Life Extension Magazine®
|
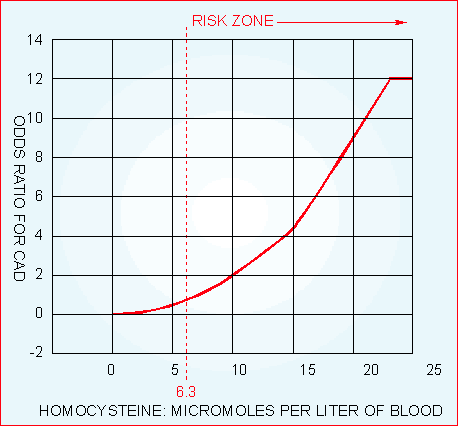
Too often, aging people fall victim to a disease that could have been prevented had the proper blood tests been done on a regular basis. Since 1983, the Life Extension Foundation has advocated annual blood testing to maintain optimal health. It was not until 1996, however, that members could order their own blood tests directly. Before 1996, members had to plead with their doctors to prescribe blood tests. In many cases, these requests were rejected because physicians did not understand the importance of screening for high-sensitivity C-reactive protein, homocysteine, DHEA, or even PSA (prostate-specific antigen). The cost of blood tests through physicians’ offices was also prohibitively expensive. The reason most people consider blood testing is to ascertain their cardiovascular risk factors. The scientific literature clearly establishes that elevated insulin, glucose, homocysteine, fibrinogen, C-reactive protein, triglycerides, and LDL dramatically increase the risk of heart attack, stroke, and other arterial diseases. What most people fail to realize is that significant changes can occur over the course of one year, meaning that previous tests may not accurately reflect current cardiovascular risk status. Now that more Foundation members than ever are ordering their own blood tests, Life Extension has been able to negotiate significantly lower prices. During the annual Blood Test Super Sale, members can help guard their precious health by gaining access to the most important blood tests at super discounted prices. The printed results of these blood tests are mailed directly to members, who can then take them to their doctors if abnormalities are revealed. If members want to discuss the blood results by telephone, they have toll-free phone access to licensed Life Extension medical doctors at no charge. In addition to identifying proven cardiovascular disease risk factors, blood tests can detect numerous other medical abnormalities that are often easy to correct if caught in time. For instance, a growing number of Americans are taking prescription drugs to treat chronic medical conditions. Many of these drugs have toxic side effects that result in the deaths of more than 100,000 Americans each year. In fact, according to the American Medical Association (AMA), adverse reactions to prescription drugs are between the fourth and sixth leading causes of death in the US. The AMA emphasizes that these deaths are occurring, even though doctors who are supposed to closely monitor their patients are prescribing the drugs. The problem is that cost-conscious HMOs and hurried physicians are not ordering blood tests that would detect drug-induced liver and kidney impairment in time to prevent these side effects from occurring. If you are taking certain prescription medications, regular blood testing is mandatory according to the drug labeling, yet doctors routinely fail to prescribe the recommended blood tests and their patients succumb to a drug-induced hospitalization, disability, or death. It is not just prescription drugs that can cause liver or kidney damage. Many factors (alcohol, over-the-counter drugs, excess niacin, hepatitis C) can make a person susceptible to liver or kidney damage. For most people, these conditions smolder for years until a life-threatening medical crisis occurs. Some people are especially vulnerable to liver and kidney damage; yet a simple blood chemistry test can detect an underlying problem in time to take corrective actions. An example of a preventable disorder that can be detected by a yearly blood test is calcium overload. This condition is caused when too much calcium is removed from the bone and deposited into the vascular system. Unless discovered by a blood test, people do not find out about calcium imbalance until after they have a crippling bone fracture, a painful kidney stone (renal calculi), or heart valve failure (due to excess valvular calcification). These diseases often manifest years after the calcium imbalance first begins, yet an inexpensive blood chemistry test could detect this problem early and enable the person to take relatively simple steps to correct the calcium imbalance before it causes irreversible damage. Excess amounts of serum iron generate free radicals that increase the risk of cancer and atherosclerosis, and probably neurological disorders such as Alzheimer’s disease and Parkinson’s disease. If a blood test shows high iron levels, there are many ways to bring it down. A blood chemistry test can detect high iron levels before an iron-induced disease manifests. The Importance of Maintaining Youthful Blood Readings When physicians review a patient’s blood test results, their only concern is when a particular result is outside the normal laboratory “reference range.” The problem is that standard reference ranges usually represent “average” populations, rather than the optimal level required to maintain good health. It now appears that most standard reference ranges are too broad to adequately detect health problems or prescribe appropriate therapy on an individual basis. In the 1960s, for instance, the upper reference range for cholesterol extended up to 300 mg/dL. This number was based on a statistical calculation indicating that it was “normal” to have total cholesterol levels as high as 300 mg/dL. At that time, it was also “normal” for men to have fatal heart attacks at a relatively young age. As greater knowledge accumulated about the risk of heart attack and high cholesterol, the upper limit reference range gradually dropped to the point where it is now 200 mg/dL.1 The same situation occurred with homocysteine reference ranges. Until recently, it was considered normal to have a homocysteine blood reading as high as 15 mm/L.2 Most reference ranges now provide a chart showing that homocysteine levels above 7 indicate an increased risk of heart attack and stroke.3 |
| Antiquated Hormone “Reference Ranges” Aging men, for instance, often suffer from excess production of insulin and estrogen, with simultaneous deficiencies of free testosterone and dehydroepiandrosterone (DHEA). If a physician were to test blood levels of all four of these hormones, the standard “reference ranges” are so wide that most men would fall into the so-called “normal” category. Standard reference ranges indicate that dangerously high insulin and estrogen levels are “normal” in elderly men (but so are heart attack, stroke, cancer, benign prostate enlargement, weight gain, type II diabetes, kidney impairment, and a host of other diseases that are associated with excess insulin and estrogen). The standard reference ranges for free testosterone and DHEA show that very low levels are perfectly “normal” for aging men. It is no coincidence that these same aging men (with low levels of testosterone/DHEA) have high rates of depression, memory loss, atherosclerosis, senility, impotency, high cholesterol, abdominal obesity, fatigue, and a host of other diseases related to low blood levels of testosterone and DHEA.4-15 Another example of flawed reference ranges can be seen in a blood test used to assess thyroid status known as the thyroid stimulating hormone (TSH) test. The TSH reference range used by many laboratories is 0.2-5.5 mU/L. A greater TSH level is indicative of a thyroid hormone deficiency. That is because the pituitary gland is over-signaling TSH due to low levels of thyroid hormone in the blood. Any reading over 5.5 alerts a doctor to a thyroid gland problem and that thyroid hormone therapy may be warranted. The trouble is that the TSH reference range is so broad that most doctors will interpret a TSH reading as low as 0.2 to be as normal as a 5.5 reading. The difference between 0.2 and 5.5, however, is 27-fold, a parameter far too great to indicate optimal or even normal thyroid function. A review of published findings about TSH levels reveals that readings over 2.0 may be indicative of adverse health problems relating to insufficient thyroid hormone output. One study showed that individuals with TSH values over 2.0 have an increased risk of developing overt hypothyroid disease over the next 20 years.16 Other studies show that TSH values over 1.9 indicate abnormal pathologies of the thyroid, specifically autoimmune attacks on the thyroid gland itself that can result in significant impairment.17 A more startling study showed that TSH values over 4.0 increase the prevalence of heart disease, after correcting other known risk factors.17 Another study showed that administration of thyroid hormone lowered cholesterol in patients with TSH ranges of 2.0-4.0 but had no effect in lowering cholesterol in patients whose TSH range was 0.2-1.9.18 It also showed that in people with elevated cholesterol, TSH values over 1.9 could indicate that a thyroid deficiency is the culprit, causing excess production of cholesterol, whereas TSH levels below 2.0 would indicate a normal thyroid hormone status. Doctors routinely prescribe cholesterol-lowering drugs to patients without properly evaluating their thyroid status. Based on the evidence presented to date, it might make sense for doctors to first attempt to correct a thyroid deficiency (based on a TSH value over 1.9) instead of resorting to cholesterol-lowering drugs. In a study done to evaluate psychological well being, impairment was found in patients with thyroid abnormalities who were nonetheless within “normal” TSH reference ranges.19 The authors of a study published in the August 3, 2002, issue of The Lancet stated that “the emerging epidemiological data begin to suggest that TSH concentrations above 2.0 (mU/L) may be associated with adverse effects.” When it comes to assessing hormone status, the use of standard reference ranges has failed aging people because reference ranges are adjusted to reflect a person’s age. Since it is normal for an aging person to have imbalances of critical hormones, standard laboratory reference ranges are not flagging dangerously high levels of estrogen and insulin or deficient levels of testosterone, thyroid, and DHEA. The table above shows standard hormone blood reference ranges for men (age 60) and compares them to what the “optimal” ranges should be.
