Life Extension Magazine®

If one were to travel back in time to the 1800s, it would have been difficult to imagine that bacterial diseases would ever cease being the leading cause of suffering and death.1
Even highly educated doctors of the era would point out that no one knew what caused tuberculosis, cholera, diphtheria, and other infectious diseases. Educated skeptics could go further and state that assuming the cause of these plagues was someday discovered—could they ever be cured?
No one could have predicted what happened during the following 100 years. Bacteria would be proven to be the cause behind these epidemic killers and antibiotics were developed to cure most of them.
The 20th century was characterized by fantastic advances in life expectancy as factors behind human mortality shifted from bacterial infections to degenerative disease.2
You’re about to discover a novel method to circumvent a major underlying cause of obesity, age-related decline and cessation of life.
What you need to know
- The science of bioenergetics is producing paradigm-shifting discoveries, including the role of AMPK in regulating the ways our bodies use and transform energy.
- AMPK is the “switch” that is the link between metabolic disease, inflammation, and longevity. This “switch” tells our cells when to store and generate energy-containing molecules such as fat, and when to “hunker down” and use existing energy stores.
- When switched “on,” AMPK triggers the use of stored energy from fats, enhances removal of fats and sugar from the blood, increases production of mitochondria, and reduces inflammation and cellular “junk.”
- Calorie restriction and vigorous exercise activate AMPK, shrinking body fat stores (especially in the belly region), lowering blood sugar and lipid levels, and producing other beneficial effects that retard the aging process.
- The drug metformin also activates AMPK, with similar body-wide results.
- A pair of natural botanical extracts have now been found to activate AMPK, reducing belly fat, cholesterol, blood sugar, and insulin levels.
- Instead of combating longevity threats using multiple drugs, supplementation with AMPK-activating botanicals can address these problems at its source.
- Age-induced risk factors can be tackled efficiently by boosting AMPK activity with Gynostemma pentaphyllum and trans-tiliroside from rose hips.
As 2015 approaches, we have a huge advantage over our ancestors who died of bacterial diseases. We already know many of the underlying causes of degenerative disease and are rapidly developing technologies to circumvent them.
In this special issue, we’re introducing you to an enzyme within our cells called AMPK. In youth, AMPK is more activated 3 and helps protect against obesity4 and diabetes.5
As we grow older, cellular AMPK activation decreases, weight gain often follows, and we are more likely to succumb to the destructive factors of aging.6-8
AMPK is found inside every cell and serves as your body’s “master regulating switch.” It determines body fat composition and how long you’ll live.
Unlike previous generations that wallowed in medical ignorance, documented methods exist right now to activate AMPK and in the process, reduce storage of body fat and protect against degenerative disorders that are today’s leading killers.
Scientists view the discovery of AMPK as a major advance in how we understand and combat aging. By reactivating cellular AMPK, we move tantalizingly closer to gaining meaningful control over deadly aging processes, such as excessive weight gain, that predispose us to so many degenerative disorders.
This article will explain the significance of AMPK in age-related disease and reveal several proven ways to enhance activation of this youth-promoting enzyme.
Why AMPK Levels Decline

Most humans today suffer chronic nutritional overload.9 When this happens, vital life functions may become disrupted, such as the proper uptake of glucose10 and fat11 from our blood for effective energy regulation.
In response to excess calorie consumption, cellular housekeeping decreases, resulting in the accumulation of cellular waste12 and damaged proteins.13 Abundant food intake impairs beneficial longevity genes while causing pro-inflammatory genes to become dominant.14
Just imagine what would happen if you never got out of bed and had all meals delivered to you. In the hospital setting, this lack of mobility increases mortality risk. Your cells are no different.
When cells are chronically over-nourished, the energy-mobilizing enzymatic activity of AMPK diminishes.15,16 The outward effects manifest in the form of unwanted weight gain, diabetes, degenerative disease, and premature death.
Few individuals are able to consistently under eat. Fortunately, scientists have identified a cellular enzyme called AMPK that, when activated, mimics many of the beneficial effects observed in calorie restrictors, including loss of surplus body fat.17
Your Cells’ “Master Switch”
A large part of the reason you are alive at this moment is because of the enzymatic reactions18 occurring in the trillions of cells20 in your body.
Cells can be described as little bundles of biochemical reactions. Enzymes often make biochemical reactions in the cell possible. Vital life functions our cells perform are dependent on cellular enzyme activity.
Over the past 30 years, scientists have been investigating the properties of an enzyme called AMPK, which is a “ master switch”21 that, in many ways, controls how our cells behave.
Extensive research shows that by increasing AMPK activation, we can reduce many of the destructive factors of aging, thus enabling cells to return to their youthful vitality.
In preclinical research, enhanced AMPK activity has been associated with a 20-30% increase in life span,7,22,10 but that’s just the beginning of the health benefits conferred by this critical cellular enzyme.
Increased AMPK activation has been shown to help reduce fat storage (especially dangerous belly fat), increase insulin sensitivity (to lower blood glucose), reduce cholesterol/triglyceride production, and suppress chronic inflammation.7,23 All of these factors underlie the lethal diseases of aging.
What Is AMPK?
 |
AMPK is found in every cell in our body.23,24 It serves as the body’s “master regulating switch” that fends off degenerative factors by revitalizing aging cells.
