Life Extension Magazine®
The public has awoken to the fact that high-glycemic carbohydrates promote weight gain by causing insulin to surge. In response to a wave of media publicity, “low-carb” diets have become enormously popular. Weight-loss books warning of insulin’s fat-inducing effects have sold millions of copies.
Anecdotal reports of people shedding huge amounts of body fat have caused dieters to avoid high-glycemic foods. Published scientific findings reveal that weight loss occurs in response to low-carb diets, but the average reduction in body fat has not been overly dramatic. Nevertheless, there are benefits to shedding (rather than gaining) fat pounds each year, along with enormous disease-preventing effects if excess insulin secretion is reduced.
This article examines the dangers of excess insulin, the role of insulin in promoting obesity, and a new dietary supplement that has been shown to reduce after-meal insulin release by 40%.
Why the Confusion Over Insulin?
Those afflicted with type I diabetes do not produce enough insulin. For these individuals, supplemental insulin is a lifesaving therapy. The public often associates insulin with healthy hormones, but insulin has an insidious dark side.
As normal aging and poor diet deprive cells of insulin sensitivity, many people produce excess insulin to force serum glucose into cells. This excessive insulin production is a contributing cause to a host of degenerative disorders, including heart disease and cancer.
The most immediate and noticeable effect of excess insulin production is unwanted weight gain. Insulin drives fat into cells, prevents fat from being released from cells, and makes people chronically hungry. High insulin levels contribute not only to obesity, but also to the disease states associated with being overweight.
While insulin saves the lives of type I diabetics who are dependent on it, it becomes a “death hormone” to aging people who secrete too much insulin. Reducing excess serum insulin is thus a critical component of any weight-reduction and longevity program.
What Is Hyperinsulinemia?
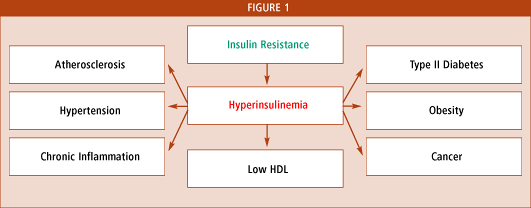
Insulin is produced by the beta cells in the pancreas. Insulin stimulates uptake of glucose (sugar) from the blood into the body’s cells. The resistance of the body’s cells to the action of insulin is called insulin resistance. As a result of insulin resistance and other factors, the pancreas produces more insulin than normal. This is called hyperinsulinemia.
In a normal person, one unit of insulin might be needed to help 10 mg of glucose go into a cell group. In hyperinsulinemia, ten units of insulin might be needed to get the same 10 mg of glucose into the cell group.1 Hyperinsulinemia and insulin resistance create myriad problems, including elevated triglycerides, low HDL, type II diabetes, and obesity.
Figure 1 below shows the pathways to obesity and several degenerative diseases associated with insulin resistance and hyperinsulinemia.
 |
Insulin and Age-Related Disease
Numerous published studies reveal that excess serum insulin (hyperinsulinemia) is a major health problem. High serum insulin promotes hypertension by impairing sodium balance.2,3 Too much insulin harms the kidneys.4 The vascular system is severely damaged by prolonged exposure to excess insulin.5,6 By acting as a catalyst in promoting cell growth, excess insulin increases the risk and progression of certain cancers.7-11 High insulin promotes the formation of beta-amyloid in brain cells and may contribute to the development of Alzheimer’s disease.12 Overproduction of insulin is even a contributory factor to prostate enlargement because of its effects in promoting the overgrowth of prostate cells.13
High serum insulin is associated with the development of abdominal obesity and the many health problems induced by abdominal obesity, including atherosclerosis and impotence.14-20 Obesity is associated with excess insulin and reduced insulin sensitivity, both risk factors for type II diabetes.
Perhaps the simplest way to evaluate the toxic effects of excess insulin is by examining its effects on human mortality. One study showed that over a 10-year period, the risk of dying was almost twice as great for those with the highest levels of insulin compared to those with the lowest.21 The study authors stated that hyperinsulinemia is associated with increased all-cause and cardiovascular mortality independent of other risk factors.
|
Why Aging People Gain Weight
The number of overweight people is staggering. A review of the published literature indicates that a significant percentage of degenerative diseases is attributed to excess body fat. Those who are overweight face a significant risk of developing type II diabetes.22-24 The treatments for obesity and type II diabetes are interrelated. By effectively treating either one of these diseases, doctors can mitigate or control the other.
