Life Extension Magazine®

This article describes findings that may enable you to rejuvenate your stem cells using low-cost approaches available right now.
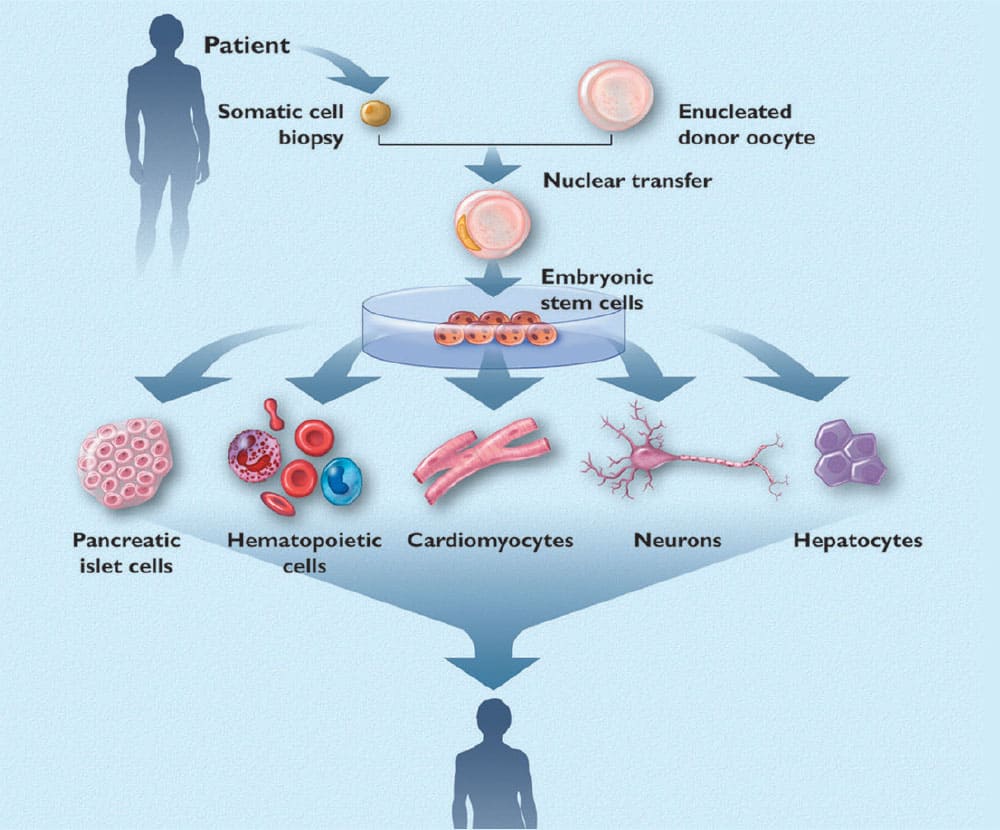
To put this into historic context, the image on this page is the March 2002 cover of Life Extension® magazine.
Our message back then was that therapeutic cloning of stem cells might enable our biology to be transported back in time to a younger state.
In that 2002 magazine article, we described how fresh, young stem cells can regenerate tissues throughout our body, thereby reversing the course of degenerative disorders.
Stem cell research, however, was torpedoed by federal edict in 2001 and has only recently regained serious momentum.
The encouraging news is that we may be able to regenerate our existing pool of stem cells today.
The significance of this cannot be overstated.
If we replenish our pool of healthy stem cells, we may regain the ability to repopulate our tissues with fresh functional cells.
The good news is that certain nutrients that readers of this magazine already supplement with have stem cell-renewing properties. This can buy precious time as ongoing research develops systemic stem cell rejuvenation therapies envisioned 20 years ago.
Stem Cells Needed to Sustain Life

Our tissues rely on functional cells to sustain organ viability.
With age, our functional cells deteriorate.
In youth, as functional cells die off, they are replaced with new cells created from stem cells present in our body.1-3
Stem cells, however, are affected by the same degenerative problems as functional cells.4
As stem cell vitality deteriorates, we lose the ability to repopulate tissues with fresh functional cells.4
What few people understand is that stem cells are capable of self-renewal, as well as producing mature functional cells.1,4
In medical practice today, stem cells are used for regenerative purposes. This is evidenced by the ability of bone marrow stem cell transplants to help leukemia patients.5
Based on the phenomenon of self-renewal, if our old stem cells can be reactivated, the effect could be whole-body rejuvenation.