Defying the Reference Ranges The Most Important Blood Tests If a serious abnormality is detected—such as elevated homocysteine, hormone imbalance, high PSA—testing should be repeated more often to determine the benefits of whatever therapy you are using to correct the potentially life-shortening abnormality. We also recommend that you consult with your physician regarding any other test that may be appropriate for your individual condition. The remainder of this article provides detailed information about individual tests and ranges that can be used to assess your health and longevity. At the end of this article, we provide information about the new lower-cost blood testing available to Life Extension members. Male and Female Testing Panels The Male and Female Testing Panels are a terrific place to begin to proactively take charge of your health. These panels comprise the most requested tests, which also happen to be the best and most comprehensive screening tests capable of identifying many common and not-so-common conditions, identifying risk factors for future disease, and offering a clinical snapshot of your current physiologic well being. Both panels consist of a full chemistry and complete blood count (CBC) measuring 35 different blood components, which assess cholesterol and triglyceride levels, blood glucose, iron and mineral levels, kidney and liver function, and blood cell components. The male and female panels also test for levels of total and free testosterone, DHEA-S (an indicator of adrenal cortical function), estradiol, homocysteine, and C-reactive protein. Both homocysteine and C-reactive protein, along with levels of cholesterol lipoproteins, are powerful predictors of cardiovascular disease. The male panel also includes the PSA (prostate-specific antigen) test, which is a very sensitive marker for prostate cancer. The female panel includes a test for progesterone levels, providing information concerning female fertility, ovulation cycles, and possible hormonal tumors. Following are snapshots of the various tests offered in the male and female test panels. Chemistry and complete blood count (CBC) PANEL The chemistry panel provides a wide range of information to assess cardiovascular, endocrine (glucose levels), hepatobiliary, and kidney function. The CBC panel provides information on the presence of infectious organisms, anemias, nutritional deficiencies, lymphoproliferative disorders (i.e., leukemia), and other hematological disorders. Cardiovascular Endocrine Hepatobiliary Kidney Hematological abnormalities and infection RBC, hematocrit, and hemoglobin Platelets WBCs and their components (lymphocytes, monocytes, neutrophils, eosinophils, and basophils)
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total and free testosterone Testosterone is produced in the testes in men, in the ovaries in women, and in the adrenal glands of both men and women. In men, testosterone production is stimulated by luteinizing (LH), which is produced by the pituitary gland and by Leydig cells in the testes. Testosterone levels normally decline with age, dropping to approximately 65% of young adult levels by age 75. This drop in testosterone is partially responsible for the significant physiologic changes seen in aging men. Less than 2% of testosterone is typically found in the “free” (uncomplexed) state in the serum of both men and women. Approx-imately 50% is bound to sex hormone-binding globulin (SHBG) and the remainder to albumin. In men, free testosterone (an androgen, or “male hormone”) levels may be used to evaluate impotence or infertility. In women, high levels of free testosterone may indicate hirsutism (excessive hair growth, especially on the face and chest), which is often indicative of polycystic ovaries and, less commonly, ovarian cancer. Increased testosterone in women also suggests low estrogen levels. Low levels of free testosterone in women can lead to loss of libido, depression, and increased risk of heart disease. Total testosterone (complexed and uncomplexed) is useful for assisting with differential diagnosis in males (LH secretion and Leydig cell function, gonadal and adrenal function, diagnosis of hypogonadism, hypopituitarism, Klinefelter syndrome, and impotence) and in females (Stein-Leventhal syndrome, masculinizing tumors of the ovary, tumors of the adrenal cortices, and congenital adrenal hyperplasia). Pregnenolone DHEA-S (dehydroepiandrosterone sulfate) Estradiol Homocysteine
C-reactive protein (CRP) These risk factors continue to facilitate the attraction and accumulation of inflammatory cells—macrophages, mast cells, and activated T-lymphocytes—within the atherosclerotic plaque. Disruption of this plaque, caused by chronic inflammation, may cause a heart attack as oxygen-deprived blood vessels become clogged with pieces of dislodged plaque material. C-reactive protein is a very sensitive marker of systemic inflammation, and has emerged as a powerful predictor of coronary heart disease21 and other cardiovascular diseases. The highly sensitive CRP test is able to measure the presence of C-reactive protein in the blood, even at very early stages of vascular disease, allowing for appropriate intervention with diet, supplements, or anti-inflammatory therapy. Elevated levels of C-reactive protein have also been found to be associated with risk of developing type II diabetes,22 loss of cognitive ability in seemingly healthy people,23 Alzheimer’s disease, and depression in the elderly. Furthermore, risk factors for atherosclerosis and heart disease, such as smoking and high blood pressure, elevate blood levels of C-reactive protein that can be detected by the high-sensitivity CRP test,24 which is part of the Male and Female Panel tests. PSA (prostate-specific antigen)free and complexed Risk of prostate cancer may be assessed by determining absolute amounts of total PSA or by calculating the percent of free PSA compared to total PSA (complexed plus uncomplexed). A study in the New England Journal of Medicine found that 25% of patients with normal digital rectal exams (DRE) and total PSA levels of 4.0–10.0 ng/ml had prostate cancer.25 In the same study group, researchers calculated that risk of prostate cancer increased with decreases in the percentage of free PSA in the serum. It should be noted that elevated levels of PSA may not necessarily signal prostate cancer, and prostate cancer may not always be accompanied by expression of PSA. Levels may be elevated in the presence of a urinary tract infection and an inflamed prostate. In another study published in the New England Journal of Medicine, investigators recommended lowering the PSA cutoff from 4.1 ng/ml (the threshold at which biopsy is currently recommended). At the current threshold, it was determined that “82 percent of cancers in younger men and 65 percent of cancers in older men would be missed.”26,27 But levels below the currently recognized cutoff of 4.1 ng/ml may not distinguish between prostate cancer and benign prostate disease. A PSA level over 2.5 ng/ml, or a PSA doubling time that occurs in less than 12 years, may be a cause for concern. Progesterone Systemic inflammation and tests for proinflammatory cytokines TNF-a, IL-6, IL-1b and IL-8 Cytokines are cellular growth factors that are synthesized by nearly every cell of the body and are generally produced only in response to “stress.” Secreted primarily from leukocytes (white blood cells), cytokines regulate the hosts’ response to infection, immune responses, inflammation, and trauma. Cytokines may be either proinflammatory (worsen disease) or anti-inflammatory (reduce inflammation and promote healing). Some studies suggest that susceptibility to disease may result from an imbalance between pro- and anti-inflammatory cytokines.29 There is also mounting evidence that depression may directly stimulate the production of proinflammatory (primarily IL-6) cytokines or indirectly stimulate production by down-regulating the cellular immune response (i.e., prolonged infection and delayed healing fuel sustained cytokine release).30 The pro-inflammatory cytokine panel detects abnormally high levels of the most dangerous inflammatory cytokines in the blood: tumor necrosis factor-a (TNF-a), interleukin-1 beta (IL-1b), interleukin-6 (IL-6), and interleukin-8 (IL-8).
