Life Extension Magazine®
Coronary heart disease (CHD) is the leading cause of death in the Western world. Although physicians and patients tend to think of CHD as a man’s disease, it is the leading cause of death among women in the US. Because of this misconception, physicians may fail to look for this condition in women or fail to recognize its warning signs, which may differ among women and men. Numerous risk factors contribute to CHD risk, including non-modifiable factors such as age and family history, and modifiable risk factors such as cholesterol level, homocysteine level, and high blood pressure. Because the incidence of CHD in women increases markedly following menopause (when levels of ovarian hormones drop dramatically), researchers have speculated that female hormones may play a cardioprotective role. Recent studies such as the Women’s Health Initiative (WHI), however, have failed to demonstrate a relationship between conventional hormone replacement therapy (HRT) and protection from CHD. As a result of such studies, many women and physicians have avoided HRT, fearing that it may increase CHD risk. We believe that trials such as the WHI failed to demonstrate a cardioprotective effect of female hormones for several reasons: namely, that these trials used forms of hormones that are not biochemically identical to those naturally found in women; that the trials used a “one-size-fits-all” dosing strategy; and that the trials did not cycle hormones to mimic the normal menstrual cycle. Our clinical experience suggests that a more refined form of HRT, which we call hormonorestorative therapy, or hormonorestoration, may indeed offer cardioprotection by helping minimize several CHD risk factors.
IntroductionIn this article, we examine the principles of hormonorestorative therapy and two case histories in which individualized hormonorestoration helped reduce multiple cardiovascular risk factors in women. Additionally, we review CHD risk factors, symptoms, and diagnosis, along with conventional and integrative strategies for heart disease prevention and treatment. We propose that an individualized strategy of hormonorestorative therapy represents a novel yet powerful approach to reducing CHD risk in women. CHD is the most common type of heart disease. According to the American Heart Association, CHD kills 241,622 women in the US each year, compared to 67,542 lives lost to lung cancer and 41,514 to breast cancer. Thirty-eight percent of US women (compared to 25% of men) die within one year of suffering a heart attack.1 CHD affects women of all racial and ethnic groups, though African-American women are more likely to die of CHD than are Caucasian women.1 Age is an important risk factor for CHD. Women tend to develop CHD later in life than men do, experiencing a greater risk after their reproductive years.2 After menopause, women have heart problems as often as men do. It is thought that naturally occurring female hormones may help protect a woman’s heart from CHD before menopause. As their life expectancy continues to increase, women spend more time in the postmenopausal phase of life, which may increase CHD risk. CHD is fast becoming a major cause of morbidity and mortality in aging women, particularly with rapid growth in the number of women aged 65 and over. The HRT ControversyIt is a well-known fact that CHD is uncommon in women before the age of 40. The biggest change in a woman’s body at this time is the declining production of most steroid hormones. This may lead to a loss of hormonal cardioprotective effects, because the cardiovascular system is controlled by multiple endocrine signals.3 Until recently, it was generally believed that the decreased incidence of CHD in women before menopause was mediated by a protective effect of estrogen on the coronary arteries, achieved by modulating levels of serum cholesterol.4 However, other research suggests that estrogen-induced improvements in serum cholesterol account for only one third of the observed clinical benefits of estrogen.4-7 It was hypothesized that an atheroprotective effect of estrogen may be mediated by this hormone’s direct effect on vascular smooth muscle cells.8 In fact, the data show that estrogen can increase dilation of arteries and inhibit the response of blood vessels to injury and the development of atherosclerosis.4 Few studies have explored the relationship between androgen levels and CHD in women. The idea that DHEA protects against atherosclerosis was proposed by Kask in 1959.9 Some data show that serum dehydroepiandrosterone sulfate (DHEA-S) and androgen levels decline with age, and that levels in the normal physiological range are correlated with lower risk of carotid artery atherosclerosis.10 There is growing support for a possible benefit of DHEA supplementation in preventing cardiovascular events in women, predominantly through an estrogenic effect.11-13 Testosterone can regulate vascular physiology directly through stimulation of androgen receptors and inhibition of plaque formation, and indirectly after conversion to estradiol.14 Nevertheless, evidence concerning the efficacy of DHEA and testosterone in protecting the cardiovascular system remains inconclusive and warrants further study.
