Life Extension Magazine®
Almost everyone knows someone who has diabetes. An estimated 18.2 million Americans and 2 million Canadians, over 6% of each nation’s population, have been diagnosed with diabetes.1,2 Millions more have the disease and are unaware of it.
The World Health Organization estimates that by 2025, the number of people with diabetes will reach 300 million—a staggering 122% increase in less than 30 years.3 At the dawn of the third millennium and 83 years after the discovery of insulin, “we are still grappling with the enormous complexity of a disease process in which almost every aspect of the body’s metabolism goes awry.”4
Diabetes is now recognized as the sixth leading cause of death and disability in the US. Its associated health care costs have spiked dramatically, more than doubling from $44 billion to $92 billion from 1997 to 2002. In Canada, diabetes treatment now consumes 15% of the national health care budget. Today, diabetes accounts for 28% of all new cases of kidney disease in Canada, and is a primary cause of adult blindness, non-trauma-related limb amputations, and major cardiovascular events.5
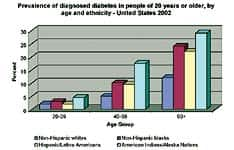 |
| Source: National Diabetes Clearinghouse. |
The disease exhibits a strong ethnic bias: Native Americans, Hispanic-Latino Americans, and African-Americans show an increased prevalence compared to the non-Hispanic white population. Unknown in Canada’s aboriginal community until recently, type II diabetes has undergone exponential growth. Within the next two decades, 27% of Canada’s First Nations (native) peoples are expected to develop the disease.5
Epidemiological data from elsewhere mirror the North American situation. Within the next 30 years, diabetes is projected to soar by almost 50% in Great Britain, 72-78% in New Zealand and Australia, and a staggering 184% in Mexico.6
Diabetes is often misunderstood as a simple sugar imbalance that can be readily corrected through medical intervention. In truth, it is a complex metabolic disorder in which a confluence of social, behavioral, dietary, and lifestyle factors unmask an underlying genetic susceptibility. The disease has serious implications for vision, cardiovascular health, and kidney and neural functions. Its expression largely depends on lifestyle issues, including diet, weight management, and physical exercise—a multifaceted combination of factors that makes treatment complex and
challenging.
The Driver: Type II Diabetes
The predominant form of diabetes is type II or non-insulin-dependent diabetes, a disease highly correlated with family history, physical inactivity, obesity, and ethnicity. Traditionally associated with middle age, the incidence of type II diabetes among younger adults and children is rising dramatically. More common in women, especially those with a history of gestational (pregnancy-related) diabetes, its prevalence in America has tripled in the last 30 years. While increasing across all age and ethnic groups, its ascendance among children and adolescents is most worrisome.
Over 90% of diabetics are type II, and it is the rapid increase in this form of the disease that is propelling the global increase in all diabetes cases. While diabetes exhibits a strong hereditary component, its rate of increase is too great to be a consequence of increased gene frequency. Instead, evidence points toward the combined influences of lifestyle, dietary, and environmental factors.7
An Emerging Epidemic: Diabetes and Children
Virtually unknown in children until recently, type II diabetes is now appearing with alarming regularity in overweight and sedentary young people. During the last three decades, the number of overweight children in the US has more than doubled. With this has come a dramatic rise in type II diabetes. In major US urban centers, the percentage of children with newly diagnosed type II diabetes has ballooned from less than 5% in 1994 to 30-50% in recent years.8
Obesity is a hallmark of the disease. A predisposition toward visceral obesity (deposition of abdominal fat) is associated with increased insulin resistance and contributes to its early onset.9 This may explain why 85% of American children who develop type II diabetes are overweight or obese at the time of diagnosis. According to Dr. Arlan Rosenbloom, chair of the American Diabetes Association Consensus Panel, “Type II diabetes in children is an emerging epidemic.”
