum total cholesterol. In studies that lasted over eight weeks, there was a significant reduction of LDL as well.247 And in a meta-analysis of 27 randomized controlled trials including 992 participants, green coffee extract at similar dosages (200 to 2,000 mg/day) for up to 24 weeks lowered serum triglycerides and increased HDL levels.248 Green coffee extract also appears to have positive effects on glucose and insulin levels, body weight, blood pressure, and cognitive function.249-252
Guggul/gum guggul
Guggul/gum guggul, the resin of the Commiphora mukul tree, has a history of traditional usage in Ayurvedic medicine and is widely used in Asia as a cholesterol-lowering agent. Guggulipid is a lipid extract of the gum that contains plant sterols (guggulsterones E and Z), the proposed bioactive compounds.125 Evidence of inconsistent quality suggest guggul supplementation may help improve the lipid profile, but better-designed trials are needed to clarify efficacy.125-131
Soluble Fibers
Soluble fibers include non-digestible and fermentable carbohydrates, and their sufficient intake has been associated with lower prevalence of cardiovascular disease.119 When included as part of a low-saturated fat/low-cholesterol diet, they can lower LDL cholesterol, generally by roughly 5‒10%, in hypercholesterolemic and diabetic patients, and may reduce LDL cholesterol in healthy individuals as well.132 The cholesterol-lowering properties of soluble oat fiber, psyllium, pectin, guar gum, beta-glucans from barley, and chitosan are substantiated by dozens of controlled human clinical trials.133-135 A 2015 review of 17 randomized controlled trials concluded that beta-glucan consumption significantly reduced total cholesterol and LDL in hypercholesterolemic individuals without any reported adverse effects.136 Soluble fibers lower cholesterol by several potential mechanisms.133 They may directly bind bile acids or dietary cholesterol, preventing/disrupting their absorption. Their high viscosities (measure of a liquid’s thickness) and effects on intestinal motility may slow or limit macronutrient uptake. They can also increase satiety, which can limit overall energy intake.
Coenzyme Q10 (CoQ10)
The generation of chemical energy in the form of ATP by mitochondria is an essential biological function. Delicate endothelial cells that line arterial walls depend on healthy mitochondrial function to control blood pressure and vascular tone. Oxidized or glycated LDL can sabotage endothelial mitochondrial function and damage the endothelial barrier, setting the stage for the atherosclerotic cascade to initiate.145,146 CoQ10 is an integral component of mitochondrial metabolism, serving as an intermediary transporter between two major check points in ATP production. Interestingly, because CoQ10 is lipid soluble, it is incorporated into LDL particles, where it serves to protect against oxidation. Insufficient levels of CoQ10 expedite atherogenesis by limiting mitochondrial efficiency in endothelial cells and leaving LDL particles vulnerable to oxidative damage. Statin drugs, which are typically used to treat high cholesterol, ironically also suppress levels of CoQ10 in the blood.147 Individuals taking a statin drug should always supplement with CoQ10.
In a randomized, controlled, double-blind trial, the addition of 200 mg CoQ10 daily in people taking statins and omega-3 fatty acids improved a range of cardiovascular risk factors. Total cholesterol, systolic blood pressure, and inflammatory markers (hs-CRP and interleukin-6) were all significantly reduced, while internal antioxidant defenses increased and statin-related adverse effects diminished.148
A 2018 review of randomized clinical trials in patients with coronary artery disease concluded that supplementation with CoQ10 reduces total cholesterol and increases HDL. This study also detected a trend for CoQ10 to reduce LDL and Lp(a).149 A previous review also found that CoQ10 supplementation reduced Lp(a).150
Carotenoids
Carotenoids are common constituents of the LDL particle. Beta-carotene is the second most abundant antioxidant in LDL; other common dietary carotenoids (lycopene, lutein) may be transported by LDL particles as well.28 Together, these three carotenoids have an indispensable role in the protection of LDL particles from oxidative damage; their serum levels have been demonstrated to be the most predictive of the degree of LDL oxidation in humans.151 Carotenoids may also possess additional lipid-lowering activities independent of their antioxidant potential. The best-studied in this respect is lycopene; an analysis of 12 human trials of lycopene revealed an average reduction in LDL cholesterol of approximately 12%.152 Potential mechanisms for this action are suppression of cholesterol synthesis by inhibition of the HMG-CoA reductase enzyme or an increase in the rate of LDL degradation.153 In an animal model, lycopene attenuated neointimal hyperplasia, a process that has a negative effect on the long-term success of balloon angioplasty of arteries.154 In another animal model, supplementing virgin olive oil with lycopene had an additive beneficial effect on blood lipids and even resulted in a decrease in body weight gain.155 A systematic review of human intervention trials that studied the consumption of tomato products and lycopene supplementation found that these interventions have beneficial effects of blood lipids and vascular function.156
Vitamin E
Natural tocopherols and tocotrienols together form vitamin E. These fat-soluble antioxidants have been studied for decades. Vitamin E inhibits the oxidation of LDL particles.157,158
Alpha-tocopherol is the best-known form of vitamin E and is found in the largest quantities in blood and tissue. It is critical, however, for anyone supplementing with vitamin E to make sure they are also getting adequate gamma-tocopherol each day. One of the most important benefits of gamma-tocopherol is its ability to improve endothelial function by increasing nitric oxide synthase, the enzyme responsible for producing vessel-relaxing nitric oxide.159 One major way it produces this effect is by sponging up destructive reactive nitrogen species, such as peroxynitrite.160 In fact, gamma-tocopherol is able to “trap” a variety of reactive nitrogen species and halt their negative effects on a host of cellular processes.161
Supplementation in humans with 100 mg per day gamma-tocopherol resulted in a reduction in several risk factors for vascular disease (eg, platelet aggregation and LDL cholesterol levels).162
Tea Polyphenols
Polyphenols are a group of over 8,000 compounds present in plants, plant-based foods, and beverages. Ingestion of polyphenols, through diet or supplementation, has been shown to exert beneficial health effects, and these compounds may favorably affect blood lipids in several ways.261,262 Epidemiological and observational studies have consistently found that higher polyphenol consumption is linked to lower incidence of chronic degenerative disease.263,264 Green and black tea, both made from the leaves of Camellia sinensis, contain a diverse array of polyphenols. Green tea is notable for its content of a type of polyphenol called catechins, particularly epigallocatechin gallate (EGCG) and epicatechin. Black tea is a source of theaflavins and thearubigins.265
A systematic review and meta-analysis examined 31 randomized controlled trials, roughly half of which were conducted in East Asian countries, and which enrolled a total of 3,321 subjects. The trials used various preparations with defined amounts of catechins. On average, green tea significantly lowered total cholesterol and LDL cholesterol compared with controls.266 An earlier meta-analysis also supports this finding for green tea, while a systematic review of trials of EGCG found that it lowered LDL cholesterol.164,267
A pilot, double-blind, placebo-controlled trial in 13 individuals with metabolic syndrome tested, for three months, the combination of 40 mg of the statin drug simvastatin and 100 mg of purified epicatechin. In the group that took the statin-epicatechin combination, total and non-HDL cholesterol improved significantly compared with baseline. There was also a significant difference compared with statin alone in change in HDL cholesterol. In the combination group, HDL cholesterol increased, while it decreased in the statin-only group. Compared with statin-only, the combination group saw a significant drop in LDL particle and small LDL particle numbers. The combination group had a significantly lower 10-year atherosclerosis and cardiovascular disease risk (a multicomponent measure of risk of a cardiac event within a 10-year window) at the end of the study compared with the statin-only group.261
Pomegranate
Pomegranate is now widely viewed as a superfruit with a myriad of health benefits, and rightfully so; dozens of placebo-controlled clinical trials have been carried out on pomegranate juice or pomegranate extract. With respect to lipid management, the efficacy of pomegranate is rivaled by very few natural compounds. The high concentration of polyphenols (particularly punicalagins) in pomegranate make it an ideal ingredient for suppressing LDL oxidation.168,169
Consumption of pomegranate polyphenols significantly lowered total and LDL cholesterol concentrations while maintaining HDL levels in subjects with elevated cholesterol profiles.170 Pomegranate also suppresses immunoreactivity against oxidized LDL, a mechanism which would be expected to limit plaque formation in the intimia.171 In fact, this is exactly what was shown in a long-term study of pomegranate consumption. Subjects received either pomegranate juice or placebo for three years; in the placebo group, carotid intima media thickness (cIMT; a measure of atherosclerosis) increased by 9% one year after study initiation, while cIMT decreased 30% in the pomegranate group. Pomegranate also significantly reduced oxidized LDL concentrations and increased serum antioxidant activity, compared with placebo, while simultaneously lowering blood pressure. Moreover, pomegranate nearly doubled the activity of paraoxonase-1 (PON-1), an antiatherogenic enzyme that optimizes HDL function and protects lipids from oxidative damage.172 Both groups continued on standard therapy that may have included statins, anti-hypertensives, etc.
