Life Extension Magazine®
What makes cells mortal? Research findings on cellular senescence may explain not only the life span potential of cells, but also cancer and human mortality.

may explain not only the life span potential
of cells, but also cancer and human mortality.
Most cells regenerate themselves by dividing to form a pair of new cells. In 1961, scientist L. Hayflick discovered that cells eventually reach a limit beyond which they cannot continue to divide (Hayflick L et al., 1961; Hayflick L, 1965). In a now-famous series of experiments, Hayflick demonstrated that cultured human fibroblasts (connective tissue cells) can divide only about 60 to 80 times. When a cell reaches this “Hayflick Limit” it enters into a twilight state called cellular senescence. Senescent cells are very much alive—yet they are distorted in both form and function.
Cultures of senescent cells cannot be mistaken for younger cells, which are uniform in appearance and line up in parallel arrays. By contrast, senescent cells exhibit a grainy appearance and take on odd shapes and sizes. They lose the ability to organize themselves in a regular pattern. These striking changes are called the senescent phenotype. A dipeptide (chemical union of two amino acids) called carnosine has been shown to rejuvenate cells displaying the senescent phenotype, quickly restoring the juvenile phenotype (McFarland GA et al., 1999; McFarland GA et al., 1994).
Senescent cells also behave in deviant ways. For example, senescent dermal (skin) cells generate more metalloproteinase enzymes that break down proteins in the surrounding extracellular matrix (the fabric that holds together cells, lymph nodes and blood vessels). They also generate more of the proinflammatory cytokine (hormone-like proteins involved in cellular signaling) interleukin 1-a. Senescent endothelial cells, which line blood vessel walls, generate higher levels of an adhesion molecule that contributes to atherosclerosis. By secreting damaging molecules and cytokines, senescent cells can disrupt the surrounding tissue microenvironment. Relatively few cells could in this way exert far-reaching deleterious effects upon tissue integrity and organ function (Campisi J, 1997).
Is cellular senescence then tantamount to aging? There are several lines of evidence supporting this conclusion. Cells from older people senesce after only a fraction of the cell divisions that fetal cells can undergo. Cells from short-lived animal species senesce faster than cells from long-lived species. Cells from people with genetic premature aging syndromes senesce prematurely, suggesting that the same genes regulate life span in the cell and the organism. Finally, senescent cells accumulate with age in organs and tissues, where they resist programmed cell death (apoptosis) and contribute to age-related degeneration (Campisi J, 1997).
The cancer connection
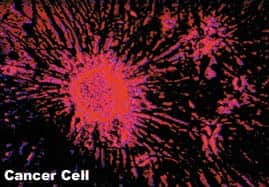
There is another direct connection between cellular senescence, aging and mortality. Surprisingly, cellular senescence appears to be controlled by tumor suppressor genes, including p53 and Rb (Bringold F et al., 2000; Campisi J, 2000, 1997). Most tumors contain cells that continue to divide beyond normal limits or indefinitely. Tumor suppressor genes are thought to act in part by inducing cellular senescence, which puts a halt to cell division. This has led scientists to the intriguing hypothesis that cellular senescence evolved as a defense against cancer. In support of this theory, recent research shows that cells can respond to carcinogenic stimuli such as DNA damage and the activation of cancer-promoting genes by entering a senescent state.
they tend to cross-link and aggregate
(as, for example, in cataracts or senile plaques).
Rapid effective proteolysis is therefore an
anti-aging mechanism.
The double-edged sword of cellular senescence thereby consigns cells to mortality in order to protect them against cancer. Ironically, cellular senescence alters the microenvironment around the cell in two ways that are thought to contribute to both aging and carcinogenesis. First, senescent cells may impair the structural integrity of the microenvironment, allowing a cell harboring an oncogenic mutation to proliferate. For example, the enzymes secreted by senescent dermal fibroblasts may be able to destroy the basement membrane and underlying stroma (the tissue framework for an organ) that keep potentially cancerous cells in check. Second, senescent cells overproduce growth factors and cytokines that could stimulate the growth of precancerous cells. These derangements of the structure and function of the cellular microenvironment could synergize with accumulating mutations to favor the early stages of tumorigenesis (Campisi J, 2000; Campisi J, 1997).