Nutritional interventions may provide an effective approach to activate dormant stem cells, thereby enhancing tissue regeneration.
Using several lines of preclinical evidence from the scientific literature, we can outline a rational approach that could allow us to reactivate aging stem cells.
www.lifeextension.com/magazine/2002/3
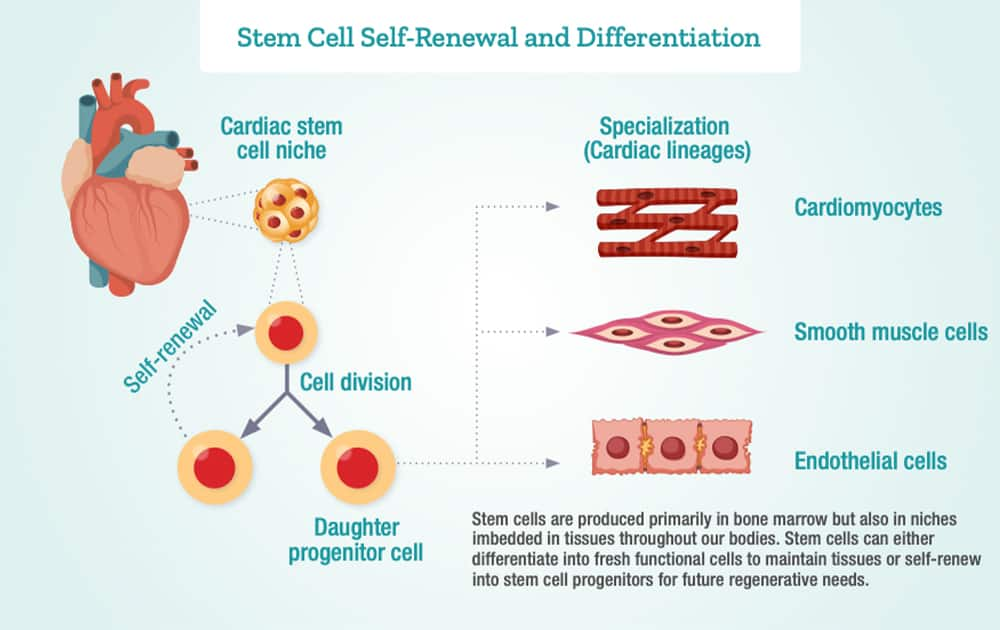
Stem Cells Are Retained with Age
Many stem cells are retained as we age and have the capacity to self-renew and differentiate into mature functional cells.4
Several factors that drive the aging process also reduce the regenerative potential of stem cells and contribute to worsening of age-related conditions.4
We now have a better understanding of specific degenerative pathways of aged tissues and how this contributes to disease, decline, and death.4,6
This gives us targets for restoring stem cell function and self-renewal using current technologies.4,6
Rejuvenating Aged Stem Cells
Normal aging (along with excess calorie ingestion) causes AMPK and NAD+ to plummet, mTOR to be imbalanced, and SIRT1 signaling to be downregulated.7-10
The impact of this is depletion of our stem cell pools and a reduced regenerative potential.11
Stem cell malfunction can be partially corrected with many of the nutrients that readers of Life Extension® magazine supplement with today.
This includes curcumin,12,13 resveratrol,14-16 Gynostemma pentaphyllum,17 NAD+ precursors,18,19 and drugs like metformin,20-22 along with sensible eating patterns.23
Stem Cell Rejuvenation in Laboratory Models

The challenge of maintaining healthy stem cells requires fighting off the same damaging factors that compromise our functional tissue cells.
These factors include damaged DNA, mitochondrial dysfunction, chronic inflammation, and oxidative stress.