Conventional HRT has been the subject of much bad publicity in recent years, which has scared many women and their doctors away from the use of all types of hormones. Since hormones are critical for the normal function and well-being of the human body, this represents a catastrophic health disaster. While many of us have heard that estrogen is dangerous, there must be something more to the story, since all men, women, and children naturally have estrogens in their bodies. Blanket statements that estrogen is harmful overlook the larger picture. In fact, the body produces several estrogens, of which the three most important are estriol, estradiol, and estrone. Estradiol and estrone are generally considered to be procarcinogenic (able to stimulate cancerous changes).15 Estriol has been touted as a much safer estrogen and, in fact, may actually decrease the risk of neoplastic changes.16 Nevertheless, it is important to note that estradiol and estrone play important roles in the body, as all hormones do, and may be dangerous only in cases of hormonal imbalance. Estrogens do not exist in isolation but instead counteract each other. They help determine female physical appearance, can improve skin and bone conditions, may help with lubrication of mucous membranes, allow ovulation, and support the central nervous system, among other actions.16 According to the critics, estrogen is not the only “bad” hormone. Progesterone also has recently come under fire in the medical community. It should be noted, however, that many of the reported ill effects ascribed to progesterone arose from synthetic progesterone analogs called progestins. Unlike progesterone, progestins are not biochemically identical to the compounds naturally found in women’s bodies. These synthetic progestins can produce side effects that generally do not occur with bioidentical progesterone.17 In fact, progesterone can be helpful for both men and women in balancing and offsetting the strong effects of estrogen. Until a few years ago, hormone replacement therapy was considered a first-line treatment for preventing CHD in women.18 After publication of the Heart and Estrogen/Progestin Replacement Follow-up (HERS II)19 and WHI20 studies, researchers claimed that HRT not only provided no cardioprotective benefit for women, but in fact did just the opposite, increasing CHD risk. Examination of the design of these studies, however, reveals that they did not use hormone forms that are bioidentical to those naturally found in the human body. Synthetic HRT typically uses 0.625 mg/d of conjugated equine estrogens and 2.5 mg/d of medroxyprogesterone acetate (progestin). The human body does not produce these hormones (Premarin® or Provera®) and thus has no deficiency of them. However, our bodies can be deficient of naturally occurring hormones such as total estrogen (which includes estriol, estrone, and estradiol) and progesterone, among others.
| ||||||||||||
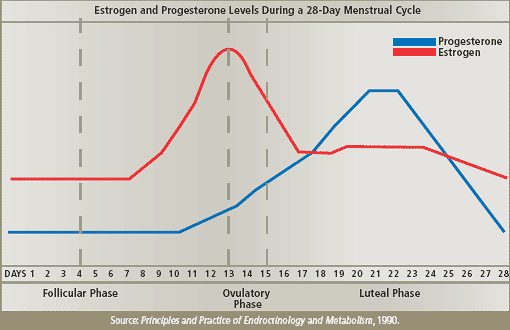
| These studies used standard doses of powerful synthetic hormones for women of different ages and weights, without evaluating each woman’s levels of various hormones either before or during treatment. Conventional HRT likewise usually uses hormones in a continuous or sequential manner. In this case, hormones were administered throughout the month or estrogen was recommended for the first two weeks alone and together with progestin for the next two weeks. Because hormone levels are constantly changing, however, the body’s physiology needs estrogens and progesterone on a daily basis, and doses must be customized to suit each woman’s particular requirements. The “one-size-fits-all” method of HRT used in the HERS II and WHI studies seems likely to generate side effects or long-term problems by not accounting for or addressing the unique needs of each participant. Thus, current problems with conventional HRT can be summarized as follows:
Principles of HormonorestorationThe chemical structure of the hormones used in hormonorestoration should be identical to human hormones. Normal ratios among hormones in each hormone group (for example, the estrogen group) are maintained. Triest, which includes all three major estrogens—estriol, estradiol, and estrone—is the preferable form of estrogens. The combination of all deficient hormones—pregnenolone, DHEA, progesterone, estrogens, and testosterone—should be used instead of just one or two hormones, the method often employed by conventional medical practitioners. Using a combination of hormones allows us to use smaller doses of each and provide hormonal balance. While many of the patented prescription hormones available in pharmacies are bioidentical, not all of the crucial estrogens are available as patented drugs. For example, estradiol is a bioidentical hormone available as a patented prescription drug, but if it is used without estriol and estrone, serious problems may result. Since estriol is not