Tumor necrosis factor-alpha Elevated levels of TNF-a have also been found in people with high blood pressure,31 and together with IL-6 may be associated with risk of heart disease.32 In a study by Verdeccia et al, levels of TNF-a were measured in persons with or without high blood pressure to ascertain if arterial flow-mediated dilation was affected by hypertension and chronic inflammation. Investigators found that regardless of whether blood pressure was controlled with antihypertensive medication, arterial flow-mediated dilation was significantly impaired in the hypertensive group. This group also showed higher levels of TNF-a, indicating persistent inflammation despite controlling blood pressure. This study showed that even when blood pressure is under control, hypertensives still suffer from continuous damage (endothelial dysfunction) to the inner lining of the arterial wall caused by a chronic inflammatory insult. These findings indicate that hypertensives should have their blood tested for TNF-a to assess how much inner wall (endothelial) arterial damage is occurring. If the level of TNF-a is high, aggressive therapies to suppress the inflammatory cascade should be considered. | ||||||||
| Interleukin-1 beta A study by Lappe et al showed that increased concentration of IL-1b significantly and independently (of CRP) predicted a step-wise increase in the risk of death or myocardial infarction.33 IL-1b is one of the key med-iators of immunobiological responses to physical stress. A study by Brambilla et al showed that IL-1b concentrations were significantly higher in patients with panic disorder both before and after alprazolam (Xanax®) pharmacotherapy, suggesting that IL-1b levels may be a marker of panic disorder that is not related to current levels of symptomology.34 Interleukin-6 Elevated IL-6 is associated with an increased risk for heart attack and stroke. A 1999 study found that individuals with high levels of both IL-6 and CRP were 2.6 times more likely to die during the nearly five-year study period than those with low levels of both measurements of inflammation.37 Elevated IL-6, along with C-reactive protein, may be predictive of development of type II diabetes.21 Pradhan et al followed 27,628 healthy (free of diagnosed type II diabetes, cardiovascular disease, and cancer) women for four years to determine whether elevated blood levels of IL-6 and CRP were associated with development of type II diabetes. Investigators found that IL-6 was significantly higher among women who subsequently developed diabetes, and that the highest levels of IL-6 increased risk for diabetes 7.5 times (women in the highest CRP ranges were 15.7 times more likely to develop type II diabetes). After adjusting for other risk factors—body mass index, family history, smoking, alcohol, exercise, and hormone replacement therapy—women with the highest levels of IL-6 were 2.3 times more likely to become diabetic (4.2 times for the highest CRP blood levels). Investigators concluded that their data supported a possible role for inflammation in the diabetes. Elevated IL-6 levels have also been found to predict risk of death from all causes, independent of other mortality risk factors.37 Harris et al followed 1,293 healthy, elderly people for 4.6 years to determine the association between IL-6, CRP, and mortality. The study found that increased levels of IL-6 were associated with a twofold greater risk of death (and, to a lesser extent, CRP), and that risk increased to 2.6 times when levels of both IL-6 and CRP were elevated. Studies have shown a link between IL-6 and insulin-like growth factor I (IGF-I), which normally declines with age. Cappola et al found that the combination of elevated IL-6 and decreased IGF-I synergistically confers a high risk for progressive disability and death in older women.38 Ferrucci et al found that elderly persons with the highest circulating levels of IL-6 were 1.76 times more likely to develop mobility-disability and 1.62 times more likely to develop mobility plus ADL (activities of daily living)-disability compared with persons with the lowest levels of IL-6.39
Interleukin-8 Additional Cardiovascular Tests Fibrinogen Fibrinogen is also an acute-phase protein reactant, meaning that it increases in response to disease processes involving tissue inflammation or damage. As discussed in the C-reactive protein section, development of atherosclerosis and heart disease are products of inflammatory processes. As such, fibrinogen, which is a measure of inflammation, can help predict risk of heart disease and stroke, and can complement tests for serum cholesterol, cholesterol lipoproteins, lipids, C-reactive protein, and inflammatory cytokines. High fibrinogen levels may indicate a risk of heart disease. Levels are also increased in other inflammatory disorders, in pregnancy, and in women taking oral contraceptives. Decreased levels are seen in patients with hereditary afibrinogenemia, intravascular coagulation, primary and secondary fibrinolysis, and liver disease. An increase in dietary fish oils may result in decreased fibrinogen levels,40 which has important implications for patients at risk for heart disease and stroke. Factor VIII Data from clinical trials have shown that persons with high levels of factor VIII are at increased risk of cardiovascular disease41 and recurrent venous thromboembolism.42 Furthermore, high levels of factor VIII were determined to be the cause, rather than a consequence, of venous thromboembolism.43 Other recent investigations support the hypothesis that there is a physiological basis to the geriatric syndrome of frailty, which is characterized as a wasting syndrome and physiological state of vulnerability to increased morbidity and mortality. Data show a significant increase in markers of inflammation (factor VIII, fibrinogen, and CRP) in the presence or absence of two prevalent chronic diseases: diabetes and cardiovascular disease. Investigators surmise that these specific physiological abnormalities “may make frail older adults more vulnerable to disease processes, functional decline, and mortality.”44 BNP (brain natriuretic peptide) Measurement of BNP involves a relatively simple, inexpensive blood test that is able to diagnose CHF in 15 minutes. CHF is the fourth leading cause of hospitalization in the US, and the leading cause of hospitalization among people over age 65. Its diagnosis is sometimes difficult, with symptoms such as shortness of breath and edema (fluid retention) that are diagnostic of several conditions, and physical examinations prone to error. Although markers such as cytokines and catecholamines (stress hormones) are elevated in CHF, they are hard to measure quickly and often are not elevated until the