Poor diet, obesity, and aging result in excessive secretion of insulin, a factor in the development of type II diabetes. Suppressing the overproduction of insulin is a crucial component of a medically supervised weight-loss program.
A noticeable effect of surplus serum insulin is constant hunger, which results in a vicious cycle in which overeating causes more and more body fat to accumulate, which in turn causes even greater amounts of unwanted insulin to be secreted from the pancreas.25 We now know that hyperinsulinemia predicts diabetes mellitus.26 Even in children, serum insulin levels are far higher in obese than in non-obese children of the same age.
The effects of consuming high-glycemic foods and the subsequent hyperinsulinemia, hunger, and weight gain that occur are the subject of an article published in the Journal of the American Medical Association titled “The Glycemic Index.”27 The article’s authors summarized their position as follows:
“It is possible that the hunger incident to hyperinsulinemia may be a cause of overeating, and therefore, the obesity that so often precedes diabetes.”
Dangerous After-Meal Sugar Levels
The excess elevation of blood sugar after eating wreaks havoc in the body via multiple pathological mechanisms. Elevations in postprandial (after-meal) blood sugar, along with the accompanying insulin surge, are major contributors to the development of diabetic and age-related disorders such as heart disease, as well as diseases of the microvasculature (small blood vessels within the eyes, kidneys, and nerves).
People who have normal fasting glucose levels but whose average postprandial glucose level exceeds 200 mg/dL are three times more likely to suffer diabetic retinopathy than those who do not.29
Growing evidence indicates that severe spikes in postprandial blood sugar are a major problem for non-diabetics as well. The two primary mechanisms by which post-meal hyperglycemia causes such problems are formation of advanced glycation end products (the binding of glucose to body proteins) and increased production of free radicals that lead to severe arterial wall damage.30,31
To investigate the relationship between glucose metabolism and the severity of heart disease, one study measured how many coronary arteries were blocked in relation to postprandial glucose-insulin blood levels and other atherogenic risk factors. All of the men participating in the study had normal fasting glucose blood readings, but in response to a glucose challenge, demonstrated significantly different rates of glucose-insulin spikes. Men with the highest levels of post-load glucose, insulin, and other measurements of glycemic imbalance had the greatest number of blocked coronary arteries.32
These studies make it abundantly clear that blunting the after-meal increase in blood sugar is an important goal for those seeking optimal longevity.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Correlating Food Type and Obesity Mounting evidence points to hyperinsulinemia as being a culprit in today’s obesity epidemic. One way to help reduce excessive insulin is by eating a low-glycemic diet. This means eliminating foods that induce the pancreas to over-secrete insulin. While people seeking to lose body fat know they should avoid sucrose and fructose, too often the craving for sugar (induced by hyperinsulinemia) results in binging on carbohydrate-rich food. The food industry misleads the public into thinking that high-glycemic foods are healthy. Fruit juices, for instance, are promoted as a concentrated source of nutrients contained in fruit. The downside to juice, however, is that it is a concentrated form of quick-release fructose that can spike serum insulin. Eating whole fruit produces a gradual release of sugar into the blood. Once most fruits are juiced, however, they become catalysts for insulin overload because of their concentration of rapidly absorbable sugar. Carrots rank high on the glycemic index, but because their glycemic load is very low, there is nothing wrong with eating carrots. Once carrots are juiced, however, the sugar is concentrated into a form that instantly hits the bloodstream and provokes an insulin spike. The moral to this story is to eat lots of fruits and vegetables, but avoid the juice. A look at the calorie content of a glass of fruit or vegetable juice confirms its fat-potentiating effects. For those concerned about obtaining adequate folic acid, vitamin C, alpha carotene, and similar nutrients, these can be obtained by eating whole fruits and vegetables and by taking supplements. As shown in the sidebar on the previous page, foods that were once considered part of a healthy “low-fat diet” have a high glycemic index and high glycemic load. Ingesting these kinds of foods helps you gain weight, even though you think you are eating right. The message for those seeking to lose some pounds is to reduce or avoid consumption of foods with a higher glycemic index/load. While this is not the entire solution to the obesity crisis, it is an important component. The problem is that it is difficult to suppress carbohydrate craving. Even when people know they should avoid sucrose-fructose foods, they too often succumb to an insulin-induced addiction and gorge themselves with ice cream, chocolate, and other sugar-laden desserts. When it comes to the problem of high-glycemic diets, two obstacles must be overcome: cutting the craving for sugar, and changing eating patterns to avoid high-glycemic foods. A partial solution is to impede the rapid absorption of sugars into the bloodstream in order to reduce the accompanying insulin spike. This reduction in postprandial insulin secretion can help induce satiety and somewhat inhibit calorie intake. After six years of intensive research, a supplement is now available that interferes with the rapid absorption of sugar, thus reducing after-meal blood glucose and insulin levels by 23% and 40%, respectively.