Insulin Resistance: the Silent Stalker
Syndrome X is the dark force behind type II diabetes. Also known as metabolic syndrome, it is a preclinical stage of the disease, believed to affect up to 25% of North American adults. A constellation of metabolic changes that progress silently over a period of years, Syndrome X is characterized by increased resistance to insulin, the regulatory hormone that suppresses hepatic (liver) glucose output and removes excess glucose from the blood.
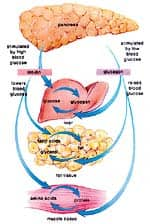 |
| Diagram of the homeostatic (insulin-glucagon) system. View enlarged image |
The onset of insulin resistance is characterized by a host of related symptoms, including:
- hypertension (high blood pressure)
- elevated blood triglycerides (fats)
- elevated LDL (“bad”) cholesterol
- reduced HDL (“good”) cholesterol
- accelerated hardening of the arteries
- proliferation of cells in the arterial walls
- development of abdominal obesity
- glycosylation (cross-linking of fats and proteins with glucose)
- hyperinsulinemia (high-blood insulin levels).
The syndrome is particularly alarming in children and adolescents because the changes, which in adults are usually spread over a number of years, are compressed into a few short years in young teens.10 The longer a person has the disease, the greater the likelihood of developing long-term disabilities. Unfortunately, physicians are now seeing more young people prematurely develop these life-threatening complications.
The prognosis is not encouraging: approximately one-third to one-half of those diagnosed with insulin resistance will develop diabetes. Of those, two-thirds will eventually die of cardiovascular complications. Compared to non-diabetics, adult diabetics are almost twice as likely to have asthma, three times more likely to have hypertension and heart disease, and four times more likely to suffer a stroke.11
Clearly, early detection of the disease is paramount.
A Delicate Balance
Insulin is produced by highly specialized beta cells in the pancreas, a lobular gland located behind the stomach. The hormone is secreted into the blood in response to elevated blood-sugar levels. Insulin helps the body utilize blood sugar by binding to specialized receptor sites on the cellular surfaces, much like a key fits into a lock. Insulin “unlocks” the receptor and glucose (a molecule too large to pass through) is transported into the cell. Once inside, glucose is used for cellular fuel, and any excess is stored as glycogen (a form of animal starch) or converted to glycerol for the formation of fat.
When blood sugar levels are low, or during times of stress, liver glycogen stores are quickly converted back to glucose by the action of glucagon, another pancreatic hormone. Glucagon also encourages the breakdown of fat in adipose tissue to glycerol and fatty acids. The liver reconverts these to glucose, releasing it into the blood. Through this intricate balancing act, insulin and glucagon perform a central role in regulating the body’s blood-sugar level.
Rising insulin resistance disrupts this balance when the normal levels of insulin no longer unlock the cellular “doors.” The beta cells of the pancreas, in a futile attempt to restore homeostasis, shift into “overdrive” and begin pumping out ever-increasing amounts of insulin. Chronically high levels of insulin further “desensitize” the cellular receptors, leading to even greater insulin resistance. A vicious and damaging cycle ensues—the genesis of type II
diabetes.
The Mechanism of Onset
What defies explanation is what causes insulin resistance in the first place. We know that virtually everyone who develops type II diabetes starts with insulin resistance. We also know that the risk of developing type II diabetes is strongly correlated with central body obesity and the ectopic (abnormal) accumulation of fat. It appears that insulin resistance may not be determined so much by the amount of body fat as by where the fat is located.4 In fact, one of the best predictive markers for insulin resistance is excess body weight, in particular weight around the waist.
McGarry presents a compelling case that the ectopic accumulation of fat in muscle and other peripheral tissues is intimately involved with the onset of insulin resistance and the gradual collapse of pancreatic beta cell function.4 Insulin sensitivity can fall dramatically without the appearance of diabetes, as long as the pancreatic beta cells compensate. It is their inevitable demise (possibly due to lipotoxicity) that leads to increased hyperglycemia, a further rise in blood lipids, and an ever-greater accumulation of fat in the muscle cell. Increasing hyperlipidemia (high blood lipids) in turn exacerbates insulin resistance and degrades liver function.