In addition, disturbances in cell cycle control due to inefficient protein removal can set the stage for cancer, as we shall see below.
The protein life cycle
The body is made up largely of proteins. The health of the body's stock of proteins depends upon its freedom from damage (as through oxidation or cross-linking), and upon its timely removal as part of normal protein turnover.
The body's antioxidant system and other lines of defense cannot completely protect proteins. Nature's second line of defense is the body's system for repairing or removing damaged proteins. While some protein repair mechanisms exist, there are no known ways to repair most protein damage, including even simple oxidative damage to the amino acids which are the building blocks of proteins. Thus it is essential for the body to efficiently remove aberrant and unneeded proteins, a process called proteolysis.
Timely proteolysis removes damaged proteins before they do significant harm, and removes undamaged proteins before they become damaged or disruptive. For example, if oxidized proteins are not broken down, they tend to cross-link and aggregate (as, for example, in cataracts or senile plaques). Rapid effective proteolysis is therefore an anti-aging mechanism (Grune T et al., 1997).
The main proteolytic enzyme complex is called the proteasome. It removes proteins that have been tagged for degradation by a peptide called ubiquitin. Through its role in protein disposal, the proteasome-ubiquitin pathway helps regulate many basic cellular processes including the cell cycle and cell division, cell differentiation, cellular signaling, cellular metabolism and DNA repair (Ciechanover A, 1998). Thus a malfunctioning proteasomal system has far-reaching consequences.
As cells age, after many cell divisions, proteasome activity declines (Sitte N et al., 2000; Merker K et al., 2000). At the same time, more and more proteins undergo damage through a process called carbonylation. Thus the proteolytic system becomes increasingly inadequate to deal with the increasing numbers of abnormal or unneeded proteins, which can irreversibly form cross-links and turn cellular processes awry.
New research shows that when the population of carbonylated proteins permanently increases—as in aging—proteasome activity is depressed (Petropoulos I et al., 2000; Keller JN et al., 2000; Burcham PC et al., 1997). A vicious circle develops of age-related decline in proteasomal activity, age-related increase in protein carbonylation and further inhibition of the proteasome. The life cycles of proteins become blocked, and the normal turnover of protein declines.
Is there a way to block this vicious circle? The body contains a dipeptide called carnosine that both protects proteins from carbonylation and helps reverse proteasomal decline. As in the aging body, proteolysis declines in cultured cells as they approach senescence. Australian scientists showed that carnosine enhances intracellular proteolytic activity in human connective tissue cells (Hipkiss AR et al., 1995). Carnosine enhanced proteolysis the most in old cells, and to a lesser extent in “middle aged” cells, compensating for age-related proteolytic decline (for details see “Carnosine—Nature's pluripotent life extension agent” from this issue).
The cell cycle
The concept of protein removal brings to mind structural proteins such as collagen that the body breaks down after a relatively long life. In order to understand the implications of proteolytic decline and buildup of aberrant proteins, it is necessary to revise this picture.
Think instead of a highly dynamic population of diverse proteins playing critical roles in the body's regulatory and signaling pathways. These proteins must be selectively synthesized at just the right moment so that they can do their precisely timed jobs, then they must be swiftly degraded at the correct point in a tightly regulated sequence of events. Normally such processes run like clockwork, but when damaged proteins accumulate the system can bog down.
It is in this way that physiological processes—both fast and slow—could become deranged by excessive buildup of proteins marked for removal by the proteasome-ubiquitin system. A case in point is the cell cycle. It consists of four phases culminating in mitosis (cell division). The key steps in this cycle are controlled by proteasomal degradation of proteins. For example, entry into the DNA synthesis phase, separation of sister chromatids, and the exit from mitosis are all dependent upon the timely removal of proteins such as cyclins by the proteasome (Hershko A, 1997; Ford HL et al., 1999).
