Life Extension Magazine®
Absorption of CoQ10
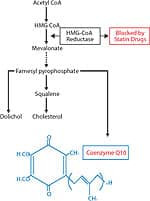
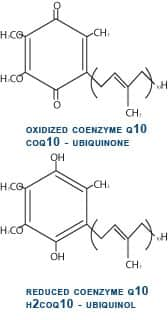
From the beginning of our experience with CoQ10 in heart failure, we have observed poor absorption of CoQ10 in patients with advanced congestive heart failure. This is extremely frustrating, because the patients who are the most ill and have the lowest plasma CoQ10 levels show minimal improvement because of their inability to absorb CoQ10. It has been our assumption that the fluid retention or edema in the intestine and liver in these critically ill patients has been responsible for this poor absorption. Up until approximately one year ago, all supplemental CoQ10 in the world has been in its oxidized or ubiquinone form. In this oxidized state, CoQ10 is stable, bright orange in color, and fat-soluble. It is this property of being fat-soluble that is responsible for the difficulty with CoQ10 absorption. A Better Form of CoQ10After ingestion, immediately after CoQ10 crosses into the first cells lining the small intestine, it is enzymatically converted to its reduced, or ubiquinol, form. So, when we measure plasma CoQ10, approximately 98-99% of the CoQ10 is in this reduced ubiquinol form (see figure 3). Kaneka Corporation of Japan has succeeded in making a stable ubiquinol formulation that we have been carefully studying since October 2006. Ubiquinol is a white powder rather than orange and is less fat-soluble, making it better absorbed. Our first patient to be treated with ubiquinol had severe heart failure with a 15% ejection fraction (a measurement of the amount of blood pumped out with each heartbeat, which is normally 60-65%). This gentleman had a sub-therapeutic plasma CoQ10 level of 2.2 mcg/mL on 450 mg per day of ubiquinone. His plasma level on 450 mg per day of ubiquinol increased dramatically to 8.5 mcg/mL and over the subsequent 10 months, his ejection fraction increased to 60% with a corresponding dramatic and perhaps life-saving clinical improvement (see figure 4). We now have a total of seven patients with advanced congestive heart failure and low plasma CoQ10 levels, despite taking as much as 600 mg per day of ubiquinone (the oxidized form of CoQ10) who have been changed over to the ubiquinol (the reduced form) formulation. Our preliminary observations have been very favorable, and it is my strong opinion that supplemental ubiquinol represents a major scientific advance in the 50-year history of CoQ10 research. Supplementing with CoQ10
I would like to make a few practical comments based on 24 years of treating thousands of heart failure patients with supplemental CoQ10 in addition to standard prescription medications. We have seen no side effects and no drug interactions from supplemental CoQ10, but we have observed a gradual lessening of the requirement for many cardiac medications that occurs with an improvement in heart muscle function. For example, we have observed a significant decrease in the need for diuretics, because of a reduced tendency for fluid retention as heart function improves. Also, we have noted a gradual improvement in hypertension that occurs as heart function improves, which may require a gradual decrease in antihypertensive medications such as angiotensin-converting enzyme (ACE) inhibitors, a class of blood pressure-lowering medicines frequently used in heart failure patients. Patients with heart disease should be followed by their physicians, particularly when there are any changes in activity, diet, prescription drugs, or