currently available as a patented “estrogen” drug, it can be obtained only by filling a doctor’s prescription at a compounding pharmacy. Thus, it may be impossible to achieve the optimal balance of estrogens using only patented prescription medications. When using the term “bioidentical,” it is important to specify that these hormones are bioidentical to those naturally found in humans. The prescription drug Premarin®, for example, contains conjugated estrogens that are “bioidentical” for horses, but not for humans. The preferable delivery system for administering bioidentical hormones is topical gels containing highly lipophilic molecules of low molecular weight that can be readily absorbed through the skin. This delivery system allows for individualized doses. Thus, some basic principles of hormonorestoration include:
Blood tests are essential in developing a safe and effective hormonorestorative program. When your doctor considers prescribing insulin, he first checks your glucose level. A similar approach should be employed for hormonorestoration. Dose recommendations for different patients should be determined by serial testing of serum hormone levels. Doses can be individually modified during treatment to produce ideal youthful physiological serum levels.
While physicians can prescribe a hormone restoration program, patients may need to adjust dosages as their clinical symptoms change. After a few months on hormono-restoration, most women are able to do this by themselves, making their own minimal changes in dosages according to their needs. When you are under stress, exercising, or performing intensive mental work, your body may require more hormones. While blood tests are important, they should always be evaluated in a clinical context. For example, we have observed that progesterone does not work always exactly as might be expected. If, during replacement therapy, you use a larger dose than your body requires, progesterone may be converted first to androstenedione and then to estradiol. This could lead to stimulation of the sympathetic nervous system, and instead of a diuretic and relaxing effect (for example, improved sleep), the opposite effect may occur. As previously noted, several risk factors contribute to the onset of CHD. If a physiologically based approach to hormone restoration helps to eliminate several of these risk factors, the risk of CHD should decrease. As also noted earlier, recent studies have found that conventional HRT actually increases CHD risk.19,20 In our experience, however, the appropriate use of hormonorestorative therapy with bioidentical hormones actually decreases several CHD risk factors. In the two cases we are about to describe, we achieved complete control of blood pressure, glucose and cholesterol levels, and psychosocial symptoms. We believe that individualized hormonorestoration of five essential steroid hormones, based on clinical symptoms and blood tests, is a more effective strategy for decreasing CHD risk than is standard HRT using two synthetic drugs. For optimal safety and efficacy, physicians and patients must monitor hormone levels regularly, usually three months after beginning treatment and then every six months thereafter. The following two cases demonstrate how an anti-aging program featuring physiological hormonorestoration can help women reduce their CHD risk factors.
| ||||||||||
Patient Case Study #1A 50-year-old Caucasian woman presented with the following complaints during her initial visit in April 1999: high blood pressure that was poorly controlled with prescription drugs; migraine; high cholesterol; depression; severe anxiety; irritability; fatigue; poor libido; low sex drive; genital herpes; poor short-term memory; trouble falling asleep; weight gain; arthritis; and irregular menstrual cycle. Her vital signs: height, 5’4”; weight, 125 pounds; blood pressure, 150/90 mmHg; pulse, 64 beats/minute. Many of these signs and symptoms are risk factors for CHD, including hypertension, elevated cholesterol, and depression/anxiety. We indicated that broad-spectrum treatment of these risk factors could dramatically reduce her chances of experiencing debilitating or deadly CHD. The patient had complained of most of her symptoms for the last 10-15 years. She did not exercise, noting that despite her desire to lose some weight, she felt tired all the time. The patient was taking the following drugs: triamterene/ hydrochlorothiazide, Procardia XL®, and Nifedical XL® (for high blood pressure); Premphase® (for hot flashes and vaginal dryness); Zoloft® (for depression); Butisol Sodium® (sedative) and Ambien® (for sleeping disorder); and Zovirax® and Valtrex® (to manage genital herpes recurrences). Initial laboratory evaluations revealed high total cholesterol (241 mg/dL). Her profile of basic steroid hormones also was significantly imbalanced. The patient’s hormone levels are shown in Table 1 (reference ranges shown in parentheses).