AMPK is involved in reducing fat storage,25 regulating glucose uptake,26,27 creating new mitochondria,28 and eliminating cellular garbage that accumulates inside aging cells.29
The discovery of how AMPK regulates aging processes has been decades in the making. A PubMed search now delivers over 7,500 published articles on various aspects of AMPK. For those who are curious, AMPK is the acronym to define the enzyme adenosine monophosphate-activated protein kinase.30
An important property of AMPK is that it induces multiple longevity factors, which have been shown to increase stress-resistance and extend life span in many organisms.31,32
One such longevity factor boosted by AMPK is the SIRT1 enzyme.7 SIRT1 operates by silencing genes that code for unhealthy responses to stress, including the inflammatory response,33,34 excess fat storage,35 and new fat production.32,36,37 SIRT1 activity is also increased by calorie restriction,38,39 the only fully proven way to increase life span in all animal models tested to date, further indicating the importance of AMPK in activating SIRT1.40,41
Resveratrol found in red wine helps activate SIRT1,42,43 but AMPK activates SIRT1 more directly.7,44,45
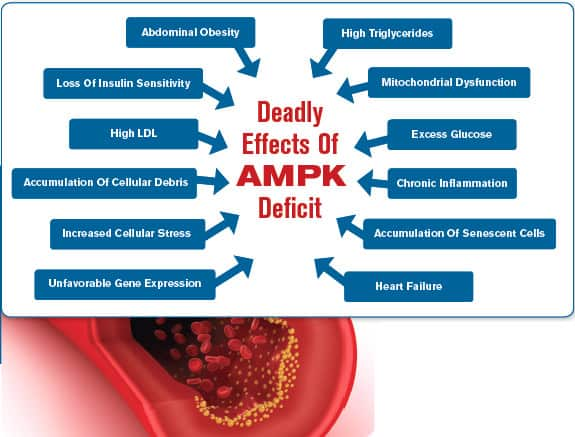
The Dangers Of Reduced AMPK
In many ways, AMPK acts as a traffic cop, efficiently moving excess fat and sugar into our cells to be burned for energy. When we are young, AMPK keeps our metabolic functions running smoothly. Ideally, we are slim and disease-free. But as we age, AMPK signaling declines, which may quickly lead to an excess buildup of blood glucose and dangerous fat accumulation.7 This turns into a lethal combination for many aging humans.
With reduced AMPK signaling, a range of damaging conditions begins to take over a previously healthy body, often leading to an early death. These damaging conditions include:
- Increased belly fat,46,47
- Chronic inflammation,7,46,47
- Elevated blood sugar,46-49
- Insulin resistance,7,46-49
- High cholesterol and triglycerides,46,47
- Decreased numbers and function of mitochondria,7,46,47
- Increased accumulations of abnormal or damaged proteins in our cells that lead to neurodegeneration.7,50
Conventional medicine diagnoses and treats each of the above conditions as a separate disease requiring separate medications when, in fact, they are all associated with one source: declining AMPK activity.
Another deadly impact of reduced AMPK activation is a decreased number of functional mitochondria28,51 and the accumulation of cellular garbage,7,52,53 which eventually renders cells nonfunctional.50,54,55
As scientists continue to unravel the mystery of AMPK, they are discovering that many of the known biochemical longevity factors (such as SIRT1, FoxO, and p53) are activated by normal AMPK function.7 For example, p53 is a tumor-suppressing gene that inhibits uncontrolled cell propagation. Without AMPK, these longevity genes fail to perform their duties, potentially resulting in premature death.7,32
Fortunately, research demonstrates that when AMPK signaling is boosted, these deteriorating processes are reversed,56 restoring more youthful metabolism, preventing related chronic diseases, and potentially adding years of useful, productive life.
How To Boost AMPK
As researchers continue to unravel the mysteries of AMPK, they have discovered four ways to boost the body’s AMPK activity:
- Exercise: AMPK activity increases with regular vigorous exercise.72 This beneficial effect of exercise on AMPK, however, may vanish in the elderly.5
- Calorie Restriction: When you under eat, you create increased AMPK activity as cells sense a requirement to function more efficiently in the presence of diminished energy (food) intake. However, when normal food intake resumes, AMPK activity declines.73,74
- Metformin: One of the drug metformin’s most beneficial mechanisms is to activate AMPK.75 This is one way it lowers elevated glucose.76 Unfortunately, most physicians only prescribe metformin for type II diabetes, making access to this drug difficult for most people. Some people also experience digestive upset in response to metformin and cannot take it.77
- Botanical Extracts: Two natural agents (the Chinese herb Gynostemma pentaphyllum78 and trans -tiliroside derived from rose hips79) have been shown to activate AMPK. Each of these agents triggers different downstream metabolic benefits, and in one study, trans-tiliroside led to an even greater glucose-lowering effect than the AMPK-activating antidiabetic drug metformin.80
With these four documented methods of boosting AMPK signaling, there is no reason for aging humans to suffer the degenerative impact caused by loss of activated AMPK.
Beneficial Effects Of Restoring AMPK
When AMPK is increased, the body kicks into high gear and functions with youthful vitality. Activated AMPK helps remove excess glucose and fats, burning them for energy instead of depositing them in the belly and elsewhere.57,58 To support all this increased fat burning, cells manufacture new mitochondria in a process known as mitochondrial biogenesis.59 These newly created mitochondria burn fuel more cleanly and efficiently than older, worn-out mitochondria,60 resulting in a higher energy output for the entire body.61-63
AMPK-activated cells are generally cleaner, healthier cells. Old cells with decreased AMPK activity tend to become clogged with cellular debris, which reduces their efficiency and shortens their life span. These junk-laden cells accelerate the aging process and contribute to neurodegenerative diseases like Alzheimer’s7 and Parkinson’s,64 and to the thickening and stiffening of vital tissues throughout the body.65,66
Cells that have been energized by AMPK empty their internal garbage cans of accumulated damaged proteins and then convert these damaged proteins for recycling into healthy new ones.55,67 The result of AMPK re-activation is enhanced efficient cellular activity. Finally, increased AMPK activation can suppress body-wide inflammatory responses such as those produced by excess belly fat.62
Over the last few years, research has shown that it is possible to revitalize AMPK to youthful levels of activity. To date, experiments have shown that increased AMPK activation is associated with:
- Extended life span by as much as 20-30% in animals,7,22,104
- Improved glucose uptake in cells, lowering blood sugar,57,68,69
- Inhibiting excess fat synthesis and increased burning of stored fat,17,70
- Reduction of blood triglycerides,71
- Decreased liver fat accumulation and fat-related chronic inflammation,62
- Increased numbers of new, healthy mitochondria.61-63,68
Fat-Loss Effects Of New AMPK-Activator
Gynostemma pentaphyllum is a plant distantly related to the cucumber.