In a randomized controlled clinical trial, women with metabolic syndrome were given polyphenol-rich pomegranate juice or water. Women who received the juice were found to have significantly lower levels of an oxidative stress marker and higher levels of healthy monounsaturated fatty acids in their red blood cell membranes. Plasma levels of the pro-inflammatory fat arachidonic acid were reduced in juice group.173 Consumption of pomegranate juice was shown to lower atherosclerosis risk factors in hemodialysis patients in another randomized placebo-controlled trial.174 In a trial on 48 overweight or obese participants, 1,000 mg pomegranate extract daily improved total cholesterol, LDL cholesterol, and HDL cholesterol levels as well as several other markers of metabolic health and inflammation.175
Curcumin
The active ingredient in turmeric (and also the main compound that gives it its bright yellow color), curcumin has a variety of protective roles in CVD, potentially reducing oxidative stress, inflammation, and the proliferation of smooth muscle cells and monocytes. Ninety-five small human trials have revealed the effects of curcumin on reducing lipid peroxidation176,177 and plasma fibrinogen,178 both factors in the progression of atherosclerosis.179 Curcumin may also reduce serum cholesterol by increasing the production of the LDL receptor,180,181 but despite successes in animal models, human data on the anti-hypercholesterolemic effects of curcumin is conflicting. A small study of 10 healthy volunteers revealed significant decreases in lipid oxidation products (-33%) and total cholesterol (-12%), with a concomitant increase in HDL cholesterol (29%) when using 500 mg curcumin daily for seven days.182 In two subsequent studies, low-dose curcumin showed a non-significant trend toward lowering total cholesterol and LDL cholesterol in acute coronary patients,183 while high dose-curcumin (1‒4 grams/day) exhibited non-significant increases in total-, LDL-, and HDL cholesterol.184
An analysis of seven randomized controlled trials concluded that turmeric and curcumin were effective in lowering LDL cholesterol and triglycerides without any serious side effects.185 When compared with placebo, supplementation with turmeric led to a reduction in body mass index, total cholesterol, and triglycerides in a study of patients with type 2 diabetes.111 A similar study involving individuals with metabolic syndrome found curcumin had a beneficial effect on lipid parameters (decreased LDL and increased HDL).186
Fish Oil
Fish oil has long been used as a nutritional supplement to support cardiovascular health. It contains the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). These fatty acids have been shown to positively modulate several aspects of cholesterol and lipid metabolism. For instance, omega-3 fatty acids may beneficially affect reverse cholesterol transport. Omega-3 fatty acids are thought to accomplish this by influencing HDL remodeling and promoting excretion of cholesterol components via the liver and gallbladder.187 Increasing omega-3 fatty acid intake also helps keep the omega-6 to omega-3 fatty acid ratio in a healthy balance. The evolutionary human diet contained roughly equal amounts of omega-6 and omega-3 fatty acids, whereas today’s Western diet typically contains much more omega-6 fats. An imbalance too heavily favoring omega-6 is associated with increased risk for a variety of diseases, including cardiovascular disease.188 In addition to eating healthful foods rich in omega-3s, supplementation is an effective method of balancing the ratio.
In a review of 22 studies on supplemental or prescription fish oil preparations that evaluated the effects of EPA and DHA on blood lipids, DHA was found to decrease triglyceride levels by 6.8% and increase HDL levels by 5.9%.189 Although DHA is known to slightly elevate LDL levels, high-dose DHA has been shown to increase LDL turnover and LDL particle size190 (larger LDL particles may not promote atherosclerosis as much as small LDL particles). In a rigorous clinical trial, 4 grams daily of a prescription-only drug form of EPA called icosapent ethyl (Vascepa) reduced the risk of cardiovascular events in people with high triglycerides who were taking statins.191 A post-hoc analysis further found that those in the icosapent ethyl group had a 34% reduction in first revascularization procedures, 36% reduction in total revascularizations, 32% fewer stenting procedures, and 39% fewer coronary artery bypass grafting procedures.235
In mid-2019, the U.S. FDA responded to a health petition and stated it would not object a qualified health claim for omega-3 fatty acids despite concluding that “the evidence is inconsistent and inconclusive.” One variant of the qualified health claim to which the FDA has not objected reads as follows:
- “Consuming EPA and DHA combined may reduce the risk of CHD (coronary heart disease) by lowering blood pressure.”
Probiotics
Probiotics, which are increasingly recognized for their role in regulating immune activity and reducing inflammation throughout the body, have attracted interest for their ability to reduce LDL cholesterol and cardiovascular risk. A review of research found that, of the probiotic strains studied, Lactobacillus reuteri192 NCIMB 30242 has been shown to lower levels of total and LDL cholesterol as well as markers of inflammation.193
In a randomized controlled trial, 114 participants with high cholesterol levels who were otherwise healthy consumed either a probiotic yogurt providing 2.8 billion colony forming units (CFUs) of microencapsulated L. reuteri NCIMB 30242 or a control yogurt daily for six weeks. The L. reuteri group had reductions in total cholesterol (9%) and LDL cholesterol (5%), levels relative to the placebo yogurt group. ApoB-100, which at high levels is associated with vascular disease, was significantly reduced in the L. reuteri group.194,195 In another controlled clinical trial on 127 healthy adults with high cholesterol levels, subjects received either L. reuteri NCIMB 30242 capsules or placebo for nine weeks. Those taking L. reuteri had a greater than 9% drop in total cholesterol and greater than 11.5% drop in LDL cholesterol levels compared with placebo. The ratio of ApoB-100 to ApoA-1 fell by 9% in the L. reuteri-supplemented group compared with placebo. The ApoB-100:ApoA-1 ratio is a strong predictor of cardiovascular risk, particularly in overweight and obese individuals.196,197 Fibrinogen and hs-CRP, additional markers of cardiovascular risk, were also significantly reduced relative to placebo.198 Interestingly, later analyses of the study results noted that subjects taking the probiotic experienced general improvement in functional gastrointestinal symptoms199 and a significant increase in vitamin D levels compared with placebo.200
Although the exact mechanism by which L. reuteri NCIMB 30242 improves lipid levels has not been fully characterized, it is known that intestinal microbes have a role in regulating cholesterol transport and metabolism, and this effect may in part depend on an ability to break down bile acids in the digestive tract.193,198 Bile acids have a close relationship with intestinal microbiota and help regulate cholesterol synthesis and lipid and glucose metabolism.201,202 By increasing bile acid breakdown and excretion, L. reuteri is thought to stimulate cholesterol-dependent bile acid production in the liver, removing cholesterol from circulation.202
An analysis of 30 randomized controlled trials encompassing over 1,600 subjects found those treated with probiotics had on average 7.8 point-lower total cholesterol and 7.3 point-lower LDL levels than those not treated.203 Another review concluded L. acidophilus-specific probiotics were particularly effective in lowering LDL levels.204 Furthermore, a systematic review of all Lactobacillus strains used in randomized controlled trials found that on average, Lactobacillus use shaved about five points off total cholesterol and four points off LDL cholesterol.192 A review of probiotic use in the type 2 diabetes population205 found that significant reductions in total cholesterol, triglycerides, LDL, and blood pressure were associated with supplementation.206
Prebiotics
Prebiotics, a subset of soluble fiber, have gained attention for their ability to be selectively fermented by gut flora for a variety of potential health-promoting benefits. The fermentation of prebiotic fibers into short-chain fatty acids such as acetate, butyrate, or propionate may inhibit cholesterol synthesis in the liver.133 In human trials, the prebiotic fibers inulin and dextrin induced reductions in serum levels of total cholesterol (-9% and -2% for inulin and dextrin, respectively), LDL cholesterol (-1 % for dextrin), and triglycerides (-21% for inulin).207,208
Red Yeast Rice
Red yeast rice is a traditional preparation of rice fermented by the yeast Monascus purpureus. The yeast produces metabolites (monacolins) that are naturally-occurring HMG-CoA reductase inhibitors (one of these, monacolin K, is chemically identical to lovastatin).209 A comprehensive review of 93 randomized trials including nearly 10,000 patients demonstrated that commercial preparations of red yeast rice produced reductions in total cholesterol, LDL cholesterol, triglycerides, and an increase in HDL cholesterol.210 A multicenter well-controlled trial which compared a red yeast rice extract called Xuezhikang to placebo demonstrated a 27% decrease in LDL.211 And a review of 22 clinical trials supported Xuezhikang’s use to reduce death and heart attack risk when added to conventional treatment for CHD.212 A long-term (4.5 year) multicenter study of nearly 5,000 patients with a previous heart attack and high total cholesterol levels demonstrated that a commercial red yeast rice preparation reduced the incidence of major coronary events, including nonfatal heart attack and cardiovascular mortality, when compared with placebo.213 Red yeast rice extracts have also been shown to be well tolerated and effective in lowering LDL in patients with statin intolerance214,215 and safe and effective in LDL-lowering even in younger individuals.216 Because of this evidence, the International Lipid Expert Panel recommended red yeast rice’s use in those whose LDL target has not been reached due to statin intolerance.124
Due to regulations regarding their labeling in the United States, standardization of commercial red yeast rice preparations for monacolins is problematic, thus levels of monacolins can vary dramatically between red yeast rice products.217
Bergamot
Likely indigenous to the Calabria region of Italy, bergamot is a fruit considered by most experts to be a hybrid of sour orange and lemon, or perhaps a natural mutation of lemon. Bergamot’s essential oils contain flavonoid glycosides which have statin-like effects6 and also naringin, which has been shown in an animal model to inhibit the oxidation of LDL and increase the fecal excretion of cholesterol.50 A small clinical trial in people with high cholesterol showed bergamot juice extract reduced total cholesterol, LDL and triglycerides; decreased the presence of small, dense LDL; and led to significant shrinkage of atherosclerotic plaque in the carotid arteries.218 A trial that enrolled 98 people with elevated blood lipids found that 12 weeks of supplementation with a formula based on bergamot extracts led to reduced triglycerides and greater weight loss compared with placebo.219 Bergamot may also complement statin drugs. An open-label placebo-controlled trial enrolled 77 people with elevated LDL cholesterol and triglycerides and randomized them into four groups: 1) placebo, 2) 10 mg or 20 mg daily of rosuvastatin, 3) bergamot polyphenols, and 4) bergamot polyphenols plus 10 mg rosuvastatin. The researchers found that the bergamot polyphenols enhanced the lipid-lowering effect of rosuvastatin.220
2025
- Jul: Updated section on tea polyphenols in Nutrients
2024
- Apr: Updated section on garlic in Nutrients
2023
- Jun: Added section on green coffee extract to Nutrients
2022
- Feb: Updated Introduction
- Feb: Updated section on lipoproteins in The Blood Lipids: Cholesterol and Triglycerides
2019
- July: Comprehensive update & review
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol. Circulation. 2018:CIR0000000000000625.