disease becomes severe. In a study of 250 patients with shortness of breath who were admitted to urgent care and emergency rooms, BNP measurements of 80 pg/ml were 95% accurate in diagnosing CHF, and lower values were 98% accurate in ruling out the condition. Furthermore, urgent care physicians missed 30 cases of CHF diagnosed by the cardiologists; a BNP test could have brought this figure down to one.49 One of this study’s co-authors remarked that the test has greater diagnostic accuracy than the PSA for prostate cancer, the mammogram for breast cancer, or a PAP smear for cervical cancer. Given that one study estimated that up to 20% of all CHF cases are misdiagnosed, the new test will enable urgent care physicians to provide a more rapid, accurate diagnosis for this group of patients.50 Millions of people worldwide with elevated blood glucose levels/type II diabetes are taking either Avandia® or Actos® (thiazolidinediones) to control hyperglycemia. Patients with type II diabetes are at increased risk of developing or exacerbating CHF, and treatment with thiazolidinediones such as Avandia® or Actos® further increases the risk.51,52 Until very recently, no way existed to identify those most likely to suffer from this devastating side effect. In a recent study, however, data showed that BNP levels were a good marker of left ventricular dysfunction (LVD)/CHF induced by pioglitazone (Actos®). The investigators concluded that type II diabetes patients treated with pioglitazone (Actos®) who had elevated BNP levels prior to the start of treatment should be carefully monitored using regular BNP testing to avoid the adverse effects of CHF.53 Dietary Tests Selenium An essential trace mineral, selenium is necessary for normal functioning of the immune system and thyroid gland, and helps protect cells against free radicals that can damage cells, contribute to chronic diseases, and promote cancers. The amount of selenium contained in plant foods is determined by soil content (e.g., high in the Dakotas, very low in some parts of China and Russia). Brazil nuts, walnuts, and bread (in the US) are high in selenium, as is meat from animals that eat grains or plants grown in selenium-rich soils. The current RDA is 55 mcg for men and women, 60 mcg for pregnant women, and 70 mcg for women who are lactating. One ounce of brazil nuts supplies 840 mcg of selenium. Selenium deficiency, common in areas with selenium-deficient soil, may lead to Keshan disease (enlarged heart and poor heart function). Low levels are also seen in patients on TPN (total parenteral nutrition) and patients with malabsorption problems (severe gastrointestinal disorders). Deficiencies can also affect thyroid function. Studies indicated that the incidence of death from cancers (lung, colorectal, and prostate) is lower among people with higher blood levels of selenium.54-60 Furthermore, areas of the US with selenium-deficient soils have higher rates of nonmelanoma skin cancer.61 Low levels of selenium may also be associated with an increased risk of heart disease, rheumatoid arthritis, and HIV/AIDS. Selenium levels should be tested in anyone who suspects occupational exposure (toxic levels) and monitored in those living in areas of selenium-deficient soil, as well as anyone with risk of (or who already has) heart disease, cancer, or arthritis. | |||||||||||||
| Vitamin B12 and folate Folic acid helps protect against chromosomal (genetic) damage; prevents atherosclerosis caused by excess homocysteine; in high doses has been shown to decrease risk of cardiovascular disease; is needed for the utilization of sugar and amino acids; may prevent some types of cancer; promotes healthier skin; and helps protect against intestinal parasites and food poisoning. It has also been established that folic acid can prevent spina bifida; therefore, women of childbearing age should increase their RDA of folic acid. Vitamin B12 and folate deficiencies are most commonly due to problems of malabsorption (B12: gastrointestinal disorders, pancreatitis, tapeworm, and alcoholism; folate: drug interference and jejeunal mucosal disease) or inadequate dietary intake (B12 in rare cases of strict vegetarian diets and folate in general malnutrition or alcoholism). Low levels of B12 are also seen in patients with multiple myeloma and iron deficiency, in those who smoke, and the elderly; in patients with cancer, aplastic anemia, and folate deficiency; in patients on hemodialysis; and in those who ingest high doses of vitamin C. High levels may be increased in acute and myelogenous leukemia, polycythemia vera, leukocytosis, and liver disease. Folic acid levels may be decreased in alcoholics; those with a chronic disease, undergoing hemodialysis, or having anorexia nervosa; and in premature infants and the elderly. Besides pregnancy, increased doses of folic acid may be indicated in hyperthyroidism, neoplasia, hemolytic anemias, and psoriasis. Endocrine DHT (5a-dihydrotestosterone) Low levels of DHT may be associated with decreased sex drive, erectile dysfunction, male pseudohermaphroditism, or pseudovaginal perineoscrotal hypospadias. Increased levels of DHT may be implicated in male-pattern baldness (alopecia), hirsutism (excessive hair growth in women), benign prostatic hyperplasia, and acne. If DHT levels are elevated, drugs such as Avodart®, Proscar®, or Propecia® may be considered. Fasting insulin and HOMAIR Insulin resistance (when the body does not respond to the insulin that it produces) is a common finding in metabolic disorders, including glucose intolerance, dyslipidemia, hyperuricemia, and hypertension,62 and is associated with an increased risk of symptomatic coronary artery disease.63 Furthermore, approximately 25% of persons with insulin resistance will go on to develop type II diabetes. According to Bonora et al, the prevalence of insulin resistance estimated by HOMA is 66% in patients with impaired glucose tolerance, 84% in NIDDM (non-insulin-dependent diabetes mellitus) subjects, 54% in persons with hypercholesterolemia, 84% in hypertriglyceridemia patients, 88% in patients with low HDL cholesterol, 63% in patients with hyperuricemia, and 58% in hypertensive patients. In patients with a combination of glucose intolerance, dyslipidemia and/or hypertension, the prevalence of insulin resistance was 95%.62 Data also show that HOMA-estimated insulin resistance is an independent predictor of cardiovascular disease in patients with type II diabetes.64 Insulin resistance may also be an indictor and likely cause of kidney disease in persons with type I diabetes, according to a study at the University of Pittsburgh. Investigators also found that because insulin resistance predicts heart disease, “it may explain the longstanding observation