Life Extension Magazine®
| While overwhelming research lies ahead, promising findings suggest that human embryonic stem cell therapy may provide the key to curing life-threatening diseases.
by Carmia Borek, Ph. D. Last summer researchers at the University of Pittsburgh Medical Center reported a scientific first-a nerve cell transplant into the brain, intended to reverse damage caused by a stroke. The patient, a 62-year-old woman with right-side paralysis and impaired speech, was injected with nerve cells (neurons). The operation was the first of 12 in a phase I clinical trial intended to investigate the safety of the procedure, and to determine whether the body would tolerate or reject the transplant through immune mechanisms. The medical team expected the transplant would improve the function of the damaged nerve cells or replace the ones that were destroyed. There were no major medical complications from the procedure; the patient reportedly felt well. Since then, other stroke patients have received transplants to help speed their recovery.The successful nerve transplant is an important clinical feat. Since the adult human brain cannot regenerate nerve tissue, replacement of damaged cells via transplants is the only technique that may help recovery. Close to one year after the initial transplant, some of patients showed different degrees of mild recovery, as reported by the investigators at a scientific meeting. This is a hopeful sign. Would it be possible to develop a regular supply of transplants to replace a damaged brain, a damaged heart or any other organ in need? Hypothetically the answer is "yes," by culturing cells from embryos that are blank, uncommitted cells, but are capable of dividing and developing (differentiating) into virtually all other cell types. Eventually these pluripotent stem cells, as they are called, would be coaxed to develop into tissues for transplantation therapy to repair damaged organs such as heart, brain, kidney and the like. Scientists have successfully cultured embryonic stem cells in mice. They directed the stem cells to develop into different cell types, studying the genes and environmental factors that regulate their development, and transplanting them as grafts that function as tissues. Non- human primate embryonic stem cells have also been cultured. But the big news came in 1998 with the demonstration that human embryonic stem cells can be grown in culture. The news caught the public imagination with visions of organs made to order, and stirred government regulatory agencies into debates on the ethics of using human embryo stem cells for research and on banning government funding for such research. While the technical achievement of growing and keeping embryonic human cells alive is an impressive feat, challenges do lie ahead. It is important to note that in order to realize the potential of stem cells for human transplantation therapy and other uses, the conditions that allow them to mature into the desired tissue must be further investigated. Some clues from stem cell development in plants and animals may help pave the way.
In certain tissues, cells are renewed constantly. Their stem cells produce differentiated cells to replace cells whose survival time is finite. Red blood cells, the body's oxygen carriers, live only 120 days. They are constantly replaced by new ones formed from stem cells in the bone marrow. At high altitudes where oxygen levels are low, such as the Andes at 14,000 feet, additional red blood cells are produced to help cope with low oxygen supplies while adapting to the new surroundings. In the digestive system, intestinal stem cells constantly differentiate into cells that line the gut, replacing those that have sloughed off. Skin stem cells make skin, and stem cells in the hair follicles make hair. Stem cells that produce a wide range of immune cells differentiate into mature immune cells in response to special signals by hormone-like substances that are increased during infection and inflammation. Embryonic stem cells, such as the ones recently cultured from human embryos, are even more fascinating. At the early stages of the embryo's development, when the fertilized egg has divided only a few times, cells cultured from these embryos-in the blastocyst stage-can give rise to essentially all cell types in the body, making them pluripotent. And, indeed, the team reporting the growth of human embryo stem cells also showed that when transplanted into mice, the human embryonic stem cells formed a variety of recognizable tissues. Injury and disease states are associated with the loss of cells or with cellular dysfunction. Stroke is associated with the death of nerve cells (neurons) and loss of brain function in the affected areas. People with Alzheimer's disease, Parkinson's disease or other neurodegenerative diseases are victims of slow degeneration and death of brain cells. A person who has just had a severe heart attack may be in a state where cells in a large area of the heart have died. In the latter case, the result is that only part of his or her heart muscle is able to pump blood. An insufficient flow of oxygen and other nutrients to the brain and other organs follows, threatening life. In many of these diseased states, cell replacement by transplantation is the desirable option to restore tissue function. Cell transplantation needs stem cells that produce identical daughter cells in a process of self reproduction, as well as create daughter cells whose destiny is to differentiate into the required specialized cell. Continuous self replication is essential for two reasons. The first is that it enables researchers to maintain transplants of tissues