| According to McGarry, in the progression from being overweight to being obese, the liver becomes resistant to insulin and the hormone’s ability to suppress hepatic (liver) glucose production. “Under these conditions, the hyperinsulinemia turns the liver into a ‘fat-producing factory’ with all of its negative downstream effects.”4 Once established, these disturbances in lipid metabolism are devastating to blood sugar balance and weight management. Accel-eration of the cycle and eventual collapse of pancreatic insulin production herald the appearance of diabetes. The answer to how these metabolic derangements begin may lie in the “thrifty gene hypothesis,” which postulates the presence of a genetic factor designed to promote extra fat deposition.12 Speculation also suggests a defect in the leptin-signaling system, a metabolic pathway that appears to mediate fatty acid metabolism in muscle tissue and acts as a kind of “fuel gauge” to monitor cellular energy status.13 (Leptin, a chemical messenger produced by the adipose, or fat-storing, cells of the body, initiates the breakdown and oxidation of stored fat.)14 Still other research points to the possible development of functional resistance to the actions of the leptin hormone itself.15,16 Whatever the mechanism, a diminished capacity to oxidize fat appears to be a pre-eminent clinical marker for insulin resistance.17-19 This is supported by animal studies, which confirm that dietary lowering of muscle triglycerides improves insulin sensitivity and reverses diabetes.20,21 The fact that two of the most effective preventive programs for diabetes are diet and exercise lends credence to the argument that onset of the disease involves a profound disturbance in lipid dynamics. Assessing Your Risk for Diabetes The new definition, developed recently by the US National Cholesterol Education Panel, incorporates five easily measured variables:
Under the new definition, a person with any three of these conditions is classified as having insulin resistance syndrome. To assess whether the new criteria could predict excess risk for heart disease and diabetes, researchers found that the risk of coronary heart disease—and, more strikingly, diabetes—rose as the number of metabolic abnormalities increased. Men with four to five features of the syndrome had almost four times the risk of coronary heart disease and 25 times the risk of diabetes compared to those with no abnormalities. The study also confirmed that C-reactive protein, an inflammatory marker, was significantly elevated in men with metabolic syndrome compared to those without metabolic syndrome.22 Current guidelines suggest that people are diabetic if their fasting glucose levels exceed 126 mg/dL (7.0 mmol/L). Levels over 109 mg/dL indicate a prediabetic state and levels below 109 are considered normal. While significantly improved from the previously used glucose tolerance test, these recommendations may still be too lenient. In October 2003, the American Diabetes Association further reduced the cut-off for impaired glucose tolerance from 109 to 100 mg/dL, meaning anyone with fasting glucose levels above 100 would be classified as prediabetic. More recently, the Life Extension Foundation has prescribed optimal fasting glucose levels of less than 86 mg/dL.23 This recommendation is based on clinical evidence that higher fasting levels can progressively and markedly increase cardiovascular risk.24 Lowering the bar on fasting glucose levels is a good thing, as it is estimated that up to one-half of diabetes sufferers have not been diagnosed.25 Screening for diabetes should begin at 45 years of age and should be repeated every three years in persons without risk factors and more frequently in individuals with risk factors.26 The Life Extension Foundation believes that high fasting blood levels of insulin (hyperinsulinemia) is the first indication of insulin resistance syndrome. Lifestyle Considerations The Obesity Connection A recent study by