- Nicholls SJ. The New Face of Hyperlipidemia and the Role of PCSK9 Inhibitors. Current cardiology reports. 2019;21(4):18.
- Banach M, Penson PE. What have we learned about lipids and cardiovascular risk from PCSK9 inhibitor outcome trials: ODYSSEY and FOURIER? Cardiovascular Research. 2019;115(3):e26-e31.
- Elhomsy G. Apolipoprotein B. Medscape. Laboratory Medicine Web site. https://emedicine.medscape.com/article/2087335-overview#a4. Published 2014. Accessed 04/22/19.
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. 2018:25709.
- Di Donna L, De Luca G, Mazzotti F, et al. Statin-like principles of bergamot fruit (Citrus bergamia): isolation of 3-hydroxymethylglutaryl flavonoid glycosides. J Nat Prod. 2009;72(7):1352-1354.
- Häggström M RD. Diagram of the pathways of human steroidogenesis. In: pathways S, ed. WikiJournal of Medicine 1 (1)2014.
- Alberts B, Johnson A, Lewis J ea. Molecular Biology of the Cell. 4th edition. New York: Garland Science; 2002. The Lipid Bilayer. In:2002: https://www.ncbi.nlm.nih.gov/books/NBK26871/. Accessed 03/12/2019.
- Fardella CE, Miller WL. Molecular biology of mineralocorticoid metabolism. Annu Rev Nutr. 1996;16(1):443-470.
- Xu K, Morgan KT, Todd Gehris A, Elston TC, Gomez SM. A whole-body model for glycogen regulation reveals a critical role for substrate cycling in maintaining blood glucose homeostasis. PLoS computational biology. 2011;7(12):e1002272-e1002272.
- Salter AM, Brindley DN. The biochemistry of lipoproteins. Journal of inherited metabolic disease. 1988;11 Suppl 1:4-17.
- Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106(25):3143-3421.
- Feingold KR, Grunfeld C. Introduction to Lipids and Lipoproteins. In: Feingold KR, Anawalt B, Boyce A, et al., eds. Endotext. South Dartmouth (MA): MDText.com, Inc.; 2000.
- Lagor W, Millar J. Overview of the LDL receptor: relevance to cholesterol metabolism and future approaches to the treatment of coronary heart disease. JRLCR. 2009;3:1-14.
- Tribble DL, Holl LG, Wood PD, Krauss RM. Variations in oxidative susceptibility among six low density lipoprotein subfractions of differing density and particle size. Atherosclerosis. 1992;93(3):189-199.
- Chait A, Brazg RL, Tribble DL, Krauss RM. Susceptibility of small, dense, low-density lipoproteins to oxidative modification in subjects with the atherogenic lipoprotein phenotype, pattern B. Am J Med. 1993;94(4):350-356.
- Liu ML, Ylitalo K, Salonen R, Salonen JT, Taskinen MR. Circulating oxidized low-density lipoprotein and its association with carotid intima-media thickness in asymptomatic members of familial combined hyperlipidemia families. Arteriosclerosis, thrombosis, and vascular biology. 2004;24(8):1492-1497.
- Koba S, Yokota Y, Hirano T, et al. Small LDL-cholesterol is superior to LDL-cholesterol for determining severe coronary atherosclerosis. Journal of atherosclerosis and thrombosis. 2008;15(5):250-260.
- Rubenfire M, American College of Cardiology. Lipoprotein(a): Diagnosis, Prognosis, Emerging Therapies. https://www.acc.org/latest-in-cardiology/ten-points-to-remember/2017/02/07/14/21/a-test-in-context-lipoproteina-diagnosis. Published 2017. Accessed Jun. 27, 2019.
- Schettler VJJ, Neumann CL, Peter C, et al. Lipoprotein apheresis is an optimal therapeutic option to reduce increased Lp(a) levels. Clinical research in cardiology supplements. 2019;14(Suppl 1):33-38.
- Thanassoulis G. Screening for High Lipoprotein(a): The Time Is Now. Circulation. 2019;139(12):1493-1496.
- Borrelli MJ, Youssef A, Boffa MB, Koschinsky ML. New Frontiers in Lp(a)-Targeted Therapies. Trends Pharmacol Sci. 2019;40(3):212-225.
- Reiner Z. Can Lp(a) Lowering Against Background Statin Therapy Really Reduce Cardiovascular Risk? Curr Atheroscler Rep. 2019;21(4):14.
- Langer C, Gansz B, Goepfert C, et al. Testosterone up-regulates scavenger receptor BI and stimulates cholesterol efflux from macrophages. Biochemical and biophysical research communications. 2002;296(5):1051-1057.
- Herbst KL, Amory JK, Brunzell JD, Chansky HA, Bremner WJ. Testosterone administration to men increases hepatic lipase activity and decreases HDL and LDL size in 3 wk. Am J Physiol Endocrinol Metab. 2003;284(6):E1112-1118.
- Turhan S, Tulunay C, Gulec S, et al. The association between androgen levels and premature coronary artery disease in men. Coronary artery disease. 2007;18(3):159-162.
- Steinberg D. Thematic review series: the pathogenesis of atherosclerosis. An interpretive history of the cholesterol controversy: part I. Journal of lipid research. 2004;45(9):1583-1593.
- Reaven PD, Witztum JL. Oxidized low density lipoproteins in atherogenesis: role of dietary modification. Annu Rev Nutr. 1996;16:51-71.
- Steinberg D. The LDL modification hypothesis of atherogenesis: an update. Journal of lipid research. 2009;50 Suppl:S376-381.
- Chouinard JA, Grenier G, Khalil A, Vermette P. Oxidized-LDL induce morphological changes and increase stiffness of endothelial cells. Experimental cell research. 2008;314(16):3007-3016.
- Fournet M, Bonte F, Desmouliere A. Glycation Damage: A Possible Hub for Major Pathophysiological Disorders and Aging. Aging Dis. 2018;9(5):880-900.
- Hegab Z, Gibbons S, Neyses L, Mamas MA. Role of advanced glycation end products in cardiovascular disease. World J Cardiol. 2012;4(4):90-102.
- Havranek EP. Primary prevention of CHD: nine ways to reduce risk. American family physician. 1999;59(6):1455-1463, 1466.
- Rizos CV, Elisaf MS, Liberopoulos EN. Effects of thyroid dysfunction on lipid profile. The open cardiovascular medicine journal. 2011;5:76-84.
- Wanjia X, Chenggang W, Aihong W, et al. A high normal TSH level is associated with an atherogenic lipid profile in euthyroid non-smokers with newly diagnosed asymptomatic coronary heart disease. Lipids in health and disease. 2012;11:44.
- van Tienhoven-Wind LJ, Dullaart RP. Low-normal thyroid function and novel cardiometabolic biomarkers. Nutrients. 2015;7(2):1352-1377.
- Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clinical chemistry. 1972;18(6):499-502.
- Bhatt DL. Non-HDL cholesterol explained. Heart Health Publishing online. https://www.health.harvard.edu/heart-health/non-hdl-cholesterol-explained. 1/2018. Accessed 5/31/2019.
- Wolska A, Remaley A. Lipoprotein Subfraction Analysis. Clinical Laboratory News online. https://www.aacc.org/publications/cln/articles/2017/january/lipoprotein-subfractionation-analysis. 1/3/2017. Accessed 5/31/2019. 2017.
- Ford ES, Ajani UA, Croft JB, et al. Explaining the decrease in U.S. deaths from coronary disease, 1980-2000. The New England journal of medicine. 2007;356(23):2388-2398.
- Schnohr P, Jensen JS, Scharling H, Nordestgaard BG. Coronary heart disease risk factors ranked by importance for the individual and community. A 21 year follow-up of 12 000 men and women from The Copenhagen City Heart Study. Eur Heart J. 2002;23(8):620-626.
- Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. The New England journal of medicine. 2008;359(21):2195-2207.
- Daviglus ML, Stamler J, Pirzada A, et al. Favorable cardiovascular risk profile in young women and long-term risk of cardiovascular and all-cause mortality. Jama. 2004;292(13):1588-1592.
- Sobal G, Menzel J, Sinzinger H. Why is glycated LDL more sensitive to oxidation than native LDL? A comparative study. Prostaglandins Leukot Essent Fatty Acids. 2000;63(4):177-186.
- Nivoit P, Wiernsperger N, Moulin P, Lagarde M, Renaudin C. Effect of glycated LDL on microvascular tone in mice: a comparative study with LDL modified in vitro or isolated from diabetic patients. Diabetologia. 2003;46(11):1550-1558.
- Toma L, Stancu CS, Botez GM, Sima AV, Simionescu M. Irreversibly glycated LDL induce oxidative and inflammatory state in human endothelial cells; added effect of high glucose. Biochemical and biophysical research communications. 2009;390(3):877-882.
- Dong Y, Wu Y, Wu M, et al. Activation of protease calpain by oxidized and glycated LDL increases the degradation of endothelial nitric oxide synthase. Journal of cellular and molecular medicine. 2009;13(9a):2899-2910.
- Calvo C. [Non-enzymatic glycosylation of lipoproteins in the pathogenesis of atherosclerosis in diabetics]. Revista medica de Chile. 1997;125(4):460-465.
- Soran H, Durrington PN. Susceptibility of LDL and its subfractions to glycation. Curr Opin Lipidol. 2011;22(4):254-261.
- Miceli N, Mondello MR, Monforte MT, et al. Hypolipidemic effects of Citrus bergamia Risso et Poiteau juice in rats fed a hypercholesterolemic diet. J Agric Food Chem. 2007;55(26):10671-10677.
- American Heart Association. Healthy Cooking Oils. https://www.heart.org/en/healthy-living/healthy-eating/eat-smart/fats/healthy-cooking-oils. Published 2018. Updated 04/24/2018. Accessed 04/30/2019.
- Association AH. The American Heart Association Diet and Lifestyle Recommendations. https://www.heart.org/en/healthy-living/healthy-eating/eat-smart/nutrition-basics/aha-diet-and-lifestyle-recommendations. Published 2015. Updated 08/15/2015. Accessed 04/30/2019.
