Life Extension Magazine®
A new blood test can accurately identify atherosclerotic plaque that is vulnerable to rupture, and help to stave off fatal cardiac events. By pinpointing individuals who are in imminent danger of an ischemic (no blood flow) event, the innovative PLAC® blood test alerts physicians and patients to the urgent need to implement aggressive protective measures. As the PLAC® test becomes more widely utilized, it promises to dramatically reduce the risk of sudden heart attack and stroke. Detecting Vulnerable PlaqueWhen Tim Russert succumbed to a fatal heart attack in June of this year, the world was shocked not only by the loss of an iconic TV news personality, but also by the unexpected nature of his death. Tim Russert had coronary artery disease that was supposedly controlled with medication and exercise. A stress test in late April was unremarkable. The autopsy revealed that cholesterol plaque rupturing in a coronary artery choked off the blood supply through that artery, causing Tim Russert’s fatal heart attack.1
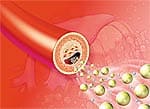
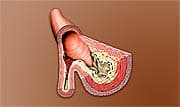
An atherosclerotic plaque, also known as an atheroma, is a deposit of harmful fats, or lipids, on the blood vessel wall. When the plaque ruptures, globules resembling chunks of oatmeal break loose and suddenly clog a blood vessel, causing heart attack or stroke. Cardiology experts noted the crucial difference between gradual narrowing of arteries, which had been controlled in Russert, versus the sudden, fatal blockage of a main coronary artery by ruptured atherosclerotic plaque on the vessel wall. Because about half of people who have a heart attack have no symptoms before the acute event,1 it is critical that they are recognized and treated before it is too late. Similarly, acute, debilitating stroke, also caused in many cases by plaque rupture, often occurs without warning. Imaging tests, such as carotid ultrasound or heart computed tomography (CT), can tell us about the current anatomical state of blood vessels supplying the brain and heart, but even they cannot identify plaque that is vulnerable to rupture.2,3 In addition, CT technology involves radiation exposure, and coronary angiography requires injection of dye that may cause complications in some individuals. In the developed world, heart disease remains the leading cause of death and disability for both men and women.4,5 While traditional risk factors such as age, gender, low-density lipoprotein (LDL), triglycerides, high-density lipoprotein (HDL), blood pressure, waist circumference, and blood sugar help predict who will develop chronic atherosclerosis, they cannot predict everyone who will suffer an acute cardiovascular event. Fortunately, the new PLAC® test, performed on a routine blood sample, can help identify individuals who are at risk for stroke or heart attack.2 PLAC® Test Identifies Atherosclerotic Plaques Ready to RuptureThe PLAC® test measures blood levels of an enzyme called lipoprotein phospholipase A2 (Lp-PLA2). The Lp-PLA2 enzyme unleashes a chain of harmful events culminating in endothelial dysfunction, which is a pathological abnormality in the blood vessel wall that sets the stage for atherosclerosis, plaque accumulation, and rupture.6 An elevated level of Lp-PLA2 may signal that an arterial plaque is susceptible to rupture, which could cause a clot to break loose, precipitating a heart attack or stroke. Traditional markers of cardiovascular risk such as cholesterol levels tell physicians very little about acute risk of stroke or heart attack.7 However, there is an important biochemical relationship between low-density lipoprotein (LDL) and Lp-PLA2. LDL particles are oxidized by oxygen free radicals, and these oxidized LDL particles penetrate into the intima, the innermost layer of the blood vessel wall.8 The PLAC® test measures the activity of the Lp-PLA2 enzyme associated with LDL particles when they are oxidized. David G. Harrison, MD, FACC, FAHA of Emory University School of Medicine in Atlanta, GA, notes, “if one has an elevated PLAC® test, it indicates that the person has [inflamed] atherosclerotic plaques in which LDL oxidation is occurring.” Lp-PLA2 is made in the plaque itself and acts as a specific marker of inflammation that appears to be directly involved in forming rupture-prone atherosclerotic plaque. Patients with coronary artery disease have markedly elevated