researchers at the Centers for Disease Control and Prevention investigated the links between obesity, diabetes, high blood pressure, high cholesterol, asthma, and arthritis. Compared to adults of a healthy weight, obese adults had twice the risk for high cholesterol, three times the risk for asthma, four times the risk for arthritis, and over six times the risk for hypertension.28 The strongest correlation was between obesity and diabetes: obese people exhibited over seven times the risk for diabetes compared with people of normal weight. The good news is that up to 90% of type II diabetes cases can be prevented with simple lifestyle changes, including diet, exercise, and smoking cessation.29 The Diabetes Prevention Program, a multi-center trial involving over 3,200 people with impaired glucose tolerance, was the first large-scale study to demonstrate conclusively that weight loss can effectively delay type II diabetes. Results showed that lifestyle intervention, consisting of calorie reduction and 30 minutes a day of mild exercise, reduced risk by 58%—almost double that conferred by the oral diabetes drug metformin (Glucophage®). The results were so convincing that the study was concluded earlier than planned. The authors surmise that up to 10 million Americans can sharply lower their risk of diabetes through simple attention to diet, exercise, and lifestyle modification.30 These findings are supported by the earlier work of Dr. Roy Walford, who demonstrated that caloric restriction aggressively lowers both fasting glucose and blood insulin levels.23 Growing evidence suggests that C-reactive protein may also play a role in the development of central body obesity and the onset of type II diabetes.31 Abdominal fat is a major source of this inflammatory agent, and the increased risk of atherosclerosis and insulin resistance associated with visceral obesity may well be a consequence of enhanced C-reactive protein secretion. The surest and safest way to remedy the situation is to lose weight. Women who completed a 12-week restricted-calorie diet lost an average of 17.4 pounds and reduced their C-reactive protein levels by 26%.32 One thing is certain: people who are overweight are already in a prediabetic state and need to take corrective action before the damage is done. Unfortunately, most diagnoses occur far too late in the game. The Value of Diet Stanford’s Dr. Gerald Reaven suggests that a diet consisting of 45% carbohydrates, 40% “good” fats, and 15% protein will benefit individuals with insulin resistance. According to Reaven, only when healthy fats replace carbohydrates will insulin levels drop and clusters of symptoms associated with insulin resistance abate.34 Unfortunately, the standard diabetic diet recommended by most physicians is very high in carbohydrates, which raises blood sugar, stimulates insulin production, and almost guarantees that the diabetic will be a patient for life. People need to pay attention not only to their total carbohydrate load, but also to the types of carbohydrates they eat. High-glycemic foods—such as white rice, white flour-based products, pasta, starchy vegetables, and many processed foods—are quickly converted to blood sugar when digested, causing insulin levels to spike. Conversely, the carbohydrates found in low-glycemic foods, such as asparagus, broccoli, cabbage, green beans, and other low-starch vegetables and fruits, are converted slowly to blood sugar and create a more gradual rise in blood insulin levels. Avoiding “white foods” is a simple recipe that can help you avoid trouble. Fats and Fiber If nothing else, simply replacing trans-fats in the diet with polyunsaturated fats will reduce the risk of diabetes dramatically. Dietary fats that are considered to be beneficial include extra virgin olive oil, fish oil, almond oil and almond butter, avocados, nuts, and seed oils such as sesame, pumpkin, sunflower, and flax. Eating a diet rich in soluble and insoluble fiber improves insulin