over-the-counter supplements. I have treated hundreds of patients with CoQ10 and have never observed an interaction with warfarin (Coumadin®). There have been anecdotal reports that the combination may increase the risk of bleeding. Individuals who use warfarin should always consult a physician before using CoQ10.28,29 Choosing a CoQ10 Formulation and DosageMost commercially available CoQ10 supplements comprise ubiquinone. Recommended daily dosages of this type of CoQ10 range from 100 mg to 600 mg. The most advanced CoQ10 formulas now contain ubiquinol, the reduced form of CoQ10, which is definitely better absorbed into the bloodstream.30-32 Recommended daily dosages of ubiquinol range from 100 mg to 300 mg. Because we know that CoQ10 levels tend to decrease with age and we live in a society that consumes very little food rich in CoQ10 (organ meats like heart, liver, and kidney), it makes sense to supplement with a modest amount of CoQ10 (ubiquinone or ubiquinol) beginning in middle age. Those who suffer from congestive heart failure or who use statin medications should aim to consume higher doses of CoQ10 (ubiquinone or ubiquinol). ConclusionCoenzyme Q10’s ability to fundamentally improve the production of energy and the antioxidant defense in every cell of the body has brought about many remarkable and unexpected improvements in all aspects of human health. This extraordinary molecule has dramatically changed my own practice of medicine and has brought joy to the treatment of previously devastating cardiovascular diseases. If you have any questions on the scientific content of this article, please call a Life Extension Health Advisor at 1-800-226-2370. | ||||||||
| References | ||||||||
| 1. Crane FL, Hatefi Y, Lester RL, Widmer C. Isolation of a quinone from beef heart mitochondria. Biochim Biophys Acta. 1957 Jul;25(1):220-1. 2. Folkers K, Littarru GP, Ho L, et al. Evidence for a deficiency of coenzyme Q10 in human heart disease. Int Z Vitaminforsch. 1970;40(3):380-90. 3. Langsjoen PH, Vadhanavikit S, Folkers K. Response of patients in classes III and IV of cardiomyopathy to therapy in a blind and crossover trial with coenzyme Q10. Proc Natl Acad Sci USA. 1985 Jun;82(12):4240-4. 4. Langsjoen PH, Langsjoen PH, Folkers K. Long-term efficacy and safety of coenzyme Q10 therapy for idiopathic dilated cardiomyopathy. Am J Cardiol. 1990 Feb 15;65(7):521-3. 5. Folkers K, Langsjoen P, Willis R, et al. Lovastatin decreases coenzyme Q levels in humans. Proc Natl Acad Sci USA. 1990 Nov;87(22):8931-4. 6. US Patent 4929437, issued May 29, 1990 and US Patent 4933165, issued June 12, 1990, both titled “Coenzyme Q10 with HMG-CoA Reductase Inhibitors.” 7. Silver MA, Langsjoen PH, Szabo S, Patil H, Zelinger A. Effect of atorvastatin on left ventricular diastolic function and ability of coenzyme Q10 to reverse that dysfunction. Am J Cardiol. 2004 Nov 15;94(10):1306-10. 8. Hindler K, Cleeland CS, Rivera E, Collard CD. The role of statins in cancer therapy. Oncologist. 2006 Mar;11(3):306-15. 9. Alsheikh-Ali AA, Maddukuri PV, Han H, Karas RH. Effect of the magnitude of lipid lowering on risk of elevated liver enzymes, rhabdomyolysis, and cancer: insights from large randomized statin trials. J Am Coll Cardiol. 2007 Jul 31;50(5):409-18. 10. Newman TB, Hulley SB. Carcinogenicity of lipid-lowering drugs. JAMA. 1996 Jan 3;275(1):55-60. 11. Li G, Larson EB, Sonnen JA, et al. Statin therapy is associated with reduced neuropathologic changes of Alzheimer disease. Neurology. 2007 Aug 28;69(9):878-85. 