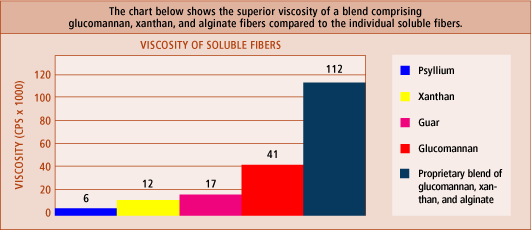
The Search for a Better Fiber The problem until now is that the large quantity of fiber required to produce a meaningful effect has resulted in poor compliance, primarily because of upper and lower gastrointestinal-related discomfort. In the early 1990s, scientists at the University of Toronto began to investigate a novel class of viscous fibers in order to identify a low-dose blend that would reduce blood glucose, insulin, cholesterol, and LDL levels. Initial studies confirmed the beneficial effects of these soluble fibers. Compared to placebo, those consuming highly viscous fibers before meals showed improvement in glycemic control, blood lipid levels, and blood pressure.35,36 The problem, however, remained—these beneficial effects could be produced only by ingesting large amounts of this fiber. To overcome this problem, the scientists tested hundreds of different fiber blends with the objective of achieving significant benefits from only a few grams of soluble fiber per meal. One of the initial findings that motivated the University of Toronto scientists to pursue this research occurred during a study that measured the glycemic-index response to different forms of fiber (or no fiber). In this study, three grams of various fibers were administered prior to a 20-gram glucose challenge. As expected, the glycemic index of the control group receiving no fiber was 100. Those receiving three grams of psyllium and xanthan showed only negligible glycemic index reductions (3% and 6%, respectively). By contrast, test subjects receiving three grams of a novel fiber blend showed a remarkable 39% reduction in their glycemic index. This finding demonstrated that consuming just three grams of this highly viscous fiber before a meal could significantly reduce the number of insulin-spiking carbohydrate calories absorbed.37 Using the Most Viscous Fibers Four to five grams of glucomannan blended into fluid or mixed with food can slow carbohydrate absorption into the bloodstream and dampen the ensuing insulin spike by up to 50%.39 Controlled clinical studies document that glucomannan can promote satiety and induce modest weight loss.40-43 It has been shown to significantly lower LDL and total cholesterol, improve diabetic control, and correct constipation.29-31,33-38,40-43 The reason glucomannan has fallen by the wayside is that in the 1980s, programs promoting quick weight loss advertised glucomannan as a supplement that could make obese people thin. The FTC stepped in and aggressively attacked those who were making exaggerated fat-loss claims for glucomannan. The subsequent negative reports by the news media caused glucomannan to be viewed by the public as a worthless dietary supplement.
