Life Extension Magazine®
|
Living longer and healthier may come from a variety of factors. One promising field is genomics, which uses new technologies to identify every human gene, and then develop precise drug "targets" that may, in turn, cure the diseases of aging. By Pamela Tames
Those kinds of drugs don't exist now, but in another decade they may, thanks to developments in genomics, an exciting new branch of biology that uses powerful technologies to identify every human gene, collectively known as the "genome." It may just be the key to discovering completely new, precise drug targets-that is, human enzymes, receptors and ion channels known to play a role in diseases, excluding those caused by pathogens. No one has been watching the surge of genetic information coming out of genomics labs more closely than pharmaceutical executives. They've been cutting million-dollar deals with so-called genomics biotech companies fast and furiously. These companies believe genomics offers an efficient way to find new drug targets. That's no small feat in a world dominated by me-too drugs-improved versions of already existing drugs. (For more on how large companies and researchers are joining to produce anti-aging drugs, see Life Extension magazine, October and November 1998.) If the genomics approach pans out, in a decade or so we could see novel treatments for age-associated diseases that are now "incurable." Right now, that possibility is hope enough for the pharmaceutical industry. Drug companies spend hundreds of millions of dollars and several years bringing a drug from lab bench to pharmacy shelf. Many companies set their sights on a growth rate of about 10% annually, but many don't have enough drugs in the pipeline to keep up that growth rate via the traditional methods of drug discovery. That is why genomics companies, making up about 5% of the biotechnology sector, are springing up everywhere. Genomics companies are going at the concept from every conceivable angle. Some have focused on developing tests for genetic mutations associated with an increased disease risk, others, on finding gene-based diagnostics. At LifeSpan BioSciences, a Seattle, Wash.-based genomics company, the dream is bigger: To better understand the links between genes and disease, with the aim of generating leads for developing new drugs that could treat not just symptoms but possibly causes, too. "When Glenna Burmer and I founded LifeSpan in 1995," says Dr. Joseph Brown, president and chief executive officer, "we were convinced genomics was the most efficient way to find new drugs. The human genome consists of about 100,000 genes. The top 200 drugs for sale today target about 50 of those genes. So there are many potential targets that haven't been discovered." The most exciting implication of genomics is the possibility of finding new, precise treatments for diseases that are now incurable. Compared to traditional techniques of drug discovery, genomics can do this faster, more efficiently and with a higher degree of predictability, explains Brown. "These are new kinds of drugs aimed at new targets. It's the difference between kicking your car and putting a diagnostic computer on it." That genomics can do all this has already been borne out. The so-called protease-inhibitor drugs used in the treatment of AIDS were developed using genomics. "A novel gene for protease was found," explains Brown, "and in just seven years a whole new class of drugs was developed and has been very effective." At LifeSpan, researchers have already found a gene that is over-expressed in breast cancer tissue samples (as compared to normal controls). The company filed a patent on that gene and has done functional assays suggesting it is required for the growth of the breast cancer. LifeSpan's ultimate goal is to find the top 1,000 genes that are attributable to disease. The company generates some of its revenue by selling to pharmaceutical companies these "gene leads" for possible drug targets. When LifeSpan was founded three years ago, the focus was initially on diseases related to aging (it's since expanded to include other major diseases). The demographics were reason enough. In 1994, 34 million people in the U.S. were over the age of 65, with big numbers in Europe and Japan as well. And, there's a strong statistical association between disease and age. The incidence of such common diseases as arthritis, hearing loss, cataracts, cancer and cardiovascular diseases all rise exponentially after age 40. Recent research findings increasingly show aging isn't just a random, wearing-out process. Rather, it appears to be controlled by specific genetic changes that affect underlying molecular mechanisms. "So my bias," says Dr. Burmer, chief scientific officer and executive vice president of LifeSpan, "is, that you won't affect human life span very much until you start treating the underlying genetic mechanisms causing disease." Burmer and Brown believe that for certain specific diseases, the underlying genetic mechanism eventually converges into a common pathway leading to symptoms. Hypothetically, this common pathway could be blocked if you find the right gene to target with a drug, effectively throwing a wrench in the machine. "The model we and others are following is to identify genes related to the progression of the disease," says Burmer. "This may not necessarily entail the primary cause of disease, although it might. But, it's somewhere in the common pathway. The principle is that, somewhere in the pathway, there's a target you can develop a drug against." Not surprisingly, finding common pathways is not easy. No matter how you look at it, disease is very complex. For one thing, genes can act very differently. Some genes are risk factors for a disease; thus, when they're activated, you're at higher risk for developing the disease. In most cases of disease, though, no single gene can be isolated. "Even when you can isolate a single gene, it may only be involved in a few cases of the disease," adds Brown. Such is the case with breast cancer. There are a number of risk factors for breast cancer, including the so-called BRCA-1 gene, age, X-rays (mammography), and estrogen levels. One or many risk factors may act on normal breast tissue, triggering a disease process, which-mediated through a common pathway-ultimately leads to cancer. Finding a "downstream target," such as an enzyme inhibitor, is potentially more effective than developing a drug to address the BRCA-1 gene (an "upstream target"), since the gene is a major cause of disease in relatively few women. 