Lp-PLA2 levels, reflecting build-up of vulnerable plaque in the blood vessel wall.7,8 An autopsy study of 25 patients with sudden coronary death used antibody to Lp-PLA2 to track its presence within diseased coronary arteries. Although early plaques had very light staining for Lp-PLA2, or none at all, ruptured plaques that had caused sudden death showed intense Lp-PLA2 staining deep within the plaque.9 Similarly, patients undergoing surgery related to atherosclerosis of the carotid artery, one of the main arteries supplying the brain, had intense staining for Lp-PLA2 within macrophages—a type of white blood cell involved in plaque inflammation. Those who had carotid artery disease without symptoms had much less intense staining for Lp-PLA2.10 PLAC® Test Predicts Acute Cardiovascular Events
High levels of Lp-PLA2 predict increased rates of coronary heart disease and stroke in multiple population-based studies. Physicians from the prestigious Mayo Clinic and other experts recently reviewed more than 25 published studies of Lp-PLA2, including a compilation of 14 prospective epidemiologic studies of 20,000 patients. They concluded that Lp-PLA2 is consistently linked to higher risk of heart attack and stroke, and that this increased risk is not significantly changed when conventional risk factors for cardiovascular disease are also considered. They determined that Lp-PLA2 is unique in being highly specific for plaque inflammation, as well as being involved in causing that inflammation.11-13 The PLAC® test is as sensitive and more specific than other markers for the prediction of acute events, allowing for enhanced ability to identify people who are at increased risk for an acute ischemic event. Lp-PLA2 has been shown to predict endothelial function in a number of studies.6,14 A striking example of this is a study showing that individuals in the highest third of Lp-PLA2 levels are more than three times as likely to have coronary endothelial dysfunction than those in the lowest third.14 In this study, Lp-PLA2 levels predicted endothelial dysfunction independently of their association with cholesterol and other cardiovascular risk factors. A review of available studies shows that individuals with high levels of Lp-PLA2 have about two to three times the risk for acute cardiovascular events, and that this risk is independent of traditional cardiovascular risk factors.2,12,13 The association of high Lp-PLA2 levels with acute cardiovascular risk holds true even in apparently healthy older adults.15 Although LDL and other lipids are not consistent predictors of stroke risk, elevated Lp-PLA2 approximately doubles stroke risk.7,16
Who Should Receive the PLAC® Test, and How Often?Currently, the most validated role for PLAC® test is in individuals with moderate or greater risk for cardiovascular events based on traditional risk factors, in whom an elevated level would prompt more aggressive risk factor modification, such as more intensive reduction of cholesterol levels and/or blood pressure, and greater emphasis on diet and lifestyle changes. Individuals in whom traditional risks are not known, such as a patient who is adopted and in whom family history is uncertain, may also particularly benefit from the PLAC® test. Based on the well-known and extensive Framingham study, risk of cardiovascular events can be calculated for a given patient.17 Some experts suggest that the PLAC® test be done in patients in whom the risk of having an acute event within 10 years is estimated to be 5-20%, according to a formula based on the Framingham study.
A recent expert consensus panel review for the clinical use of Lp-PLA2 testing7 recommends that the test be used in moderate and high-risk patients,12 but not as a screening test for individuals at low risk of cardiovascular disease and stroke. The consensus panel simplified the definition of moderate risk from that based on the Framingham study, and defined it as any one or more of the following: age 65 or older, currently smoking, metabolic syndrome, fasting blood sugar over 99, or any two or more traditional risk factors for cardiovascular disease.7 In patients with metabolic syndrome, the PLAC® test is clearly more specific for prediction of risk than the C-reactive protein test, which appears to increase in response to the presence of metabolic syndrome.7 The PLAC® test should be done once each year in persons who are obese, who have high blood