Life Extension Magazine®
As longevity scientists look to target and stop aging, many researchers are focusing on geroprotectors, compounds capable of preventing or even reversing aging at the cellular level.
Senescent cells are particularly troublesome when they enter the stage in which they can no longer properly divide and function. As cells become dysfunctional, organ health markedly deteriorates.
Another problem caused by cell senescence is release of proinflammatory cytokines that systemically damage tissues.
Compounds capable of identifying and eliminating senescent cells are categorized as senolytics.
Clinical research on geroprotectors and senolytics is complicated by the fact that many decades of time may be required to determine human longevity benefits.
A novel way to accelerate the research is via the strategic use of high-speed computer programs employing artificial intelligence biomedical algorithms. This technology has advanced to where it can identify natural compounds that activate anti-aging pathways throughout the body.
Seeing the enormous potential of this kind of deep-learning artificial intelligence, Life Extension® has partnered with Insilico Medicine to identify nutrient cocktails that function as geroprotectors. The objective is to develop better ways to potentially slow or even stop certain aspects of aging processes.
This scientific collaboration has resulted in the identification of a geroprotector formulation consisting of four nutrients with various complementary and reinforcing properties. These include effects on cell signaling pathways with the potential to prevent the degenerative progression of damaged cells to a senescent state—and eliminate those that have reached irreversible senescence.1,2
With the discovery of geroprotective compounds, there are now greater opportunities to more effectively intervene into cellular aging processes.
By harnessing artificial intelligence advances with existing scientific data, a powerful new weapon to combat degenerative aging has emerged.
Using Artificial Intelligence to Solve Aging
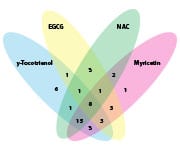
Activation - Figure 1.
The numerals within the diagram
indicate the number of pathways
activated by the four short-
listed nutrients.
Life Extension and Insilico Medicine researchers ran computer simulations of more than 200 potential geroprotective compounds and narrowed the list down to four.1,2 While most of these candidates were known natural nutrients, their full potential as geroprotectors remained elusive.
Scientists found that all four of these nutrients work together, but in very different ways, to beneficially influence key anti-aging pathways. Together, they combat numerous aging factors throughout the body.
These compounds all modulate specific biological pathways responsible for keeping us young and healthy.
When combined, these ingredients promote anti-aging mechanisms at the cellular level throughout the body, acting by multiple pathways, some unique, and some overlapping.
Together, these four natural compounds represent the beginning of the future: anti-aging cocktails identified using artificial intelligence under expert human supervision.
Geroprotectors and Senolytic Agents Extend Lifespan

The concept of geroprotection, meaning prevention of cells from entering an aged, senescent, inflammation-promoting state, originated in Soviet-era Russia in the late 1970s.
Modern science has uncovered a variety of natural substances capable of extending healthy lifespan, and these agents have been called geroprotectors.
Scientists then discovered that several of these nutrients function as senolytic agents.
The term senolytic means the removal of useless senescent cells from tissues.3,4 Cellular senescence is characterized by the aged cell’s inability to divide, yet the senescent cell continues to generate a variety of proinflammatory compounds as well as other harmful cellular mediators that can potentiate damage to healthy tissues.
Cancer researchers were first intrigued by the idea of developing senolytic drugs that could selectively kill malignant cancer cells without damaging normal cells in the body.5,6
Based on this research, scientists have realized that senolytics can enhance healthspan by eliminating aging cells from our tissues, making room for newer, active cells that support youthful tissue, organ, and systems functioning. The result is a younger, healthier and better-functioning body.
Why should we care about getting rid of these senescent cells?
Because they contribute to virtually all known age-associated disorders: heart disease, stroke, cancer, diabetes, obesity, liver and kidney disease, osteoporosis, and neurodegeneration.4,7-22 In fact, senescent cells have recently been labeled “…drivers of age-related pathologies.”22
There’s already evidence that removing senescent cells from old animals through nutrients and drugs promotes longevity and improves the function of aging body systems.4
Using sophisticated genetic engineering, scientists in 2011 showed they could identify and eliminate senescent cells in mice that naturally age rapidly.23 After lifelong administration of this therapy, senescent cells were swept clean from fat, muscle, and eye tissues, resulting in delayed onset of age-related disorders in those areas. Furthermore, treating mice late in life slowed the progression of such disorders that had already appeared.
Following up on this initial research, the same group in 2016 showed that clearing senescent cells using this approach delayed cancer development and slowed age-related deterioration of kidney, heart, and fat tissues, and extended median lifespan in mice.24
Similar findings resulted from a 2015 study using existing drugs and nutrients, in this case the combination of dasatinib (a cancer drug) plus the polyphenol nutrient quercetin.4 Lab studies showed that this combination reduced the numbers of senescent cells in normally aged mice and in age-accelerated mice, as well as in those exposed to radiation.4
This study showed that in the living animals, healthspans were also improved, as determined by scores of age and wellbeing and by individual symptom scores for common symptoms of aging. (Figure 2 on next page.)
What You Need to Know
The Remarkable Potential of Geroprotectors
- The discovery of ways to counteract cellular senescence is ushering in a new era in health promotion and disease prevention.
- Senescent cells are old cells that have lost the ability to replicate, but instead of dying, they hunker down in tissues and secrete proinflammatory substances that promote aging throughout the body.
- Many stimuli trigger cells to become senescent, including oxidative stress, DNA damage, mitochondrial dysfunction, elevated blood glucose, and others.
- Because the senescence-promoting stimuli are the same throughout the body, and because those stimuli operate via a number of known biochemical signaling pathways, it has now become possible to slow aging in all tissues of the body at once.
- This ability is especially pronounced in a handful of nutrients called geroprotectors.
- These geroprotective nutrients were characterized through a computer algorithm that compared senescence-promoting pathways with those modulated by each of hundreds of candidate compounds.
- The “final four” compounds, myricetin, NAC, gamma tocotrienol, and EGCG each modulate a different but overlapping set of signaling pathways.
- Separately and together, these compounds work to slow cellular aging in all tissues in the body.
- Supplementing with these nutrients in combination seems likely to reduce the body’s burden of senescent cells, promoting youthful function and preventing not one or two, but literally all of the known age-related disorders that threaten human lifespan and healthspan.
What Are Signaling Pathways?
Researchers from Insilico Medicine identified nutrients based on their abilities to modulate certain biochemical signaling pathways that control development of cellular senescence.1,2 To grasp the breakthrough nature of this approach, it’s vital to understand what biologists mean by “signaling pathways.”
Cells need to send and receive information about their environment, nutrient status, stress level, and many other important factors. Without this constant communication, cells could not perform their various life-sustaining duties.
Cells communicate by producing and sending vast amounts of signaling molecules, and receiving other molecules at specific receptors on their membranes and in their nuclei. This network of molecular events is called a signaling pathway.25 When beneficial pathways fail, or destructive ones prevail, cells are pushed toward the senescent state,26 and degenerative aging processes begin to unfold.