The published scientific studies on glucomannan, however, are quite impressive. Although it does not make fat people thin, a double-blind trial showed that compared to placebo, obese subjects taking one gram of glucomannan before each meal lost 5.5 pounds after only eight weeks.40 The subjects were instructed not to change their eating or exercise patterns. Total cholesterol and LDL also were reduced (by 21.7 and 15.0 mg/dL, respectively) in the glucomannan-supplemented group. No adverse reactions to glucomannan were reported. Several other published studies confirm that glucomannan modestly reduces weight compared to placebo or diet alone.41-43 Total cholesterol and LDL, along with after-meal insulin and glucose blood levels, are significantly reduced when glucomannan is taken before meals.41-43 With this knowledge of glucomannan as a foundation, University of Toronto scientists led by Vladimir Vuksan, PhD, combined glucomannan with two other viscous fibers (xanthan and alginate) in an exact ratio to increase the viscosity of the original glucomannan material by 2.5-5 times.44 A mulberry concentrate (20:1) was added to enhance the glycemic-control and lipid-lowering effects.48 The primary benefit of this proprietary fiber blend lies in its superior viscosity. This means that it is better able to expand in the gastrointestinal tract to inhibit sugar absorption and bind cholesterol. This enables much smaller quantities to be taken than of other viscous dietary fibers to achieve comparable health benefits. The fiber blend’s trade name is PGX™, which stands for “polyglycoplex.”
Studies Confirm Effectiveness The second study was performed over a three-week period to better reflect real-life experiences. Study subjects took three grams of the fiber blend three times a day before meals. After three weeks, there was a 23% reduction in postprandial glucose, a 40% reduction in after-meal insulin release, and a 55.9% improvement in whole-body insulin sensitivity scores. In addition, this proprietary fiber blend reduced body fat by 2.8% from baseline by the end of the three-week study period. As a result of these findings, a large, longer-term clinical study has been initiated to further evaluate this unique fiber blend’s effects on weight loss.
| ||||||||||
| Using Fiber to Aid Weight Loss
The new PGX™ high-viscosity fiber blend may provide better results than ordinary fibers used in previous studies. Clinical studies have repeatedly shown that after-meal blood sugar levels decrease as soluble fiber viscosity increases.34,56 This relationship has also been shown with improved weight control and diminished appetite.40-43 We are anxiously awaiting the results of the ongoing weight-loss study of PGX™. In the meantime, however, the dramatic effects shown by the PGX™ fiber blend in reducing insulin, glucose, LDL, and total cholesterol make it a common-sense supplement to take before meals. Any reduction of calorie absorption should benefit overall health. Taking steps to reduce excess insulin secretion may help protect against a wide range of degenerative processes. Fiber: Limits to Weight Loss The problem is that age-associated weight gain is a multifactorial process that can be only partially addressed with existing approaches. The scientific protocol on obesity in Life Extension’s Disease Prevention and Treatment book describes why it is difficult for people to lose significant amounts of body fat. There is help on the way, but the total solution in a pill is not yet here. In the meantime, Life Extension recommends that people seeking to lose weight consume the following supplements:
Consider asking your doctor to prescribe 500 mg two to three times daily of the insulin-normalizing drug metformin.58-67 Men (without prostate cancer) should restore testosterone to youthful levels to help reduce dangerous abdominal obesity.68-108 Thyroid hormone deficiencies should be corrected.109,110 Make some effort to reduce consumption of high-glycemic foods, and to exercise, if possible.111-114
Summary In today’s gluttonous world, overproduction of insulin has become a major health problem. Even in people who eat a healthy diet, the aging process often causes insulin resistance that results in higher-than-desired insulin levels. An overwhelming number of scientific studies shows that hyperinsulinemia is an underlying culprit for many of today’s killer diseases and infirmities. While pharmaceutical companies are investigating insulin-suppressing drugs, the good news is that fiber supplements have been shown to enhance blood sugar control, decrease after-meal insulin levels, and reduce the number of calories absorbed by the body. The best fiber sources for reducing after-meal blood sugar-insulin levels, lowering cholesterol levels, and promoting weight loss are those that are rich in water-soluble fibers such as glucomannan, psyllium, guar gum, and pectin. When taken with water before meals, these fiber sources bind to the water in the stomach and small intestine to form a gelatinous, viscous mass that not only slows down the absorption of glucose, but also induces a sense of satiety (fullness) and reduces the absorption of calories. With the introduction of a new highly viscous fiber blend trademarked under the name PGX™, it may now be possible to achieve the multiple documented benefits of fiber by swallowing only a few capsules before each meal. The longevity potential associated with reducing after-meal glucose and insulin blood levels, lowering total cholesterol and LDL, and losing some weight is enormous. | ||||||
| References |
|