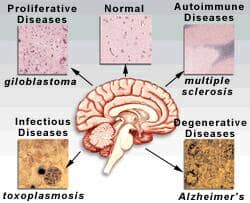 When Tissue is the Issue Many investigators are tracking the genes in these common, multi-step disease pathways. But, like the blind men and the elephant, everyone may be somewhat correct but not have the whole picture yet. "We anticipate that companies like ours that focus on gene discovery will start seeing different parts of the picture," says Burmer. "In aging skin, for example, we see a whole cluster of genes that are up-regulated (turned on) or down-regulated (turned off) in coordinated fashion. So we're hoping to fill in many more pieces of the puzzle and put names to each step."Analyzing changes in gene expression and identifying key genes at which to aim drugs isn't the same as finding a longevity drug. "When you think about a cure to aging," says Burmer, "what you're really talking about in technical terms is a form of controlled regeneration. That's a tough nut to crack. After a tissue undergoes regeneration, it's a question of the genetic code in the cells of that tissue being altered in such a way that it will, for example, double the number of stem cells being produced. It this could happen, it would be, in effect, the real cure to aging." Even then, there are problems. Activating the "on switch" (if there actually is one) for continued stem-cell doubling in a tissue could increase the cancer rate in different organs. And how would you design a clinical trial to establish if a possible aging cure actually works? "You can design a therapeutic trial to measure a drug's effect on a disease," says Brown, "but how do you measure if a drug increases life expectancy? It's very difficult."
|
|
Which is why the goal at LifeSpan isn't to find the switch that doubles stem cell populations; it's to discover gene targets on disease pathways. These are two discrete research paths, and the latter is equally important. "For many people," says Brown, "the goal is avoiding debilitating diseases of aging or at least controlling and treating them, rather than just extending the number of years they are able to live." In order to find the common pathways implicated in disease, you have to sift through huge volumes of genetic information. Then you have to determine which of the many gene targets that you've identified are specifically disease-associated or causative, and thus possible drug targets. LifeSpan researchers accomplish this by correlating gene expression to changes in actual human tissue samples representing diseased versus normal (aging) states. LifeSpan has an on-site human tissue bank containing 1 million normal and disease samples. The samples are anonymous and were taken from hospital pathology departments. They include more than 175 different types of tissues from most body organs, and represent all age groups. There are samples for more than 500 different disease categories at every stage of disease progression, including aging diseases, autoimmune diseases, infectious diseases, degenerative diseases, cancer and benign proliferative diseases, and genetic diseases. Any individual tissue expresses about 20,000 to 50,000 of the approximately 100,000 genes that make up the entire human genome. Further, genes that are expressed in normal tissue may be turned off in disease states, and different genes may be turned on instead. In order to analyze hundreds of genes simultaneously and to compare patterns of gene expression in diseased and healthy tissue samples, LifeSpan researchers use proprietary "high density array" technology. A high-density array is a glass or nylon square plate that supports a filter of "DNA spots," with each spot representing a single gene (the DNA comes from cloned genes). An array can have up to 10,000 spots per filter, allowing the testing of a highly efficient 10,000 genes at a time. To analyze the genes from a tissue sample, researchers first extract what's known as the messenger RNA, or mRNA. The mRNA is labeled so it acts as a probe when you place it on the filter. "Each gene hybridizes (or binds) to its complementary mRNA probe and you get an array of spots," explains Brown. "The darker the spot, the more gene expression in that tissue." The process yields a river of genetic and biological information. "We have a million tissues and will be generating tens of millions of data points on gene expression," says Brown. In order to make sense of it all and put it into useful forms (a computing science called bioinformatics), LifeSpan has developed proprietary DNA sequence analysis software, which can search through a million different genes in less than 24 hours and identify novel gene-disease associations or gene-drug associations. The flux of gene expression can be visualized as a series of lists of DNA sequences, 3-D bar graphs, or "topograms," in which the heights of the peaks represent the amount of gene expression. "You see different genes being over-expressed in young versus old tissues and in different tissue types," explains Brown. "Each tissue type gives a characteristic gene pattern, and in disease you get more subtle