pressure, type 2 diabetes mellitus, high cholesterol, or a strong family history of stroke and coronary heart disease. Regular smokers also need to be tested. In patients receiving heart transplants, Lp-PLA2 levels also predict risk of heart attack and cardiac death related to inflammation in the arteries of the transplanted heart, a condition known as cardiac allograft vasculopathy.18 PLAC® Test Helps Guide Cardiovascular Risk ManagementAlthough Lp-PLA2 is not a primary target of therapy, the expert consensus panel7 recognizes that patients with high Lp-PLA2 have evidence of vascular inflammation and should therefore be treated more intensively with preventive therapy to lower risk of a cardiovascular event. The panel recommends that patients with high Lp-PLA2 levels be upgraded from moderate risk to high risk, or from high risk to very high risk. In these patients, a suitable goal is to lower LDL to 100 mg/dL in high-risk patients and to 70 mg/dL in very high-risk patients.7 Patients with a cluster of cardiovascular risk factors known as the metabolic syndrome, namely overweight, abnormal blood lipids, high blood sugar, and high blood pressure, may also benefit from monitoring Lp-PLA2 and from treatment strategies, such as intensified lifestyle changes and combination lipid-lowering therapy, that reduce Lp-PLA2.2,12,19 In addition to its use to gauge the intensity of lifestyle and pharmacologic interventions needed, the PLAC® test can be used serially to assess the response to these therapies. Statins for elevated cholesterol typically reduce Lp-PLA2 levels by 20-30%, and the addition of niacin or a fibrate drug in combination can achieve an additional 5-20% lowering. In addition to statins and fibrates, orlistat and other lipid-lowering drugs also lower Lp-PLA2, allowing progress of therapy to be monitored with the PLAC® test.11,16,19,20 Blood pressure medications known as angiotensin-converting enzyme (ACE) inhibitors also lower peak phospholipase A2, and some scientists have suggested that the therapeutic efficacy of cardiovascular drugs may depend, to some degree, on their ability to lower markers of vascular inflammation such as Lp-PLA2.2,10 Omega-3 fatty acids and niacin show promise in reducing plasma Lp-PLA2.21,22 Additionally, a protein isolated from the turmeric spice (curcumin) inhibits a form of phospholipase A2 found in the animal kingdom.23 |
||||||
Advantages of the PLAC® TestLp-PLA2 is not influenced by acute illness such as colds and bacterial infections (as occurs with C-reactive protein), and thus serves as a clinically useful biomarker for risk of an acute cardiovascular event. False-positive PLAC® test results may occur in people with high-density lipoprotein (HDL) levels greater than 110 mg/dL, which ordinarily reflects lower cardiovascular risk. This may be attributed to possible genetic differences in individuals with HDL in this range.24
Lp-PLA2 has a valuable predictive value—only 5% of even high-risk patients seem to have a recurrent cardiovascular event in the next four to six years when Lp-PLA2 are in the lower ranges. Physicians can thus monitor Lp-PLA2 levels in high-risk patients to figure out when arterial plaque has been stabilized. A recent study confirms that low levels of Lp-PLA2, in conjunction with other less conventional markers of cardiovascular risk, indicate a very low risk of acute coronary syndrome.25 It should be noted that Lp-PLA2 in an untreated person may rise in time, so a low Lp-PLA2 in apparently healthy people should not be used to assure someone that their lifetime risk is low. Regular monitoring remains important for life-long cardiovascular event prevention. Although LDL is widely used as a marker of cardiovascular risk, it is far less predictive than Lp-PLA2,26,27 which does not rise until plaque is inflamed and advanced. Only 1 in 500 LDL particles may be associated with Lp-PLA2, and LDL may be elevated for decades before Lp-PLA2 rises, signaling a change from chronic atherosclerosis to acute risk for plaque rupture. Lipids levels alone provide little information on the status of artery wall health, whereas Lp-PLA2 is a direct measure of it. Rick Lanman, MD, chief medical officer and executive vice president of diaDexus, manufacturer of the PLAC® test, notes, “You can have a patient with coronary-artery bypass graft surgery who has low Lp-PLA2 because they are on a statin and their plaque has become stable.”