sensitivity and reduces circulating insulin levels. Fiber impedes gastric emptying and the passage of food through the gut, slows the breakdown of high-glycemic starchy foods, and delays glucose uptake into the blood. In a recent study reported in the New England Journal of Medicine, researchers conclude that a high-fiber diet significantly improves glycemic control, decreases insulin levels, and lowers plasma lipid concentrations in as little as six weeks.36 Exercise Is Essential Physical exercise burns calories, and as energy expenditure is increased, the incidence of diabetes is found to decrease. Moreover, this protective effect appears to be most pronounced in individuals who are at greatest risk for developing the disease.38 Physically fit people also secrete less insulin. Results from the Nurses’ and Physicians’ Health Studies, conducted in the early 1990s, reveal that insulin response is more attenuated in physically fit individuals than in people who are less fit.39,40 Researchers recently demonstrated that when individuals are introduced to a regular exercise program, they experience a striking decrease in their risk of developing diabetes.41 In fact, regular exercise, when combined with weight loss, can reduce the insulin requirements of type II diabetics by up to 100%.42,43 A low-glycemic, reduced-calorie diet with healthy fats and fiber, along with regular exercise, is a safe and effective means to prevent and treat diabetes, as well as to shed excess weight. |
| The Role of Supplements Alpha-lipoic acid enhances the breakdown of glucose through intervention at several points along the central metabolic pathway. Supplementation provides a significant boost in glucose disposal and insulin sensitivity.44 Biotin assists in the metabolism of fats, proteins, and carbohydrates, and directly influences blood glucose levels by enhancing glucokinase activity.45 (Glucokinase is an enzyme that controls the first step of glucose oxidation.) Supplementation at high levels significantly improves glucose control and reduces the risk of diabetic neuropathy.46 L-carnitine deficiency is common in diabetics and is associated with cataract formation and cardiomyopathy (weakening of the heart muscle). Supplementation improves insulin sensitivity, in-creases glucose storage, and optimizes carbohydrate metabolism.47 Carnitine also appears to protect against diabetic neuropathy by lowering sorbitol levels in nerve cells. Carnosine, an amino acid peptide, prevents cross-linking of glucose with proteins (glycation) and arrests the formation of advanced glycation end-products in the cell.48 Glycation accelerates the aging process and is problematic in diabetics. Carnosine also bolsters proteolytic (protein-destroying) pathways to dispose of damaged proteins created through glycation. Chromium is essential in modulating glucose metabolism and boosting glucose sensitivity.49 It enhances insulin-dependent transport of glucose into the cell, likely by facilitating the binding of insulin to the receptor site. Supplementation at 1000 micrograms per day has been found to markedly reduce insulin resistance.50 As over 90% of adults are deficient in chromium, daily supplementation is warranted. Coenzyme Q10, an important component of the cell’s central metabolic pathway, enhances cellular energy production and protects cells from damage by free radicals. Japanese researchers cite its ability to boost respiratory chain function in pancreatic beta cells and improve glycemic control.51 Magnesium lowers blood glucose, increases insulin sensitivity, reduces stress response (which promulgates diabetes), and assists in the maintenance of healthy beta cells.52 Low magnesium status is common among type II diabetics53 and is believed to disrupt insulin secretion and the hormone’s capacity to transport glucose. Supplementation has been found to increase the number and sensitivity of cellular insulin receptors50 and to mitigate carbohydrate intolerance.45 N-acetyl-L-cysteine