The patient had an extremely high level of total estrogen and low levels of the four other steroid hormones. She demonstrated a relative dominance of estrogens, which can stimulate sympathetic system activity and might explain why she had serious difficulties correcting her blood pressure. The patient’s initial treatment program focused on correcting what we considered her “foundational problem”: hormonal imbalance. The patient started the following treatment with bioidentical hormones taken in the morning:
Additional supplements included in her treatment were: Life Extension Mix, three tablets taken three times daily; omega-3 fatty acids, 1000 mg taken in the morning; glucosamine sulfate, 2000 mg taken in the morning; phosphatidylserine, 200 mg taken in the morning; and NutriCology® ProGreens® (containing green foods, plant fibers, bioflavonoids, herbal extracts, and probiotics), one scoop taken in the morning. After three days on the program, the patient discontinued her use of Premphase®. During the first month of treatment, the patient’s blood pressure improved to 130/90 mmHg, her migraines decreased in frequency and severity, and her joint pain disappeared completely. We increased her dose of DHEA to 100 mg in the morning and 50 mg at noon, and added 0.2 ml of progesterone and 420 mg of magnesium citrate to be taken one hour before bedtime. During the next three months, the patient’s depression and anxiety were so improved that she decreased and then discontinued her use of Zoloft® and Butisol Sodium®. She later stopped taking Ambien® because her sleeping disorder had resolved. The patient reported that she was on only one drug for hypertension (Procardia XL®) and that her energy level had improved tremendously. She started exercising four to five times weekly. At this time, we added to the patient’s regimen human growth hormone (HGH), 0.5 IU daily taken six days per week, and androstenedione, 50 mg taken 30 minutes before exercise. We decreased DHEA to 50 mg and increased pregnenolone to 200 mg taken in the morning. After one year of treatment, the patient had experienced no recurrences of genital herpes and thus decided to discontinue her prescription medications for herpes. Her quality of life had vastly improved, with no occurrence of migraine, which she described as the “first sustained relief in several years.” Her total cholesterol had dropped to 187 mg/dL and her blood pressure was 120-130/70 mmHg, though she no longer was using any medication to control blood pressure. She also reported no memory problems. Today, the patient enjoys a healthy lifestyle, exercises regularly, eats a balanced diet, and continues her hormonorestorative therapy and supplements.
Patient Case Study #2A 56-year-old Caucasian woman presented in September 1999 with a history of chronic fatigue syndrome, obesity, severe shortness of breath, hypertension, type II diabetes, depression, anxiety, panic attacks, insomnia, arthritis, body aches, hot flashes, vaginal dryness, no sex life, vaginal yeast infection, short-term memory problems, and “chocoholism.” Her height was 4’10,” her weight 232 pounds, her blood pressure 168/86 mmHg, and her pulse 72 beats/minute.