Emerging data indicate that interventions that blunt the effects of excessive calorie intake can rejuvenate some lineages of stem cells by:11,23-25
- Activating AMPK
- Suppressing mTOR
- Boosting sirtuins
Sirtuins are indispensable for DNA repair, controlling inflammation and other life sustaining processes. Resveratrol activates sirtuins but requires NAD+ for optimal functionality.26
In aged mice, treatment with the NAD+ precursor nicotinamide riboside rejuvenated muscle stem cells.19
This study showed that boosting NAD+ improved mitochondrial function in muscle stem cells and inhibited stem cell senescence. The researchers also showed that boosting NAD+ decreased senescence of brain and skin stem cells.19
An ongoing clinical trial may reveal neurological improvement in response to aggressive NAD+ boosting therapy.27
Clinical trials have demonstrated that oral administration of NAD+ precursors results in increased NAD+ levels,28,29 which are vital for stem cell functionality.19
AMPK + Resveratrol + NAD+ = Stem Cell Rejuvenation

In response to resveratrol, cells express proteins called sirtuins that provide several benefits including generating new mitochondria.30
Sirtuins are dependent on NAD+ to interact with FoxO, a beneficial transcription factor, to promote healthy gene expression.26,31
The cellular enzyme AMPK has a dynamic interaction with sirtuin 1 (SIRT1).
The potential combined benefit of boosting AMPK, NAD+, SIRT1 and FoxO is the favorable impact this might have in promoting stem cell health.
Moreover, AMPK activity helps to normalize excess mTOR, which can impede stem cell functionality.23
Multi-Modal Approach to Stem Cell Renewal
We now know of several factors involved in the maintenance and potential rejuvenation of our aging stem cells.
The encouraging aspect of all this is we can target these stem cell renewal processes today via:
- AMPK activation35
- Sirtuin activation24,54
- FoxO activation55,56
- NAD+ replenishment19
- mTOR regulation (via AMPK activation)6,57
This approach may enable elderly individuals to rejuvenate their aged stem cells, which would then repopulate senile tissues with fresh, functional (somatic) cells.
Understanding the Role of mTOR in Obesity and Aging
“mTOR” stands for the mechanistic target of rapamycin.
It is a protein found inside most cells and is responsible for regulating cellular growth by sensing and integrating diverse nutritional and environmental cues.7
Excessive activation of cellular mTOR is involved in diseases plaguing aging populations, such as cancer, type II diabetes, and obesity.7
Regulating mTOR activity extends lifespan in laboratory models by delaying the development of chronic diseases, including cancer.32
Maintenance of stem cell pools requires a finely tuned balance between stem cell renewal and differentiation.6
When mTOR is excessively activated in certain stem cell lineages, the pool of stem cells becomes exhausted.32 This diminishes our ability to regenerate our tissues with fresh functional cells.6
When properly balanced, mTOR will not adversely impact cellular aging.33-37
Enhancing autophagy in hematopoietic stem cells improves their regenerative capacity.38
One way of inducing autophagy is suppression of excess mTOR via AMPK activation.38
According to a report published in the journal Nature:
“…it will be exciting to test whether rejuvenation interventions aimed at activating autophagy in unhealthy autophagy-inactivated oHSCs [old hematopoietic stem cells] will improve the health of the aging blood system.”38
Most People Need to Lower mTOR
Regulation of mTOR represents a viable approach to preserve the stem cell pool.
This, in turn, would help maintain functionality of our tissues and organs over time.
When calorie intake is reduced, mTOR activity diminishes, and autophagy is beneficially activated.
This process (autophagy) cleans up accumulated cellular waste products and preserves cell function.39
The autophagy-regulating signaling network that includes AMPK and mTOR serves to maintain this delicate autophagy balance.40
Interventions that activate AMPK serve to balance mTOR and enable optimal levels of cellular autophagy.
Boost Cell AMPK To Lower mTOR
AMPK was first identified in 1973 for its role in fat metabolism.41
Based on evidence from preclinical studies, it is expected that when people practice severe calorie restriction, AMPK activity increases, which confers protective effects.42
One of AMPK’s benefits is to signal cells to consume stored fat. One way that AMPK performs this fat-removing process is by down-regulating mTOR.43
AMPK is a master energy sensor in cells.35
When AMPK is activated by compounds like metformin or Gynostemma pentaphyllum,35,44 cells think they are energy deprived.
The desired effect for most aging people is to prompt cells to turn down excess mTOR and utilize fat stores for energy production.