- Sacks FM, Lichtenstein AH, Wu JHY, et al. Dietary Fats and Cardiovascular Disease: A Presidential Advisory From the American Heart Association. Circulation. 2017;136(3):e1-e23.
- LANE MA, BLACK A, INGRAM DK, ROTH GS. Calorie Restriction in Nonhuman Primates: Implications for Age-Related Disease Risk. Journal of Anti-Aging Medicine. 1998;1(4):315-326.
- Walford RL, Mock D, Verdery R, MacCallum T. Calorie restriction in biosphere 2: alterations in physiologic, hematologic, hormonal, and biochemical parameters in humans restricted for a 2-year period. The journals of gerontology Series A, Biological sciences and medical sciences. 2002;57(6):B211-224.
- Meyer TE, Kovacs SJ, Ehsani AA, Klein S, Holloszy JO, Fontana L. Long-term caloric restriction ameliorates the decline in diastolic function in humans. Journal of the American College of Cardiology. 2006;47(2):398-402.
- Fontana L, Klein S, Holloszy JO, Premachandra BN. Effect of long-term calorie restriction with adequate protein and micronutrients on thyroid hormones. J Clin Endocrinol Metab. 2006;91(8):3232-3235.
- Fontana L, Meyer TE, Klein S, Holloszy JO. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc Natl Acad Sci U S A. 2004;101(17):6659-6663.
- Stewart TM, Bhapkar M, Das S, et al. Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy Phase 2 (CALERIE Phase 2) screening and recruitment: methods and results. Contemporary clinical trials. 2013;34(1):10-20.
- Lefevre M, Redman LM, Heilbronn LK, et al. Caloric restriction alone and with exercise improves CVD risk in healthy non-obese individuals. Atherosclerosis. 2009;203(1):206-213.
- Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease. Circulation.0(0):CIR.0000000000000678.
- Nassef Y, Lee KJ, Nfor ON, Tantoh DM, Chou MC, Liaw YP. The Impact of Aerobic Exercise and Badminton on HDL Cholesterol Levels in Adult Taiwanese. Nutrients. 2019;11(3).
- Cho AR, Moon JY, Kim S, et al. Effects of alternate day fasting and exercise on cholesterol metabolism in overweight or obese adults: A pilot randomized controlled trial. Metabolism: clinical and experimental. 2019;93:52-60.
- Stefanick ML, Mackey S, Sheehan M, Ellsworth N, Haskell WL, Wood PD. Effects of Diet and Exercise in Men and Postmenopausal Women with Low Levels of HDL Cholesterol and High Levels of LDL Cholesterol. New England Journal of Medicine. 1998;339(1):12-20.
- Glenney SS, Brockemer DP, Ng AC, Smolewski MA, Smolgovskiy VM, Lepley AS. Effect of Exercise Training on Cardiac Biomarkers in At-Risk Populations: A Systematic Review. J Phys Act Health. 2017;14(12):968-989.
- Patel H, Alkhawam H, Madanieh R, Shah N, Kosmas CE, Vittorio TJ. Aerobic vs anaerobic exercise training effects on the cardiovascular system. World journal of cardiology. 2017;9(2):134-138.
- Barter PJ, Rye KA. Cardioprotective properties of fibrates: which fibrate, which patients, what mechanism? Circulation. 2006;113(12):1553-1555.
- Devaraj S, Siegel D, Jialal I. Statin therapy in metabolic syndrome and hypertension post-JUPITER: what is the value of CRP? Curr Atheroscler Rep. 2011;13(1):31-42.
- Fung EC, Crook MA. Statin myopathy: a lipid clinic experience on the tolerability of statin rechallenge. Cardiovascular therapeutics. 2012;30(5):e212-218.
- Meador BM, Huey KA. Statin-associated myopathy and its exacerbation with exercise. Muscle Nerve. 2010;42(4):469-479.
- Hammersley D, Signy M. Ezetimibe: an update on its clinical usefulness in specific patient groups. Therapeutic advances in chronic disease. 2017;8(1):4-11.
- Davidson MH. Therapies targeting exogenous cholesterol uptake: new insights and controversies. Curr Atheroscler Rep. 2011;13(1):95-100.
- Toth PP, Patti AM, Giglio RV, et al. Management of Statin Intolerance in 2018: Still More Questions Than Answers. American journal of cardiovascular drugs : drugs, devices, and other interventions. 2018;18(3):157-173.
- Brown JE, Rice-Evans CA. Luteolin-rich artichoke extract protects low density lipoprotein from oxidation in vitro. Free Radic Res. 1998;29(3):247-255.
- Englisch W, Beckers C, Unkauf M, Ruepp M, Zinserling V. Efficacy of Artichoke dry extract in patients with hyperlipoproteinemia. Arzneimittelforschung. 2000;50(3):260-265.
- Bundy R, Walker AF, Middleton RW, Wallis C, Simpson HC. Artichoke leaf extract (Cynara scolymus) reduces plasma cholesterol in otherwise healthy hypercholesterolemic adults: a randomized, double blind placebo controlled trial. Phytomedicine. 2008;15(9):668-675.
- Rondanelli M, Giacosa A, Opizzi A, et al. Beneficial effects of artichoke leaf extract supplementation on increasing HDL-cholesterol in subjects with primary mild hypercholesterolaemia: a double-blind, randomized, placebo-controlled trial. International journal of food sciences and nutrition. 2013;64(1):7-15.
- Nazni P, Vijayakumar TP, Alagianambi P, Amirthaveni M. Hypoglycemic and hypolipidemic effect of Cynara scolymus among selected type 2 diabetic individuals. Pak J Nutr. 2006;5(2):147-151.
- Zema MJ. Gemfibrozil, nicotinic acid and combination therapy in patients with isolated hypoalphalipoproteinemia: a randomized, open-label, crossover study. Journal of the American College of Cardiology. 2000;35(3):640-646.
- McCarty MF. Inhibition of acetyl-CoA carboxylase by cystamine may mediate the hypotriglyceridemic activity of pantethine. Med Hypotheses. 2001;56(3):314-317.
- Bertolini S, Donati C, Elicio N, et al. Lipoprotein changes induced by pantethine in hyperlipoproteinemic patients: adults and children. Int J Clin Pharmacol Ther Toxicol. 1986;24(11):630-637.
- Gaddi A, Descovich GC, Noseda G, et al. Controlled evaluation of pantethine, a natural hypolipidemic compound, in patients with different forms of hyperlipoproteinemia. Atherosclerosis. 1984;50(1):73-83.
- Donati C, Bertieri RS, Barbi G. [Pantethine, diabetes mellitus and atherosclerosis. Clinical study of 1045 patients]. Clin Ter. 1989;128(6):411-422.
- Tonutti L, Taboga C, Noacco C. [Comparison of the efficacy of pantethine, acipimox, and bezafibrate on plasma lipids and index of cardiovascular risk in diabetics with dyslipidemia]. Minerva Med. 1991;82(10):657-663.
- Rumberger JA, Napolitano J, Azumano I, Kamiya T, Evans M. Pantethine, a derivative of vitamin B(5) used as a nutritional supplement, favorably alters low-density lipoprotein cholesterol metabolism in low- to moderate-cardiovascular risk North American subjects: a triple-blinded placebo and diet-controlled investigation. Nutr Res. 2011;31(8):608-615.
- Yadav SS, Singh MK, Singh PK, Kumar V. Traditional knowledge to clinical trials: A review on therapeutic actions of Emblica officinalis. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie. 2017;93:1292-1302.
- Akhtar MS, Ramzan A, Ali A, Ahmad M. Effect of Amla fruit (Emblica officinalis Gaertn.) on blood glucose and lipid profile of normal subjects and type 2 diabetic patients. International journal of food sciences and nutrition. 2011;62(6):609-616.
- Kim HY, Okubo T, Juneja LR, Yokozawa T. The protective role of amla (Emblica officinalis Gaertn.) against fructose-induced metabolic syndrome in a rat model. The British journal of nutrition. 2010;103(4):502-512.
- Gopa B, Bhatt J, Hemavathi KG. A comparative clinical study of hypolipidemic efficacy of Amla (Emblica officinalis) with 3-hydroxy-3-methylglutaryl-coenzyme-A reductase inhibitor simvastatin. Indian journal of pharmacology. 2012;44(2):238-242.
- Usharani P, Fatima N, Muralidhar N. Effects of Phyllanthus emblica extract on endothelial dysfunction and biomarkers of oxidative stress in patients with type 2 diabetes mellitus: a randomized, double-blind, controlled study. Diabetes, metabolic syndrome and obesity : targets and therapy. 2013;6:275-284.
- Khanna S, Das A, Spieldenner J, Rink C, Roy S. Supplementation of a standardized extract from Phyllanthus emblica improves cardiovascular risk factors and platelet aggregation in overweight/class-1 obese adults. Journal of medicinal food. 2015;18(4):415-420.
- Gauhar R, Hwang SL, Jeong SS, et al. Heat-processed Gynostemma pentaphyllum extract improves obesity in ob/ob mice by activating AMP-activated protein kinase. Biotechnology letters. 2012;34(9):1607-1616.
- Nguyen PH, Gauhar R, Hwang SL, et al. New dammarane-type glucosides as potential activators of AMP-activated protein kinase (AMPK) from Gynostemma pentaphyllum. Bioorganic & medicinal chemistry. 2011;19(21):6254-6260.
- Winder WW, Hardie DG. AMP-activated protein kinase, a metabolic master switch: possible roles in type 2 diabetes. The American journal of physiology. 1999;277(1 Pt 1):E1-10.
- Park SH, Huh TL, Kim SY, et al. Antiobesity effect of Gynostemma pentaphyllum extract (actiponin): a randomized, double-blind, placebo-controlled trial. Obesity (Silver Spring). 2014;22(1):63-71.