that in type I diabetes, kidney disease predicts heart disease. In other words, insulin resistance may be the ‘common ground’ for both complications.”65 Early detection of insulin resistance may, therefore, help prevent potentially serious complications that may result from metabolic disorders, including type I and II diabetes, dyslipidemia, hyperuricemia, and hypertention. Somatomedin-C (insulin-like growth factor/IGF-1) IGF-1 is critical in mediating the growth of muscle and other tissues, and normal levels steadily increase until 12-15 years of age, and then begin to decline. Up to one-third of skeletal muscle mass and strength is lost between the ages of 30 and 80.66 A study by Barton-Davis et al showed that IGF-1 overexpression in the muscle cells of mice can preserve the characteristics (morphological and functional) of the skeletal muscles of old mice such that they are equivalent to those of young adult muscles.66 Ruiz-Torres et al showed that when IGF-1 levels in older (over 70) males were similar to levels in younger males (up to 39 years), the older males do not show age-dependent decreases in serum testosterone and lean body mass, nor increases in fat body mass.67 Low levels of IGF-1 have been implicated in the development of atherosclerosis. Van den Beld et al found that high free IGF-1 concentrations appeared to be correlated with reduced risk of atherosclerosis, suggesting that IGF-1 (along with endogenous testosterone and estrone) may play a protective role in the development of atherosclerosis in aging men.68 A study by Carro et al suggests a role for IGF-1 as a neuroprotective hormone. Data show a correlation between lower levels of IGF-1 and higher levels of amyloid-B accumulation in the brains of Alzheimer’s patients. In studies of mutant mice, high amyloid-B levels are seen when serum IGF-1 levels are low. Conversely, the amyloid-B burden can be decreased by increasing levels of serum IGF-1. Investigators suggested that “circulating IGF-1 is a physiological regulator of brain amyloid levels with therapeutic potential.”69 Elevated levels of IGF-1 may be indicative of acromegaly (gigantism) and diabetic retinopathy. Although it has been suspected that high levels of IGF-1 are associated with increased risk of prostate cancer, recent data suggest that IGF-1 may be serving as a tumor marker rather than an etiologic factor for the disease.70 The IGF-1 test (decreased levels) may also be used to evaluate pituitary insufficiency and hypothalamic lesions in children (diagnosis of dwarfism and response to therapy). Low levels have also been found in patients with amyotrophic lateral sclerosis.71 A study on asymptomatic HIV-1-infected subjects tested the hypothesis that oral administration of 3 grams per day of acetyl-L-carnitine (ALCAR) could significantly affect IGF-1 levels. The researchers found that while ALCAR did not raise total IGF-1 levels, it significantly increased the levels of free IGF-1 (the bioactive component of total IGF-1) in treated patients. None of the subjects investigated reported any toxicity directly or indirectly related to ALCAR administration. Remarkably, all treated patients reported subjectively, without exception, an improved sense of well being by the second to third week of ALCAR therapy.72 Thyroid Thyroid stimulating hormone (TSH) is secreted by the pituitary gland and serves to control thyroid hormone secretion in the thyroid. Thyroxine (T4) and triiodothyronine (T3, free) are hormones that are released from the thyroid. Iodine that is taken up by the thyroid is incorporated in T3 and T4 (so called because they have three and four iodine atoms, respectively), which serves to increase the body’s basal metabolic rate, regulate growth and development, increase cardiac output, increase the metabolism of cholesterol, increase the number of LDL receptor sites in the liver, and inhibit TSH secretion. Normally, a decrease in T3 and T4 stimulates TSH release from the pituitary that, in turn, stimulates T3 and T4 production and secretion, and growth of the thyroid gland. When T3 and T4 levels are increased, TSH production is shut down via negative feedback channels. When TSH, T3, or T4 levels fall above or below normal, this is referred to as hypothyroidism (low thyroid activity) or hyperthyroidism (increased thyroid activity, also called thyrotoxicosis). Overt hyper- or hypothyroidism is generally easy to diagnose, but subclinical disease is a bit more elusive. In the National Health and Nutrition Examination Survey (NHANES III), hypothyroidism was found in 4.6% (4.3% mild and 0.3% clinical disease) of a cross-sectional population in the US and hyperthyroidism in 1.3% (0.5% clinical and 0.7% mild) of the same study group. Because mild (or “subclinical”) symptoms may be nonspecific (or absent) and progress slowly, and thryroid functions are not routinely screened, people with mild hyper- or hypothyroidism may go undiagnosed. Undiagnosed mild disease may progress to clinical disease states. People with hypothyroidism and elevated serum cholesterol and LDL have an increased risk of atherosclerosis. Mild hypothyroidism (low thyroid gland function) may be associated with reversible hyper-cholesterolemia (high blood cholesterol) and cognitive dysfunction, as well as such nonspecific symptoms as fatigue, depression, cold intolerance, dry skin, constipation, and weight gain. Mild hyperthyroidism is often associated with atrial fibrillation and reduced bone mineral density and nonspecific symptoms such as fatigue, weight loss, heat intolerance, nervousness, insomnia, muscle weakness, dyspnea, and palpitations, among others. Measurement of TSH is the best test for assessing thyroid function. Currently, the American Thyroid Association recommends TSH testing beginning at age 35, and every five years thereafter.73 Comparing the ratios between TSH, T3, and T4 blood levels, though, may elucidate definitive diagnosis. This is extremely important, given that the majority of people with mild hypo- or hyperthyroidism are asymptomatic, and levels of thyroid hormones may be depressed or elevated only slightly. Although the normally “accepted” upper range for TSH is 5.50 mcIU/mL, investigations have shown that blood levels equal and greater than 2.0 mcIU/mL may actually indicate adverse health effects:
On the positive side, when TSH levels are 2.0-4.0 mcIU/mL, cholesterol levels decline in response to T4 therapy.76 The table below summarizes characteristic thyroid panel results from persons with overt or mild hypo- or hyperthyroidism. Free T3 is valuable in confirming the diagnosis of hyperthyroidism when an elevated free or total T4 level is found. Abnormal concentrations may be seen in T3 toxicosis in the presence of normal T4 levels.