In traditional Asian medicine, it’s used to promote longevity.81 Today’s scientists have discovered why Asian doctors prescribed G. pentaphyllum to address age-related health issues: It promotes AMPK activation.78,82
G. pentaphyllum not only activates AMPK, but it also shuttles excess fats into the mitochondria to be utilized for energy and safe disposal.82 The result is efficient energy production and a sharp reduction in unnecessary fat storage.
Results of G. pentaphyllum-induced AMPK activation include increased fat burning, as well as an increase in cellular glucose uptake.78,82 Extracts of G. pentaphyllum have other beneficial properties as well, including the ability to prolong cellular life in the face of stresses induced by oxidation, fat accumulation, and diabetes.83,84
When scientists began exploring the benefits of G. pentaphyllum for AMPK activation, they turned to animal studies. What they found was that leaf extracts of G. pentaphyllum activate AMPK, resulting in reduced body weight gain and fat accumulation.78 In a preclinical study, obese mice supplemented with G. pentaphyllum showed impressive declines in markers associated with obesity and its related diseases.
In another study, this time using diabetic rats, three weeks of G. pentaphyllum supplementation resulted in improved glucose tolerance by 35% and reduced new glucose production in the liver by 29%, with a reduction in liver glycogen, the storage form of sugar.85
These results show the enormous beneficial impact of reducing circulating sugar and fats in response to AMPK activation by G. pentaphyllum.
Human studies have confirmed what many of the researchers had found in the lab: G. pentaphyllum boosts AMPK activity and provides important longevity benefits.
In a compelling human study, type II diabetics who were not using diabetic medications drank a tea made with G. pentaphyllum. The results compared to controls were:86
- A 5-fold reduction in fasting glucose,
- A 10-fold reduction in hemoglobin A1c, a measure of chronic glucose exposure,
- A near 3-fold decrease in insulin resistance,
- No dangerously low blood sugar episodes, which can often occur with certain oral antidiabetic drugs (especially sulfonylurea class drugs).
In another human study, those taking G. pentaphyllum significantly boosted the effects of a sulfonylurea antidiabetic drug, producing an additional fasting glucose reduction of 52.2 mg/dL compared with just 16.2 mg/dL for the drug alone.87
None of these findings should be surprising since the prescription drug metformin, which is an AMPK activator, produces many of these same benefits.88,89
AMPK-Boosting Effects Of Trans-Tiliroside
The second natural compound found to increase AMPK activity is trans- tiliroside, extracted from plants such as rose hips.
Trans- tiliroside also boosts AMPK signaling, but it triggers different downstream metabolic benefits than G. pentaphyllum.79,90,91 Combining these two bioactives (G. pentaphyllum and trans-tiliroside) provides broader AMPK activation effects than each one separately.
Scientists are now finding that trans-tiliroside increases the GLUT4 transporter in cell membranes, which helps pull excess sugar out of the blood and into cells, where it is burned for energy, thereby reducing circulating blood glucose.91
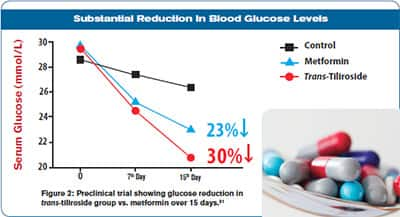
In animal studies, trans-tiliroside supplementation significantly reduces dangerous after-meal glucose spikes while also suppressing surges in insulin.92 A laboratory study of insulin-resistant human liver cells found that trans -tiliroside boosted cellular glucose consumption in a manner that compared favorably to metformin, a widely used antidiabetic drug.93
Not surprisingly, in mouse models of diabetes, daily oral administration of trans-tiliroside reduced fasting blood sugar by up to nearly 30% after 15 days of treatment, while diabetic mice treated with metformin had a near 23% reduction.80 Supplemented animals also had significant reductions in serum triglycerides and total cholesterol, while experiencing beneficial increases in HDL cholesterol levels.
And in obese-diabetic mice, supplementation with trans-tiliroside increased fat burning, lowered plasma insulin, lowered free fatty acids, and lowered triglycerides, while increasing levels of adiponectin, a protein hormone that regulates glucose and breaks down fat.90 Moreover, oral supplementation of trans-tiliroside in mice for two weeks with the human equivalent dose of 56 mg/day significantly reduced plasma glucose levels.94
Along with reducing the dangers of excess blood glucose, trans-tiliroside has been shown to battle obesity, especially by reducing cellular fat. In a laboratory experiment, trans-tiliroside also demonstrated reduced plasma cholesterol and a lower ratio of LDL to HDL (which means beneficial HDL levels increased in relation to atherogenic LDL).80
Another study showed that rose hip powder prevented obesity in lean mice on a high-fat diet and reversed weight gain and body fat mass increases in obese mice on the same diet.95 Baseline levels of glucose and insulin were lowered and glucose tolerance was improved.