protects pancreatic beta cells from oxidative damage. Free radicals flourish in fatty tissue and when circulating glucose levels are high (two conditions common in insulin resistance syndrome and diabetes). Supple-mentation with N-acetyl-L-cysteine lowers blood sugar levels, protects beta cells from glucose toxicity, and suppresses beta cell death.54 Vitamin C (ascorbic acid) lowers blood glucose, inhibits glycation,55 prevents the accumulation of sorbitol (which causes cataracts), and synergizes and replenishes other antioxidants. By reducing the level of C-reactive protein, ascorbic acid helps quench the inflammatory response in diabetics and lowers cardiovascular risk. Because vitamin C is the principal antagonist for the excessive free-radical activity observed in diabetes, its presence is critical.45 As most diabetics are deficient in this important nutrient, supplementation is essential. Vitamin E (alpha-tocopherol), a potent lipid-phase antioxidant, quenches lipid peroxidation, increases insulin sensitivity, and enhances glucose transport. High-dose vitamin E supplementation at 1200 milligrams per day has been found to reduce vascular inflammation and lower C-reactive protein levels.56 Vitamin K appears to play an important role in the regulation of blood sugar57 and the reduction of interleukin-6 (IL-6), an inflammatory marker for diabetes; however, people on anticoagulant drugs, such as Coumadin®, should not take vitamin K. Insulin resistance and diabetes can also be treated successfully with plant-based remedies indigenous to many native cultures. Several herbal remedies are remarkably effective and, unlike drug therapies, have few or no side effects. Hormone therapies, using testosterone and dehydroepiandrosterone (DHEA), show considerable promise. For a detailed description of these complementary therapies, readers are encouraged to consult the protocols in the Life Extension Foundation’s Disease Prevention and Treatment book,58 as well as Michael Murray’s excellent treatise in his book, Healing Power of Herbs.59 |
| References |
| 1. Diabetes Division, Bureau of Cardio- Respiratory Diseases and Diabetes, Laboratory Centre for Disease Control, Health Protection Branch. Diabetes in Canada: National Statistics and Opportunities for Improved Surveillance, Prevention, and Control. 1999. Health Canada. 2. Diabetes Overview. National Diabetes Information Clearinghouse (NDIC) web- site. November, 2003. Available at: http://diabetes.niddk.nih.gov/dm/pubs/overview/ index.htm. Accessed Jan 31, 2004. 3. Canadian Diabetes Association. About Diabetes: The Prevalence and Costs of Diabetes. Canadian Diabetes Association website. Available at: http://www.diabetes.ca/ Section_About/prevalence.asp. Accessed Jan 31, 2004. 4. McGarry JD. Banting lecture 2001: dysregu- lation of fatty acid metabolism in the etiolo- gy of type 2 diabetes. Diabetes. 2002 Jan;51(1):7-18. 5. Canadian Diabetes Association. Diabetes: impact of disease staggering. Globe and Mail. November 1, 2000: C-1. 6. World Health Organization. Total of People with Diabetes. WHO website. October 31, 2003. Available at: http://www.who.int/ncd/dia/ databases4.htm. Accessed February 2, 2004. 7. Rosenbloom A, Arslanian S, Brink S, et al. Type 2 diabetes in children and adolescents. Diabetes Care. 2000 Mar;23(3):381-9. 8. Rocchini AP. Childhood obesity and a dia- betes epidemic. N Engl J Med. 2002 Mar;346(11):854-5. 9. Widen E, Lehto M, Kanninen T, Walston J, Shuldiner AR, Groop LC. Association of a polymorphism in the beta 3-adrenergic- receptor gene with features of the insulin resistance syndrome in Finns. N Engl J Med. 1995 Aug;333(6):348-51. 10. Grundy SM, Howard B, Smith S Jr, Eckel R, Redberg R, Bonow RO. Prevention Conference VI: Diabetes and Cardiovascular Disease: executive summary: conference proceeding for healthcare pro- fessionals from a special writing group of the American Heart Association. Circulation. 2002 May;105(18):2231-9. 