| |||||||||||||||||||||||||||||
| Gastric Tests (Stomach) Helicobactor pylori, IgG antibodies Hepatobiliary Tests (Liver) Gamma glutamyl transpeptidase (GGT) Cancer Markers Carcinoembryonic antigen (CEA) Production usually halts before birth, and blood levels are undetectable in healthy adults. CEA can be seen in people who are heavy smokers, and in patients with certain cancers, particularly those of the pancreas, colon, rectum, breast, and lung. A tumor marker, CEA is not used to diagnose disease, but to determine the extent of disease and patient prognosis, and to monitor treatment efficacy. CEA is often the first marker to elevate in response to a cancer relapse and may show up months before patients become symptomatic or show a response with other laboratory tests. CEA levels may also be elevated in patients with inflammations (i.e., infections, inflammatory bowel disease, and pancreatitis), hypothyroidism, and cirrhosis. Cancer Antigen (CA) 15-3 have antigen levels in the same range as healthy individuals. Increased CA 15-3 levels may also be seen in cancers of the pancreas, lung, ovary, and liver, as well as in (non-malignant) hepatitis and cirrhosis. Skeletal Disorders Women in particular are at increased risk for development of osteoporosis (decrease in bone density). At risk are persons of low body weight (relative to height), cigarette smokers, alcohol abusers, individuals with low dietary calcium and vitamin D intake, women undergoing early menopause, young women who are not menstruating, and persons not getting enough weight-bearing exercise. Family history and advanced age are also risk factors. People with osteoporosis are at increased risk for bone fractures, which may lead to chronic pain, disability, and even death. Osteoporosis can be prevented by determining risk through laboratory testing of biochemical markers, and then taking the proper steps (diet, exercise, drug therapy, supplementation) to reduce bone loss. A highly accurate and inexpensive test of bone resorption (breakdown) is Pyrilinks-D or Dpd (deoxypyridinoline), which involves testing of the second voided urine specimen of the day. DpD, along with pyridinoline (Pyd), forms the rigid crosslinks of mature Type I collagen in bone. During bone resorption, DpD is released into the circulation and is excreted unmetabolized in the urine. Increased levels of Dpd in the urine have been correlated to risk of osteoporosis77 and response to hormone replacement therapy.78 Interleukin-6 (IL-6), which is produced in a variety of tissues including bone, stimulates the differentiation and proliferation of osteoclasts (cells that play an active role in bone resorption), which may then lead to increased bone resorption. Increased blood levels of IL-6 have been found to be a major predictor of bone loss in postmenopausal women, specifically through the first postmenopausal decade.79 Increased levels have also been seen in women with hyperparathyroidism with subsequent bone loss.80 Biochemical markers for bone remodeling (growth) can assess a patient’s status or risk for significant decrease in bone mass by providing a means of measuring bone turnover. In a clinical trial of 7,598 women, increased Ntx (bone resorption marker) was associated with increased risk of hip fractures. While bone densitometry can give an accurate snapshot of bone density, one to two years between evaluations are necessary to detect a bone loss of only 3-5%. Changes to bone metabolism can be assessed using biochemical markers beginning three to six months after initiation of therapy. Bone markers may be classified as bone formation or resorption. Resorption, the breaking down of collagen, occurs prior to bone formation, and biochemical markers provide a direct indication of efficacy of antiresorptive therapy. No change in marker levels may indicate therapy is ineffective (or the patient is noncompliant). An increase in marker levels indicates bone loss (ineffective therapy); a decrease in marker levels indicates therapy is working. Summary To get a clear understanding of one’s physiological well being, simple and noninvasive blood analysis should be performed annually, or more often if assessment of therapeutic efficacy (or for other reasons) is indicated. Especially when test values are at the high or low end of the normal range, periodic testing will be able to pick up positive or negative trends. For example, moderately high cholesterol or C-reactive protein levels may not be flagged in a one-time test, but over a period of time a slight increase will send up a red flag, alerting a patient to a potential problem—increased risk of heart disease. Conversely, following trends will also let you know that you are on the correct path following diet, supplement, or pharmaceutical intervention. Preventable disorders often can be detected years before they manifest as problems with deleterious, and sometimes irreversible, consequences that can substantially impair your quality of life. A yearly blood test is a relatively inexpensive investment (compared to the cost of disease-related health care and prescription medications) to protect your health, increase your quality of life now and in the future, and protect your most precious commodity—you! Get more information about mail order blood testing, including the recommended male and female panels. Penny Baron is a retrovirologist at a prominent New York City hospital, and has been researching HIV/AIDS since 1983. She holds masters degrees in microbiology and clinical nutrition. |
| References |
1. ADVANCEDATA, from Vital & Health Statistics of The National Center for Health Statistics, US Department of Health and Education Welfare, No. 5, February 22, 1977, Public Health Service- Health Resources Administration. “A Comparison of Levels of Serum Cholesterol of Adults 18-74 Years of Age in the Untied States in 1960-62 and 1971- 74.” 2. Mahanonda N, Leowattana W, Kangkagate C, Lolekha P, Pokum S.Homocysteine and restenosis after percutaneous coronary intervention. J Med Assoc Thai.2001 Dec;84 Suppl 3:S636-44. 3. Robinson K, Mayer EL, Miller DP, et al. Hyperhomocysteinemia and low pyridoxal phosphate. Common and inde- pendent reversible risk factors for coronary artery disease. Circulation. 1995 Nov 15;92(10):2825-30. 4. Tenover JS. Effects of testosterone supple- mentation in the aging male. J Clin Endocrinol Metab. 1992 Oct;75(4):1092-8. 5. Phillips GB, Pinkernell BH, Jing TY. The association of hypotestosteronemia with coronary artery disease in men. Arterioscler Thromb. 1994 May;14(5):701-6. 6. Gelfand MM, Wiita B. Androgen and estrogen-androgen hormone replacement therapy: a review of the safety literature, 1941 to 1996. Clin Ther. 1997 May-Jun;19(3):383-404; discussion 367-8. 7. Gooren LJ Endocrine aspects of ageing in the male. Mol Cell Endocrinol. 