By studying signaling pathways that influence development of cellular senescence, scientists can target specific pathways to slow progression of senescence and decrease the number of senescent cells. With the help of artificial intelligence and technology, we can determine which pathways are modulated by a single nutrient—and how nutrients can modulate multiple pathways.1,2
Attacking cellular senescence by modulating multiple independent and overlapping pathways is a broad-spectrum approach to geroprotection and the removal of senescent cells.
Figure 2. Effects of Senolytic Treatment on Mice
Aging Scores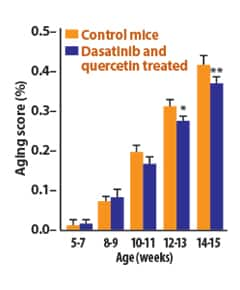
Aging scores of control mice vs. those of mice treated with dasatinib+quercetin. Significant reductions in score are seen at 12-13 and 14-15 weeks, indicated by “*.” |
Symptom Scores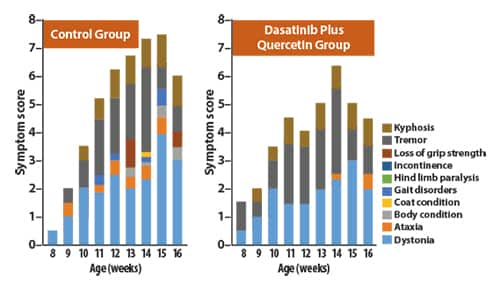
Symptom scores for multiple signs of aging. Kyphosis = humped back; Ataxia = poor balance; Dystonia = loss of muscle tone.4 |
Identifying Geroprotective Nutrients

The basic idea behind geroprotector screening is to compare large numbers of molecular profiles of human tissues of patients of all ages as well as tissues that contain various diseases to identify the changes implicated in degenerative aging.
Unlike studies done in the glass dishes of a laboratory (in vitro studies), or those done in living animals (in vivo studies), research done inside a computer exists purely in virtual form, in the silicon chips of the processor, and is referred to as an “in silico” study.
Searching for natural compounds that could work on many different levels to halt the aging process, researchers resorted to this innovative “in silico” type of study and identified four age-reversing substances.1,2
How the Selected Nutrients Perform as Geroprotectors
Each of the nutrients identified using in silico type of research showed an impressive record at geroprotection.1,2
- Myricetin, a plant-derived polyphenol, is revealing a wide array of pathway modulation in age-related disorders.
- N-acetyl-cysteine (NAC) is a natural sulfur-containing molecule best known for its free-radical scavenging capabilities.
- Gamma tocotrienol is now showing a wide range of signaling pathway modulation that produces health benefits that far exceed those of simple oxidant-reducing nutrients.35-38
- Epigallocatechin-gallate (EGCG) is a polyphenol with known anti-inflammatory properties, but new studies are showing that EGCG also regulates multiple pathways that influence aging in a broad range of tissues.
In particular, myricetin is known to regulate the p38 MAPK family of stress-responsive signaling molecules that are known to regulate aging in many tissues.27,28
Myricetin also promotes cell differentiation and self-repair, and regulates pathways involved in metabolic processes.29-32
NAC is proving useful for its ability to upregulate signaling pathways that boost natural, cellular protections against oxidative stress that promotes cellular senescence.33
In addition, NAC has shown powerful effects on reducing pathways that promote inflammation, adding further anti-aging benefits to this versatile molecule.33,34
A unique pathway modulated by gamma tocotrienol is the mevalonate pathway that controls cholesterol production, cancer promotion, and bone formation.35-37
EGCG uniquely regulates the Wnt pathway, which is vital in determining developing cells’ proper fate and preventing cancer.39
EGCG also prevents sugar-induced damage to tissues throughout the body, helping to suppress their pro-aging effects.40
Scientists found that these compounds reduced cellular aging and various processes that contributed to aging by beneficially modulating a group of signaling pathways that led to the formation of senescent cells.1,2
Most of these pathways are known to contribute to, or protect against, development of senescent cells, and all have been shown to protect against aging at the cellular level.
Table 1 (below) illustrates these pathways in context, showing both considerable overlap (e.g., antioxidant and anti-inflammatory pathways) as well as several pathways unique to each nutrient.
Table 1. How Geroprotective Nutrients Team Up To Slow Aging
Part 1
| Nutrients → | NAC | Myricetin | ||
| ↓Body System or Condition↓ | Pathways Modulated | Health Impact | Pathways Modulated | Health Impact |
| Bone (Target: Slowing Osteoporosis by ↑Bone Formation and/or ↓Bone Resorption) |
↑Antioxidant ↓Inflammatory |
↓Death of bone-producing cells ↓Birth of bone-resorbing cells |
↑P38-MAPK28 ↓MMP (Protein-melting enzymes)53 |
↑Birth of bone-forming cells28 ↓Birth of bone-resorbing cells53 |
| Cancer (Target: Reduce Incidence and Severity) |
↑Antioxidant ↓Inflammatory57 ↓Cancer cell telomerase58 ↓Growth factors59 ↑ER-stress60 |
Sensitizes cancer to chemo57 ↑Cancer cell death58,60↓Cancer growth59 |
↑Antioxidant ↑P38-MAPK61,62 ↓VEGF63 ↓Akt46 |
↑Cancer cell death61-63 ↓Replication61,62 ↓Malignant transformation46 ↓New blood vessels46,63 |
| Cardiovascular (Target: Slow Atherosclerosis) |
↑Antioxidant ↓Inflammatory68 |
↓Hypertension69 ↓Arrythmia70 | ↑Antioxidant ↑Self-repair29,30,71 ↓Activity of calcium channels72 |
↓Vessel constriction72,73 |
| Diabesity (Targets: ↓Weight, body fat, lipid glucose, ↓tissue damage, inflammation) |
↑Antioxidant ↓Inflammatory ↓PPAR-gamma ↑FXR ↑Adiponectin43 |
↓Obesity ↓Atherosclerosis ↓Blood sugar ↑Insulin sensitivity ↑Fat burning ↓Liver fat43 ↓Cataracts ↑Wound healing85,86 |
↓Inflammatory87,88 ↑IRS-1-associated PI3-kinase ↑GLUT431 ↓NFkB, STAT1, NrF289 ↓CCAAT/Enhancer-binding protein PPAR-gamma42 |
↑Insulin sensitivity ↑Glucose uptake ↓Insulin87,88 ↓Fat cell maturation42 ↓Body weight90 ↓Fat storage91 |
| Liver and pancreas (Targets: ↓Liver fat, ↓Inflammation, ↑Liver function, ↓Cancer risk) ↓Fibrosis |
↑Antioxidant103 ↑Mitochondrial function104 |