Balancing mTOR activity and autophagy can be achieved via increasing cellular AMPK in the following six ways:
-
Reduce calorie intake and more specifically, avoid sugars and simple carbohydrates. High blood levels of glucose (and insulin) fuel excess mTOR activity.45
-
Brief periods (3-5 days) of calorie restriction per month have shown great benefits indicative of balanced mTOR,46 but compliance is difficult.
-
Consider “intermittent fasting” for 14-18 hours five days a week, based on voluminous data, including a fascinating report published in the December 26, 2019 issue of the New England Journal of Medicine.47
-
Preclinical studies show that calorie restriction mimetics such as resveratrol and NAD+ can be used to support SIRT1 and FoxO function.26,48
-
AMPK activators such as the drug metformin and/or nutrients such as Gynostemma pentaphyllum extract44,49 and hesperidin50 help support mTOR activity and autophagy.
-
Increased physical activity can meaningfully boost AMPK.51
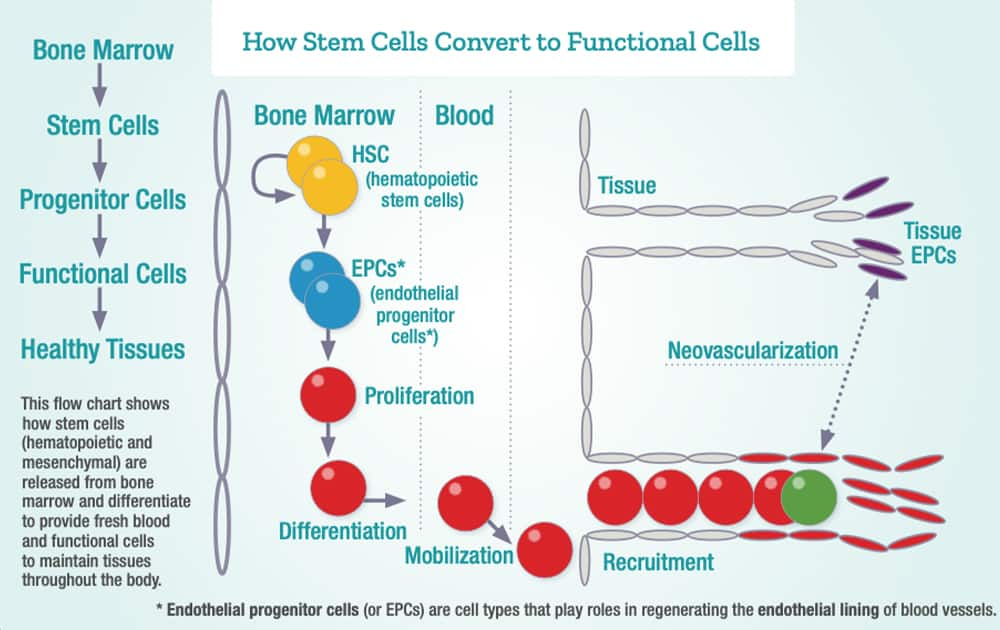
Rejuvenating Bone Marrow Stem Cells
The impact of boosting AMPK and lowering excess mTOR may enable rejuvenation of aging bone marrow (hematopoietic) stem cells.
A safe way of balancing mTOR is to boost cellular AMPK activity.
Increasing AMPK regulates mTOR, which facilitates removal of cellular debris (via autophagy).52,53
As it relates to combatting aging, activating autophagy appears to be a critical factor for the rejuvenation of aged hematopoietic stem cells.25
Hematopoietic stem cells are crucial for producing new immune cells, platelets and red blood cells.
Middle aged and elderly people today have ready access to low-cost approaches to help preserve their bone marrow stem cell pools.
When the bone marrow stem cell niche becomes exhausted, life can no longer be sustained.
That’s because oxygen-carrying red blood cells, immune-protecting white cells and hemorrhage-guarding platelets must to be continually produced in the bone marrow for systemic existence.
Increase SIRT1 with Resveratrol
Resveratrol activates SIRT1 inside cells, which is linked to many of the same longevity-enhancing benefits as calorie restriction.26
Based on our interpretation of emerging evidence, age control could be enhanced by modest doses of resveratrol, with adequate NAD+ replenishment to ensure sirtuin functionality.