An additional manifestation of AMPK activation resulted in a reduction in liver fat stores, the result of reduced fat production.95 Still more impressively, studies have now shown that trans-tiliroside can significantly inhibit the accumulation of visceral fat and gain in body weight in mice after just two weeks’ supplementation, without any change in food intake at the human equivalent dose of 56 mg/day.94
Are AMPK Activators Weight-Loss Agents?

When AMPK is activated, cells go into a survival mode, making and storing no new fat, while burning up available fat from storage, pumping glucose in from the blood for additional energy, building new mitochondria for more efficient energy use, and recycling damaged or dysfunctional proteins. These are all beneficial anti-aging mechanisms.
The net effects of AMPK activation closely resemble those of calorie restriction and vigorous exercise. This is because both calorie restriction and vigorous exercise activate AMPK in response to energy needs that exceed supply. The drug metformin independently activates AMPK, producing similar benefits, which include reduction in dangerous belly fat stores, general weight loss, and reductions in blood sugar and fat levels.77,96,97
G. pentaphyllum and trans-tiliroside have induced profound weight-loss effects in controlled studies. The question is how well will they work in real-world settings, where overweight individuals often over-consume so many calories that no drug, hormone, or supplement can induce meaningful fat loss.
The answer to the question of the weight-loss benefits of AMPK activators can be analogized to cancer patients who take aggressive steps to boost immune function. Restoring optimal immune status alone will not cure most malignancies, but failing to address immune deficits places cancer patients at significantly greater risks of dying.
Likewise, we suspect that G. pentaphyllum and trans-tiliroside by themselves are not going to enable significantly overweight individuals to become normal weight. But if one is serious about shedding excess body fat, and they don’t take steps to reactivate their cellular AMPK, it may be less likely that they will be able to achieve their fat-loss objectives.
In response to aging, vigorous exercise may not induce an AMPK activating effect5 and most people are unable to consistently restrict their caloric intake.
It’s abundantly clear that those seeking to shed excess fat (especially in the belly) should take measures to activate their AMPK so that they may assist the body in restoring the youthful enzymatic activity needed to help safely manage body weight.
Deadly Delays…
 |
What History Has Taught Us
In 1796, Edward Jenner demonstrated that inoculation using fluid from cowpox lesions could safely prevent smallpox.101
This research was initially rejected, but by 1840, the British government provided Jenner’s smallpox vaccine free of charge.
What few people realize is that 700 years before Jenner’s discovery, the Chinese had developed a crude form of inoculation. Yet no one in Europe paid attention despite widespread smallpox outbreaks that ravaged all social classes.
Even more surprising are the 300 million people who perished from smallpox in the 20th century102 because of the delay in implementing universal smallpox vaccination.
When Edward Jenner discovered the smallpox vaccine, no one knew that viruses existed. They only observed that those inoculated with his vaccine were immune from smallpox for life.
Gynostemma pentaphyllum was first described in a Chinese medical book in 1406.103 Doctors in the following centuries discovered the multiple disease-fighting benefits this herb conveyed, but they had no idea how it worked in the body. Back in those days, no one knew about blood glucose and certainly nothing was understood about something as sophisticated as AMPK.
All doctors could do in early centuries was observe the beneficial effects that occurred when patients were treated with Gynostemma pentaphyllum.
Today we know that declining AMPK activity predisposes humans to degenerative diseases, and we know of validated methods to markedly increase AMPK signaling. One low-cost approach to activating AMPK is to take 450 mg of standardized Gynostemma pentaphyllum extract daily plus 56 mg of trans -tiliroside for optimal effects.
Summary
Activating AMPK represents a major advance in our ability to combat degenerative disease and obesity. AMPK is now being intensively studied in the prevention and treatment of many age-related disorders.98,99
When AMPK is activated, cells decrease production and storage of new fat and burn stored body fat. In addition, AMPK activation results in glucose being pumped out of the blood and into cells for additional energy. Mitochondrial biogenesis occurs, leading to more efficient energy use while cellular debris and dysfunctional proteins are removed.
The net effects of AMPK activation closely resemble those of calorie restriction and vigorous exercise. This occurs because both calorie restriction and vigorous exercise activate AMPK in response to energy needs that exceed supply. (Exercise may not increase AMPK in elderly individuals.)5
Gynostemma pentaphyllum and trans-tiliroside, two naturally sourced extracts, uniquely increase AMPK activation without drugs, severe calorie restriction, or exercise. Studies show that these supplements significantly reduce risky belly fat accumulation, promote weight loss, normalize blood sugar, improve insulin resistance, and lower serum lipid levels.
When looking at the chart on this page, one can see that human life expectancy has more than doubled since the founding years of our country.
What’s encouraging is that these averages include people who don’t take care of themselves (including tobacco smokers and those who eat an unhealthy diet). This means that individuals who follow healthier lifestyles, which includes taking steps to activate AMPK, may add many more healthy years to their projected life spans.
According to the latest data, a man reaching age 65 today can expect to live on average until age 84.3. Women have it better and can expect to live to 86.6 if they make it to age 65. One out of every four 65-year-olds will live past age 90.100 Life Extension®’s healthy longevity objectives stretch far beyond this.
Activation of the AMPK enzyme is a critical component in warding off degenerative disease. As stated in the beginning of this article, vital life functions are dependent on cellular enzyme activity.
By activating cellular AMPK, we provide our body with the opportunity to reverse physical and mental decline associated with aging, buying us time to take advantage of future longevity breakthroughs that will markedly extend healthy human life spans.