↓Fatty liver disease, toxic damage105-107 ↓Liver cancer formation108 |
↓Oxidation, inflammation91 ↑Detox enzymes109 ↓DNA damage, ↑DNA repair, ↓JAK1-STAT, CDK1, PAK145-47 |
↓Fatty liver disease91 ↓Fibrosis110 ↓Liver cancer formation45,46 ↑Cancer cell death47 |
| Neuroprotection (Targets: Cognition, Memory, ↓Alzheimer's, Parkinson's) Strok protection |
↑Antioxidant, ↓Inflammatory ↓Toxic protein accumulation ↑Mitochondrial function121-124 ↑Brain cell survival path-ways125,126 |
↑Cognitive function122-126 | ↓Excitotoxicity by ↓calcium overload, ↓glutamate stimulation, ↓capase-3127 ↓Glutamate release52 ↓Deposits of toxic beta-amyloid protein128 ↑GABA protective pathways129 |
↓Cognitive deficts, ↓decrease depressive behavior130,131 ↑Learning and memory132 |
| Kidney (Targets: ↓kidney failure, stone formation, ↑kidney function) |
↑Oxidant protection ↓Inflammation33,138,139 |
↓Urine protein loss33 ↓Kidney stones138 ↓Uremic anemia139 |
↑Antioxidant140 ↓SREBP, VEGF ↑PPAR141 |
↓Glomerular thickening (↑Kidney filtering function)141 ↓Diabetic kidney damage140 |
Table 1. How Geroprotective Nutrients Team Up To Slow Aging
Part 2
| Nutrients → | Gamma Tocotrienol | EGCG | ||
| ↓Body System or Condition↓ | Pathways Modulated | Health Impact | Pathways Modulated | Health Impact |
| Bone (Target: Slowing Osteoporosis by ↑Bone Formation and/or ↓Bone Resorption) |
↓Mevalonate35,36 | ↑Activity of bone-forming cells35,36 ↓Activity of bone-resorbing cells35,36,54 ↑Bone quality36 |
↑Wnt39 ↓HSP27 ↓GSK-3beta ↓Akt55 |
↑Birth of bone-forming cells56 |
| Cancer (Target: Reduce Incidence and Severity) |
↓Mevalonate37,38 ↓Inflammatory64 |
↓Replicaton37,38 ↓New blood vessels37 ↑Cancer cell death37,38 |
↓Growth factors65 ↓NFkB66 ↑miRNA67 |
↓Cell division65 ↑Cancer cell death66 ↓Cancer growth67 |
| Cardiovascular (Target: Slow Atherosclerosis) |
↓Inflammatory74 ↑Antioxidant75 ↓Mevalonate76 ↑Autophagy50 |
↓Plaque75 ↓Cholesterol76 ↓Ischemic damage50 |
↓Inflammatory ↑Antioxidant77 ↑AMPK49 |
↓Lipids ↓DNA Damage ↑Fat burning ↓Fat storage ↑Endothelial function ↓BP78-84 |
| Diabesity (Targets: ↓Weight, body fat, lipid glucose, ↓tissue damage, inflammation) |
↑Autophagy ↑AMPK ↓PPAR gamma44 ↑Fat cell death92 ↓NFkB93-95 |
↓Fat cell formation ↑Fat burning44 ↓Body fat ↓Risk of diabetes ↓Impact on tissues92 |
↑AMPK96 ↑Cellular antioxidants97 ↓DNA Damage97 |
↓Fat accumulation ↑Fat burning ↓Blood sugar ↓Body weight, BMI, Waist circumference.78,98-102 |
| Liver and pancreas (Targets: ↓Liver fat, ↓Inflammation, ↑Liver function, ↓Cancer risk) ↓Fibrosis |
↑Mitochondrial failure in senescent pancreas cells111 ↓NFkB112 |
Improved NAFLD113 Senolytic in pancreas111 |
↑AMPK, oxidant protection ↓Inflammation.114-116 |
↓Fibrosis ↑Markers of liver function ↑Liver regeneration after injury.114,117-120 |
| Neuroprotection (Targets: Cognition, Memory, ↓Alzheimer's, Parkinson's) Strok protection |
↑Antioxidant, ↓Inflammatory133 |
Improved cell viability133 | ↓Inflammation ↓Oxidant stress ↑DNA damage repair48,134 ↑Cleanup of toxic proteins51 |
↑Learning and memory in neurodegenerative disease, stroke, aging135-137 |
| Kidney (Targets: ↓kidney failure, stone formation, ↑kidney function) |
↓TGF-beta142 ↓Inflammation (↓NFkB)94 ↓Oxidative stress143 ↑Mitochondrial function144 |
↓Diabetic kidney damage94 ↑Kidney function, filtering efficiency143 ↓Structural damage from toxins143 |
↓Thromboxane145 ↓RAGE, AGEs40 ↓DNA damage48 ↑Oxidative stress protection146 ↓NFkB147 |
↑Lifespan in rats by ↑kidney function147 ↑Kidney function145 ↓Kidney stone formation146↑Kidney structure, function148 ↓Kidney fibrosis149 |
Some key points about the information presented in Table 1:
- All four nutrients excel at protecting cells against oxidative stress, as well as against inflammation. These processes are known to promote formation of senescent cells.26,41
- Myricetin, NAC, and gamma tocotrienol all prevent energy from being stored as fat through the PPAR-gamma pathway.42-44
- Myricetin and EGCG excel at preventing senescence-inducing DNA damage and promoting its repair.45-48
- Gamma tocotrienol and EGCG activate the AMPK pathway that promotes youthful cellular function, reduced fat storage, and improved entry of glucose into cells.44,49
- Gamma tocotrienol and EGCG both promote autophagy, the removal of debris (junk proteins) that accumulates inside of aged cells.50,51
- Myricetin reduces the effects of glutamate, the excitatory neurotransmitter implicated in brain aging.52
These nutrients have also shown the potential to be senolytic agents, meaning that they not only prevent cellular senescence, but may contribute directly to removal of age-accelerating senescent cells from tissues.1,6
Combining Geroprotectors to Slow Aging
The discovery and characterization of geroprotective nutrients is changing the ways we think about aging. The new paradigm is to look at signaling pathways that effect aging at cellular, and indeed, subcellular levels.
By utilizing this technology, whether aging is occurring in the brain, the heart, the intestinal tract, the muscle, or anywhere else in the body is irrelevant. Instead, we can see how myriad biochemical signaling pathways are operating in every cell in the body, ultimately contributing to accumulation of senescent cells that produce symptoms of aging and degenerative illnesses.
These advances mean that we can begin to identify specific nutrients, the pathways that they modulate, and their long-term effects in cells. This data is then used to develop anti-aging mixtures capable of defeating cellular senescence wherever it occurs.
NAC, myricetin, gamma tocotrienol, and EGCG are demonstrating the ability, between them, to modulate more than a score of signaling pathways that prevent cells from deteriorating into age-promoting, senescent cells, while also preventing those cells from taking the alternate route of becoming malignant.