Most people over age 40 should initiate supplementation with the oral NAD+ precursor (nicotinamide riboside) in the daily dose of 300 mg to 600 mg, along with 100 mg to 300 mg of resveratrol and AMPK-activating compounds.
By targeting known regulators of stem cell self-renewal and differentiation, we are proposing a unique protocol to rejuvenate your own stem cells.
Factors that Confer Stem Cell Health
As it relates to stem cell regeneration, the following processes are intimately involved:
- DNA repair pathways affected by:58
- SIRT1
- NAD+
- FoxO
- Protein synthesis affected by:55
- AMPK
- mTOR
- FoxO
- Mitochondrial function affected by:59
- SIRT1
- NAD+
- FoxO
Hallmarks of degenerative aging include dysregulation of AMPK, FoxO and SIRT1, depletion of NAD+, and excessive activation of mTOR.7,60
In what may be a unified approach to living healthier, the ability to reactivate aged stem cells is already being practiced by some enlightened people today.
This includes those who take steps to balance AMPK, SIRT1, FoxO and NAD+ while normalizing excess mTOR.
Summary Overview
Adult stem cells lose their ability to repopulate tissues with fresh functional cells.
The result is systemic deterioration of tissues throughout our aging bodies.
Treatments that are currently being used to slow aging, such as boosting AMPK and sirtuins, appear likely to facilitate stem cell rejuvenation.
Regulation of mTOR enhances the regenerative capacity of hematopoietic stem cells in aged mice.25
mTOR is hyperactive in the bone marrow stem cell niche of aged mice.25 Excess mTOR can be balanced by increasing AMPK.
Published data indicate that agents that boost NAD+,19 sirtuins24,54 and FoxO,56 along with compounds that increase AMPK35,44 (and down-regulate mTOR6,25) may work together to improve stem cell function.
Metformin is an FDA-approved drug with potent AMPK-activating properties. It and other compounds that activate AMPK (like Gynostemma pentaphyllum) represent a potential option for induction of stem cell rejuvenation in adult stem cell therapies.
The combined application of these mechanistic approaches in clinical medical practice could induce systemic rejuvenation of dysfunctional stem cells.
How FoxO Enhances Stem Cell Health
Bone-marrow-derived stem cells are termed hematopoietic stem cells and are essential to sustaining life processes.
FoxO (Forkhead box) are cell proteins that play an important role in stem cell biology.
FoxO help regulate the expression of genes involved in cell growth, proliferation, differentiation, insulin regulation, and longevity.
During aging, the removal and regeneration of functional cells becomes disturbed mainly due to a decrease in the regenerative potential of adult stem cells.
Deletion of FoxO1/3a/4 in the bone marrow of mice leads to apoptosis (death) of hematopoietic stem cells. This prevents the re-population of critical bone-marrow-derived stem cells.
Aged mice in which FoxO3a was deleted display reduced regenerative potential61 and depletion of the stem cell pool.62
Treatment of FoxO-deficient mice with N-acetylcysteine restored the hematopoietic stem cell compartment.
These observations correlate with the idea that decreased function of adult stem cells is involved in the onset of age-related diseases.2
Current evidence favorably implicates FoxO transcription factors in longer human lifespans You can learn technical details about FoxO by searching Google: “FoxO and aging.”
For simplicity’s sake, it’s good to know that boosting NAD+ and sirtuin expression (with resveratrol) promotes favorable FoxO genetic transcription.
In this Month’s Issue…
A multi-year investigation using deep-learning AI technology has led to the discovery of three natural compounds that favorably modulate signaling pathways associated with stem cell health.
One of these compounds was shown to promote hematopoietic stem cell expansion in a laboratory model, while the other demonstrated reductions in metabolic parameters (like reduced fasting insulin).
This nutrient formula is described on page 46 of this month’s issue.
Page 65 describes a published clinical trial in which biological aging was reversed on average by 2.5 years using three compounds that have a long history of use.
Page 34 is an Age Reversal Update that introduces new human studies, one that aims to achieve meaningful and statistically significant rejuvenation effects in only 12 months.