PLAC® Test: Sensitive, Reliable, and Non-InvasiveCurrently, the PLAC® Test is the only blood test cleared by the US Food and Drug Administration (FDA) to aid in assessing risk for both coronary heart disease and ischemic stroke associated with atherosclerosis. The FDA’s 2005 approval of the Lp-PLA2 blood test16 is based on results from the National Heart Lung and Blood Institute’s Atherosclerosis Risk in Communities (ARIC) study, which involved more than 1,300 patients.28 The groundbreaking ARIC study showed that individuals with high levels of Lp-PLA2 have twice the risk of atherosclerotic stroke over the next six to eight years compared with individuals with normal Lp-PLA2 levels. The study also found that individuals with high levels of both C-reactive protein and Lp-PLA2 had the highest risk for future coronary events and stroke, after adjusting for traditional risk factors.28 Imaging tests typically cost much more than the PLAC® test. Other drawbacks of most imaging tests are that they are time consuming and involve radiation exposure. All that is needed for the PLAC® test is a blood sample. Additionally, the PLAC® test is covered by Medicare and many major commercial insurers. No fasting is needed for the PLAC® test, and the patient can be tested while taking commonly used medications such as aspirin, Tylenol®, Benadryl®, and Pravachol®. Interpreting PLAC® Test ResultsAlthough the units measuring Lp-PLA2 levels are different from those used to measure total cholesterol, the ranges associated with different levels of risk are similar. Predictive Lp-PLA2 levels for acute cardiovascular events are:
Levels over 200 or 220 ng/mL are very highly correlated with endothelial dysfunction, which in turn is highly correlated with cardiovascular events and predisposition to atherosclerosis. In practical terms, the PLAC® test can help physicians determine whether someone is at the high end of the low-risk group or at moderate risk. If they have a low Lp-PLA2, less aggressive management strategies may be indicated, while those with a higher level of Lp-PLA2 would more likely need intensive treatment. ConclusionCardiovascular disease prevention is essential for maintaining a long and healthy life span. Yet monitoring risk factors like cholesterol levels and blood pressure has not been enough to predict which individuals are likely to suffer from acute ischemic events. The PLAC® test offers a reliable, inexpensive, and non-invasive means of determining which individuals are at high risk of suffering from potentially deadly cardiovascular events such as heart attack and stroke. The valuable information garnered from this revolutionary technology can help paint a more accurate picture of an individual’s true risk of succumbing to an ischemic event—allowing physicians and patients to implement aggressive risk management strategies to help avert the tragedy of premature cardiovascular death and disability. If you have any questions on the scientific content of this article, please call a Life Extension Health Advisor at 1-800-226-2370. | |||
| References | |||
1. Available at: http://www.webmd.com/heart-disease/news/20080616/tim-russerts-death-questions-answers. Accessed June 16, 2008. 2. Weintraub HS. Identifying the vulnerable patient with rupture-prone plaque. Am J Cardiol. 2008 Jun 16;101(12A):3F-10F. 3. Wilensky RL, Hamamdzic D. The molecular basis of vulnerable plaque: potential therapeutic role for immunomodulation. Curr Opin Cardiol. 2007 Nov;22(6):545-51. 4. Naghavi M, Falk E, Hecht HS, Shah PK. The First SHAPE (Screening for Heart Attack Prevention and Education) Guideline. Crit Pathw Cardiol. 2006 Dec;5(4):187-90. 5. Clarke J. Heart disease and gender in mass print media. Menopause Int. 2008 Mar;14(1):18-20. 6. Lavi S, McConnell JP, Rihal CS, et al. Local production of lipoprotein-associated phospholipase A2 and lysophosphatidylcholine in the coronary circulation: association with early coronary atherosclerosis and endothelial dysfunction in humans. Circulation. 2007 May 29;115(21):2715-21. 7. Davidson MH, Corson MA, Alberts MJ, et al. Consensus panel recommendation for incorporating lipoprotein-associated phospholipase A2 testing into cardiovascular disease risk assessment guidelines. Am J Cardiol. 2008 Jun 16;101(12A):51F-7F. 8. Adibhatla RM, Dempsy R, Hatcher JF. Integration of cytokine biology and lipid metabolism in stroke. Front Biosci. 2008;13:1250-70. 