If you have any questions on the scientific content of this article, please call a Life Extension® Wellness Specialist at 1-866-864-3027.
References
- Nelson, KE, Williams, CM. Infectious Disease Epidemiology. Burlington, MA: Jones & Bartlett Publishers; 2014.
- De Flora S, Quaglia A, Bennicelli C, Vercelli M. The epidemiological revolution of the 20th century. FASEB J. 2005;19:892–7.
- Hardman SE, Hall DE, Cabrera AJ, Hancock CR, Thomson DM. The effects of age and muscle contraction on AMPK activity and heterotrimer composition. Exp Gerontol. 2014 Jul;55:120-8.
- Yang Z, Kahn BB, Shi H, Xue BZ. Macrophage alpha1 AMP-activated protein kinase (alpha1AMPK) antagonizes fatty acid-induced inflammation through SIRT1. J Biol Chem. 2010 Jun 18;285(25):19051-9.
- Mortensen B, Poulsen P, Wegner L, et al. Genetic and metabolic effects on skeletal muscle AMPK in young and older twins. Am J Physiol Endocrinol Metab. 2009;297:E956–E64.
- Liu F, Benashski SE, Persky R, Xu Y, Li J, McCullough LD. Age-related changes in AMP-activated protein kinase after stroke. Age (Dordr). 2012;34(1):157-68.
- Salminen A, Kaarniranta K. AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res Rev. 2012 Apr;11(2):230-41.
- Rojas J, Arraiz N, Aguirre M, Velasco M, Bermúdez V. AMPK as target for intervention in childhood and adolescent obesity. J Obes. 2011;2011:252817.
- Ford ES, Dietz WH. Trends in energy intake among adults in the United States: findings from NHANES. Am J Clin Nutr. 2013;97(4):848-53.
- Büsing KA, Schönberg SO, Brade J, Wasser K. Impact of blood glucose, diabetes, insulin, and obesity on standardized uptake values in tumors and healthy organs on 18F-FDG PET/CT. Nucl Med Biol. 2013 Feb;40(2):206-13.
- Blaak EE. Fatty acid metabolism in obesity and type 2 diabetes mellitus. Proc Nutr Soc. 2003 Aug;62(3):753-60.
- Choi JH, Kim D. Effects of age and dietary restriction on life span and oxidative stress of SAMP8 mice with learning and memory impairments. J Nutr Health Aging. 2000;4(3):182-6.
- Poon HF, Shepherd HM, Reed TT, Calabrese V, Stella AMG, Pennisi G, et al. Proteomics analysis provides insight into caloric restriction mediated oxidation and expression of brain proteins associated with age-related impaired cellular processes: Mitochondrial dysfunction, glutamate dysregulation and impaired protein synthesis. Neurobiol Aging. 2006 Jul;27(7):1020-34.
- Chalkiadaki A, Guarente L. High-fat diet triggers inflammation-induced cleavage of SIRT1 in adipose tissue to promote metabolic dysfunction. Cell Metab. 2012 Aug 8;16(2):180-8.
- Coughlan KA, Valentine RJ, Ruderman NB, Saha AK. Nutrient excess in AMPK downregulation and insulin resistance. J Endro Diabetes Obes. 2013; 123(7):2764–72.
- Saha AK, Xu XJ, Balon TW, Brandon A, Kraegen EW, Ruderman NB. Insulin resistance due to nutrient excess: is it a consequence of AMPK downregulation? Cell Cycle. 2011 Oct 15;10(20):3447-51.
- Luo Z, Zang M, Guo W. AMPK as a metabolic tumor suppressor: control of metabolism and cell growth. Future Oncol. 2010 Mar;6(3):457-70.
- Gurung N, Ray S, Bose S, Rai V. A broader view: microbial enzymes and their relevance in industries, medicine, and beyond. Biomed Res Int. 2013;2013:329121.
- Available at: http://publications.nigms.nih.gov/chemhealth/act.htm. Accessed August 23, 2014.
- Available at: http://www.smithsonianmag.com/smart-news/there-are-372-trillion-cells-in-your-body-4941473/?no-ist. Accessed August 23, 2014.
- Winder WW, Hardie DG. AMP-activated protein kinase, a metabolic master switch: possible roles in type 2 diabetes. Am J Physiol. 1999 Jul;277(1 Pt 1):E1-10.
- Stenesen D, Suh JM, Seo J, et al. Adenosine nucleotide biosynthesis and AMPK regulate adult life span and mediate the longevity benefit of caloric restriction in flies. Cell Metab. 2013 Jan 8;17(1):101-12.
- Towler MC, Hardie DG. AMP-activated protein kinase in metabolic control and insulin signaling. Circ Res. 2007 Feb 16;100(3):328-41.
- Zelenak C, Föller M, Velic A, et al. Proteome analysis of erythrocytes lacking AMP-activated protein kinase reveals a role of PAK2 kinase in eryptosis. J Proteome Res. 2011 Apr 1;10(4):1690-7.
- Villena JA, Viollet B, Andreelli F, Kahn A, Vaulont S, Sul HS. Induced adiposity and adipocyte hypertrophy in mice lacking the AMP-activated protein kinase-alpha2 subunit. Diabetes. 2004 Sep;53(9):2242-9.
- Friedrichsen M, Mortensen B, Pehmøller C, Birk JB, Wojtaszewski JF. Exercise-induced AMPK activity in skeletal muscle: role in glucose uptake and insulin sensitivity. Mol Cell Endocrinol. 2013 Feb 25;366(2):204-14.
- Rutter GA, Da Silva Xavier G, Leclerc I. Roles of 5’-AMP-activated protein kinase (AMPK) in mammalian glucose homoeostasis. Biochem J. 2003 Oct 1;375(Pt 1):1-16.