As a result, these nutrients have been combined into the first-ever geroprotective formulation based on artificial intelligence with far-reaching effects on age-related disorders to which senescent cells contribute. This improves our ability to develop strategies aimed at preventing cellular aging throughout the body.
The real world upshot is an opportunity to lengthen healthy human lifespans.
Summary

Senescent cells lose the ability to contribute meaningfully to body function. Instead of dying they go into an abnormal secretory state, pumping out destructive pro-inflammatory molecules that contribute to destructive aging throughout the body.
In partnership with researchers at Insilico Medicine, Life Extension scientists reviewed the biochemical pathways that push aging cells into senescence, and then screened hundreds of drugs and nutrients capable of favorably modifying those pathways.
The result was a short-list of four specific nutrients, myricetin, NAC, gamma tocotrienol, and EGCG, all of which modify senescence-inducing pathways, slowing down the development of senescent cells and exhibiting characteristics of senolytics.
Each nutrient not only has its own unique set of pathways that it regulates, but also overlaps with similar pathways modulated by the others, providing comprehensive protection against aging at the cellular level.
That ability, now known as geroprotection, makes it possible to better prevent or decelerate aging throughout the body, rather than on an organ-by-organ, or disease-by-disease basis.
This work is only the beginning.
Future studies will no doubt identify other cocktails with geroprotective properties and will fine-tune our understanding of how manipulation of senescence-inducing pathways can operate.
In time, new studies will produce further evidence of the intriguing notion of senolytic compounds, those capable of sweeping the body clean of existing senescent cells – a possibility that could facilitate meaningful age reversal.
If you have any questions on the scientific content of this article, please call a Life Extension® Wellness Specialist at 1-866-864-3027.
References
- Aliper A, Belikov AV, Garazha A, et al. In search for geroprotectors: in silico screening and in vitro validation of signalome-level mimetics of young healthy state. Aging (Albany NY). 2016;8(9):2127-52.
- Geroprotective Properties of Gamma Tocotrienol. Insilico Medicine. Data on File. 2016.
- Kirkland JL, Tchkonia T. Clinical strategies and animal models for developing senolytic agents. Exp Gerontol. 2015;68:19-25.
- Zhu Y, Tchkonia T, Pirtskhalava T, et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14(4):644-58.
- Campisi J, d’Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol. 2007;8(9):729-40.
- Malavolta M, Pierpaoli E, Giacconi R, et al. Pleiotropic Effects of Tocotrienols and Quercetin on Cellular Senescence: Introducing the Perspective of Senolytic Effects of Phytochemicals. Curr Drug Targets. 2016;17(4):447-59.
- Carracedo J, Buendia P, Merino A, et al. Cellular senescence determines endothelial cell damage induced by uremia. Exp Gerontol. 2013;48(8):766-73.
- Chinta SJ, Woods G, Rane A, et al. Cellular senescence and the aging brain. Exp Gerontol. 2015;68:3-7.
- Clements ME, Chaber CJ, Ledbetter SR, et al. Increased cellular senescence and vascular rarefaction exacerbate the progression of kidney fibrosis in aged mice following transient ischemic injury. PLoS One. 2013;8(8):e70464.
- D’Mello MJ, Ross SA, Briel M, et al. Association between shortened leukocyte telomere length and cardiometabolic outcomes: systematic review and meta-analysis. Circ Cardiovasc Genet. 2015;8(1):82-90.
- Erusalimsky JD, Kurz DJ. Cellular senescence in vivo: its relevance in ageing and cardiovascular disease. Exp Gerontol. 2005;40(8-9):634-42.
- Farr JN, Fraser DG, Wang H, et al. Identification of Senescent Cells in the Bone Microenvironment. J Bone Miner Res. 2016;31(11):1920-9.
- Gutierrez-Reyes G, del Carmen Garcia de Leon M, Varela-Fascinetto G, et al. Cellular senescence in livers from children with end stage liver disease. PLoS One. 2010;5(4):e10231.
- Matjusaitis M, Chin G, Sarnoski EA, et al. Biomarkers to identify and isolate senescent cells. Ageing Res Rev. 2016;29:1-12.
- Nishimatsu H, Suzuki E, Saito Y, et al. Senescent Cells Impair Erectile Function through Induction of Endothelial Dysfunction and Nerve Injury in Mice. PLoS One. 2015;10(4):e0124129.
- Palmer AK, Tchkonia T, LeBrasseur NK, et al. Cellular Senescence in Type 2 Diabetes: A Therapeutic Opportunity. Diabetes. 2015;64(7):2289-98.
- Ramakrishna G, Rastogi A, Trehanpati N, et al. From cirrhosis to hepatocellular carcinoma: new molecular insights on inflammation and cellular senescence. Liver Cancer. 2013;2(3-4):367-83.
- Seki E, Brenner DA. Recent advancement of molecular mechanisms of liver fibrosis. J Hepatobiliary Pancreat Sci. 2015;22(7):512-8.
- Testa R, Genovese S, Ceriello A. Nutritional imbalances linking cellular senescence and type 2 diabetes mellitus. Curr Opin Clin Nutr Metab Care. 2014;17(4):338-42.
- Velarde MC, Demaria M, Campisi J. Senescent cells and their secretory phenotype as targets for cancer therapy. Interdiscip Top Gerontol. 2013;38:17-27.
- Yeh JK, Wang CY. Telomeres and Telomerase in Cardiovascular Diseases. Genes (Basel). 2016;7(9).
- Ovadya Y, Krizhanovsky V. Senescent cells: SASPected drivers of age-related pathologies. Biogerontology. 2014;15(6):627-42.
- Baker DJ, Wijshake T, Tchkonia T, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479(7372):232-6.
- Baker DJ, Childs BG, Durik M, et al. Naturally occurring p16 (Ink4a)-positive cells shorten healthy lifespan. Nature. 2016;530(7589):184-9.
- Available at: https://www.ncbi.nlm.nih.gov/books/NBK9870/. Accessed January 10, 2017.
- Sasaki M, Ikeda H, Sato Y, et al. Proinflammatory cytokine-induced cellular senescence of biliary epithelial cells is mediated via oxidative stress and activation of ATM pathway: a culture study. Free Radic Res. 2008;42(7):625-32.
- Segales J, Perdiguero E, Munoz-Canoves P. Regulation of Muscle Stem Cell Functions: A Focus on the p38 MAPK Signaling Pathway. Front Cell Dev Biol. 2016;4:91.
- Hsu YL, Chang JK, Tsai CH, et al. Myricetin induces human osteoblast differentiation through bone morphogenetic protein-2/p38 mitogen-activated protein kinase pathway. Biochem Pharmacol. 2007;73(4):504-14.
- Scarabelli TM, Mariotto S, Abdel-Azeim S, et al. Targeting STAT1 by myricetin and delphinidin provides efficient protection of the heart from ischemia/reperfusion-induced injury. FEBS Lett. 2009;583(3):531-41.