11. Stagnitti MN. Statistical Brief #34: The Prevalence of Obesity and Other Chronic Health Conditions among Diabetic Adults in the U.S. Community Population, 2001. Medical Expenditure Panel Survey. Agency for Healthcare Research and Quality, Department of Health and Human Services webpage. Available at: http://www.meps.ahrq.gov/PrintProducts/ PrintProd_Detail.asp. Accessed Jan 30, 2004. 12. Neel JV. Diabetes mellitus: a “thrifty” genotype rendered detrimental by “progress”? Am J Hum Genet. 1962 Dec;14:353-62. 13. Minokoshi Y, Kim YB, Peroni OD, et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature. 2002 Jan;415(6869):339-43. 14. Deus P. Leptin: The next big thing. Mind and Muscle Magazine [serial online]. Oct 26, 2001; Issue 3. Available at: http://mindand muscle.net/magazine/i3leptin.html. Accessed February 12, 2004. 15. Sinha MK, Caro JF. Clinical aspects of lep- tin. Vitam Horm. 1998;54:1-30. 16. Eiden S, Daniel C, Steinbrueck A, Schmidt I, Simon E. Salmon calcitonin—a potent inhibitor of food intake in states of impaired leptin signalling in laboratory rodents. J Physiol. 2002 Jun;541(Pt 3):1041-8. 17. Astrup A, Buemann B, Christensen NJ, Toubro S. Failure to increase lipid oxidation in response to increasing dietary fat content in formerly obese women. Am J Physiol. 1994 Apr;266(4 Pt 1):E592-E599. 18. Froidevaux F, Schutz Y, Christin L, Jequier E. Energy expenditure in obese women before and during weight loss, after refeed- ing, and in the weight-relapse period. Am J Clin Nutr. 1993 Jan;57(1):35-42. 19. Ravussin E, Lillioja S, Knowler WC, et al. Reduced rate of energy expenditure as a risk factor for body-weight gain. N Engl J Med. 1988 Feb;318(8):467-72. 20. Man ZW, Hirashima T, Mori S, Kawano K. Decrease in triglyceride accumulation in tis- sues by restricted diet and improvement of diabetes in Otsuka Long-Evans Tokushima fatty rats, a non-insulin-dependent diabetes model. Metabolism. 2000 Jan;49(1):108-14. 21. Ohneda M, Inman LR, Unger RH. Caloric restriction in obese pre-diabetic rats prevents beta-cell depletion, loss of beta-cell GLUT 2 and glucose incompetence. Diabetologia. 1995 Feb;38(2):173-9. 22. Sattar N, Gaw A, Scherbakova O, et al. Metabolic syndrome with and without C- reactive protein as a predictor of coronary heart disease and diabetes in the West of Scotland Coronary Prevention Study. Circulation. 2003 Jul;108(4):414-9. 23. Faloon W. What you don’t know about blood sugar. Life Extension Magazine. January 2004:11-20. 24. Bjornholt JV, Erikssen G, Aaser E, et al. Fasting blood glucose: an underestimated risk factor for cardiovascular death. Results from a 22-year follow-up of healthy nondia- betic men. Diabetes Care. 1999 Jan;22(1):45-49. 25. Harris MI, Hadden WC, Knowler WC, Bennett PH. Prevalence of diabetes and impaired glucose tolerance and plasma glu- cose levels in US population aged 20-74 yr. Diabetes. 1987 Apr;36(4):523-34. 26. Mayfield J. Diagnosis and classification of diabetes mellitus: new criteria. Am Fam Physician. 1998 Oct;58(6):1355-70. 27. Allison DB, Fontaine KR, Manson JE, Stevens J, VanItallie TB. Annual deaths attributable to obesity in the United States. JAMA. 1999 Oct;282(16):1530-8. 28. Mokdad AH, Ford ES, Bowman BA, et al. Prevalence of obesity, diabetes, and obesity- related health risk factors, 2001. JAMA. 2003 Jan;289(1):76-9. 29. Hu FB, Manson JE, Stampfer MJ, et al. Diet, lifestyle, and the risk of type 2 diabetes mel- litus in women. N Engl J Med. 2001 Sep;345(11):790-7. 30. Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or met- formin. N Engl J Med. 2002 Feb;346(6):393-403. 31. Lemieux I, Pascot A, Prud’homme D, et al. Elevated C-reactive protein: another compo- nent of the atherothrombotic profile of abdominal obesity. Arterioscler Thromb Vasc Biol. 2001 Jun;21(6):961-7. 32. Heilbronn LK, Noakes M, Clifton PM. Energy restriction and weight loss on very- low-fat diets reduce C-reactive protein con- centrations in obese, healthy women. Arterioscler Thromb Vasc Biol. 2001 Jun;21(6):968-70. 