- Yan W, Zhang H, Liu P, Wang H, Liu J, Gao C, et al. Impaired mitochondrial biogenesis due to dysfunctional adiponectin-AMPK-PGC-1α signaling contributing to increased vulnerability in diabetic heart. Basic Res Cardiol. 2013 May;108(3):329.
- Mihaylova MM, Shaw RJ. The AMPK signaling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol. 2011 Sep 2;13(9):1016-23.
- Jeong KJ, Kim GW, Chung SH. AMP-activated protein kinase: An emerging target for ginseng. J Ginseng Res. 2014 Apr;38(2):83-8.
- Liu WY, Jiang RS. Advances in the research of AMPK and its subunit genes. PJBS. Nov 15 2013;16(22):1459-68.
- Salminen A, Hyttinen JM, Kaarniranta K. AMP-activated protein kinase inhibits NF-kB signaling and inflammation: impact on health span and life span. J Mol Med. 2011;89(7):667-76.
- Hwang JW, Yao H, Caito S, Sundar IK, Rahman I. Redox regulation of SIRT1 in inflammation and cellular senescence. Free Radic Biol Med. 2013 Mar 27;61C:95-110.
- Yang H, Zhang W, Pan H, Feldser HG, Lainez E, Miller C, et al. SIRT1 activators suppress inflammatory responses through promotion of p65 deacetylation and inhibition of NF-κB activity. PLoS One. 2012;7(9):e46364.
- Picard F, Kurtev M, Chung N, Topark-Ngarm A, Senawong T, Machado De Oliveira R, et al. Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-gamma. Nature. 2004 Jun 17;429(6993):771-6.
- Um JH, Pendergast JS, Springer DA, et al. AMPK regulates circadian rhythms in a tissue- and isoform-specific manner. PLoS One. 2011;6(3):e18450.
- Jin Q, Zhang F, Yan T, Liu Z, Wang C, Ge X, Zhai Q. C/EBPalpha regulates SIRT1 expression during adipogenesis. Cell Res. 2010 Apr;20(4):470-9.
- Yu W, Zhou HF, Lin RB, Fu YC, Wang W. Short term calorie restriction activates SIRT1 4 and 7 in cardiomyocytes in vivo and in vitro. Mol Med Rep. 2014 Apr;9(4):1218-24.
- Chen D, Bruno J, Easlon E, Lin SJ, Cheng HL, Alt FW, Guarente L. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 2008 Jul 1;22(13):1753-7.
- Cantó C, Auwerx J. Caloric restriction, SIRT1 and longevity . Trends Endocrinol Metab. 2009 Sep;20(7):325-31.
- Cohen DE , Supinski AM,Bonkowski MS,Donmez G, Guarente LP. Neuronal SIRT1 regulates endocrine and behavioral responses to calorie restriction. Genes Dev. 2009 Dec 15;23(24): 2812-7.
- Ghosh S, Liu B, Zhou Z. Resveratrol activates SIRT1 in a lamin A-dependent manner. Cell Cycle. 2013 Mar 15;12(6):872-6.
- Liu B, Zhou Z. Activation of SIRT1 by resveratrol requires lamin A. Aging (Albany NY). 2013 Feb;5(2):94-5.
- Srivastava RA, Pinkosky SL, Filippov S, Hanselman JC, Cramer CT, Newton RS. AMP-activated protein kinase: an emerging drug target to regulate imbalances in lipid and carbohydrate metabolism to treat cardio-metabolic diseases. J Lipid Res. 2012 Dec;53(12):2490-514.
- Lee CW, Wong LL, Tse EY, Liu HF, Leong VY, Lee JM, et al. AMPK promotes p53 acetylation via phosphorylation and inactivation of SIRT1 in liver cancer cells. Cancer Res. 2012 Sep 1;72(17):4394-404.
- Steinberg GR, Kemp BE. AMPK in health and disease. Physiol Rev. 2009 89: 1025-78.
- Viollet B, Horman S, Leclerc J, Lantier L, Foretz M, Billaud M, et al. AMPK inhibition in health and disease. Crit Rev Biochem Mol Biol. 2010 Aug; 45(4): 276-95.
- Ruderman N, Prentki M. AMP kinase and malonyl-CoA: targets for therapy of the metabolic syndrome. Nat Rev Drug Discov. 2004 Apr;3(4):340-51.
- Dugan LL, You YH, Ali SS, Diamond-Stanic M, Miyamoto S, DeCleves AE, et al. AMPK dysregulation promotes diabetes-related reduction of superoxide and mitochondrial function. J Clin Invest. 2013 Nov 1;123(11):4888-99.
- Poels J, Spasic MR, Callaerts P, Norga KK. An appetite for destruction: from self-eating to cell cannibalism as a neuronal survival strategy. Autophagy. 2012 Sep;8(9):1401-3.
- Jornayvaz FR, Shulman GI. Regulation of mitochondrial biogenesis. Essays Biochem. 2010;47:69-84.
- Hardie DG. AMPK and autophagy get connected. EMBO J. 2011 Feb 16;30(4):634-5.
- Possik E, Jalali Z, Nouët Y, Yan M, Gingras MC, Schmeisser K, et al. Folliculin regulates AMPK-dependent autophagy and metabolic stress survival. PLoS Genet. 2014 Apr 24;10(4):e1004273.
- She C, Zhu LQ, Zhen YF, Wang XD, Dong QR. Activation of AMPK protects against hydrogen peroxide-induced osteoblast apoptosis through autophagy induction and NADPH maintenance: new implications for osteonecrosis treatment? Cell Signal. 2014 Jan;26(1):1-8.