- Qiu Y, Cong N, Liang M, et al. Systems Pharmacology Dissection of the Protective Effect of Myricetin Against Acute Ischemia/Reperfusion-Induced Myocardial Injury in Isolated Rat Heart. Cardiovasc Toxicol. 2016.
- Liu IM, Tzeng TF, Liou SS, et al. Improvement of insulin sensitivity in obese Zucker rats by myricetin extracted from Abelmoschus moschatus. Planta Med. 2007;73(10):1054-60.
- Semwal DK, Semwal RB, Combrinck S, et al. Myricetin: A Dietary Molecule with Diverse Biological Activities. Nutrients. 2016;8(2):90.
- Fujii S, Zhang L, Kosaka H. Albuminuria, expression of nicotinamide adenine dinucleotide phosphate oxidase and monocyte chemoattractant protein-1 in the renal tubules of hypertensive Dahl salt-sensitive rats. Hypertens Res. 2007;30(10):991-8.
- de Andrade KQ, Moura FA, dos Santos JM, et al. Oxidative Stress and Inflammation in Hepatic Diseases: Therapeutic Possibilities of N-Acetylcysteine. Int J Mol Sci. 2015;16(12):30269-308.
- Chin KY, Mo H, Soelaiman IN. A review of the possible mechanisms of action of tocotrienol - a potential antiosteoporotic agent. Curr Drug Targets. 2013;14(13):1533-41.
- Chin KY, Ima-Nirwana S. The biological effects of tocotrienol on bone: a review on evidence from rodent models. Drug Des Devel Ther. 2015;9:2049-61.
- Wada S. Chemoprevention of tocotrienols: the mechanism of antiproliferative effects. Forum Nutr. 2009;61:204-16.
- Loganathan R, Selvaduray KR, Nesaretnam K, et al. Tocotrienols promote apoptosis in human breast cancer cells by inducing poly(ADP-ribose) polymerase cleavage and inhibiting nuclear factor kappa-B activity. Cell Prolif. 2013;46(2):203-13.
- Wang D, Wang Y, Xu S, et al. Epigallocatechin-3-gallate Protects against Hydrogen Peroxide-Induced Inhibition of Osteogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stem Cells. Stem Cells Int. 2016;2016:7532798.
- Yamabe N, Kang KS, Hur JM, et al. Matcha, a powdered green tea, ameliorates the progression of renal and hepatic damage in type 2 diabetic OLETF rats. J Med Food. 2009;12(4):714-21.
- Suzuki E, Takahashi M, Oba S, et al. Oncogene- and oxidative stress-induced cellular senescence shows distinct expression patterns of proinflammatory cytokines in vascular endothelial cells. ScientificWorldJournal. 2013;2013:754735.
- Wang Q, Wang ST, Yang X, et al. Myricetin suppresses differentiation of 3 T3-L1 preadipocytes and enhances lipolysis in adipocytes. Nutr Res. 2015;35(4):317-27.
- Ma Y, Gao M, Liu D. N-acetylcysteine Protects Mice from High Fat Diet-induced Metabolic Disorders. Pharm Res. 2016;33(8):2033-42.
- Zhao L, Ha JH, Okla M, et al. Activation of autophagy and AMPK by gamma-tocotrienol suppresses the adipogenesis in human adipose derived stem cells. Mol Nutr Food Res. 2014;58(3):569-79.
- Abalea V, Cillard J, Dubos MP, et al. Repair of iron-induced DNA oxidation by the flavonoid myricetin in primary rat hepatocyte cultures. Free Radic Biol Med. 1999;26(11-12):1457-66.
- Devi KP, Rajavel T, Habtemariam S, et al. Molecular mechanisms underlying anticancer effects of myricetin. Life Sci. 2015;142:19-25.
- Iyer SC, Gopal A, Halagowder D. Myricetin induces apoptosis by inhibiting P21 activated kinase 1 (PAK1) signaling cascade in hepatocellular carcinoma. Mol Cell Biochem. 2015;407(1-2):223-37.
- Orsolic N, Sirovina D, Gajski G, et al. Assessment of DNA damage and lipid peroxidation in diabetic mice: effects of propolis and epigallocatechin gallate (EGCG). Mutat Res. 2013;757(1):36-44.
- Cai Y, Zhao L, Qin Y, et al. EGCG Blocked Phenylephrin-Induced Hypertrophy in H9C2 Cardiomyocytes, by Activating AMPK-Dependent Pathway. Korean J Physiol Pharmacol. 2015;19(3):203-10.
- Lekli I, Ray D, Mukherjee S, et al. Co-ordinated autophagy with resveratrol and gamma-tocotrienol confers synergetic cardioprotection. J Cell Mol Med. 2010;14(10):2506-18.
- Lee JH, Moon JH, Kim SW, et al. EGCG-mediated autophagy flux has a neuroprotection effect via a class III histone deacetylase in primary neuron cells. Oncotarget. 2015;6(12):9701-17.
- Chang Y, Chang CY, Wang SJ, et al. Myricetin inhibits the release of glutamate in rat cerebrocortical nerve terminals. J Med Food. 2015;18(5):516-23.
- Ko SY. Myricetin suppresses LPS-induced MMP expression in human gingival fibroblasts and inhibits osteoclastogenesis by downregulating NFATc1 in RANKL-induced RAW 264.7 cells. Arch Oral Biol. 2012;57(12):1623-32.
- Deng L, Ding Y, Peng Y, et al. gamma-Tocotrienol protects against ovariectomy-induced bone loss via mevalonate pathway as HMG-CoA reductase inhibitor. Bone. 2014;67:200-7.
- Natsume H, Adachi S, Takai S, et al. (-)-Epigallocatechin gallate attenuates the induction of HSP27 stimulated by sphingosine 1-phosphate via suppression of phosphatidylinositol 3-kinase/Akt pathway in osteoblasts. Int J Mol Med. 2009;24(2):197-203.
- Kaida K, Honda Y, Hashimoto Y, et al. Application of Green Tea Catechin for Inducing the Osteogenic Differentiation of Human Dedifferentiated Fat Cells in Vitro. Int J Mol Sci. 2015;16(12):27988-8000.
- Qanungo S, Uys JD, Manevich Y, et al. N-acetyl-L-cysteine sensitizes pancreatic cancers to gemcitabine by targeting the NFkappaB pathway. Biomed Pharmacother. 2014;68(7):855-64.
- Li P, Wu M, Wang J, et al. NAC selectively inhibit cancer telomerase activity: A higher redox homeostasis threshold exists in cancer cells. Redox Biol. 2016;8:91-7.
- Lee MF, Chan CY, Hung HC, et al. N-acetylcysteine (NAC) inhibits cell growth by mediating the EGFR/Akt/HMG box-containing protein 1 (HBP1) signaling pathway in invasive oral cancer. Oral Oncol. 2013;49(2):129-35.
- Guan D, Xu Y, Yang M, et al. N-acetyl cysteine and penicillamine induce apoptosis via the ER stress response-signaling pathway. Mol Carcinog. 2010;49(1):68-74.