- Dong Y, Zhang M, Wang S, et al. Activation of AMP-activated protein kinase inhibits oxidized LDL-triggered endoplasmic reticulum stress in vivo. Diabetes. 2010;59(6):1386-1396.
- Umeno A, Horie M, Murotomi K, Nakajima Y, Yoshida Y. Antioxidative and Antidiabetic Effects of Natural Polyphenols and Isoflavones. Molecules (Basel, Switzerland). 2016;21(6).
- Devi KP, Rajavel T, Nabavi SF, et al. Hesperidin: A promising anticancer agent from nature. Industrial crops and products. 2015;76:582-589.
- Li C, Schluesener H. Health-promoting effects of the citrus flavanone hesperidin. Crit Rev Food Sci Nutr. 2017;57(3):613-631.
- Roohbakhsh A, Parhiz H, Soltani F, Rezaee R, Iranshahi M. Neuropharmacological properties and pharmacokinetics of the citrus flavonoids hesperidin and hesperetin--a mini-review. Life Sci. 2014;113(1-2):1-6.
- Jia S, Hu Y, Zhang W, et al. Hypoglycemic and hypolipidemic effects of neohesperidin derived from Citrus aurantium L. in diabetic KK-A(y) mice. Food Funct. 2015;6(3):878-886.
- Rizza S, Muniyappa R, Iantorno M, et al. Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J Clin Endocrinol Metab. 2011;96(5):E782-792.
- Zhang J, Sun C, Yan Y, et al. Purification of naringin and neohesperidin from Huyou (Citrus changshanensis) fruit and their effects on glucose consumption in human HepG2 cells. Food Chem. 2012;135(3):1471-1478.
- Homayouni F, Haidari F, Hedayati M, Zakerkish M, Ahmadi K. Hesperidin Supplementation Alleviates Oxidative DNA Damage and Lipid Peroxidation in Type 2 Diabetes: A Randomized Double-Blind Placebo-Controlled Clinical Trial. Phytother Res. 2017.
- Sun Q, Wedick NM, Tworoger SS, et al. Urinary Excretion of Select Dietary Polyphenol Metabolites Is Associated with a Lower Risk of Type 2 Diabetes in Proximate but Not Remote Follow-Up in a Prospective Investigation in 2 Cohorts of US Women. J Nutr. 2015;145(6):1280-1288.
- Adab Z, Eghtesadi S, Vafa MR, et al. Effect of turmeric on glycemic status, lipid profile, hs-CRP, and total antioxidant capacity in hyperlipidemic type 2 diabetes mellitus patients. Phytother Res. 2019.
- Salden BN, Troost FJ, de Groot E, et al. Randomized clinical trial on the efficacy of hesperidin 2S on validated cardiovascular biomarkers in healthy overweight individuals. Am J Clin Nutr. 2016;104(6):1523-1533.
- Haidari F, Heybar H, Jalali MT, Ahmadi Engali K, Helli B, Shirbeigi E. Hesperidin supplementation modulates inflammatory responses following myocardial infarction. J Am Coll Nutr. 2015;34(3):205-211.
- Amir Shaghaghi M, Abumweis SS, Jones PJ. Cholesterol-lowering efficacy of plant sterols/stanols provided in capsule and tablet formats: results of a systematic review and meta-analysis. Journal of the Academy of Nutrition and Dietetics. 2013;113(11):1494-1503.
- Salo P, Wester I. Low-fat formulations of plant stanols and sterols. The American journal of cardiology. 2005;96(1a):51d-54d.
- Weingartner O, Bohm M, Laufs U. Controversial role of plant sterol esters in the management of hypercholesterolaemia. Eur Heart J. 2009;30(4):404-409.
- Calpe-Berdiel L, Escola-Gil JC, Blanco-Vaca F. New insights into the molecular actions of plant sterols and stanols in cholesterol metabolism. Atherosclerosis. 2009;203(1):18-31.
- Shrestha S, Freake HC, McGrane MM, Volek JS, Fernandez ML. A combination of psyllium and plant sterols alters lipoprotein metabolism in hypercholesterolemic subjects by modifying the intravascular processing of lipoproteins and increasing LDL uptake. J Nutr. 2007;137(5):1165-1170.
- Badimon L, Vilahur G, Padro T. Nutraceuticals and atherosclerosis: human trials. Cardiovascular therapeutics. 2010;28(4):202-215.
- Normen L, Holmes D, Frohlich J. Plant sterols and their role in combined use with statins for lipid lowering. Current opinion in investigational drugs (London, England : 2000). 2005;6(3):307-316.
- Berger A, Jones PJ, Abumweis SS. Plant sterols: factors affecting their efficacy and safety as functional food ingredients. Lipids in health and disease. 2004;3:5.
- Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). Jama. 2001;285(19):2486-2497.
- Lichtenstein AH, Appel LJ, Brands M, et al. Summary of American Heart Association Diet and Lifestyle Recommendations revision 2006. Arteriosclerosis, thrombosis, and vascular biology. 2006;26(10):2186-2191.
- Banach M, Patti AM, Giglio RV, et al. The Role of Nutraceuticals in Statin Intolerant Patients. Journal of the American College of Cardiology. 2018;72(1):96-118.
- Ulbricht C, Basch E, Szapary P, et al. Guggul for hyperlipidemia: a review by the Natural Standard Research Collaboration. Complementary therapies in medicine. 2005;13(4):279-290.
- Singh RB, Niaz MA, Ghosh S. Hypolipidemic and antioxidant effects of Commiphora mukul as an adjunct to dietary therapy in patients with hypercholesterolemia. Cardiovasc Drugs Ther. 1994;8(4):659-664.
- Szapary PO, Wolfe ML, Bloedon LT, et al. Guggulipid for the treatment of hypercholesterolemia: a randomized controlled trial. Jama. 2003;290(6):765-772.
- Nohr LA, Rasmussen LB, Straand J. Resin from the mukul myrrh tree, guggul, can it be used for treating hypercholesterolemia? A randomized, controlled study. Complementary therapies in medicine. 2009;17(1):16-22.
- Patti AM, Al-Rasadi K, Katsiki N, et al. Effect of a Natural Supplement Containing Curcuma Longa, Guggul, and Chlorogenic Acid in Patients With Metabolic Syndrome. Angiology. 2015;66(9):856-861.
- Das S, Datta A, Bagchi C, Chakraborty S, Mitra A, Tripathi SK. A Comparative Study of Lipid-Lowering Effects of Guggul and Atorvastatin Monotherapy in Comparison to Their Combination in High Cholesterol Diet-Induced Hyperlipidemia in Rabbits. Journal of dietary supplements. 2016;13(5):495-504.
- Amato A, Caldara GF, Nuzzo D, et al. NAFLD and Atherosclerosis Are Prevented by a Natural Dietary Supplement Containing Curcumin, Silymarin, Guggul, Chlorogenic Acid and Inulin in Mice Fed a High-Fat Diet. Nutrients. 2017;9(5).
- Anderson JW, Randles KM, Kendall CW, Jenkins DJ. Carbohydrate and fiber recommendations for individuals with diabetes: a quantitative assessment and meta-analysis of the evidence. J Am Coll Nutr. 2004;23(1):5-17.
- Brown L, Rosner B, Willett WW, Sacks FM. Cholesterol-lowering effects of dietary fiber: a meta-analysis. Am J Clin Nutr. 1999;69(1):30-42.
- AbuMweis SS, Jew S, Ames NP. beta-glucan from barley and its lipid-lowering capacity: a meta-analysis of randomized, controlled trials. European journal of clinical nutrition. 2010;64(12):1472-1480.
- Baker WL, Tercius A, Anglade M, White CM, Coleman CI. A meta-analysis evaluating the impact of chitosan on serum lipids in hypercholesterolemic patients. Ann Nutr Metab. 2009;55(4):368-374.
- Zhu X, Sun X, Wang M, et al. Quantitative assessment of the effects of beta-glucan consumption on serum lipid profile and glucose level in hypercholesterolemic subjects. Nutr Metab Cardiovasc Dis. 2015;25(8):714-723.
- Sangle GV, Chowdhury SK, Xie X, Stelmack GL, Halayko AJ, Shen GX. Impairment of mitochondrial respiratory chain activity in aortic endothelial cells induced by glycated low-density lipoprotein. Free radical biology & medicine. 2010;48(6):781-790.
- Roy Chowdhury SK, Sangle GV, Xie X, Stelmack GL, Halayko AJ, Shen GX. Effects of extensively oxidized low-density lipoprotein on mitochondrial function and reactive oxygen species in porcine aortic endothelial cells. Am J Physiol Endocrinol Metab. 2010;298(1):E89-98.
- Littarru GP, Langsjoen P. Coenzyme Q10 and statins: biochemical and clinical implications. Mitochondrion. 2007;7 Suppl:S168-174.
- Tóth Š, Šajty M, Pekárová T, et al. Addition of omega-3 fatty acid and coenzyme Q10 to statin therapy in patients with combined dyslipidemia. Journal of basic and clinical physiology and pharmacology. 2017;28(4):327-336.
- Jorat MV, Tabrizi R, Mirhosseini N, et al. The effects of coenzyme Q10 supplementation on lipid profiles among patients with coronary artery disease: a systematic review and meta-analysis of randomized controlled trials. Lipids in health and disease. 2018;17(1):230.
- Sahebkar A, Simental-Mendía LE, Stefanutti C, Pirro M. Supplementation with coenzyme Q10 reduces plasma lipoprotein(a) concentrations but not other lipid indices: A systematic review and meta-analysis. Pharmacol Res. 2016;105:198-209.
- Karppi J, Nurmi T, Kurl S, Rissanen TH, Nyyssonen K. Lycopene, lutein and beta-carotene as determinants of LDL conjugated dienes in serum. Atherosclerosis. 2010;209(2):565-572.
- Ried K, Fakler P. Protective effect of lycopene on serum cholesterol and blood pressure: Meta-analyses of intervention trials. Maturitas. 2011;68(4):299-310.