The patient began having most of these symptoms after the age of 44, soon after her divorce. Her body weight at that time was 118 pounds. At the age of 30, she had a complete hysterectomy secondary to fibroids. This patient had numerous risk factors that, left untreated, could lead to a major cardiovascular event. Heart disease does not happen in isolation, but instead results from a system-wide breakdown in the body. We indicated that by intervening and diminishing her risk factors, we could help her decrease her risk for CHD. At the time of her first visit, she was taking Glucophage® and glyburide (for type II diabetes), Zestril® and hydrochlorothiazide (for high blood pressure), Wellbutrin® (for depression), Premarin® (for hot flashes and sex disorder), Tylenol® (for pain), multi-vitamins, liquid minerals, vitamin E, niacin, and two weight-loss supplements. In this case, we again found a high estrogen level (699 pg/mL) and low levels of progesterone (0.2 ng/mL), testosterone (16 ng/dL), DHEA-S (35 ug/dL), and pregnenolone (less than 10 ng/dL). We recommended the following program (all taken in the morning except where noted):
After two days on the program, the patient discontinued her use of Premarin®. We recommended that she decrease her carbohydrate intake and begin an exercise program. One month later, she returned to the clinic, reporting no complaints whatsoever. Her energy levels had improved significantly, she had lost 10 pounds, and she had elected to discontinue her use of Wellbutrin® and Tylenol®. We added to her regimen glucosamine sulfate (2250 mg taken in the morning), androstenedione (50 mg taken 30 minutes before exercise), and 0.2 ml of progesterone and 420 mg of magnesium citrate, taken one hour before bedtime. After four months on the program, the patient had lost an additional 28 pounds and said that she felt like a different person. Her blood pressure had normalized to 120/80 mmHg and her blood glucose was stable. At this time, we added the following supplements to her regime: conjugated linoleic acid (CLA), 8 grams taken in the morning before breakfast; chitosan, two capsules taken before lunch and two capsules before dinner; hydroxycitric acid (HCA), one (1000-mg) capsule taken three times daily before meals (for weight loss); B-complex vitamins, one tablet daily; and omega-3 fatty acids, 3000 mg taken twice daily. We also suggested a one-month parasite-cleansing program using Unicity™ Paraway® Pack. At the patient’s one-year follow-up visit, she reported that she was no longer taking prescription drugs for high blood pressure or type II diabetes, and that her blood pressure and blood glucose levels were normal and stable. | |||||||||||||
ConclusionIn the cases just described, each woman had several risk factors for CHD before initiating an individualized anti-aging program of hormone restoration therapy. We believe that hormone restoration may help prevent the development of CHD in women by helping to eliminate cardiovascular risk factors such as high blood pressure, being overweight or obese, elevated blood lipids and blood sugar, and abnormal responses to stress. We further believe that individualized hormonorestorative therapy using bioidentical hormones applied in a cyclical manner and balancing levels of all the body’s major steroid hormones may help women achieve the reduced risk of cardiovascular disease associated with the premenopausal years. Hormone restoration is not yet widely practiced because it is not always easy to implement, requiring as it does an individualized regimen tailored to the needs of each patient. The two cases just described may help us understand what can be done to reduce or eliminate CHD risk factors in women through the application of hormonorestorative therapy. |
| References |