- Sun F, Zheng XY, Ye J, et al. Potential anticancer activity of myricetin in human T24 bladder cancer cells both in vitro and in vivo. Nutr Cancer. 2012;64(4):599-606.
- Huang H, Chen AY, Ye X, et al. Myricetin inhibits proliferation of cisplatin-resistant cancer cells through a p53-dependent apoptotic pathway. Int J Oncol. 2015;47(4):1494-502.
- Huang H, Chen AY, Rojanasakul Y, et al. Dietary compounds galangin and myricetin suppress ovarian cancer cell angiogenesis. J Funct Foods. 2015;15:464-75.
- Shin-Kang S, Ramsauer VP, Lightner J, et al. Tocotrienols inhibit AKT and ERK activation and suppress pancreatic cancer cell proliferation by suppressing the ErbB2 pathway. Free Radic Biol Med. 2011;51(6):1164-74.
- Chen D, Wan SB, Yang H, et al. EGCG, green tea polyphenols and their synthetic analogs and prodrugs for human cancer prevention and treatment. Adv Clin Chem. 2011;53:155-77.
- Singh BN, Shankar S, Srivastava RK. Green tea catechin, epigallocatechin-3-gallate (EGCG): mechanisms, perspectives and clinical applications. Biochem Pharmacol. 2011;82(12):1807-21.
- Zhu K, Wang W. Green tea polyphenol EGCG suppresses osteosarcoma cell growth through upregulating miR-1. Tumour Biol. 2016;37(4):4373-82.
- Silva LA, Silveira PC, Pinho CA, et al. N-acetylcysteine supplementation and oxidative damage and inflammatory response after eccentric exercise. Int J Sport Nutr Exerc Metab. 2008;18(4):379-88.
- Vasdev S, Mian T, Longerich L, et al. N-acetyl cysteine attenuates ethanol induced hypertension in rats. Artery. 1995;21(6):312-6.
- Gu WJ, Wu ZJ, Wang PF, et al. N-Acetylcysteine supplementation for the prevention of atrial fibrillation after cardiac surgery: a meta-analysis of eight randomized controlled trials. BMC Cardiovasc Disord. 2012;12:10.
- Tiwari R, Mohan M, Kasture S, et al. Cardioprotective potential of myricetin in isoproterenol-induced myocardial infarction in Wistar rats. Phytother Res. 2009;23(10):1361-6.
- Fusi F, Sgaragli G, Saponara S. Mechanism of myricetin stimulation of vascular L-type Ca2+ current. J Pharmacol Exp Ther. 2005;313(2):790-7.
- Angelone T, Pasqua T, Di Majo D, et al. Distinct signalling mechanisms are involved in the dissimilar myocardial and coronary effects elicited by quercetin and myricetin, two red wine flavonols. Nutr Metab Cardiovasc Dis. 2011;21(5):362-71.
- Qureshi AA, Reis JC, Papasian CJ, et al. Tocotrienols inhibit lipopolysaccharide-induced pro-inflammatory cytokines in macrophages of female mice. Lipids Health Dis. 2010;9:143.
- Teoh MK, Chong JM, Mohamed J, et al. Protection by tocotrienols against hypercholesterolaemia and atheroma. Med J Malaysia. 1994;49(3):255-62.
- McAnally JA, Gupta J, Sodhani S, et al. Tocotrienols potentiate lovastatin-mediated growth suppression in vitro and in vivo. Exp Biol Med (Maywood). 2007;232(4):523-31.
- Tipoe GL, Leung TM, Hung MW, et al. Green tea polyphenols as an anti-oxidant and anti-inflammatory agent for cardiovascular protection. Cardiovasc Hematol Disord Drug Targets. 2007;7(2):135-44.
- Bogdanski P, Suliburska J, Szulinska M, et al. Green tea extract reduces blood pressure, inflammatory biomarkers, and oxidative stress and improves parameters associated with insulin resistance in obese, hypertensive patients. Nutr Res. 2012;32(6):421-7.
- Brown AL, Lane J, Coverly J, et al. Effects of dietary supplementation with the green tea polyphenol epigallocatechin-3-gallate on insulin resistance and associated metabolic risk factors: randomized controlled trial. Br J Nutr. 2009;101(6):886-94.
- Widlansky ME, Hamburg NM, Anter E, et al. Acute EGCG supplementation reverses endothelial dysfunction in patients with coronary artery disease. J Am Coll Nutr. 2007;26(2):95-102.
- Jang HJ, Ridgeway SD, Kim JA. Effects of the green tea polyphenol epigallocatechin-3-gallate on high-fat diet-induced insulin resistance and endothelial dysfunction. Am J Physiol Endocrinol Metab. 2013;305(12):E1444-51.
- Nogueira LP, Nogueira Neto JF, Klein MR, et al. Short-term Effects of Green Tea on Blood Pressure, Endothelial Function, and Metabolic Profile in Obese Prehypertensive Women: A Crossover Randomized Clinical Trial. J Am Coll Nutr. 2016:1-8.
- Samavat H, Newman AR, Wang R, et al. Effects of green tea catechin extract on serum lipids in postmenopausal women: a randomized, placebo-controlled clinical trial. Am J Clin Nutr. 2016;104(6):1671-82.
- Suzuki-Sugihara N, Kishimoto Y, Saita E, et al. Green tea catechins prevent low-density lipoprotein oxidation via their accumulation in low-density lipoprotein particles in humans. Nutr Res. 2016;36(1):16-23.
- Park NY, Lim Y. Short term supplementation of dietary antioxidants selectively regulates the inflammatory responses during early cutaneous wound healing in diabetic mice. Nutr Metab (Lond). 2011;8(1):80.
- Jain AK, Lim G, Langford M, et al. Effect of high-glucose levels on protein oxidation in cultured lens cells, and in crystalline and albumin solution and its inhibition by vitamin B6 and N-acetylcysteine: its possible relevance to cataract formation in diabetes. Free Radic Biol Med. 2002;33(12):1615-21.
- Choi HN, Kang MJ, Lee SJ, et al. Ameliorative effect of myricetin on insulin resistance in mice fed a high-fat, high-sucrose diet. Nutr Res Pract. 2014;8(5):544-9.
- Kandasamy N, Ashokkumar N. Protective effect of bioflavonoid myricetin enhances carbohydrate metabolic enzymes and insulin signaling molecules in streptozotocin-cadmium induced diabetic nephrotoxic rats. Toxicol Appl Pharmacol. 2014;279(2):173-85.
- Cho BO, Yin HH, Park SH, et al. Anti-inflammatory activity of myricetin from Diospyros lotus through suppression of NF-kappaB and STAT1 activation and Nrf2-mediated HO-1 induction in lipopolysaccharide-stimulated RAW264.7 macrophages. Biosci Biotechnol Biochem. 2016;80(8):1520-30.