By inhibiting the proteasome, carbonylated proteins could interfere with cell cycle progression and control. To understand how this can happen, consider an engine whose oil isn't changed regularly. When the detergent in the oil is used up, contaminants precipitate and sludge forms on vital engine parts. The sludge accumulates, impairing engine performance, until finally the engine dies.
The body too needs an efficient sludge removal system. When protein “sludge” accumulates, the gears of the cell cycle can get clogged up. This could impair the efficiency of cell division, and perhaps more importantly, enable damaged cells to reproduce. The result is increasing chromosomal instability, leading to degeneration and cancer (Schmutte C et al., 1999). Another possible outcome is cellular senescence, when the cell cycle grinds to a halt. Protein carbonylation thus becomes a potentially terminal condition.
Cell cycle control represents one more pathway along which damage to, and inefficient removal of protein could contribute to both cellular mortality and the cellular immortalization seen in cancer. In this scenario, the buildup of carbonylated protein feeds a vicious circle of proteasomal impairment, chromosomal instability, and increasing numbers of defective and senescent cells, which the body cannot remove. Insofar as the cellular life cycle is bound up with the life cycles of proteins, it behooves us to maintain healthy intact proteins and to ensure their timely turnover.
Skin aging
changes only subtly with age,
profound changes take place in the
dermis (inner skin layer).
The aging processes discussed above—cellular senescence, protein carbonylation and proteasomal decline—play leading roles in the changes that aging brings to the skin. While the epidermis (outer skin layer) changes only subtly with age, profound changes take place in the dermis (inner skin layer). There, the population of fibroblasts (connective tissue cells) is cut in half by age 80. Collagen becomes disorganized with broken fibers, while the extracellular matrix shows widespread destruction (West MD, 1994).
Protein carbonylation damages all components of the epidermis and dermis, leading to loss of elasticity, wrinkles, macromolecular disorganization, loss of extracellular matrix and reduced capacity for wound repair—all of which are primary characteristics of aged skin. Protein carbonylation rises with age in the epidermis and in cultured keratinocytes (dividing cells that migrate into the epidermis). As elsewhere in the body, it results from protein oxidation, glycation (protein-sugar reactions) and reactions with lipid peroxidation products (Petropoulos I et al., 2000).
Collagen, the protein substance of connective tissue, tends to cross-link with age. It is well known that collagen is cross-linked in the course of glycation and the consequent formation of advanced glycation end products (AGEs). This robs the skin of elasticity and youthful tone. Recent laboratory research demonstrates that this problem can be self perpetuating. Once AGEs form, they can directly induce the cross-linking of collagen—even in the absence of glucose and oxidation reactions (Sajithlal GB et al., 1998). The researchers found that neither antioxidants nor metal chelators could inhibit direct cross-linking of collagen by AGEs. Only an anti-glycating agent, in this case the drug aminoguanidine, could inhibit this process. The natural dipeptide carnosine offers a superior efficacy and toxicity profile compared to aminoguanidine (Munch G et al., 1997; Preston JE et al., 1998; Burcham PC, 2000).
The researchers propose that reactive carbonyl compounds have the ability to induce collagen cross-linking regardless of oxidative conditions. Their findings underline the importance of preventing protein carbonylation and in particular glycation before the cycle of collagen cross-linking gets started.
Proteasomal decline in Alzheimer's disease
The brain depends upon the proteasome for timely disposal of proteins. When the proteasome is inhibited, oxidized and aggregated proteins accumulate, and neurons degenerate and die. A bottleneck in protein removal could shift the balance toward the accumulation of deposits characteristic of Alzheimer's disease.
A new study at the University of Kentucky provides the first direct evidence of reduced proteasome activity in a neurodegenerative disorder. The scientists compared proteasome activity in five brain regions of normal and Alzheimer's disease brains, using specimens removed during autopsies. They found the activity of the proteasome significantly reduced in three brain regions showing severe degeneration in Alzheimer's disease. By contrast, proteasome activity was not reduced in two brain regions showing less or no degenerative change in Alzheimer's disease (Keller JN et al., 2000).
