Life Extension Magazine®
Progesterone (women)Progesterone, a hormone that is synthesized in a biochemical pathway from cholesterol, is a precursor to other hormones, including cortisol, estrogen, and testosterone. In women, progesterone is produced in the corpus luteum of the ovary, in smaller amounts by the adrenal glands, and in copious amounts by the placenta during pregnancy. Progesterone levels, which decline with increasing age, may be used to track cycles of ovulation and menstruation, evaluate corpus luteum function in infertility patients, and assess placental function during pregnancy. In women, progesterone is responsible for maintaining a normal pregnancy. According to a recent study, a single serum progesterone measurement may be a useful indicator of pregnancy outcome in women undergoing in-vitro fertilization or intracytoplasmic sperm injection. Investigators measured serum progesterone levels 14 days after egg retrieval and performed ultrasound at eight weeks of gestation. In women with normally progressing pregnancies, progesterone levels remained high. Women with abnormal pregnancies or who could not conceive had significantly lower progesterone levels, despite progesterone supplementation.22 At the University of Adelaide Exercise Physiology Laboratory in Australia, researchers reported that progesterone may boost fat metabolism in women. Women who exercised during times of peak (mid-month) progesterone and estrogen levels had increased rates of fat metabolism as well as lower perceived exertion levels. These findings suggest that progesterone and estrogen improve exercise performance, possibly by promoting the use of fats as energy sources to support physical activity.23 All of the blood hormone tests discussed so far are included in the popular Male and Female Panels. The only exception is progesterone, which is included only in the Female Panel.
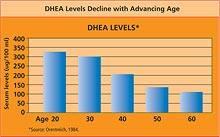
SPECIALIZED TESTSPregnenolonePregnenolone functions mainly as a hormone intermediate in the body. Made directly from cholesterol, pregnenolone is the precursor for DHEA and other hormones, including testosterone, estrogen, cortisol, and aldosterone. The body produces pregnenolone primarily in the adrenal glands, liver, testes (in men), and ovaries (in women), as well as in the brain. Pregnenolone levels tend to diminish with age, leading to a decline in the “downstream” hormones for which it acts as a precursor. Pregnenolone levels tend to be elevated in congenital adrenal hyperplasia and idiopathic hirsutism (increased hair growth in women). Investigators have shown that pregnenolone may help reduce fatigue while enhancing endurance.24 In an experimental model, pregnenolone promoted the growth of brain cells.25 In an animal study, pregnenolone administration helped to enhance memory. While DHEA and testosterone yielded similar effects, the hormones estradiol, estrone, and progesterone did not benefit memory. The investigators suggested that pregnenolone may exert its effects by acting as a precursor for numerous neurosteroid hormones. They concluded, “low serum levels of pregnenolone in aging and the increases of cancer and behavioral disorders in individuals receiving drugs that block synthesis of cholesterol, the immediate precursor of pregnenolone, suggest possible clinical utility for pregnenolone.”26 Testing one’s blood to measure pregnenolone levels is becoming very popular, as low pregnenolone can cause a wide range of hormone imbalances.
Total Estrogens (women)The total estrogens test measures the sum of three estrogens: estradiol, estriol, and estrone. Women produce estrogens mainly in the ovaries (and in the placenta during pregnancy). The adrenal glands also produce small amounts. These hormones shift over the course of a woman’s life, strongly influencing her health and well-being. Estradiol is the most commonly measured type of estrogen. Its level varies throughout the menstrual cycle and falls to a low but constant level after menopause. Increased levels of estradiol in woman suggest an increased risk of breast or endometrial cancer. Low levels may increase risk for bone fractures. Estriol is considered the “weakest” of the three primary estrogens, yet women with higher levels of estriol may have far lower risks of breast cancer.27 European doctors routinely prescribe estriol to menopausal and postmenopausal women in place of more potent estrone and estradiol drugs. If a blood test reveals low estriol levels, then a woman may ask her doctor for a prescription estriol drug (2-8 mg/day). Estriol drugs are available through compounding pharmacies. Estrone, the major source of estrogen in postmenopausal women, may help assess adrenal or ovarian conditions. The body manufactures small amounts of estrone in most tissues, especially in fat and muscle. Doctors use the total estrogen laboratory assessment to evaluate fertility problems, menstrual disorders, menopausal status, and sexual maturity in women. It is also used to help detect fetal birth defects