- Su HM, Feng LN, Zheng XD, et al. Myricetin protects against diet-induced obesity and ameliorates oxidative stress in C57BL/6 mice. J Zhejiang Univ Sci B. 2016;17(6):437-46.
- Xia SF, Le GW, Wang P, et al. Regressive Effect of Myricetin on Hepatic Steatosis in Mice Fed a High-Fat Diet. Nutrients. 2016;8(12).
- Zhao L, Fang X, Marshall MR, et al. Regulation of Obesity and Metabolic Complications by Gamma and Delta Tocotrienols. Molecules. 2016;21(3).
- Kuhad A, Bishnoi M, Tiwari V, et al. Suppression of NF-kappabeta signaling pathway by tocotrienol can prevent diabetes associated cognitive deficits. Pharmacol Biochem Behav. 2009;92(2):251-9.
- Kuhad A, Chopra K. Attenuation of diabetic nephropathy by tocotrienol: involvement of NFkB signaling pathway. Life Sci. 2009;84(9-10):296-301.
- Kim Y, Wang W, Okla M, et al. Suppression of NLRP3 inflammasome by gamma-tocotrienol ameliorates type 2 diabetes. J Lipid Res. 2016;57(1):66-76.
- Li Y, Zhao S, Zhang W, et al. Epigallocatechin-3-O-gallate (EGCG) attenuates FFAs-induced peripheral insulin resistance through AMPK pathway and insulin signaling pathway in vivo. Diabetes Res Clin Pract. 2011;93(2):205-14.
- Choi SW, Yeung VT, Collins AR, et al. Redox-linked effects of green tea on DNA damage and repair, and influence of microsatellite polymorphism in HMOX-1: results of a human intervention trial. Mutagenesis. 2015;30(1):129-37.
- Brown AL, Lane J, Holyoak C, et al. Health effects of green tea catechins in overweight and obese men: a randomised controlled cross-over trial. Br J Nutr. 2011;106(12):1880-9.
- Basu A, Sanchez K, Leyva MJ, et al. Green tea supplementation affects body weight, lipids, and lipid peroxidation in obese subjects with metabolic syndrome. J Am Coll Nutr. 2010;29(1):31-40.
- Venables MC, Hulston CJ, Cox HR, et al. Green tea extract ingestion, fat oxidation, and glucose tolerance in healthy humans. Am J Clin Nutr. 2008;87(3):778-84.
- Fukino Y, Ikeda A, Maruyama K, et al. Randomized controlled trial for an effect of green tea-extract powder supplementation on glucose abnormalities. Eur J Clin Nutr. 2008;62(8):953-60.
- Suliburska J, Bogdanski P, Szulinska M, et al. Effects of green tea supplementation on elements, total antioxidants, lipids, and glucose values in the serum of obese patients. Biol Trace Elem Res. 2012;149(3):315-22.
- Chen Y, Johansson E, Yang Y, et al. Oral N-acetylcysteine rescues lethality of hepatocyte-specific Gclc-knockout mice, providing a model for hepatic cirrhosis. J Hepatol. 2010;53(6):1085-94.
- Gonzalez R, Ferrin G, Hidalgo AB, et al. N-acetylcysteine, coenzyme Q10 and superoxide dismutase mimetic prevent mitochondrial cell dysfunction and cell death induced by d-galactosamine in primary culture of human hepatocytes. Chem Biol Interact. 2009;181(1):95-106.
- Lai IK, Dhakal K, Gadupudi GS, et al. N-acetylcysteine (NAC) diminishes the severity of PCB 126-induced fatty liver in male rodents. Toxicology. 2012;302(1):25-33.
- Lasram MM, El-Golli N, Lamine AJ, et al. Changes in glucose metabolism and reversion of genes expression in the liver of insulin-resistant rats exposed to malathion. The protective effects of N-acetylcysteine. Gen Comp Endocrinol. 2015;215:88-97.
- El-Lakkany NM, Seif El-Din SH, Sabra AA, et al. Co-administration of metformin and N-acetylcysteine with dietary control improves the biochemical and histological manifestations in rats with non-alcoholic fatty liver. Res Pharm Sci. 2016;11(5):374-82.
- Garcia-Roman R, Salazar-Gonzalez D, Rosas S, et al. The differential NF-kB modulation by S-adenosyl-L-methionine, N-acetylcysteine and quercetin on the promotion stage of chemical hepatocarcinogenesis. Free Radic Res. 2008;42(4):331-43.
- Guo J, Meng Y, Zhao Y, et al. Myricetin derived from Hovenia dulcis Thunb. ameliorates vascular endothelial dysfunction and liver injury in high choline-fed mice. Food Funct. 2015;6(5):1620-34.
- Geng Y, Sun Q, Li W, et al. The common dietary flavonoid myricetin attenuates liver fibrosis in carbon tetrachloride-treated mice. Mol Nutr Food Res. 2016.
- Vaquero EC, Rickmann M, Molero X. Tocotrienols: balancing the mitochondrial crosstalk between apoptosis and autophagy. Autophagy. 2007;3(6):652-4.
- Muto C, Yachi R, Aoki Y, et al. Gamma-tocotrienol reduces the triacylglycerol level in rat primary hepatocytes through regulation of fatty acid metabolism. J Clin Biochem Nutr. 2013;52(1):32-7.
- Magosso E, Ansari MA, Gopalan Y, et al. Tocotrienols for normalisation of hepatic echogenic response in nonalcoholic fatty liver: a randomised placebo-controlled clinical trial. Nutr J. 2013;12(1):166.
- Krishnan TR, Velusamy P, Srinivasan A, et al. EGCG mediated downregulation of NF-AT and macrophage infiltration in experimental hepatic steatosis. Exp Gerontol. 2014;57:96-103.
- Santamarina AB, Carvalho-Silva M, Gomes LM, et al. Decaffeinated green tea extract rich in epigallocatechin-3-gallate prevents fatty liver disease by increased activities of mitochondrial respiratory chain complexes in diet-induced obesity mice. J Nutr Biochem. 2015;26(11):1348-56.
- Santamarina AB, Oliveira JL, Silva FP, et al. Green Tea Extract Rich in Epigallocatechin-3-Gallate Prevents Fatty Liver by AMPK Activation via LKB1 in Mice Fed a High-Fat Diet. PLoS One. 2015;10(11):e0141227.
- Hung GD, Li PC, Lee HS, et al. Green tea extract supplementation ameliorates CCl4-induced hepatic oxidative stress, fibrosis, and acute-phase protein expression in rat. J Formos Med Assoc. 2012;111(10):550-9.
- Liu HW, Chan YC, Wang MF, et al. Dietary (-)-Epigallocatechin-3-gallate Supplementation Counteracts Aging-Associated Skeletal Muscle Insulin Resistance and Fatty Liver in Senescence-Accelerated Mouse. J Agric Food Chem. 2015;63(38):8407-17.
- Pezeshki A, Safi S, Feizi A, et al. The Effect of Green Tea Extract Supplementation on Liver Enzymes in Patients with Nonalcoholic Fatty Liver Disease. Int J Prev Med. 2016;7:28.