New research from France suggests a mechanism by which proteasomal impairment could in turn increase production of the amyloid-beta material that makes up senile plaques. Proteins called presenilins influence the production of amyloid-beta from its “parent” protein called amyloid precursor protein, or APP. Mutations in the presenilin genes that lead to early onset Alzheimer's disease upset the balance between production of amyloid-beta and of a neuroprotective derivative of APP called secreted APP. These mutations especially favor production of a long form of amyloid-beta that more readily collects into aggregates and eventually plaques.
There is considerable evidence that presenilin protein concentrations are regulated by the proteasome. The French research shows that inhibition of the proteasome increases production of the long aggregable form of amyloid-beta. Amyloid-beta in turn inhibits the proteasome. The researchers propose that proteasome activators could reverse this imbalance, potentially in sporadic as well as genetic forms of Alzheimer's disease (Checler F et al., 2000; Marambaud P et al., 1998).
The dynamic fibroblast
Connective tissue cells, called fibroblasts, play the leading role in the ongoing regeneration of the dermis. In order to function properly, fibroblasts must strike a delicate balance between destruction of extracellular protein and synthesis of new protein.
Normally fibroblasts are quiescent, dividing at a low rate. They produce only small amounts of the matrix metalloproteinase enzymes (collagenase and stromelysin) that break down the surrounding extracellular matrix, and large amounts of matrix metalloproteinase inhibitors (TIMP-1 and TIMP-2). But in response to various stimuli including wounding and inflammation, they undergo a drastic transformation into activated fibroblasts. They then secrete large amounts of enzymes that break down collagen and destroy the extracellular matrix.

activity declines in keratinocytes and
epidermal cells with age.
Cellular senescence locks fibroblasts and keratinocytes into an approximation of this activated state (West MD, 1994). They switch from a matrix-producing to a matrix-degrading mode, secreting more matrix metalloproteinases and less matrix metalloproteinase inhibitors. Senescent fibroblasts and keratinocytes are known to accumulate in aging skin, as demonstrated by a biomarker of skin cell senescence (Dimri GP et al., 1995). In addition to breaking down the extracellular matrix, they secrete proinflammatory mediators such as interleukin-1 alpha and growth factors such as heregulin (regulator of breast and epithelial cell growth) whose influence extends far beyond the cell secreting them (Campisi J, 1998; Campisi J, 1997).
Proteolysis of connective tissue is a normal part of skin cell development and wound healing. Proteolytic enzymes and their inhibitors sculpt structural proteins and break them down at the appropriate times. Unfortunately, as aging skin cells senesce and increase their proteolytic activity, the proteasome (the main enzyme complex for protein degradation) enters an age-related decline. The balance between protein creation and destruction is again upset, compromising the integrity and regeneration of skin tissue.
New research confirms that proteasome activity declines in keratinocytes and epidermal cells with age. At the same time, protein carbonyl levels are rising and the increasing numbers of senescent cells are secreting more proteolytic enzymes. This has been demonstrated clearly in keratinocytes, both in culture and in specimens from humans, where there is an inverse relationship between proteasome content and biomarkers of cellular senescence (Petropoulos I et al., 2000).
The skin makes visible the changes that occur throughout the body as damaged proteins and senescent cells accumulate. As we have seen, the life cycles of cells and proteins may regulate both our appearance as we age and how long we live. Preserving the integrity and regular turnover of protein is thus a key defense against the downward spirals of degeneration in the later years. Carnosine is the only agent that has shown multi-modal protective effects against protein degradation and cellular senescence.
References
Bringold F, Serrano M. Tumor suppressors and oncogenes in cellular senescence. Exp Gerontol. 2000; 35(3):317-29.
Burcham PC, Kerr PG, Fontaine F. The antihypertensive hydralazine is an efficient scavenger of acrolein. Redox Rep. 2000; 5(1):47-9.