- Egan DF, Shackelford DB, Mihaylova MM, et al. Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science. 2011;331:456-61.
- McCarty MF. AMPK activation--protean potential for boosting healthspan. Age (Dordr). 2014 Apr;36(2):641-63.
- Habegger KM, Hoffman NJ, Ridenour CM, Brozinick JT, Elmendorf JS. AMPK enhances insulin-stimulated GLUT4 regulation via lowering membrane cholesterol. Endocrinology. 2012 May;153(5):2130-41.
- Abbott MJ, Constantinescu S, Turcotte LP. AMP-activated protein kinase alpha2 is an essential signal in the regulation of insulin-stimulated fatty acid uptake in control-fed and high-fat-fed mice. Exp Physiol. 2012 May;97(5):603-17.
- Zong H, Ren JM, Young LH, Pypaert M, Mu J, Birnbaum MJ, et al. AMP kinase is required for mitochondrial biogenesis in skeletal muscle in response to chronic energy deprivation. Proc Natl Acad Sci USA. 2002 Dec 10;99(25):15983-7.
- Calvani R, Joseph AM, Adhihetty PJ, Miccheli A, Bossola M, Leeuwenburgh C, Bernabei R, Marzetti E. Mitochondrial pathways in sarcopenia of aging and disuse muscle atrophy. Biol Chem. 2013 Mar;394(3):393-414.
- Niemann B, Pan R, Teschner M, Boening A, Silber RE, Rohrbach S. Age and obesity-associated changes in the expression and activation of components of the AMPK signaling pathway in human right atrial tissue. Exp Gerontol. 2013 Jan;48(1):55-63.
- Bijland S, Mancini SJ, Salt IP. Role of AMP-activated protein kinase in adipose tissue metabolism and inflammation. Clin Sci (Lond). 2013 Apr;124(8):491-507.
- Turdi S, Fan X, Li J, et al. AMP-activated protein kinase deficiency exacerbates aging-induced myocardial contractile dysfunction. Aging Cell. 2010 Aug;9(4):592-606.
- Hindle JV. Ageing, neurodegeneration and Parkinson’s disease. Age Ageing. 2010 Mar;39(2):156-61.
- Patel AS, Lin L, Geyer A, Haspel JA, An CH, et al. Autophagy in Idiopathic Pulmonary Fibrosis. PLoS One. 2012;7(7):e41394
- Hermida N, Markl A, Hamelet J, et al. HMGCoA reductase inhibition reverses myocardial fibrosis and diastolic dysfunction through AMP-activated protein kinase activation in a mouse model of metabolic syndrome. Cardiovasc Res. 2013 Jul 1;99(1):44-54.
- Mao K, Klionsky DJ. AMPK activates autophagy by phosphorylating ULK1. Circ Res. 2011 Apr 1;108(7):787-8.
- O’Neill HM, Maarbjerg SJ, Crane JD, et al. AMP-activated protein kinase (AMPK) beta1beta2 muscle null mice reveal an essential role for AMPK in maintaining mitochondrial content and glucose uptake during exercise. Proc Natl Acad Sci USA. 2011 Sep 20;108(38):16092-7.
- Andrade BM, Cazarin J, Zancan P, Carvalho DP. AMP-activated protein kinase upregulates glucose uptake in thyroid PCCL3 cells independent of thyrotropin. Thyroid. 2012 Oct;22(10):1063-8.
- Brusq JM, Ancellin N, Grondin P, et al. Inhibition of lipid synthesis through activation of AMP kinase: an additional mechanism for the hypolipidemic effects of berberine. J Lipid Res. 2006 Jun;47(6):1281-8.
- Aguilera-Mendez A, Fernandez-Mejia C. The hypotriglyceridemic effect of biotin supplementation involves increased levels of cGMP and AMPK activation. Biofactors. 2012 Sep-Oct;38(5):387-94.
- Hardie DG. AMP-activated protein kinase: a key system mediating metabolic responses to exercise. Med Sci Sports Exerc. 2004 Jan;36(1):28-34.
- Lee SH, Min KJ. Caloric restriction and its mimetics. BMB Rep. 2013 Apr;46(4):181-7.
- Ribaric S. Diet and aging. Oxid Med Cell Longev. 2012;2012: 741468.
- Hawley SA, Gadalla AE, Olsen GS, Hardie DG. The antidiabetic drug metformin activates the AMP-activated protein kinase cascade via an adenine nucleotide-independent mechanism. Diabetes. 2002 Aug;51(8):2420-5.
- Kristensen JM, Treebak JT, Schjerling P, Goodyear L, Wojtaszewski JF. Two weeks of metformin treatment induces AMPK-dependent enhancement of insulin-stimulated glucose uptake in mouse soleus muscle. Am J Physiol Endocrinol Metab. 2014 May 15;306(10):E1099-109.
- Fowler MJ. Diabetes treatment, part 2: oral agents for glycemic management. Clin Diabet. 2007;25(4):131-4.
- Gauhar R, Hwang SL, Jeong SS, et al. Heat-processed Gynostemma pentaphyllum extract improves obesity in ob/ob mice by activating AMP-activated protein kinase. Biotechnol Lett. 2012 Sep;34(9):1607-16.
- Nagatomo A, Nishida N, Matsuura Y, Shibata N. Rosehip Extract Inhibits Lipid Accumulation in White Adipose Tissue by Suppressing the Expression of Peroxisome Proliferator-activated Receptor Gamma. Prev Nutr Food Sci. 2013 Jun;18(2):85-91.