- Fuhrman B, Elis A, Aviram M. Hypocholesterolemic effect of lycopene and beta-carotene is related to suppression of cholesterol synthesis and augmentation of LDL receptor activity in macrophages. Biochemical and biophysical research communications. 1997;233(3):658-662.
- Mao M, Lei H, Liu Q, et al. Lycopene inhibits neointimal hyperplasia through regulating lipid metabolism and suppressing oxidative stress. Molecular medicine reports. 2014;10(1):262-268.
- Aidoud A, Ammouche A, Garrido M, Rodriguez AB. Effect of lycopene-enriched olive and argan oils upon lipid serum parameters in Wistar rats. Journal of the science of food and agriculture. 2014;94(14):2943-2950.
- Cheng HM, Koutsidis G, Lodge JK, Ashor A, Siervo M, Lara J. Tomato and lycopene supplementation and cardiovascular risk factors: A systematic review and meta-analysis. Atherosclerosis. 2017;257:100-108.
- Badiou S, Cristol JP, Morena M, et al. Vitamin E supplementation increases LDL resistance to ex vivo oxidation in hemodialysis patients. International journal for vitamin and nutrition research Internationale Zeitschrift fur Vitamin- und Ernahrungsforschung Journal international de vitaminologie et de nutrition. 2003;73(4):290-296.
- Tesoriere L, D'Arpa D, Butera D, et al. Oral supplements of vitamin E improve measures of oxidative stress in plasma and reduce oxidative damage to LDL and erythrocytes in beta-thalassemia intermedia patients. Free Radic Res. 2001;34(5):529-540.
- Li D, Saldeen T, Romeo F, Mehta JL. Relative Effects of alpha- and gamma-Tocopherol on Low-Density Lipoprotein Oxidation and Superoxide Dismutase and Nitric Oxide Synthase Activity and Protein Expression in Rats. J Cardiovasc Pharmacol Ther. 1999;4(4):219-226.
- McCarty MF. Gamma-tocopherol may promote effective no synthase function by protecting tetrahydrobiopterin from peroxynitrite. Med Hypotheses. 2007;69(6):1367-1370.
- Saito F, Iwamoto S, Yamauchi R. Reaction products of gamma-tocopherol with (E)-4-oxo-2-nonenal in acidic acetonitrile. Bioscience, biotechnology, and biochemistry. 2010;74(1):168-174.
- Singh I, Turner AH, Sinclair AJ, Li D, Hawley JA. Effects of gamma-tocopherol supplementation on thrombotic risk factors. Asia Pac J Clin Nutr. 2007;16(3):422-428.
- Onakpoya I, Spencer E, Heneghan C, Thompson M. The effect of green tea on blood pressure and lipid profile: a systematic review and meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis. 2014;24(8):823-836.
- Aviram M, Dornfeld L, Kaplan M, et al. Pomegranate juice flavonoids inhibit low-density lipoprotein oxidation and cardiovascular diseases: studies in atherosclerotic mice and in humans. Drugs under experimental and clinical research. 2002;28(2-3):49-62.
- Sezer ED, Akcay YD, Ilanbey B, Yildirim HK, Sozmen EY. Pomegranate wine has greater protection capacity than red wine on low-density lipoprotein oxidation. Journal of medicinal food. 2007;10(2):371-374.
- Esmaillzadeh A, Tahbaz F, Gaieni I, Alavi-Majd H, Azadbakht L. Cholesterol-lowering effect of concentrated pomegranate juice consumption in type II diabetic patients with hyperlipidemia. International journal for vitamin and nutrition research Internationale Zeitschrift fur Vitamin- und Ernahrungsforschung Journal international de vitaminologie et de nutrition. 2006;76(3):147-151.
- Fuhrman B, Volkova N, Aviram M. Pomegranate juice inhibits oxidized LDL uptake and cholesterol biosynthesis in macrophages. J Nutr Biochem. 2005;16(9):570-576.
- Aviram M, Rosenblat M, Gaitini D, et al. Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clin Nutr. 2004;23(3):423-433.
- Kojadinovic MI, Arsic AC, Debeljak-Martacic JD, et al. Consumption of pomegranate juice decreases blood lipid peroxidation and levels of arachidonic acid in women with metabolic syndrome. Journal of the science of food and agriculture. 2017;97(6):1798-1804.
- Shema-Didi L, Kristal B, Sela S, Geron R, Ore L. Does Pomegranate intake attenuate cardiovascular risk factors in hemodialysis patients? Nutr J. 2014;13:18.
- Hosseini B, Saedisomeolia A, Wood LG, Yaseri M, Tavasoli S. Effects of pomegranate extract supplementation on inflammation in overweight and obese individuals: A randomized controlled clinical trial. Complementary therapies in clinical practice. 2016;22:44-50.
- Ramirez-Boscá A, Soler A, Gutierrez MAC, Alvarez JL, Almagro EQ. Antioxidant curcuma extracts decrease the blood lipid peroxide levels of human subjects. Age. 1995;18(4):167-169.
- Ramirez Bosca A, Carrion Gutierrez MA, Soler A, et al. Effects of the antioxidant turmeric on lipoprotein peroxides: Implications for the prevention of atherosclerosis. Age (Omaha). 1997;20(3):165-168.
- Ramirez Bosca A, Soler A, Carrion-Gutierrez MA, et al. An hydroalcoholic extract of Curcuma longa lowers the abnormally high values of human-plasma fibrinogen. Mech Ageing Dev. 2000;114(3):207-210.
- Wongcharoen W, Phrommintikul A. The protective role of curcumin in cardiovascular diseases. Int J Cardiol. 2009;133(2):145-151.
- Dou X, Fan C, Wo L, Yan J, Qian Y, Wo X. Curcumin up-regulates LDL receptor expression via the sterol regulatory element pathway in HepG2 cells. Planta Med. 2008;74(11):1374-1379.
- Peschel D, Koerting R, Nass N. Curcumin induces changes in expression of genes involved in cholesterol homeostasis. J Nutr Biochem. 2007;18(2):113-119.
- Soni KB, Kuttan R. Effect of oral curcumin administration on serum peroxides and cholesterol levels in human volunteers. Indian journal of physiology and pharmacology. 1992;36(4):273-275.
- Alwi I, Santoso T, Suyono S, et al. The effect of curcumin on lipid level in patients with acute coronary syndrome. Acta medica Indonesiana. 2008;40(4):201-210.
- Baum L, Cheung SK, Mok VC, et al. Curcumin effects on blood lipid profile in a 6-month human study. Pharmacological research : the official journal of the Italian Pharmacological Society. 2007;56(6):509-514.
- Qin S, Huang L, Gong J, et al. Efficacy and safety of turmeric and curcumin in lowering blood lipid levels in patients with cardiovascular risk factors: a meta-analysis of randomized controlled trials. Nutr J. 2017;16(1):68.
- Yang YS, Su YF, Yang HW, Lee YH, Chou JI, Ueng KC. Lipid-lowering effects of curcumin in patients with metabolic syndrome: a randomized, double-blind, placebo-controlled trial. Phytother Res. 2014;28(12):1770-1777.
- Pizzini A, Lunger L, Demetz E, et al. The Role of Omega-3 Fatty Acids in Reverse Cholesterol Transport: A Review. Nutrients. 2017;9(10).
- Simopoulos AP. Evolutionary Aspects of the Dietary Omega-6/Omega-3 Fatty Acid Ratio: Medical Implications. In: Alvergne A, Jenkinson C, Faurie C, eds. Evolutionary Thinking in Medicine: From Research to Policy and Practice. Cham: Springer International Publishing; 2016:119-134.
- Jacobson TA, Glickstein SB, Rowe JD, Soni PN. Effects of eicosapentaenoic acid and docosahexaenoic acid on low-density lipoprotein cholesterol and other lipids: a review. Journal of clinical lipidology. 2012;6(1):5-18.
- Allaire J, Vors C, Tremblay AJ, et al. High-Dose DHA Has More Profound Effects on LDL-Related Features Than High-Dose EPA: The ComparED Study. J Clin Endocrinol Metab. 2018;103(8):2909-2917.
- Bhatt DL, Steg PG, Miller M, et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England journal of medicine. 2019;380(1):11-22.
- Wu Y, Zhang Q, Ren Y, Ruan Z. Effect of probiotic Lactobacillus on lipid profile: A systematic review and meta-analysis of randomized, controlled trials. PLoS One. 2017;12(6):e0178868.
- DiRienzo DB. Effect of probiotics on biomarkers of cardiovascular disease: implications for heart-healthy diets. Nutrition reviews. 2014;72(1):18-29.
- Jones ML, Martoni CJ, Parent M, Prakash S. Cholesterol-lowering efficacy of a microencapsulated bile salt hydrolase-active Lactobacillus reuteri NCIMB 30242 yoghurt formulation in hypercholesterolaemic adults. The British journal of nutrition. 2012;107(10):1505-1513.
- Semekovich CF, Goldman L. Goldman-Cecil Medicine. Twenty-fifth edition. Chapter 206: Disorders of Lipid Metabolism. 1389-1397..e.3. Copyright 2016 by Saunders, an imprint of Elsevier Inc. www.clinicalkey.com. Accessed 7/7/2016.
- Lu M, Lu Q, Zhang Y, Tian G. ApoB/apoA1 is an effective predictor of coronary heart disease risk in overweight and obesity(). Journal of biomedical research. 2011;25(4):266-273.
- Walldius G, Frank S, Kostner G. Biochemistry, Genetics, and Molecular Biology. Chapter 5: The apoB/apoA-I Ratio is a Strong Predictor of Cardiovascular Risk. http://www.intechopen.com/books/lipoproteins-role-in-health-and-diseases/the-apob-apoa-i-ratio-is-a-strong-predictor-of-cardiovascular-risk. 10/3/2012. Accessed 7/7/2016.