needed for treatment for long periods of time. The second reason is that it enables the production and maintenance of large amounts of immature cells that, in principle, could be transplanted into adult tissue and become functional mature cells, incorporating themselves into the tissue. In the case of red blood cells that live only 120 days, effective transplantation of blood cells could be achieved by initially re-establishing a red blood cell population that needs a boost, and continuing with replenishment of the red blood cell population as the old ones die. This is possible only with transferred stem cells that can self-renew for long periods of time. Bone marrow-cell transfers used to replace cancerous leukemic blood cells with normal cells are perhaps the best known examples of cell replacement to restore tissue function in a diseased tissue. There have been many efforts to establish human stem cell cultures from specific adult tissues that have met with little success. An alternative method attempted was to culture human pluripotent stem cells that can develop into different cell types. Pluripotent stem cells with the ability to produce all types of fetal and adult cells exist only in the early embryo and in a stem cell tumor called teratocarcinoma. Cultures from teratocarcinoma cells have been in use for some time in experimental investigations. Though their cancerous origin makes them unpopular for therapeutic purposes in humans, they actually do not behave as cancer cells upon differentiation into mature cells. In fact, the transplanted nerve cells used in the stroke patient were from altered teratocarcinoma cells. Human embryonic stem cells that can divide and replicate for long periods of time have reportedly been grown in culture in two instances. The first, reported in November 1998 by Dr. James Thomson of the University of Wisconsin and collaborators, showed that cells from early embryos that have only a few cells (blastocysts) could be cultured in vitro and maintained as a culture of dividing cells for more than eight months. These embryos were a result of in vitro fertilization and were donated by the individuals involved. The cells had all the characteristics of normal early embryonic stem cells. Thomson and colleagues found that when injected into mice deprived of their immune systems, to avoid graft rejection, the human embryonic cells formed many recognizable human tissues. These included cells of the lining of the gut, bone, cartilage, muscle and nerve tissue. Even when kept in dishes under controlled conditions, the embryonic stem cells differentiated into distinct embryonic tissues that could further develop into specialized tissues in appropriate growth conditions. Thus, the growth and differentiation in vitro could enable scientists to study developmental events, especially in the early stages, that cannot be studied directly in the human embryo. These may be useful in investigating problems associated with infertility and loss of pregnancy, as well as birth defects. The second report came from a laboratory more than 1,000 miles away from Wisconsin, at the Johns Hopkins University School of Medicine in Baltimore. Dr. John Gearhart and collaborators reported the isolation and culturing of human pluripotent stem cells from embryonic tissue that, after further development, become ovaries or testes (primordial germ cells). The tissues originated from five- to nine-week-old embryos of aborted pregnancies. The cells kept dividing in culture for more than 20 weeks and showed characteristics of pluripotent stem cells. They were able to differentiate into further stages of embryo development when kept in culture. How the cells behave when injected into animals has not yet been reported. More recently, a research team from Milan, Italy, succeeded in isolating stem cells from a human embryonic brain. Depending on the conditions in which they were grown, the cells had the ability to mature into different types of brain cells. The successful culturing of these cells shows promise for clinical brain transplantation from a source of normal specialized embryonic brain stem cells. Though the technical achievement of growing human stem cells is exciting, there is still much to be done. In several areas, additional research must be conducted to further explore therapy and transplantation possibilities. Differentiation occurs in a disorganized manner, producing a mixture of many cell types. Mixed populations of cells are unsuitable for transplantation into specific tissues. When there are many cell types, they may compete with one another to enter the tissue, preventing the cell of interest from getting into the intended tissue area that needs repair. In addition, if upon transplantation the undifferentiated stem cell remains undifferentiated-dividing indefinitely without maturing- it may develop into a stem cell tumor. There is a lot to learn about the conditions required for the stem cell to form other tissues for long periods of time, i.e. remain pluripotent; much to learn about the substances and conditions that will drive a stem cell to differentiate into the specific mature cell type needed. Lack of genetic matching may result in graft rejection, hence the task of matching the identity of the embryonic source and the patient requiring the transplant. In the future, this hurdle may be overcome by culturing adult cells from a patient and using the genetic material of the patient's cells to create embryonic stem cells. To illustrate an example, let's take a hypothetical situation. Someone by the name of Mark has a severe heart attack. A technician takes some of Mark's skin cells and separates them, strips