- Qiao W, Zhao C, Qin N, Zhai HY, Duan HQ. Identification of trans-tiliroside as active principle with anti-hyperglycemic, anti-hyperlipidemic and antioxidant effects from Potentilla chinesis. J Ethnopharmacol. 2011 May 17;135(2):515-21.
- Li XL, Wang ZH, Zhao YX, Luo SJ, Zhang DW, Xiao SX, et al. Isolation and antitumor activities of acidic polysaccharide from Gynostemma pentaphyllum Makino. Carbohydr Polym. 2012 Jul 1;89(3):942-7.
- Nguyen PH, Gauhar R, Hwang SL, et al. New dammarane-type glucosides as potential activators of AMP-activated protein kinase (AMPK) from Gynostemma pentaphyllum. Bioorg Med Chem. 2011 Nov 1;19(21):6254-60.
- Lobo SN, Qi YQ, Liu QZ. The effect of Gynostemma pentaphyllum extract on mouse dermal fibroblasts. ISRN Dermatol. 2014 Mar 4.
- Muller C, Gardemann A, Keilhoff G, Peter D, Wiswedel I, Schild L. Prevention of free fatty acid-induced lipid accumulation, oxidative stress, and cell death in primary hepatocyte cultures by a Gynostemma pentaphyllum extract. Phytomedicine. 2012 Mar 15;19(5):395-401.
- Yassin K, Huyen VT, Hoa KN, Ostenson CG. Herbal extract of gynostemma pentaphyllum decreases hepatic glucose output in type 2 diabetic goto-kakizaki rats. Int J Biomed Sci. 2011 Jun;7(2):131-6.
- Huyen VT, Phan DV, Thang P, Hoa NK, Ostenson CG. Antidiabetic effect of Gynostemma pentaphyllum tea in randomly assigned type 2 diabetic patients. Horm Metab Res. 2010 May;42(5):353-7.
- Huyen VT, Phan DV, Thang P, Ky PT, Hoa NK, Ostenson CG. Antidiabetic Effects of Add-On Gynostemma pentaphyllum extract therapy with sulfonylureas in type 2 diabetic patients. Evid Based Complement Alternat Med. 2012;2012:452313.
- Chilton M, Dunkley A, Carter P, Davies MJ, Khunti K, Gray LJ. The effect of antiobesity drugs on waist circumference: a mixed treatment comparison. Diabetes Obes Metab. 2014 Mar;16(3):237-47.
- Diabetes Prevention Program Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care. 2012 Apr;35(4):731-7.
- Goto T, Teraminami A, Lee JY, et al. Tiliroside, a glycosidic flavonoid, ameliorates obesity-induced metabolic disorders via activation of adiponectin signaling followed by enhancement of fatty acid oxidation in liver and skeletal muscle in obese-diabetic mice. J Nutr Biochem. 2012 Jul;23(7):768-76.
- Shi L, Qin N, Hu L, Liu L, Duan H, Niu W. Tiliroside-derivatives enhance GLUT4 translocation via AMPK in muscle cells. Diabetes Res Clin Pract. 2011 May;92(2):e41-6.
- Goto T, Horita M, Nagai H, et al. Tiliroside, a glycosidic flavonoid, inhibits carbohydrate digestion and glucose absorption in the gastrointestinal tract. Mol Nutr Food Res. 2012 Mar;56(3):435-45.
- Zhu Y, Zhang Y, Liu Y, Chu H, Duan H. Synthesis and biological activity of trans-tiliroside derivatives as potent anti-diabetic agents. Molecules. 2010;15(12):9174-83.
- Ninomiya K, Matsuda H, Kubo M, Morikawa T, Nishida N, Yoshikawa M. Potent anti-obese principle from Rosa canina: structural requirements and mode of action of trans-tiliroside. Bioorg Med Chem Lett. 2007 Jun 1;17(11):3059-64.
- Andersson U, Henriksson E, Strom K, Alenfall J, Goransson O, Holm C. Rose hip exerts antidiabetic effects via a mechanism involving downregulation of the hepatic lipogenic program. Am J Physiol Endocrinol Metab. 2011 Jan;300(1):E111-21.
- Matsui Y, Hirasawa Y, Sugiura T, Toyoshi T, Kyuki K, Ito M. Metformin reduces body weight gain and improves glucose intolerance in high-fat diet-fed C57BL/6J mice. Biol Pharm Bull. 2010;33(6):963-70.
- Yanovski JA, Krakoff J, Salaita CG, et al. Effects of metformin on body weight and body composition in obese insulin-resistant children: a randomized clinical trial. Diabetes. 2011 Feb;60(2):477-85.
- Samuel MA, Voinescu PE, Lilley BN, et al. LKB1 and AMPK regulate synaptic remodeling in old age. Nat Neurosci. 2014 Aug 3.
- North BJ, Sinclar DA. The intersection between aging and cardiovascular disease. Circ Res.2012;110: 1097-1108.
- Available at: http://www.ssa.gov/planners/lifeexpectancy.htm. Accessed August 23, 2014.
- Riedel S. Edward Jenner and the history of smallpox and vaccination. Proc (Bayl Univ Med Cent). 2005 Jan;18(1):21-5.
- Available at: http://www.bbc.co.uk/history/british/empire_seapower/smallpox_01.shtml. Accessed August 23, 2014.
- Mishra RN, Joshi D. Jiao gu lan (Gynostemma pentaphyllum): The Chinese rasayan-current research scenario. IJRPBS. 2011;2(4)1483-1502.
- Ulgherait M, Rana A, Rera M, Graniel J, Walker DW. AMPK modulates tissue and organismal aging in a non-cell-autonomous manner. Cell Rep. 2014 Sep 3.