33. Challem J, Berkson B, Smith M. Syndrome X. New York: John Wiley & Sons; 2000. 34. Reaven GM. Syndrome X. New York: Simon and Schuster; 2000. 35. Salmeron J, Hu FB, Manson JE, et al. Dietary fat intake and risk of type 2 diabetes in women. Am J Clin Nutr. 2001 Jun;73(6):1019-26. 36. Chandalia M, Garg A, Lutjohann D, von Bergmann K, Grundy SM, Brinkley LJ. Beneficial effects of high dietary fiber intake in patients with type 2 diabetes mellitus. N Engl J Med. 2000 May;342(19):1392-8. 37. Greiger L. Syndrome X. Heart Information Network webpage. Available at: http://www.heartinfo.com/nutrition/ syndx072999.htm. Accessed Nov 15, 2000. 38. Helmrich SP, Ragland DR, Leung RW, Paffenbarger RS, Jr. Physical activity and reduced occurrence of non-insulin-depen- dent diabetes mellitus. N Engl J Med. 1991 Jul;325(3):147-52. 39. Manson JE, Rimm EB, Stampfer MJ, et al. Physical activity and incidence of non- insulin-dependent diabetes mellitus in women. Lancet. 1991 Sep;338(8770):774-778. 40. Manson JE, Nathan DM, Krolewski AS, Stampfer MJ, Willett WC, Hennekens CH. A prospective study of exercise and inci- dence of diabetes among US male physi- cians. JAMA. 1992 Jul;268(1):63-7. 41. Uusitupa M, Louheranta A, Lindstrom J, et al. The Finnish Diabetes Prevention Study. Br J Nutr. 2000 Mar;83(Suppl 1):S137-42. 42. Nieman DC. Fitness and Sports Medicine. 3rd ed. Palo Alto, CA: Bull Publishing; 1995. 43. Diabetes Type II and the Syndrome X Connection. Life Extension Foundation website. Available at: http://www.lifeextension.com/protocols/prtcls-text/t-prctl-042.html. Accessed Jan 27, 2004. 44. Jacob S, Streeper RS, Fogt DL, et al. The antioxidant alpha-lipoic acid enhances insulin-stimulated glucose metabolism in insulin-resistant rat skeletal muscle. Diabetes. 1996 Aug;45(8):1024-9. 45. Murray MT. Encyclopedia of Nutritional Supplements. Rocklin, CA: Prima Publishing; 1996. 46. Koutsikos D, Agroyannis B, Tzanatos- Exarchou H. Biotin for diabetic peripheral neuropathy. Biomed Pharmacother. 1990;44(10):511-4. 47. Crayhon R. The Carnitine Miracle. New York: M. Evans; 1999. 48. Hipkiss AR, Brownson C. A possible new role for the anti-ageing peptide carnosine. Cell Mol Life Sci. 2000 May;57(5):747-53. 49. McCarty MF. Toward a wholly nutritional therapy for type 2 diabetes. Med Hypotheses. 2000 Mar;54(3):483-7. 50. Diabetes Type II and the Syndrome X Connection. Life Extension Foundation website. Available at: http://www.lifeextension.com/ protocols/prtcls-text/t-prctl-042.html. Accessed Jan 27, 2004. 51. McCarty MF. Can correction of sub-optimal coenzyme Q status improve beta-cell func- tion in type II diabetics? Med Hypotheses. 1999 May;52(5):397-400. 52. Kowluru A, Chen HQ, Modrick LM, Stefanelli C. Activation of acetyl-CoA car- boxylase by a glutamate-and magnesium- sensitive protein phosphatase in the islet beta-cell. Diabetes. 2001 Jul;50(7):1580-7. 53. Paolisso G, Passariello N, Pizza G, et al. Dietary magnesium supplements improve B- cell response to glucose and arginine in elderly non-insulin dependent diabetic sub- jects. Acta Endocrinol (Copenh). 1989 Jul;121(1):16-20. 54. Kaneto H, Kajimoto Y, Miyagawa J, et al. Beneficial effects of antioxidants in diabetes: possible protection of pancreatic beta-cells against glucose toxicity. Diabetes. 1999 Dec;48(12):2398-2406. 55. Emekli N. Nonenzymatic glycosylation of tis- sue and blood proteins. J Marmara Univ Dent Fac. 1996 Sep;2(2-3):530-534. 56. Devaraj S. Vitamin E shows promise in treating diabetes. Washington, DC: Hearst Newspapers. June 5, 2001. Available at: http://www.ithyroid.com/diabetes.htm. Accessed January 27, 2004. 57. Sakamoto N, Nishiike T, Iguchi H, Sakamoto K. Relationship between acute insulin response and vitamin K intake in healthy young male volunteers. Diabetes Nutr Metab. 1999 Feb;12(1):37-41. 58. Segala M, ed. Disease Prevention and Treatment. 4th ed. Hollywood, FL: Life Extension Media; 2003. 59. Murray MT. Healing Power of Herbs. 2nd ed. Rocklin, CA: Prima Publishing; 1995. |