- Jones ML, Martoni CJ, Prakash S. Cholesterol lowering and inhibition of sterol absorption by Lactobacillus reuteri NCIMB 30242: a randomized controlled trial. European journal of clinical nutrition. 2012;66(11):1234-1241.
- Jones ML, Martoni CJ, Ganopolsky JG, Sulemankhil I, Ghali P, Prakash S. Improvement of gastrointestinal health status in subjects consuming Lactobacillus reuteri NCIMB 30242 capsules: a post-hoc analysis of a randomized controlled trial. Expert Opin Biol Ther. 2013;13(12):1643-1651.
- Jones ML, Martoni CJ, Prakash S. Oral supplementation with probiotic L. reuteri NCIMB 30242 increases mean circulating 25-hydroxyvitamin D: a post hoc analysis of a randomized controlled trial. J Clin Endocrinol Metab. 2013;98(7):2944-2951.
- Staels B, Fonseca VA. Bile Acids and Metabolic Regulation: Mechanisms and clinical responses to bile acid sequestration. Diabetes Care. 2009;32(Suppl 2):S237-245.
- Kumar M, Nagpal R, Kumar R, et al. Cholesterol-lowering probiotics as potential biotherapeutics for metabolic diseases. Experimental diabetes research. 2012;2012:902917.
- Cho YA, Kim J. Effect of Probiotics on Blood Lipid Concentrations: A Meta-Analysis of Randomized Controlled Trials. Medicine. 2015;94(43):e1714.
- Sun J, Buys N. Effects of probiotics consumption on lowering lipids and CVD risk factors: a systematic review and meta-analysis of randomized controlled trials. Ann Med. 2015;47(6):430-440.
- Zhao H, Song A, Zhang Y, Shu L, Song G, Ma H. Effect of Resveratrol on Blood Lipid Levels in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Obesity (Silver Spring). 2019;27(1):94-102.
- He J, Zhang F, Han Y. Effect of probiotics on lipid profiles and blood pressure in patients with type 2 diabetes: A meta-analysis of RCTs. Medicine. 2017;96(51):e9166.
- Brighenti F, Casiraghi MC, Canzi E, Ferrari A. Effect of consumption of a ready-to-eat breakfast cereal containing inulin on the intestinal milieu and blood lipids in healthy male volunteers. European journal of clinical nutrition. 1999;53(9):726-733.
- Li S, Guerin-Deremaux L, Pochat M, Wils D, Reifer C, Miller LE. NUTRIOSE dietary fiber supplementation improves insulin resistance and determinants of metabolic syndrome in overweight men: a double-blind, randomized, placebo-controlled study. Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme. 2010;35(6):773-782.
- Cunningham E. Is red yeast rice safe and effective for lowering serum cholesterol? J Am Diet Assoc. 2011;111(2):324.
- Liu J, Zhang J, Shi Y, Grimsgaard S, Alraek T, Fonnebo V. Chinese red yeast rice (Monascus purpureus) for primary hyperlipidemia: a meta-analysis of randomized controlled trials. Chinese medicine. 2006;1:4.
- Moriarty PM, Roth EM, Karns A, et al. Effects of Xuezhikang in patients with dyslipidemia: a multicenter, randomized, placebo-controlled study. Journal of clinical lipidology. 2014;8(6):568-575.
- Shang Q, Liu Z, Chen K, Xu H, Liu J. A systematic review of xuezhikang, an extract from red yeast rice, for coronary heart disease complicated by dyslipidemia. Evidence-based complementary and alternative medicine : eCAM. 2012;2012:636547.
- Lu Z, Kou W, Du B, et al. Effect of Xuezhikang, an extract from red yeast Chinese rice, on coronary events in a Chinese population with previous myocardial infarction. The American journal of cardiology. 2008;101(12):1689-1693.
- Becker DJ, Gordon RY, Halbert SC, French B, Morris PB, Rader DJ. Red yeast rice for dyslipidemia in statin-intolerant patients: a randomized trial. Ann Intern Med. 2009;150(12):830-839, w147-839.
- Halbert SC, French B, Gordon RY, et al. Tolerability of red yeast rice (2,400 mg twice daily) versus pravastatin (20 mg twice daily) in patients with previous statin intolerance. The American journal of cardiology. 2010;105(2):198-204.
- Kasliwal RR, Bansal M, Gupta R, et al. ESSENS dyslipidemia: A placebo-controlled, randomized study of a nutritional supplement containing red yeast rice in subjects with newly diagnosed dyslipidemia. Nutrition (Burbank, Los Angeles County, Calif). 2016;32(7-8):767-776.
- Gordon RY, Cooperman T, Obermeyer W, Becker DJ. Marked variability of monacolin levels in commercial red yeast rice products: buyer beware! Arch Intern Med. 2010;170(19):1722-1727.
- Toth PP, Patti AM, Nikolic D, et al. Bergamot Reduces Plasma Lipids, Atherogenic Small Dense LDL, and Subclinical Atherosclerosis in Subjects with Moderate Hypercholesterolemia: A 6 Months Prospective Study. Frontiers in pharmacology. 2015;6:299.
- Cai Y, Xing G, Shen T, Zhang S, Rao J, Shi R. Effects of 12-week supplementation of Citrus bergamia extracts-based formulation CitriCholess on cholesterol and body weight in older adults with dyslipidemia: a randomized, double-blind, placebo-controlled trial. Lipids in health and disease. 2017;16(1):251.
- Gliozzi M, Walker R, Muscoli S, et al. Bergamot polyphenolic fraction enhances rosuvastatin-induced effect on LDL-cholesterol, LOX-1 expression and protein kinase B phosphorylation in patients with hyperlipidemia. Int J Cardiol. 2013;170(2):140-145.
- D'Andrea E, Hey SP, Ramirez CL, Kesselheim AS. Assessment of the Role of Niacin in Managing Cardiovascular Disease Outcomes: A Systematic Review and Meta-analysis. JAMA Netw Open. 2019;2(4):e192224.
- Kamanna VS, Kashyap ML. Mechanism of Action of Niacin. The American journal of cardiology. 2008;101(8, Supplement):S20-S26.
- Garg A, Sharma A, Krishnamoorthy P, et al. Role of Niacin in Current Clinical Practice: A Systematic Review. Am J Med. 2017;130(2):173-187.
- Neelakantan N, Seah JYH, van Dam RM. The Effect of Coconut Oil Consumption on Cardiovascular Risk Factors: A Systematic Review and Meta-Analysis of Clinical Trials. Circulation. 2020.
- Hoppstädter J, Valbuena Perez JV, Linnenberger R, et al. The glucocorticoid-induced leucine zipper mediates statin-induced muscle damage. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 2020:10.1096/fj.201902557RRR.
- tctMD. FDA Approves New Lipid-Lowering Drug, Bempedoic Acid. https://www.tctmd.com/news/fda-approves-new-lipid-lowering-drug-bempedoic-acid. Published 2020. Accessed February 26, 2020.
- Goldberg AC, Leiter LA, Stroes ESG, et al. Effect of Bempedoic Acid vs Placebo Added to Maximally Tolerated Statins on Low-Density Lipoprotein Cholesterol in Patients at High Risk for Cardiovascular Disease: The CLEAR Wisdom Randomized Clinical Trial. JAMA. 2019;322(18):1780-1788.
- Lalwani ND, Hanselman JC, MacDougall DE, Sterling LR, Cramer CT. Complementary low-density lipoprotein-cholesterol lowering and pharmacokinetics of adding bempedoic acid (ETC-1002) to high-dose atorvastatin background therapy in hypercholesterolemic patients: A randomized placebo-controlled trial. Journal of Clinical Lipidology. 2019;13(4):568-579.
- Ballantyne CM, Davidson MH, Macdougall DE, et al. Efficacy and safety of a novel dual modulator of adenosine triphosphate-citrate lyase and adenosine monophosphate-activated protein kinase in patients with hypercholesterolemia: results of a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial. J Am Coll Cardiol. 2013;62(13):1154-1162.
- Ballantyne CM, Laufs U, Ray KK, et al. Bempedoic acid plus ezetimibe fixed-dose combination in patients with hypercholesterolemia and high CVD risk treated with maximally tolerated statin therapy. European journal of preventive cardiology. 2019:2047487319864671-2047487319864671.
- Khan SU, Michos ED. Bempedoic acid and ezetimibe – better together. European Journal of Preventive Cardiology. 2019:2047487319864672.
- Ray KK, Bays HE, Catapano AL, et al. Safety and Efficacy of Bempedoic Acid to Reduce LDL Cholesterol. New England Journal of Medicine. 2019;380(11):1022-1032.
- Bilen O, Ballantyne CM. Bempedoic Acid (ETC-1002): an Investigational Inhibitor of ATP Citrate Lyase. Current atherosclerosis reports. 2016;18(10):61-61.
- Burke AC, Telford DE, Huff MW. Bempedoic acid: effects on lipoprotein metabolism and atherosclerosis. Curr Opin Lipidol. 2019;30(1):1-9.
- Reduction of Revascularizations in Patients with Hypertriglyceridemia With Icosapent Ethyl: Insights From REDUCE-IT REVASC [press release]. Society for Cardiovascular Angiography and Interventions.2020.