An article on page 24 reveals how type II diabetics may delay or prevent the onset of disabling neuropathy.
In the meantime, readers of Life Extension® magazine should appreciate that many of the nutrients they take today are demonstrating beneficial effects on the health of their stem cell pools.
For longer life,
William Faloon, Co-Founder
Life Extension Buyers Club
References
- Available at: https://stemcells.nih.gov/info/basics/1.htm. Accessed December 9, 2019.
- Boyette LB, Tuan RS. Adult Stem Cells and Diseases of Aging. J Clin Med. 2014 Jan 21;3(1):88-134.
- Ren R, Ocampo A, Liu GH, et al. Regulation of Stem Cell Aging by Metabolism and Epigenetics. Cell Metab. 2017 Sep 5;26(3):460-74.
- Oh J, Lee YD, Wagers AJ. Stem cell aging: mechanisms, regulators and therapeutic opportunities. Nat Med. 2014 Aug;20(8):870-80.
- Available at: https://www.cancer.org/cancer/acute-myeloid-leukemia/treating/bone-marrow-stem-cell-transplant.html. Accessed December 9, 2019.
- Ito K, Suda T. Metabolic requirements for the maintenance of self-renewing stem cells. Nat Rev Mol Cell Biol. 2014 Apr;15(4):243-56.
- Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012 Apr 13;149(2):274-93.
- Salminen A, Kaarniranta K, Kauppinen A. Age-related changes in AMPK activation: Role for AMPK phosphatases and inhibitory phosphorylation by upstream signaling pathways. Ageing Res Rev. 2016 Jul;28:15-26.
- Imai SI, Guarente L. It takes two to tango: NAD(+) and sirtuins in aging/longevity control. NPJ Aging Mech Dis. 2016 08/18/online;2:16017.
- Massudi H, Grant R, Braidy N, et al. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLoS One. 2012;7(7):e42357.
- Khorraminejad-Shirazi M, Farahmandnia M, Kardeh B, et al. Aging and stem cell therapy: AMPK as an applicable pharmacological target for rejuvenation of aged stem cells and achieving higher efficacy in stem cell therapy. Hematol Oncol Stem Cell Ther. 2018 Dec;11(4):189-94.
- Sharifi S, Zununi Vahed S, Ahmadian E, et al. Stem Cell Therapy: Curcumin Does the Trick. Phytother Res. 2019 Nov;33(11):2927-37.
- Ormond DR, Shannon C, Oppenheim J, et al. Stem cell therapy and curcumin synergistically enhance recovery from spinal cord injury. PLoS One. 2014;9(2):e88916.
- Safaeinejad Z, Kazeminasab F, Kiani-Esfahani A, et al. Multi-effects of Resveratrol on stem cell characteristics: Effective dose, time, cell culture conditions and cell type-specific responses of stem cells to Resveratrol. Eur J Med Chem. 2018 Jul 15;155:651-7.
- Safaeinejad Z, Nabiuni M, Peymani M, et al. Resveratrol promotes human embryonic stem cells self-renewal by targeting SIRT1-ERK signaling pathway. Eur J Cell Biol. 2017 Oct;96(7):665-72.
- Suvorova, II, Knyazeva AR, Petukhov AV, et al. Resveratrol enhances pluripotency of mouse embryonic stem cells by activating AMPK/Ulk1 pathway. Cell Death Discov. 2019;5:61.
- Dong L, Yang KQ, Fu WY, et al. Gypenosides protected the neural stem cells in the subventricular zone of neonatal rats that were prenatally exposed to ethanol. Int J Mol Sci.2014 Nov 28;15(12):21967-79.
- Igarashi M, Miura M, Williams E, et al. NAD(+) supplementation rejuvenates aged gut adult stem cells. Aging Cell. 2019 Jun;18(3):e12935.
- Zhang H, Ryu D, Wu Y, et al. NAD(+) repletion improves mitochondrial and stem cell function and enhances life span in mice. Science. 2016 Jun 17;352(6292):1436-43.
- Ma J, Zhang ZL, Hu XT, et al. Metformin promotes differentiation of human bone marrow derived mesenchymal stem cells into osteoblast via GSK3beta inhibition. Eur Rev Med Pharmacol Sci. 2018 Nov;22(22):7962-8.