1. Available at: http://www.americanheart.org/presenter.jhtml?identifier=2859. Accessed May 27, 2005. 2. Lewis V, Hoeger K. Prevention of coronary heart disease: a nonhormonal approach. Semin Reprod Med. 2005 May;23(2):157-66. 3. Baxter JD, Young WF, Jr., Webb P. Cardiovascular endocrinology: introduction. Endocr Rev. 2003 Jun;24(3):253-60. 4. Mendelsohn ME, Karas RH. The protective effects of estrogen on the cardiovascular system. N Engl J Med. 1999 Jun 10;340(23):1801-11. 5. Bush TL, Barrett-Connor E, Cowan LD, et al. Cardiovascular mortality and noncontraceptive use of estrogen in women: results from the Lipid Research Clinics Program Follow-up Study. Circulation. 1987 Jun;75(6):1102-09. 6. Grady D, Rubin SM, Petitti DB, et al. Hormone therapy to prevent disease and prolong life in postmenopausal women. Ann Intern Med. 1992 Dec 15;117(12):1016-37. 7. Mendelsohn ME and Karas RH. Estrogen and the blood vessel wall. Curr Opin Cardiol. 1994 Sep;9(5):619-26. 8. Karas RH, Patterson BL, Mendelsohn ME. Human vascular smooth muscle cells contain functional estrogen receptor. Circulation. 1994 May;89(5):1943-50. 9. Kask E. 17-Ketosteroids and arteriosclerosis. Angiology. 1959 Oct;10:358-68. 10. Bernini GP, Sgro’ M, Moretti A, et al. Endogenous androgens and carotid intimal-medial thickness in women. J Clin Endocrinol Metab. 1999 Jun;84(6):2008-12. 11. Ebeling P, Koivisto VA. Physiological importance of dehydroepiandrosterone. Lancet. 1994 Jun 11;343(8911):1479-81. 12. Vermeulen A. Dehydroepiandrosterone sulfate and aging. Ann NY Acad Sci. 1995 Dec 29;774:121-7. 13. Labrie F, Belanger A, Luu-The V, et al. DHEA and the intracrine formation of androgens and estrogens in peripheral target tissues: its role during aging. Steroids. 1998 May;63(5-6):322-8. 14. Wu FC, von EA. Androgens and coronary artery disease. Endocr Rev. 2003 Apr;24(2):183-217. 15. Yager JD. Endogenous estrogens as carcinogens through metabolic activation. J Natl Cancer Inst Monogr. 2000;(27):67-73. 16. Head KA. Estriol: safety and efficacy. Altern Med Rev. 1998 Apr;3(2):101-13. 17. Apgar BS, Greenburg G. Using progestins in clinical practice. Am Fam Physician. 2002 Oct 15;62(8):1839-46, 1849-50. 18. Mosca LJ. Contemporary management of hyperlipidemia in women. J Womens Health Gend Based Med. 2002 Jun;11(5):423-32. 19. Grady D, Herrington D, Bittner V, et al. Cardiovascular disease outcomes during 6.8 years of hormone therapy: Heart and Estrogen/progestin Replacement Study follow-up (HERS II). JAMA. 2002 Jul 3;288(1):49-57. 20. Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results From the Women’s Health Initiative randomized controlled trial. JAMA. 2002 Jul 17;288(3):321-33. 21. Ford ES, Giles WH, Dietz WH. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA. 2002 Jan 16;287(3):356-9. 22. Lakka HM, Laaksonen DE, Lakka TA, et al. The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA. 2002 Dec 4;288(21):2709-16. 23. Mosca L, Grundy SM, Judelson D, et al. Guide to Preventive Cardiology for Women. AHA/ACC Scientific Statement Consensus panel statement. Circulation. 1999 May 11;99(18):2480-4. 24. Clarke R, Daly L, Robinson K, et al. Hyperhomocysteinemia: an independent risk factor for vascular disease. N Engl J Med. 1991 Apr 25;324(17):1149-55. 25. Robinson K, Mayer E, Jacobsen DW. Homocysteine and coronary artery disease. Cleve Clin J Med. 1994 Nov;61(6):438-50. 26. Boushey CJ, Beresford SA, Omenn GS, Motulsky AG. A quantitative assessment of plasma homocysteine as a risk factor for vascular disease. Probable benefits of increasing folic acid intakes. JAMA. 1995 Oct 4;274(13):1049-57. 27. Mitra B, Panja M. High sensitive C-reactive protein: a novel biochemical markers and its role in coronary artery disease. J Assoc Physicians India. 2005 Jan;53:25-32. 28. Ridker PM, Morrow DA. C-reactive protein, inflammation, and coronary risk. Cardiol Clin. 2003 Aug;21(3):315-25. 29. Merriman S, Haw C, Kirk J, Stubbs J. Risk factors for coronary heart disease among inpatients who have mild intellectual disability and mental illness. J Intellect Disabil Res. 2005 May;49(Pt 5):309-16. 30. Gupta R, Sarna M, Thanvi J, et al. High prevalence of multiple coronary risk factors in Punjabi Bhatia community: Jaipur Heart Watch-3. Indian Heart J. 2004 Nov;56(6):646-52. 31. Cassidy AE, Bielak LF, Zhou Y, et al. Progression of subclinical coronary atherosclerosis: does obesity make a difference? Circulation. 2005 Apr 19;111(15):1877-82. 32. Bunker SJ, Colquhoun DM, Esler MD, et al. “Stress” and coronary heart disease: psychosocial risk factors. Med J Aust. 2003 Mar 17;178(6):272-6. 33. Banerjee A, Chisti Y, Banerjee UC. Streptokinase—a clinically useful thrombolytic agent. Biotechnol Adv. 2004 Feb;22(4):287-307. |