- Sniderman AD, Navar AM, Thanassoulis G. Apolipoprotein B vs Low-Density Lipoprotein Cholesterol and Non-High-Density Lipoprotein Cholesterol as the Primary Measure of Apolipoprotein B Lipoprotein-Related Risk: The Debate Is Over. JAMA Cardiol. Nov 13 2021;doi:10.1001/jamacardio.2021.5080
- Johannesen CDL, Mortensen MB, Langsted A, Nordestgaard BG. Apolipoprotein B and Non-HDL Cholesterol Better Reflect Residual Risk Than LDL Cholesterol in Statin-Treated Patients. Journal of the American College of Cardiology. 2021/03/23/ 2021;77(11):1439-1450. doi:https://doi.org/10.1016/j.jacc.2021.01.027
- StatPearls. Biochemistry, Apolipoprotein B. Accessed 12/29/2021, https://www.statpearls.com/articlelibrary/viewarticle/17778
- Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). European Heart Journal. 2019;41(1):111-188. doi:10.1093/eurheartj/ehz455
- Marston NA, Giugliano RP, Melloni GEM, et al. Association of Apolipoprotein B–Containing Lipoproteins and Risk of Myocardial Infarction in Individuals With and Without Atherosclerosis: Distinguishing Between Particle Concentration, Type, and Content. JAMA Cardiology. 2021;doi:10.1001/jamacardio.2021.5083
- Shapiro MD, Fazio S. Apolipoprotein B-containing lipoproteins and atherosclerotic cardiovascular disease. F1000Research. 2017;6:134-134. doi:10.12688/f1000research.9845.1
- Sniderman AD, Thanassoulis G, Glavinovic T, et al. Apolipoprotein B Particles and Cardiovascular Disease: A Narrative Review. JAMA Cardiology. 2019;4(12):1287-1295. doi:10.1001/jamacardio.2019.3780
- Pimpley V, Patil S, Srinivasan K, Desai N, Murthy PS. The chemistry of chlorogenic acid from green coffee and its role in attenuation of obesity and diabetes. Prep Biochem Biotechnol. 2020;50(10):969-978. doi:10.1080/10826068.2020.1786699. https://pubmed.ncbi.nlm.nih.gov/32633686/
- Ye X, Li J, Gao Z, Wang D, Wang H, Wu J. Chlorogenic Acid Inhibits Lipid Deposition by Regulating the Enterohepatic FXR-FGF15 Pathway. Biomed Res Int. 2022;2022:4919153. doi:10.1155/2022/4919153. https://pubmed.ncbi.nlm.nih.gov/35257010/
- Wu C, Luan H, Zhang X, et al. Chlorogenic acid protects against atherosclerosis in ApoE-/- mice and promotes cholesterol efflux from RAW264.7 macrophages. PLoS One. 2014;9(9):e95452. doi:10.1371/journal.pone.0095452. https://pubmed.ncbi.nlm.nih.gov/25187964/
- Ding F, Ma B, Nazary-Vannani A, et al. The effects of green coffee bean extract supplementation on lipid profile in humans: A systematic review and meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis. Jan 3 2020;30(1):1-10. doi:10.1016/j.numecd.2019.10.002. https://pubmed.ncbi.nlm.nih.gov/31748178/
- Asbaghi O, Sadeghian M, Nasiri M, et al. The effects of green coffee extract supplementation on glycemic indices and lipid profile in adults: a systematic review and dose-response meta-analysis of clinical trials. Nutr J. Jul 14 2020;19(1):71. doi:10.1186/s12937-020-00587-z. https://pubmed.ncbi.nlm.nih.gov/32665012/
- Morvaridi M, Rayyani E, Jaafari M, Khiabani A, Rahimlou M. The effect of green coffee extract supplementation on cardio metabolic risk factors: a systematic review and meta-analysis of randomized controlled trials. J Diabetes Metab Disord. Jun 2020;19(1):645-660. doi:10.1007/s40200-020-00536-x. https://pubmed.ncbi.nlm.nih.gov/32550217/
- Pourmasoumi M, Hadi A, Marx W, Najafgholizadeh A, Kaur S, Sahebkar A. The Effect of Green Coffee Bean Extract on Cardiovascular Risk Factors: A Systematic Review and Meta-analysis. Adv Exp Med Biol. 2021;1328:323-345. doi:10.1007/978-3-030-73234-9_21. https://pubmed.ncbi.nlm.nih.gov/34981487/
- Bosso H, Barbalho SM, de Alvares Goulart R, Otoboni A. Green coffee: economic relevance and a systematic review of the effects on human health. Crit Rev Food Sci Nutr. 2023;63(3):394-410. doi:10.1080/10408398.2021.1948817. https://pubmed.ncbi.nlm.nih.gov/34236263/
- Roshan H, Nikpayam O, Sedaghat M, Sohrab G. Effects of green coffee extract supplementation on anthropometric indices, glycaemic control, blood pressure, lipid profile, insulin resistance and appetite in patients with the metabolic syndrome: a randomised clinical trial. Br J Nutr. Feb 2018;119(3):250-258. doi:10.1017/s0007114517003439. https://pubmed.ncbi.nlm.nih.gov/29307310/
- Saitou K, Ochiai R, Kozuma K, et al. Effect of Chlorogenic Acids on Cognitive Function: A Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients. Sep 20 2018;10(10)doi:10.3390/nu10101337. https://pubmed.ncbi.nlm.nih.gov/30241302/
- Murali S, Smith ER, Tiong MK, Tan SJ, Toussaint ND. Interventions to Attenuate Cardiovascular Calcification Progression: A Systematic Review of Randomized Clinical Trials. J Am Heart Assoc. Dec 5 2023;12(23):e031676. doi:10.1161/jaha.123.031676. https://pubmed.ncbi.nlm.nih.gov/38014685/
- Imaizumi VM, Laurindo LF, Manzan B, et al. Garlic: A systematic review of the effects on cardiovascular diseases. Crit Rev Food Sci Nutr. 2023;63(24):6797-6819. doi:10.1080/10408398.2022.2043821. https://pubmed.ncbi.nlm.nih.gov/35193446/
- Fu Z, Lv J, Gao X, et al. Effects of garlic supplementation on components of metabolic syndrome: a systematic review, meta-analysis, and meta-regression of randomized controlled trials. BMC Complement Med Ther. Jul 22 2023;23(1):260. doi:10.1186/s12906-023-04038-0. https://pubmed.ncbi.nlm.nih.gov/37481521/
- Ried K. Garlic Lowers Blood Pressure in Hypertensive Individuals, Regulates Serum Cholesterol, and Stimulates Immunity: An Updated Meta-analysis and Review. J Nutr. Feb 2016;146(2):389s-396s. doi:10.3945/jn.114.202192. https://pubmed.ncbi.nlm.nih.gov/26764326/
- Baik JS, Min JH, Ju SM, et al. Effects of Fermented Garlic Extract Containing Nitric Oxide Metabolites on Blood Flow in Healthy Participants: A Randomized Controlled Trial. Nutrients. Dec 8 2022;14(24)doi:10.3390/nu14245238. https://pubmed.ncbi.nlm.nih.gov/36558397/
- Villaño D, Marhuenda J, Arcusa R, et al. Effect of Black Garlic Consumption on Endothelial Function and Lipid Profile: A Before-and-After Study in Hypercholesterolemic and Non-Hypercholesterolemic Subjects. Nutrients. Jul 14 2023;15(14)doi:10.3390/nu15143138. https://pubmed.ncbi.nlm.nih.gov/37513556/
- Rais N, Ved A, Ahmad R, et al. S-Allyl-L-Cysteine — A garlic Bioactive: Physicochemical Nature, Mechanism, Pharmacokinetics, and health promoting activities. Journal of Functional Foods. 2023/08/01/ 2023;107:105657. doi:https://doi.org/10.1016/j.jff.2023.105657. https://www.sciencedirect.com/science/article/pii/S1756464623002578
- Nirvanashetty S, Panda S. High Potency Aged Garlic Extract reduces Cardiovascular Disease Risk Factors in Healthy Participants: A Randomized, Double-Blind, Placebo Controlled Study. Open Acc J Comp & Alt Med. 10/20/2023 2023;5(1):585-593. doi:10.32474/OAJCAM.2023.05.000203. https://lupinepublishers.com/complementary-alternative-medicine-journal/pdf/OAJCAM.MS.ID.000202.pdf
- Ormiston CK, Pazargadi A, Rosander A, Ceballos G, Villarreal F, Taub PR. Enhancement of Statin Effects on Lipid Lowering and Reduction of Cardiovascular Risk Score by (−)-Epicatechin in Proof-of-Concept Pilot Study. Clinical and Translational Science. 2025;18(5):e70236. doi:https://doi.org/10.1111/cts.70236. https://pubmed.ncbi.nlm.nih.gov/40387782/
- Rana A, Samtiya M, Dhewa T, Mishra V, Aluko RE. Health benefits of polyphenols: A concise review. J Food Biochem. Oct 2022;46(10):e14264. doi:10.1111/jfbc.14264. https://pubmed.ncbi.nlm.nih.gov/35694805/
- Espín JC, Jarrín-Orozco MP, Osuna-Galisteo L, Ávila-Gálvez MÁ, Romo-Vaquero M, Selma MV. Perspective on the Coevolutionary Role of Host and Gut Microbiota in Polyphenol Health Effects: Metabotypes and Precision Health. Molecular Nutrition & Food Research. 2024;68(22):2400526. doi:https://doi.org/10.1002/mnfr.202400526. https://pubmed.ncbi.nlm.nih.gov/39538982/
- Cory H, Passarelli S, Szeto J, Tamez M, Mattei J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front Nutr. 2018;5:87. doi:10.3389/fnut.2018.00087. https://pubmed.ncbi.nlm.nih.gov/30298133/
- Khan N, Mukhtar H. Tea Polyphenols in Promotion of Human Health. Nutrients. 2018;11(1):39. https://pubmed.ncbi.nlm.nih.gov/30585192/
- Xu R, Yang K, Li S, Dai M, Chen G. Effect of green tea consumption on blood lipids: a systematic review and meta-analysis of randomized controlled trials. Nutr J. May 20 2020;19(1):48. doi:10.1186/s12937-020-00557-5. https://pubmed.ncbi.nlm.nih.gov/32434539/
- Momose Y, Maeda-Yamamoto M, Nabetani H. Systematic review of green tea epigallocatechin gallate in reducing low-density lipoprotein cholesterol levels of humans. Int J Food Sci Nutr. Sep 2016;67(6):606-13. doi:10.1080/09637486.2016.1196655. https://pubmed.ncbi.nlm.nih.gov/27324590/