during pregnancy and fetal distress in high-risk pregnancies. Occasionally, doctors use the test in men to assess abnormal sexual characteristics such as enlarged breasts (gynecomastia). Hormone imbalance may be a cause of migraine headaches in women. Declining estrogen levels during menstruation and meno-pause may trigger migraine head-aches. By contrast, sustained high levels of estrogen, as occur during pregnancy, frequently provide relief from headaches. Estrogen produces changes in body levels of prostaglandins and opioids, which may account for its effects in relieving headaches.9,10 Researchers at the North Central Mississippi Regional Cancer Center in Greenwood, MS, and later at the Life Extension Foundation, used an innovative treatment to correct hormone imbalances associated with migraine headaches. They administered hormone restorative therapy to 23 women suffering from migraine. In 100% of cases, hormone restoration resolved headaches, and the headaches did not recur. Additionally, symptoms associated with migraine, such as body pains, insomnia, depression, and fatigue, also resolved entirely. (See “An Innovative New Treatment for Migraine,” Life Extension, September 2004.)11 Thyroid Stimulating Hormone (TSH)The thyroid stimulating hormone (TSH) test assesses thyroid gland status. Thyroid hormones plays a crucial role in regulating growth, development, and meta-bolism. Thyroid hormones increase oxygen consumption and heat production, which are measured as resting metabolic rate. Elevated TSH is associated with hypothyroidism, a condition marked by lethargy, weakness, slow speech, constipation, dry skin, and weight gain. Low TSH suggests hyperthyroidism, a condition characterized by increased heart rate, hyperactivity, nervousness, palpitations, and heat intolerance. While overt hypothyroidism can increase cardiovascular disease risk, a possible risk from subclinical hypothyroidism is controversial. Scientists investigated the relationship of thyroid status and cardiovascular health in women participating in the Rotterdam Study. Women with subclinical hypothyroidism, as determined by a high-normal TSH level and normal serum free thyroxine level, had a greater prevalence of aortic atherosclerosis and myocardial infarction. Adjustment for body mass index, total cholesterol, HDL (high-density lipoprotein), and blood pressure did not affect these results. The study authors concluded, “subclinical hypothyroidism is a strong indicator of risk for atherosclerosis and myocardial infarction in elderly women.”28
Dihydrotestosterone (DHT)DHT is an activated form of testosterone generated by the enzyme 5-alpha reductase. Scientists estimate that DHT is 2-10 times more potent than testosterone.29-31 DHT is responsible for the development and maintenance of the male external genitalia and the prostate gland. Elevated DHT levels may indicate hypergonadism or hirsutism. Additionally, elevated DHT may increase the risk of prostate enlargement or cancer. Agents that block the conversion of testosterone to dihydrotestosterone by inhibiting the 5-alpha reductase enzyme are being studied as therapeutics for managing benign prostatic enlargement. Such agents improve urinary flow and reduce the risk of acute urinary retention or the necessity for surgical management of an enlarged prostate.32 In addition, researchers are studying 5-alpha reductase inhibitors for their potential role in reducing prostate cancer risk.33 ConclusionMen and women no longer need suffer the effects of diminished hormone levels and the accompanying onset of degenerative decline and disease. Blood testing is a powerful tool for assessing hormone status and implementing appropriate therapeutics. By restoring optimal hormone levels, you can reduce your risk for many of the most common diseases of aging, including life-threatening conditions such as osteoporosis, Alzheimer’s disease, heart disease, and depression. | |||||
| References | |||||
| 1. Barrett-Connor E, Mueller JE, von Muhlen DG, et al. Low levels of estradiol are associated with vertebral fractures in older men, but not women: the Rancho Bernardo Study. J Clin Endocrinol Metab. 2000 Jan;85(1):219-23. 2. Amin S, Zhang Y, Sawin CT, et al. Association of hypogonadism and estradiol levels with bone mineral density in elderly men from the Framingham study. Ann Intern Med. 2000 Dec 19;133(12):951-63. 3. Rapuri PB, Gallagher JC, Haynatzki G. Endogenous levels of serum estradiol and sex hormone binding globulin determine bone mineral density, bone remodeling, the rate of bone loss, and response to treatment with estrogen in elderly women. J Clin Endocrinol Metab. 2004 Oct;89(10):4954-62. 4. Barrett-Connor E, von Muhlen D, Laughlin GA, Kripke A. Endogenous levels of dehydroepiandrosterone sulfate, but not other sex hormones, are associated with depressed mood in older women: the Rancho Bernardo Study. J Am Geriatr Soc. 1999 Jun;47(6):685-91. 5. Tsai EC, Boyko EJ, Leonetti DL, Fujimoto WY. Low serum testosterone level as a predictor of increased visceral fat in Japanese-American men. Int J Obes Relat Metab Disord. 