12. Golomb BA, Jaworski B. Statins and dementia. Arch Neurol. 2001 Jul;58(7):1169-70. 13. Padala KP, Padala PR, Potter JF. Simvastatin-induced decline in cognition. Ann Pharmacother. 2006 Oct;40(10):1880-3. 14. Kurian KC, Rai P, Sankaran S, et al. The effect of statins in heart failure: beyond its cholesterol-lowering effect. J Card Fail. 2006 Aug;12(6):473-8. 15. Phillips PS, Haas RH, Bannykh S, et al. Statin-associated myopathy with normal creatine kinase levels. Ann Intern Med. 2002 Oct 1;137(7):581-5. 16. Langsjoen PH, Langsjoen JO, Langsjoen AM, Lucas LA. Treatment of statin adverse effects with supplemental Coenzyme Q10 and statin drug discontinuation. Biofactors. 2005;25(1-4):147-52. 17. Bartnik M, Norhammar A, Rydén L. Hyperglycaemia and cardiovascular disease. J Intern Med. 2007 Aug;262(2):145-56. 18. Le NA, Walter MF. The role of hypertriglyceridemia in atherosclerosis. Curr Atheroscler Rep. 2007 Aug;9(2):110-5. 19. Available at: http://circ.ahajournals.org/cgi/content/abstract.Accessed November 29, 2007. 20. Allen Maycock CA, Muhlestein JB, Horne BD, et al. Statin therapy is associated with reduced mortality across all age groups of individuals with significant coronary disease, including very elderly patients. J Am Coll Cardiol. 2002 Nov 20;40(10):1777-85. 21. Sorrentino S, Landmesser U. Nonlipid-lowering effects of statins. Curr Treat Options Cardiovasc Med. 2005 Dec;7(6):459-66. 22. Langsjoen PH, Langsjoen AM. Overview of the use of CoQ10 in cardiovascular disease. Biofactors. 1999;9(2-4):273-84. 23. Langsjoen PH, Littarru GP, Silver MA. Role of concomitant coenzyme Q10 with statins for patients with hyperlipidemia. Curr Topics Nutr Res. 2005;3(3):149-58. 24. Langsjoen PH, Langsjoen AM. Coenzyme Q10 in cardiovascular disease with emphasis on heart failure and myocardial ischaemia. Asia Pacific Heart J. 1998;7(3):160-8. 25. Permanetter B, Rössy W, Klein G, Weingartner F, Seidl KF, Blömer H. Ubiquinone (coenzyme Q10) in the long-term treatment of idiopathic dilated cardiomyopathy. Eur Heart J. 1992 Nov;13(11):1528-33. 26. Watson PS, Scalia GM, Gaibraith AJ, Burstow DJ, Aroney CN, Bett JH. Is coenzyme Q10 helpful for patients with idiopathic cardiomyopathy? Med J Aust. 2001 Oct 15;175(8):447; author reply 447-8. 27. Khatta M, Alexander BS, Krichten CM, et al. The effect of coenzyme Q10 in patients with congestive heart failure. Ann Intern Med. 2000 Apr 18;132(8):636-40. 28. Shalansky S, Lynd L, Richardson K, Ingaszewski A, Kerr C. Risk of warfarin-related bleeding events and supratherapeutic international normalized ratios associated with complementary and alternative medicine: a longitudinal analysis. Pharmacotherapy. 2007 Sep;27(9):1237-47. 29. Engelsen J, Nielsen JD, Winther K. Effect of coenzyme Q10 and ginkgo biloba on warfarin dosage in stable, long-term warfarin treated outpatients. A randomised, double blind, placebo-crossover trial. Thromb Haemost. 2002 Jun;87(6):1075-6. 30. Hosoe K, Kitano M, Kishida H, Kubo H, Fujii K, Kitahara M. Study on safety and bioavailability of ubiquinol (Kaneka QH (trade mark)) after single and 4-week multiple oral administration to healthy volunteers. Regul Toxicol Pharmacol. 2007 Feb;47(1):19-28. 31. Shults CW, Oakes D, Kieburtz K, et al. Effects of coenzyme Q10 in early Parkinson disease: evidence of slowing of the functional decline. Arch Neurol. 2002 Oct;59(10):1541-50. 32. Shults CW, Flint BM, Song D, Fontaine D. Pilot trial of high dosages of coenzyme Q10 in patients with Parkinson’s disease. Exp Neurol. 2004 Aug;188(2):491-4. |