1998 Oct 25;145(1-2):153-9. 8. Barrett-Connor E, Von Muhlen DG, Kritz- Silverstein D. Bioavailable testosterone and depressed mood in older men: the Rancho Bernardo Study. J Clin Endocrinol Metab. 1999 Feb;84(2):573- 7. 9. Schweiger U, Deuschle M, Weber B, et al. Testosterone, gonadotropin, and cortisol secretion in male patients with major depression. Psychosom Med. 1999 May- Jun;61(3):292-6. 10. Rabkin JG; Wagner GJ; Rabkin R. Testosterone therapy for human immuno-deficiency virus-positive men with and with out hypogonadism. J Clin Psychopharmacol. 1999 Feb;19(1):19-27. 11. Seidman SN, Walsh BT. Testosterone and depression in aging men. Am J Geriatr Psychiatry. 1999 Winter;7(1):18-33. 12. The Testosterone Syndrome, Eugene Shippen and Fryer, W. P. 116. 13. Maximize Your Vitality & Potency, Jonathan Wright and Lenard, L., pp. 146- 47. 14. Super T, Karlis Ullis, MD, Shackman, J, and Ptacek, G. pp. 43-44. 15. Tan RS, Pu SJ. The andropause and memo- ry loss: is there a link between androgen decline and dementia in the aging male? Asian J Androl. 2001 Sep;3(3):169-74. 16. Vanderpump MP, Tunbridge WM, French JM, et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf). 1995 Jul;43(1):55-68. 17. Hak AE, Pols HA, Visser TJ, Drexhage HA, Hofman A, Witteman JC. Subclinical hypothyroidism is an independent risk fac- tor for atherosclerosis and myocardial infarction in elderly women: the Rotterdam Study. Ann Intern Med. 2000 Feb 15;132(4):270-8. 18. Michalopoulou G, Alevizaki M, Piperingos G, et al. High serum cholesterol levels in persons with “high-normal” TSH levels: should one extend the definition of subclin- ical hypothyroidism? Eur J Endocrinol. 1998 Feb;138(2):141-45. 19. Pollock MA, Sturrock A, Marshall K, et al. Thyroxine treatment in patients with symp- toms of hypothyroidism but thyroid func- tion tests within the reference range: ran- domized double blind placebo controlled crossover trial. BMJ. 2001 Oct 20;323(7318):891-5. 20. Bjelland I, Tell GS, Vollset SE, Refsum H, Ueland PM. Folate, vitamin B12, homocysteine, and the MTHFR 677CT polymorphism in anxiety and depression. The Hordaland Homocysteine Study. Archives of General Psychiatry. 2003 Jun;60(6):618-26. 21. Koenig W, Sund M, Fröhlich M, et al. C- reactive protein, a sensitive marker of inflammation, predicts future risk of coronary heart disease in initially healthy middle-aged men. Results from the MONICA (Monitoring Trends and Determinants in Cardiovascular Disease) Augsburg Cohort Study, 1984 to 1992. Circulation. 1999 Jan 19;99(2):237-42 22. Pradhan AD, Manson JE, Rifai N, Buring JE, Ridket PM. C-reactive protein, interleukin 6, and risk of developing Type II diabetes mellitus. JAMA. 2001 Jul 18;286(3):327-34. 23. Teunissen CE, van Boxel MP, Bosma H, et al. Inflammation markers in relation to cog- nition in a healthy aging population. J Neuroimmunol. 2003 Jan (1-2);134:142-50. 24. Pearson TA, Mensah GA, Alexander RW, et al. Markers of inflammation and cardiovas- cular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation. 2003 Jan 28;107(3):499-511. 25. Catalona WJ, Partin AW, Slawin KM, et al. Use of percentage of free prostate-specific antigen to enhance differentiation of prostate cancer from benign prostatic dis- ease. A prospective multicenter clinical trial. JAMA. 1998 May 20; 279(19):1542-7. 26. Punglia RS, D’Amico AV, Catalona WJ, Roehl KA, Karen M., Kuntz KM. Effect of verification bias on screening for prostate cancer by measurement of prostate- specific antigen. N Engl J Med. 2003 Jul 24;349(4):335-42. 27. Reuters Health: Precursor of PSA accurate ly detects prostate cancer. July 15, 2003. 28. Reuters Health: Exercise tougher at begin ning of menstrual cycle. July 5, 2002. 29. Dinarello CA. Proinflammatory cytokines. Chest. 2000;118:503-508. 30. Kiecolt-Glaser JK, Preacher KJ, MacCallum RC, Atkinson C, Malarkey WB, Glaser R. Chronic stress and age-related increases in the proinflammatory cytokine IL-6. Proc Natl Acad Sci. 2003 Jul 22;100(15):9090-5. 31. Verdecchia P, Reboldi G, Porcellati C, et al. Risk of cardiovas cular disease in relation to achieved office and ambulatory blood pres- sure control in treated hypertensive sub- jects. J Am Coll Cardiol. 2002 Mar 6;39(5):878-85. 32. Mendall MA, Patel P, Asante M, et al. Relation of serum cytokine concentrations to cardiovascular risk factors and coronary heart disease. Heart. 1997 Sep;78(3):273-7. 33. Lappe JM, Muhlestein JB, Carlquist JF, Davis BF, Horne BD, Anderson JL. Inlerleukin-1 beta predicts death or myocardial infarction independently of high-sensitivity C-reactive protein and stan- dard risk factors in patients with coronary artery disease. Abstracts – Myocardial Ischemia and Infarction 841-2. J Am Coll Cardiol. 2002. 34. Brambilla F, Bellodi L, Perna G, Bertani A, Panerai A, Sacerdote P. Plasma Interleukin-1beta concentrations in panic disorder. Psychiatry Res. 1994 Nov;54(2):135-42. 35. Ishimi Y, Miyaura C, Jin CH, Akatsu T, Abe E, Nakamura Y, Yamaguchi A, Yoshiki S, Matsuda T, Hirano T, et al. IL-6 is pro- duced by osteoblasts and induces bone resorption. J Immunol. 1990 Nov 15;145(10):3297-303. 36. Lou W, Ni Z, Dyer K, Tweardy DJ, Gao AC. Interleukin-6 induces prostate cancer cell growth accompanied by activation of stat3 signaling pathway. Prostate. 2000 Feb 15;42(3):239-42. 37. Harris TB, Ferrucci L, Tracy RP, et al. Associations of elevated interleukin-6 and C-reactive protein levels with mortality in the elderly. Am J Med. 1999 May;106(5):506-12. 38. Cappola AR, Xue Q-L, Ferrucci L, Guralnik JM, Volpato S, Fried LP. Insulin- like growth factor I and interleukin-6 con- tribute synergistically to disability and mor- tality in older women. J Clin Endocrinol Metab. 2003 May;88(5):2019-25. 39. Ferruci L, Harris TB, Guralnik JM, et al. Serum IL-6 level and the development of disability in older persons. J Am Geriatr Soc. 1999 Jun;47(6):639-46. 40. Radack K, Deck C, Huster G. Dietary sup- plementation with low-dose fish oils lowers fibrinogen levels. Ann Intern Med. 1989 Nov 1;111(9):757-8. 41. Folsom AR, Wu KK, Shahar E, Davis CE. Association of hemostatic variables with prevalent cardiovascular disease and asymp- tomatic carotid artery atherosclerosis. The Atherosclerosis Risk in Communities (ARIC) Study Investigators. Arterioscler Thromb. 1993 Dec;13(12):1829-36. 42. Kyrle PA, Minar E, Hirschl M, et al. High plasma levels of factor VIII and the risk of recurrent venous thromboembolism. N Engl J Med.2000 Aug 17;343(7):457-62. 43. Walston J, McBurnie MA, Newman A, et al. Frailty and activation of the inflamma tion and coagulation systems with and with out clinical comorbidities. Arch Intern Med. 2002 Nov 11;162(20):2333-41. 44. Burguera JL, Burguera M, Gallignani M, Alarcon OM, Burgueera JA. Blood serum selenium in the province of Merida, Venezuela, related to sex, cancer incidence and soil selenium content. J Trace Elem Electrolytes Health Dis. 1990 Jun;4(2):73-7. 