Burcham PC, Kuhan YT. Diminished susceptibility to proteolysis after protein modification by the lipid peroxidation product malondialdehyde: inhibitory role for crosslinked and noncrosslinked adducted proteins. Arch Biochem Biophys. 1997; 340(2):331-7.
Campisi J. Aging and cancer: the double-edged sword of replicative senescence. J Am Geriatr Soc. 1997; 45(4):482-8.
Campisi J. The role of cellular senescence in skin aging. J Investig Dermatol Symp Proc. 1998; 3(1):1-5.
Campisi J. Cancer, aging and cellular senescence. In Vivo. 2000; 14(1):183-8.
Checler F, da Costa CA, Ancolio K, et al. Role of the proteasome in Alzheimer's disease. Biochim Biophys Acta. 2000; 1502(1):133-8.
Ciechanover A. The ubiquitin-proteasome pathway: on protein death and cell life. EMBO J. 1998; 17(24):7151-60.
Dimri GP, Lee X, Basile G, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA. 1995; 92(20):9363-7.
Ford HL, Pardee AB. Cancer and the cell cycle. J Cell Biochem. 1999; Suppl 32-33:166-72.
Grune T, Reinheckel T, Davies KJ. Degradation of oxidized proteins in mammalian cells. FASEB J. 1997; 11(7):526-34.
Hayflick L. The limited in vitro lifetime of human diploid cell strains. Exp Cell Res. 1965; 37:614-36.
Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961; 25:585-621.
Hershko A. Roles of ubiquitin-mediated proteolysis in cell cycle control. Curr Opin Cell Biol. 1997; 9(6):788-99.
Hipkiss AR, Michaelis J, Syrris P, et al. Strategies for the extension of human life span. Perspect Hum Biol. 1995; 1:59-70.
Keller JN, Hanni KB, Markesbery WR. Impaired proteasome function in Alzheimer's disease. J Neurochem. 2000; 75:436-9.
McFarland GA, Holliday R. Retardation of the senescence of cultured human diploid fibroblasts by carnosine. Exp Cell Res. 1994; 212(2):167-75.
McFarland GA, Holliday R. Further evidence for the rejuvenating effects of the dipeptide L-carnosine on cultured human diploid fibroblasts. Exp Gerontol. 1999; 34(1):35-45.
Marambaud P, Alves da Costa C, Ancolio K, et al. Alzheimer's disease-linked mutation of presenilin 2 (N141I-PS2) drastically lowers APPalpha secretion: control by the proteasome. Biochem Biophys Res Commun. 1998; 252(1):134-8.
Merker K, Sitte N, Grune T. Hydrogen peroxide-mediated protein oxidation in young and old human MRC-5 fibroblasts. Arch Biochem Biophys. 2000; 375(1):50-4.
Munch G, Mayer S, Michaelis J, et al. Influence of advanced glycation end-products and AGE-inhibitors on nucleation-dependent polymerization of beta-amyloid peptide. Biochim Biophys Acta. 1997; 1360(1):17-29.
Petropoulos I, Conconi M, Wang X, et al. Increase of oxidatively modified protein is associated with a decrease of proteasome activity and content in aging epidermal cells. J Gerontol A Biol Sci Med Sci. 2000; 55(5):B220-7.
Preston JE, Hipkiss AR, Himsworth DT, et al. Toxic effects of beta-amyloid(25-35) on immortalised rat brain endothelial cell: protection by carnosine, homocarnosine and beta-alanine. Neurosci Lett. 1998; 242(2):105-8.
Sajithlal GB, Chithra P, Chandrakasan G. Advanced glycation end products induce crosslinking of collagen in vitro. Biochim Biophys Acta. 1998; 1407(3):215-24.
Schmutte C, Fishel R. Genomic instability: first step to carcinogenesis. Anticancer Res. 1999; 19(6A):4665-96.
Sitte N, Merker K, von Zglinicki T, et al. Protein oxidation and degradation during proliferative senescence of human MRC-5 fibroblasts. Free Radic Biol Med. 2000; 28(5):701-8.
West MD. The cellular and molecular biology of skin aging. Arch Dermatol. 1994; 130(1):87-95.