2000 Apr;24(4):485-91. 6. Villareal DT, Holloszy JO. Effect of DHEA on abdominal fat and insulin action in elderly women and men: a randomized controlled trial. JAMA. 2004 Nov 10;292(18):2243-8. 7. Malkin CJ, Pugh PJ, Jones RD, et al. The effect of testosterone replacement on endogenous inflammatory cytokines and lipid profiles in hypogonadal men. J Clin Endocrinol Metab. 2004 Jul;89(7):3313-8. 8. Malkin CJ, Pugh PJ, Morris PD, et al. Testosterone replacement in hypogonadal men with angina improves ischaemic threshold and quality of life. Heart. 2004 Aug;90(8):871-6. 9. Silberstein SD. The role of sex hormones in headache. Neurology. 1992 Mar;42(3 Suppl 2):37-42. 10. Silberstein SD. Sex hormones and headache. Rev Neurol (Paris). 2000;156 Suppl 44S30-4S41. 11. Available at: http://www.lifeextension.com/magazine/mag2004/sep2004_report_migraine_01.htm. Accessed March 25, 2005. 12. Rigano A, Rigano M, Cancellieri F, Pulle C. Sexually and well-being in early menopause. Effect of transdermal estradiol therapy. Panminerva Med. 2001 Jun;43(2):115-8. 13. Corsini E, Lucchi L, Meroni M, et al. In vivo dehydroepiandrosterone restores age-associated defects in the protein kinase C signal transduction pathway and related functional responses. J Immunol. 2002 Feb 15;168(4):1753-8. 14. Greendale GA, Edelstein S, Barrett-Connor E. Endogenous sex steroids and bone mineral density in older women and men: the Rancho Bernardo Study. J Bone Miner Res. 1997 Nov;12(11):1833-43. 15. Available at: http://www.jeanhailes.org.au/research/pub_abs_endo2004.htm. Accessed March 25, 2005. 16. Fiore C, Inman DM, Hirose S, et al. Treatment with the neurosteroid dehydroepiandrosterone promotes recovery of motor behavior after moderate contusive spinal cord injury in the mouse. J Neurosci Res. 2004 Feb 1;75(3):391-400. 17. Laaksonen DE, Niskanen L, Punnonen K, et al. Testosterone and sex hormone-binding globulin predict the metabolic syndrome and diabetes in middle-aged men. Diabetes Care. 2004 May;27(5):1036-41. 18. Niskanen L, Laaksonen DE, Punnonen K, Mustajoki P, Kaukua J, Rissanen A. Changes in sex hormone-binding globulin and testosterone during weight loss and weight maintenance in abdominally obese men with the metabolic syndrome. Diabetes Obes Metab. 2004 May;6(3):208-15. 19. Hayashi T, Boyko EJ, Leonetti DL, et al. Visceral adiposity is an independent predictor of incident hypertension in Japanese Americans. Ann Intern Med. 2004 Jun 15;140(12):992-1000. 20. Moffat SD, Zonderman AB, Metter EJ, et al. Free testosterone and risk for Alzheimer disease in older men. Neurology. 2004 Jan 27;62(2):188-93. 21. Shifren JL, Braunstein GD, Simon JA, et al. Transdermal testosterone treatment in women with impaired sexual function after oophorectomy. N Engl J Med. 2000 Sep 7;343(10):682-8. 22. Ioannidis G, Sacks G, Reddy N, et al. Day 14 maternal serum progesterone levels predict pregnancy outcome in IVF/ICSI treatment cycles: a prospective study. Hum Reprod. 2005 Mar;20(3):741-6. 23. Available at: http://www.adelaide.edu.au/news/news352.html. Accessed March 25, 2005. 24. Roberts E. Pregneolone—from Selye to Alzheimer and a model of the pregnenolone sulfate binding site on the GABAA receptor. Biochem Pharmacol. 1995 Jan 6;49(1):1-16. 25. Mayo W, George O, Darbra S, et al. Individual differences in cognitive aging: implication of pregnenolone sulfate. Prog Neurobiol. 2003 Sep;71(1):43-8. 26. Flood JF, Morley JE, Roberts E. Memory-enhancing effects in male mice of pregnenolone and steroids metabolically derived from it. Proc Natl Acad Sci USA. 1992 Mar 1;89(5):1567-71. 27. Sitieri PK, Sholtz PI, Cirillo PM, et al. Prospective study of estrogens during pregnancy and the risk of breast cancer. Unpublished study performed at the Public Health Institute in Oakland, California, and funded by the US Army Medical Research and Material Command under DAMD 17-99-1-9358. 28. Hak AE, Pols HA, Visser TJ, et al. Subclinical hypothyroidism is an independent risk factor for atherosclerosis and myocardial infarction in elderly women: the Rotterdam Study. Ann Intern Med. 2000 Feb 15;132(4):270-8. 29. Geller J, Sionit L. Castration-like effects on the human prostate of a 5 alpha-reductase inhibitor, finasteride. J Cell Biochem Suppl. 1992; 16H:109-12. 30. Deslypere JP, Young M, Wilson JD, McPhaul MJ. Testosterone and 5 alpha-dihydrotestosterone interact differently with the androgen receptor to enhance transcription of the MMTV-CAT reporter gene. Mol Cell Endocrinol. 1992 Oct;88(1-3):15-22. 31. Wright AS, Thomas LN, Douglas RC, Lazier CB, Rittmaster RS. Relative potency of testosterone and dihydrotestosterone in preventing atrophy and apoptosis in the prostate of the castrated rat. J Clin Invest.1996 Dec 1;98(11):2558-63. 32. Foley CL, Kirby RS. 5 alpha-reductase inhibitors: what’s new? Curr Opin Urol. 2003 Jan;13(1):31-7. 33. Schulman C. Use of 5-alpha-reducatase inhibitors in the prevention of prostate cancer. Ann Urol (Paris). 2004 Dec;38 Suppl 2:S35-42. |