45. Mukoyama M, Nakao K, Hosoda K, et al. Brain natriuretic peptide as a novel cardiac hormone in humans: evidence for an exqui site dual natriuretic peptide system, atrial natriuretic peptide and brain natriuretic peptide. J Clin Invest. 1991 Apr:87(4):1402-12. 46. Carillo-Jimenez R, Treadwell TL, Goldfine H, Buenano A, Lamas GA, Hennekens CH. Brain natriuretic peptide and HIV-related cardiomyopathy. AIDS Read. 2002 Nov;12(11):501-8. 47. Kawai K, Hata K, Takaoka H, Kawai H, Yokoyama M. Plasma brain natriuretic pep- tide as a novel therapeutic indicator in idio- pathic dilated cardiomyopathy during beta- blocker therapy: a potential of hormone- guided treatment. Am Heart J. 2001 Jun;141(6):925-32. 48. Richards AM, Nicholls MG, Yandle TG, et al. Plasma N-terminal pro-brain natriuretic peptide and adrenomedullin: new neuro- hormonal predictors of left ventricular func- tion and prognosis after myocardial infarc- tion. Circulation. 1998 May 19;97(19):1921- 9. 49. Dao Q, Krishnaswamy P, Kazanegra R, et al. Utility of B-type natriuretic peptide in the diagnosis of congestive heart failure in an urgent-care setting. J Am Coll Cardiol. 2001 Feb;37(2):379-85. 50. Available at: http://www.lifeextension.com/what shot /2001_02.html#hpfi. Accessed March 10, 2004. 51. Cheng-Lai A, Levine A. Rosiglitazone: an agent from the thiazolidinedione class for the treatment of type 2 diabetes. Heart Dis. 2000 Jul-Aug;2(4):326-33. 52. Krentz AJ, Bailey CJ, Melander A. Thiazolidinediones for type 2 diabetes. BMJ. 2000 Jul 29;321(7256):252-3. 53. Ogawa S, Takeuchi K, Ito S. Plasma BNP levels in the treatment of type 2 diabetes with piglitazone. J Clin Endocrinol Metab. 2003 Aug;88(8):3993-6. 54. Burguera JL, Burguera M, Gallignani M, Alarcon OM, Burgueera JA. Blood serum selenium in the province of Merida, Venezuela, related to sex, cancer incidence and soil selenium content. J Trace Elem Electrolytes Health Dis. 1990 Jun;4(2):73-7. 55. Fleet JC. Dietary selenium repletion may reduce cancer incidence in people at high risk who live in areas with low soil selenium. Nutr Rev. 1997 Jul;55(7):277-9. 56. Knekt P, Marniemi J, Teppo L, Heliovaara M, Aromaa A. Is low selenium status a risk factor for lung cancer? Am J Epidemiol. 1998 Nov 15;148(10):975-82. 57. Patterson BH, Levander OA. Naturally occurring selenium compounds in cancer chemoprevention trials: A workshop sum- mary. Cancer Epidemiol Prev. 1997 Jan;6(1):63-9. 58. Russo MW, Murray SC, Wurzelmann JI, Woosley JT, Sandler RS. Plasma selenium levels and the risk of colorectal adenomas. Nutr Cancer. 1997 ;28(2):125-9. 59. Shamberger RJ. The genotoxicity of seleni- um. Mutat Res.1985 Jul;154(1):29-48. 60. Young KJ, Lee PN. Intervention studies on cancer. Eur J Cancer Prev. 1999 Apr;8(2):91-103. 61. Fleet JC. Dietary selenium repletion may reduce cancer incidence in people at high risk who live in areas with low soil selenium. Nutr Rev. 1997 Jul;55(7):277-9. 62. Bonora E, Kiechl S, Willeit J, et al. Prevalence of insulin resistance in metabolic disorders: the Bruneck Study. Diabetes. 1998 Oct;47(10):1643-9. 63. Arad Y, Newstein D, Cadet F, Roth M, Guerci AD. Association of multiple risk factors and insulin resistance with increased prevalence of asymptomatic coronary artery disease by an electon-beam computed tomographic study. Arterioscler Thromb Vasc Biol. 2001 Dec;21(12):2051-8. 64. Bonora E, Formentini G, Calcaterra F, et al. HOMA-estimated insulin resistance is an independent predictor of cardiovascular disease in Type II diabetic subjects. Prospective data from the Verona Diabetes Complications Study. Diabetes Care. 2002 Jul;25(7):1135-41. 65. Available at:http://www.discover.pitt.edu/ media/pcc020916/sciInsulinResistant.html. Accessed March 10, 2004. 66. Barton-Davis ER, Shoturma DI, Musaro A, Rosenthal N, Sweeney HL. Viral mediated expression of insulin-like growth factor I blocks the aging-related loss of skeletal muscle function. Proc Natl Acad Sci. 1998 Dec 22;95(26):15603-7. 67. Ruiz-Torres A, Soares de Meo Kirzner M. Ageing and longevity are related to growth hormone/insulin-like growth factor-1 secre- tion. Gerontology. 2002 Nov-Dec;48(6);401- 7. 68. van den Beld AW, Bots ML, Janssen JA, Pols HA, Lamberts SW, Grobbee DE. Endogenous hormones and carotid atherosclerosis in elderly men. Am J Epidemiol. 2004 Jan 1;157(1):25-31. 69. Carro E, Trejo JL, Gomez-Isla T, LeRoith D, Torres-Aleman I. Serum insulin-like growth factor I regulates brain amyloid-B levels. Nature Medicine. 2002 Dec;8(12):1390-7. 70. Woodson K, Tangrea JA, Pollak M, et al. Serum insulin-like growth factor I: tumor marker or etiologic factor? A prospective study of prostate cancer among Finnish men. Cancer Res. 2003 Jul 15;63(14):3991-4. 71. Wilczak N, de Vos RAI, De Keyser J. Free insulin-like growth factor (IGF)-1 and IGF binding proteins 2,5, and 6 in spinal motor neurons in amyotrophic lateral sclerosis. Lancet. 2003 Mar 22;361(9362):1007-11. 72. Di Marzio L, Moretti S, D’Alo S, et al. Acetyl-L-carnitine administration increases insulin-like growth factor 1 levels in asymp- tomatic HIV-1-infected subjects: correlation with its suppressive effect on lymphocyte apoptosis and ceramide generation. Clin Immunol. 1999 Jul;92(1):103-10. 73. Ladenson PW, Singer PA, Ain KB, et al. American Thyroid Association guidelines for detection of thyroid dysfunction. Arch Intern Med. 2000 Jun 12;160(11):1573-5. 74. Vanderpump MP, Tunbridge WM, French JM et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol. 1995 Jul;43(1):55-68. 75. Hak AE, Pols HA, Visser TJ, Drexhage HA, Hofman A, Witteman JC. Subclinical hypothyroidism is an independ- ent risk factor for atherosclerosis and myocardial infarction in elderly women: The Rotterdam Study. Ann Intern Med. 2000 Feb 15;132(4):270-8. 76. Michalopoulou G, Alevizaki M, Piperingos G. et al. High serum cholesterol levels in persons with “high-normal” TSH levels: should one extend the definition of subclini- cal hypothyroidism. Eur J Endocrinol. 1998 Feb;138(2):141-5. 77. Garnero P, Hausherr E, Chapey MC, et al. Markers of bone resorption predict hip fracture in elderly women: The EPIDOS prospective study. J Bone Min Res. 1996 Oct;11(10):1531-8. 78. Hesley RP, Shepard KA, Jankins DK, Riggs BL. Monitoring estrogen replacement ther- apy and identifying rapid bone losers with an immunoassay for deoxypyridinoline. Osteoporos Int.1998;8(2):159-64. 79. Scheidt-Nave C, Bismar H, Leidig-Bruckner G, et al. Serum inter- leukin 6 is a major predictor of bone loss in women specific to the first decade past menopause. J Clin Endocrinol Metab. 2001 May;86(5):2032-42. 80. Lakatos P, Foldes J, Horvath C, et al. Serum interleukin-6 and bone metabolism in patients with thyroid function disorders. J Clin Endocrinol Metab. 1997 Jan;82(1):78-81. |