9. Kolodgie FD, Burke AP, Skorija KS, et al. Lipoprotein-associated phospholipase A2 protein expression in the natural progression of human coronary atherosclerosis. Arterioscler Thromb Vasc Biol. 2006 Nov;26(11):2523-9. 10. Mannheim D, Herrmann J, Versari D, et al. Enhanced expression of Lp-PLA2 and lysophosphatidylcholine in symptomatic carotid atherosclerotic plaques. Stroke. 2008 May;39(5):1448-55. 11. Lerman A, McConnell JP. Lipoprotein-associated phospholipase A2: a risk marker or a risk factor? Am J Cardiol. 2008 Jun 16;101(12A):11F-22F. 12. Anderson JL. Lipoprotein-associated phospholipase A2: an independent predictor of coronary artery disease events in primary and secondary prevention. Am J Cardiol. 2008 Jun 16;101(12A):23F-33F. 13. Corson MA, Jones PH, Davidson MH. Review of the evidence for the clinical utility of lipoprotein-associated phospholipase A2 as a cardiovascular risk marker. Am J Cardiol. 2008 Jun 16;101(12A):41F-50F. 14. Yang EH, McConnell JP, Lennon RJ, et al. Lipoprotein-associated phospholipase A2 is an independent marker for coronary endothelial dysfunction in humans. Arterioscler Thromb Vasc Biol. 2006 Jan;26(1):106-11. 15. Daniels LB, Laughlin GA, Sarno MJ, et al. Lipoprotein-associated phospholipase A2 is an independent predictor of incident coronary heart disease in an apparently healthy older population: the Rancho Bernardo Study. J Am Coll Cardiol. 2008 Mar 4;51(9):913-9. 16. Gorelick PB. Lipoprotein-associated phospholipase A2 and risk of stroke. Am J Cardiol. 2008 Jun 16;101(12A):34F-40F. 17. Sullivan LM, Massaro JM, D’Agostino RB Sr. Presentation of multivariate data for clinical use: The Framingham Study risk score functions. Stat Med. 2004 May 30;23(10):1631-60. 18. Raichlin E, McConnell JP, Bae JH, et al. Lipoprotein-associated phospholipase A2 predicts progression of cardiac allograft vasculopathy and increased risk of cardiovascular events in heart transplant patients. Transplantation. 2008 Apr 15;85(7):963-8. 19. Rosenson RS. Fenofibrate reduces lipoprotein associated phospholipase A2 mass and oxidative lipids in hypertriglyceridemic subjects with the metabolic syndrome. Am Heart J. 2008 Mar;155(3):499. 20. Filippatos TD, Gazi IF, Liberopoulos EN, et al. The effect of orlistat and fenofibrate, alone or in combination, on small dense LDL and lipoprotein-associated phospholipase A2 in obese patients with metabolic syndrome. Atherosclerosis. 2007 Aug;193(2):428-37. 21. Schmidt EB, Koenig W, Khuseyinova N, Christensen JH. Lipoprotein-associated phospholipase A2 concentrations in plasma are associated with the extent of coronary artery disease and correlate to adipose tissue levels of marine n-3 fatty acids. Atherosclerosis. 2008 Jan;196(1):420-4. 22. Kuvin JT, Dave DM, Sliney KA, et al. Effects of extended-release niacin on lipoprotein particle size, distribution, and inflammatory markers in patients with coronary artery disease. Am J Cardiol. 2006 Sep 15;98(6):743-5. 23. Chethankumar M, Srinivas L. New biological activity against phospholipase A2 by Turmerin, a protein from Curcuma longa L. Biol Chem. 2008 Mar;389(3):299-303. 24. Kao WT, Yen YC, Lung FW. The effects of beta2 adrenergic receptor gene polymorphism in lipid profiles. Lipids Health Dis. 2008;720. 25. Mockel M, Danne O, Muller R, et al. Development of an optimized multimarker strategy for early risk assessment of patients with acute coronary syndromes. Clin Chim Acta. 2008 Jul;393(2):103-9. 26. Khush KK, Waters D. Lessons from the PROVE-IT trial. Higher dose of potent statin better for high-risk patients. Cleve Clin J Med. 2004 Aug;71(8):609-16. 27. Robins SJ, Collins D, Nelson JJ, Bloomfield HE, Asztalos BF. Cardiovascular events with increased lipoprotein-associated phospholipase A(2) and low high-density lipoprotein-cholesterol: the Veterans Affairs HDL Intervention Trial. Arterioscler Thromb Vasc Biol. 2008 Jun;28(6):1172-8. 28. Ballantyne CM, Hoogeveen RC, Bang H, et al. Lipoprotein-associated phospholipase A2, high-sensitivity C-reactive protein, and risk for incident ischemic stroke in middle-aged men and women in the Atherosclerosis Risk in Communities (ARIC) study. Arch Intern Med. 2005 Nov 28;165(21):2479-84. 29. Brilakis ES, Khera A, McGuire DK, et al. Influence of race and sex on lipoprotein-associated phospholipase A2 levels: observations from the Dallas Heart Study. Atherosclerosis. 2008 Jul;199(1):110-5. 30. El-Saed A, Sekikawa A, Zaky RW, et al. Association of lipoprotein-associated phospholipase A2 with coronary calcification among American and Japanese men. J Epidemiol. 2007 Nov;17(6):179-85. |