changes. So, we look for these subtle differences." According to Brown, by this stage in the search for candidate genes, "You've narrowed the field from 100,000 genes to about 10,000 interesting genes for which there is some evidence they are related to the disease process, either because they're over-expressed or mutated. In effect, you've gone from deep-sea fishing to fly-fishing. But, the big question still remains: Which of these candidate genes would make a good drug target?" LifeSpan researchers use a variety of methods to help narrow the field even further. These include functional assays in cell cultures (you specifically inhibit a gene in culture, or in vitro); functional assays in what's known as a "knockout mouse" (the gene is mutated in fruit fly or another organism and you analyze the effect of that on cells, organs, and organism); and "target validation." To validate a target, researchers make specific probes or antibodies that bind selectively to the gene or protein under study. Then they compare antibody-gene binding patterns in diseased and normal tissues. This pinpoints precisely where in the cell or organ the gene is expressed and how it's associated with the actual disease pathology. By this stage, you've teased out 1,000 most-likely drug targets. You still don't have a final answer, says Brown, but you have enough good leads to proceed to the next step-actually picking a target to test. By narrowing the field and validating targets, LifeSpan helps reduce some of the financial risk involved in choosing a target to test and develop-typically, a $10 million expenditure for drug companies. It's not perfect, but it's better than traditional drug discovery, which starts with a natural substance, such as a plant, and randomly screens compounds extracted from it in an attempt to find ones that have potential therapeutic activity. This usually takes years and isn't always fruitful. Once you do find something you think has therapeutic activity, the next step is extensive animal testing. Fewer than one compound in 10,000 makes it through this process, which typically takes 12 to 15 years and costs about $300 million. With genomics, there's a better chance you'll find the right target. "By identifying every part of the pathway, a drug company can choose different parts to manipulate," says Burmer. "Once a target is chosen, it's basically traditional drug development from there, albeit with more efficient combinatorial chemistry. It's a powerful new way to construct giant libraries of potential drugs by synthesizing thousands of variations on each chemical theme." Big pharmaceutical companies aren't the only ones interested in new targets. So are cosmetics companies, most of which have aging research groups. LifeSpan is in discussion with a number of them; their goal is to investigate the normal process of skin aging in an attempt to develop new therapies called "cosmeceuticals," pharmacologic agents that act at a cellular level, stimulating actual change. Examples include retin-A for wrinkled skin, and minoxidil for hair loss. Compounds such as these and others than may be developed can have a dramatic effect on aging, says Brown. "So there's a realization in this industry that there are compounds that can have an actual affect on aging skin and the goal is to find these. Our process can do that because we're looking specifically at gene expression in young and older skin, and finding genes that are differentially expressed and thus potential targets for cosmeceuticals," he adds. When genomics first hit, venture capitalists, the big drug companies and Wall Street swooned over the commercial possibilities, and poured millions into startups. However, it's clear genomics is in its infancy and everything takes longer than anticipated. Genetic information is swelling databases at a prodigious rate, but there's still a lot that is unknown about the relationship between DNA sequences and helping people live longer, healthier lives. Add to that the length of time required for gene discovery and drug development. "It takes one to two years to filter through your initial discovery and focus on the ones that are truly important to a disease. From gene discovery to drug discovery is about 10 years, even after you've found the gene. We don't anticipate a radical shortening of this process. Even if an aging-related gene were discovered, it would take a minimum of 10 years before it hit the market." One problem is that genomics emphasizes the front end of drug discovery, so drug companies may end up with a logjam of potential drugs to take into development. Currently, the tail end of drug development isn't getting a similar push from other breakthrough technologies. That's not about to slow research at LifeSpan. "Every time we discover a gene that looks interesting, we follow the lead," says Burmer. "We do more functional genomics to see if it's really involved in aging disease or aging normal tissue or aging mouse. We're hoping for clues to treat the chronic diseases of aging, so that one can improve the quality of life during the later years." No one is anticipating a major leap in how diseases of aging are treated in the near future. What's more likely, says Burmer, is that treatments will be developed one disease at a time. Eventually, you could end up with multiple drugs to target different steps in the pathway and multiple drugs to target the individual diseases of aging. These probably won't be preventive agents, since diseases such as Alzheimer's only affect a small percentage of the population. "So, in many cases, it will be precise diagnosis and very early treatment," explains Brown. "We can't be taking 25 different pills to cover all the possible diseases of aging. You're much better off if you can control or treat the ones you do end up getting." |