them of their genetic material and injects it into a donated human egg that, too, has had all of its genetic material removed. The egg is stimulated to divide in vitro and the embryo develops in the dish. In the embryo's early stage, cells are removed and cultured as pluripotent stem cells that can develop into many types of cells, including heart cells. A medical team maintains the cells and grows them under conditions that will coax them to develop into heart cells only. Since the cells are a perfect genetic match to Mark's tissue, they can be transplanted into his heart without the risk of rejection by immune cells that attack foreign cells. The cells will replace the ones that died in the heart attack. It may be possible in the future to have a bank of frozen cells taken early in life and used later to repair a variety of damaged organs. The first transplant of nerve cells into a stroke patient, described earlier, was done before the reported successful culturing of normal human embryonic stem cells. The nerve transplant that "marks a transition in stroke medicine from damage limitations to restoration of lost brain function," as described by the team leader Dr. Kondziolka, was done by using nerve cells developed from a human stem cell tumor, a teratocarcinoma. First, the stroke lesion was located in the brain by radiological means (computer tomography-CT, or nuclear magnetic resonance-NM). Then, at least two million nerve cells (neurons) isolated from a teratocarcinoma cell line (teratocarcinoma cells that had been in culture for some time and available commercially) were injected close to the stroke lesion, in several sites. The transplantation grafts of cells were produced by treating the tumor cells with the vitamin A-related compound retinoic acid, which stimulates the stem cells to produce fully differentiated non-dividing neurons. The simple chemical achieves its dramatic effect on the cells by stimulating a set of genes that is used only by neurons. Simultaneously, the retinoic acid suppresses other genes that instruct cells to differentiate into other tissues. Once differentiated and no longer dividing, the transplanted cells were considered safe for the patient, i.e. noncancerous. In rats given similar transplants, the grafts survived for more than a year without showing any sign of reverting back to their cancerous state. The patients that were treated with the nerve cell transplants are still being monitored. Similar studies on rats suffering from an perimentally produced stroke showed that grafts of differentiated teratocarcinoma cells resulted in significant recovery in the brain. Injection of millions of cells into the brain reversed damage and restored lost motor activity and cognitive function. Injection of smaller numbers of cells resulted in improvement that lasted only a month, but by increasing the cell number and treating the animals with cyclosporin to lower the risk of graft rejection by immune cells, long term recovery was observed. The potential clinical applications of successfully culturing human embryonic stem cells are broad. One immediate application of pluripotent cells that can grow indefinitely and differentiate into human cell types is drug screening. This may enable the screening of drugs on fully mature human cell lines to test for drug toxicity on different tissue types. Because cultured human embryonic cells resemble cells in developing human embryos, they could be used to identify drugs that may interfere with normal embryonic development during pregnancy. Large quantities of disease-free cells may be produced and used in transplants, bringing hope to patients with all kinds of ills. Cells of the pancreas that make insulin could be developed for treating diabetes, nerve cell implants for repairing damage in specific areas of the brain for treating Parkinson's, stroke, and patients with dementia including Alzheimer's disease. Cells could be differentiated into liver or kidney cells to repair diseased and damaged organs. Embryonic cells could be exposed to substances that stimulate them to produce the wide range of cells found in the blood, a possible alternative to bone marrow transplants. A challenge to cell transplantation is that the immune system tends to reject cells that are different and foreign to the recipient. In the case of the treatment of the stroke patient with injected nerve cells, cyclosporin, an immune suppressive chemical, was used to prevent rejection. Animal experiments suggest that embryonic cells could be modified to be accepted by the immune system and not recognized by the recipient's immune cells as foreign. In principle, this could be achieved by altering the embryonic cells genetically so they function as "universal donors," acceptable to all patients. Such strategies would involve the difficult course of altering genes and treating the cells with different drugs and different growth conditions to select the cells with the desirable altered genes. Alternatively, embryonic stem cells that are genetically identical to the patient's cells could be created, as described earlier in treating a heart muscle damaged from a severe heart attack. This strategy would require a step used in cloning. A technician would take cells, such as skin cells, from a patient and grow them in culture. Using a hollow glass called a pipette that is a tenth of the diameter of a human hair, the technician would transfer the nucleus of a cell that contains the cell's DNA, its genetic material, into an unfertilized human egg. The egg would be activated by an electric shock and begin to develop into an embryo. The only genetic information in this unfertilized egg would be from