- Saito Y, Mori H, Takasu C, et al. Beneficial effects of green tea catechin on massive hepatectomy model in rats. J Gastroenterol. 2014;49(4):692-701.
- Clark J, Clore EL, Zheng K, et al. Oral N-acetyl-cysteine attenuates loss of dopaminergic terminals in alpha-synuclein overexpressing mice. PLoS One. 2010;5(8):e12333.
- Martinez M, Hernandez AI, Martinez N. N-Acetylcysteine delays age-associated memory impairment in mice: role in synaptic mitochondria. Brain Res. 2000;855(1):100-6.
- Jayalakshmi K, Singh SB, Kalpana B, et al. N-acetyl cysteine supplementation prevents impairment of spatial working memory functions in rats following exposure to hypobaric hypoxia. Physiol Behav. 2007;92(4):643-50.
- Kamboj SS, Chopra K, Sandhir R. Neuroprotective effect of N-acetylcysteine in the development of diabetic encephalopathy in streptozotocin-induced diabetes. Metab Brain Dis. 2008;23(4):427-43.
- Kanwar SS, Nehru B. Modulatory effects of N-acetylcysteine on cerebral cortex and cerebellum regions of ageing rat brain. Nutr Hosp. 2007;22(1):95-100.
- Moreira PI, Harris PL, Zhu X, et al. Lipoic acid and N-acetyl cysteine decrease mitochondrial-related oxidative stress in Alzheimer disease patient fibroblasts. J Alzheimers Dis. 2007;12(2):195-206.
- Shimmyo Y, Kihara T, Akaike A, et al. Three distinct neuroprotective functions of myricetin against glutamate-induced neuronal cell death: involvement of direct inhibition of caspase-3. J Neurosci Res. 2008;86(8):1836-45.
- Shimmyo Y, Kihara T, Akaike A, et al. Multifunction of myricetin on A beta: neuroprotection via a conformational change of A beta and reduction of A beta via the interference of secretases. J Neurosci Res. 2008;86(2):368-77.
- Zhang XH, Ma ZG, Rowlands DK, et al. Flavonoid Myricetin Modulates GABA(A) Receptor Activity through Activation of Ca(2+) Channels and CaMK-II Pathway. Evid Based Complement Alternat Med. 2012;2012:758097.
- Ma Z, Wang G, Cui L, et al. Myricetin Attenuates Depressant-Like Behavior in Mice Subjected to Repeated Restraint Stress. Int J Mol Sci. 2015;16(12):28377-85.
- Wang QM, Wang GL, Ma ZG. Protective effects of myricetin on chronic stress-induced cognitive deficits. Neuroreport. 2016;27(9):652-8.
- Lei Y, Chen J, Zhang W, et al. In vivo investigation on the potential of galangin, kaempferol and myricetin for protection of D-galactose-induced cognitive impairment. Food Chem. 2012;135(4):2702-7.
- Selvaraju TR, Khaza’ai H, Vidyadaran S, et al. The neuroprotective effects of tocotrienol rich fraction and alpha tocopherol against glutamate injury in astrocytes. Bosn J Basic Med Sci. 2014;14(4):195-204.
- Mandel SA, Weinreb O, Amit T, et al. Molecular mechanisms of the neuroprotective/neurorescue action of multi-target green tea polyphenols. Front Biosci (Schol Ed). 2012;4:581-98.
- Rezai-Zadeh K, Arendash GW, Hou H, et al. Green tea epigallocatechin-3-gallate (EGCG) reduces beta-amyloid mediated cognitive impairment and modulates tau pathology in Alzheimer transgenic mice. Brain Res. 2008;1214:177-87.
- Li Q, Zhao H, Zhao M, et al. Chronic green tea catechins administration prevents oxidative stress-related brain aging in C57BL/6J mice. Brain Res. 2010;1353:28-35.
- Schimidt HL, Vieira A, Altermann C, et al. Memory deficits and oxidative stress in cerebral ischemia-reperfusion: neuroprotective role of physical exercise and green tea supplementation. Neurobiol Learn Mem. 2014;114:242-50.
- Fishman AI, Green D, Lynch A, et al. Preventive effect of specific antioxidant on oxidative renal cell injury associated with renal crystal formation. Urology. 2013;82(2):489.e1-7.
- Hsu SP, Chiang CK, Yang SY, et al. N-acetylcysteine for the management of anemia and oxidative stress in hemodialysis patients. Nephron Clin Pract. 2010;116(3):c207-16.
- Ozcan F, Ozmen A, Akkaya B, et al. Beneficial effect of myricetin on renal functions in streptozotocin-induced diabetes. Clin Exp Med. 2012;12(4):265-72.
- Kandasamy N, Ashokkumar N. Renoprotective effect of myricetin restrains dyslipidemia and renal mesangial cell proliferation by the suppression of sterol regulatory element binding proteins in an experimental model of diabetic nephropathy. Eur J Pharmacol. 2014;743:53-62.
- Siddiqui S, Ahsan H, Khan MR, et al. Protective effects of tocotrienols against lipid-induced nephropathy in experimental type-2 diabetic rats by modulation in TGF-beta expression. Toxicol Appl Pharmacol. 2013;273(2):314-24.
- Khan MR, Siddiqui S, Parveen K, et al. Nephroprotective action of tocotrienol-rich fraction (TRF) from palm oil against potassium dichromate (K 2 Cr 2 O 7)-induced acute renal injury in rats. Chem Biol Interact. 2010;186(2):228-38.
- Nowak G, Bakajsova D, Hayes C, et al. gamma-Tocotrienol protects against mitochondrial dysfunction and renal cell death. J Pharmacol Exp Ther. 2012;340(2):330-8.
- Rhee SJ, Kim MJ, Kwag OG. Effects of green tea catechin on prostaglandin synthesis of renal glomerular and renal dysfunction in streptozotocin-induced diabetic rats. Asia Pac J Clin Nutr. 2002;11(3):232-6.
- Jeong BC, Kim BS, Kim JI, et al. Effects of green tea on urinary stone formation: an in vivo and in vitro study. J Endourol. 2006;20(5):356-61.
- Niu Y, Na L, Feng R, et al. The phytochemical, EGCG, extends lifespan by reducing liver and kidney function damage and improving age-associated inflammation and oxidative stress in healthy rats. Aging Cell. 2013;12(6):1041-9.
- Zhou P, Yu JF, Zhao CG, et al. Therapeutic potential of EGCG on acute renal damage in a rat model of obstructive nephropathy. Mol Med Rep. 2013;7(4):1096-102.
- Kanlaya R, Khamchun S, Kapincharanon C, et al. Protective effect of epigallocatechin-3-gallate (EGCG) via Nrf2 pathway against oxalate-induced epithelial mesenchymal transition (EMT) of renal tubular cells. Sci Rep. 2016;6:30233.