the DNA of the patient's cell. Once it develops into an early embryo in the blastocyst stage, the human embryonic stem cells would be cultured. The differentiated cells-genetically identical to the donor's- would be produced for therapy. The technique of nuclear transfer from an adult cell into an egg was the one used in cloning the sheep Dolly, but here the egg containing the genetic material of Dolly's mother was implanted into a uterus and allowed to develop into what was to become Dolly, rather than grown in a dish into an early embryo for the purpose of culturing the desired stem cells. Though the human embryonic stem cells are pluripotent and under different conditions could mature into different tissues, they cannot form an embryo. Research using human embryos has met with both public and governmental support and resistance. So far, the research that resulted in the two first reports on the culturing of human embryonic stem cells was supported by private funds from the Geron Corporation. A U.S. Congressional ban on cloning human embryos exists. However, earlier this year, the National Institutes of Health announced that embryo stem cell research has been exempted by the U.S. Department of Health and Human Services, but is limited to research on stem cells that have already been extracted from the embryo through non-governmental support. A ban on the extraction and culturing of cells from the embryo remains. The NIH explained the reason for the decision through the results of Thomson, who derived human stem cells from the inner cells of blastocysts from left-over fertility studies and treatments. Although the stem cells retain the ability to differentiate into many kinds of cells, they are unable to form an embryo if implanted into a womb. Thus the NIH statement says that "even if the cells are derived from a human embryo, they are not themselves a human embryo." To grow human stem cells in a dish, researches must first remove the outer layer of cells of the blastocyst used to culture the embryo. These outer cells, called trophoblasts, are essential for the development of the placenta, the tissue that nourishes the embryo and protects it in the womb from rejection by the mother's immune system. Thus, by removing this outer layer the researcher prevents the remaining inner cells from developing in the womb. The work of Gearhart-culturing stem cells from primordial germ cells, the tissues that give rise to ovaries and testes-was done using older embryos from terminated pregnancies. At the Senate hearing, witnesses who implored the Senate to exempt stem cell research from the federal ban on embryo research included a man diagnosed with Parkinson's disease at the age of 27, and a Harvard University professor who had a seven-year-old child with diabetes. Both diseases are among those that could benefit from stem cell research and cell therapy. The scientific and medical community cheered the decision of the Department of Health and Human Services allowing the research on isolated stem cells to proceed. However, a spokesman for the National Conference of Catholic Bishops protested, saying that the decision may provide an incentive for embryo destruction. As scientists gear up to pursue new avenues to study embryonic development and cure disease by stem cell therapy, the debate on embryonic cell research continues. References: Bonn, D First cell transplant aimed to reverse stroke damage. The Lancet 1998;352:119 Butler D. Breakthrough stirs US embryo debate. . . Nature 1998;396:104-105 Gearhart, J. New potential for human embryonic stem cells. Science 1998;282:1061-1062 Shamblot JM Derivation of pluripotent stem cells from cultured human primordial germ cells. Proc. Natl. Acad. Sci. 1998;95:13726-13731 Thomson JA et al. Embryonic stem cell lines derived from human blastocysts. Science 1998;282: 1145-1147 Vescovi AL et al. Exp Neurol. 1999;156:71-83 Wadman M. Embryonic stem-cell research exempt from ban, NIH is told. Nature 1999;397:185-186 |

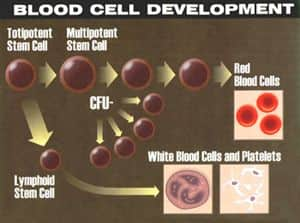 The simplest definition of a stem cell is that of an immature cell that can reproduce itself and produce daughter cells that are mature, differentiated cells. There are various types of stem cells-all divide but not all can function as special cells, including heart, kidney or nerve cells. Yet, when stem cells divide into replica daughter cells, under certain favorable conditions, some of these daughter cells go on to "differentiate." They then become committed to functioning as cells of the heart, kidney, brain and other tissues. The capacity to divide as undifferentiated cells, and produce one or more differentiated cell types, is the core feature of stem cells' function in the body. These functions are needed for the development of the embryo and for repairing the everyday wear and tear of the tissues throughout life.
The simplest definition of a stem cell is that of an immature cell that can reproduce itself and produce daughter cells that are mature, differentiated cells. There are various types of stem cells-all divide but not all can function as special cells, including heart, kidney or nerve cells. Yet, when stem cells divide into replica daughter cells, under certain favorable conditions, some of these daughter cells go on to "differentiate." They then become committed to functioning as cells of the heart, kidney, brain and other tissues. The capacity to divide as undifferentiated cells, and produce one or more differentiated cell types, is the core feature of stem cells' function in the body. These functions are needed for the development of the embryo and for repairing the everyday wear and tear of the tissues throughout life.