- Dadwal P, Mahmud N, Sinai L, et al. Activating Endogenous Neural Precursor Cells Using Metformin Leads to Neural Repair and Functional Recovery in a Model of Childhood Brain Injury. Stem Cell Reports. 2015 Aug 11;5(2):166-73.
- Gilbert EAB, Morshead CM. Metformin Activates Neural Stem and Progenitor Cells in the Spinal Cord and Improves Functional Outcomes Following Injury. The FASEB Journal. 2019;33(1_supplement):208.3-.3.
- Mihaylova MM, Sabatini DM, Yilmaz OH. Dietary and metabolic control of stem cell function in physiology and cancer. Cell Stem Cell. 2014 Mar 6;14(3):292-305.
- Yuan HF, Zhai C, Yan XL, et al. SIRT1 is required for long-term growth of human mesenchymal stem cells. J Mol Med (Berl). 2012 Apr;90(4):389-400.
- Chen C, Liu Y, Liu Y, et al. mTOR regulation and therapeutic rejuvenation of aging hematopoietic stem cells. Sci Signal. 2009 Nov 24;2(98):ra75.
- Houtkooper RH, Pirinen E, Auwerx J. Sirtuins as regulators of metabolism and healthspan. Nat Rev Mol Cell Biol. 2012 Mar 7;13(4):225-38.
- Available at: https://clinicaltrials.gov/ct2/show/NCT02942888. Accessed December 11, 2019.
- Trammell SA, Schmidt MS, Weidemann BJ, et al. Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nat Commun. 2016 Oct 10;7:12948.
- Airhart SE, Shireman LM, Risler LJ, et al. An open-label, non-randomized study of the pharmacokinetics of the nutritional supplement nicotinamide riboside (NR) and its effects on blood NAD+ levels in healthy volunteers. PLoS One. 2017;12(12):e0186459.
- Price NL, Gomes AP, Ling AJ, et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012 May 2;15(5):675-90.
- Feige JN, Auwerx J. Transcriptional targets of sirtuins in the coordination of mammalian physiology. Curr Opin Cell Biol. 2008 Jun;20(3):303-9.
- Papadopoli D, Boulay K, Kazak L, et al. mTOR as a central regulator of lifespan and aging. F1000Res. 2019;8:F1000 Faculty Rev-998.
- Deblon N, Bourgoin L, Veyrat-Durebex C, et al. Chronic mTOR inhibition by rapamycin induces muscle insulin resistance despite weight loss in rats. Br J Pharmacol. 2012 Apr;165(7):2325-40.
- Kim YC, Guan KL. mTOR: a pharmacologic target for autophagy regulation. J Clin Invest. 2015 Jan;125(1):25-32.
- Burkewitz K, Zhang Y, Mair WB. AMPK at the nexus of energetics and aging. Cell Metab. 2014 Jul 1;20(1):10-25.
- Viollet B, Horman S, Leclerc J, et al. AMPK inhibition in health and disease. Crit Rev Biochem Mol Biol. 2010 Aug;45(4):276-95.
- Chakrabarti P, Kandror KV. The role of mTOR in lipid homeostasis and diabetes progression. Curr Opin Endocrinol Diabetes Obes. 2015 Oct;22(5):340-6.
- Ho TT, Warr MR, Adelman ER, et al. Autophagy maintains the metabolism and function of young and old stem cells. Nature. 2017 Mar 9;543(7644):205-10.
- Madeo F, Zimmermann A, Maiuri MC, et al. Essential role for autophagy in life span extension. J Clin Invest. 2015 Jan;125(1):85-93.
- Alers S, Loffler AS, Wesselborg S, et al. Role of AMPK-mTOR-Ulk1/2 in the regulation of autophagy: cross talk, shortcuts, and feedbacks. Mol Cell Biol. 2012 Jan;32(1):2-11.
- Goodman M, Liu Z, Zhu P, et al. AMPK Activators as a Drug for Diabetes, Cancer and Cardiovascular Disease. Pharm Regul Aff. 2014 Jul;3(2):118.
- Canto C, Auwerx J. Calorie restriction: is AMPK a key sensor and effector? Physiology (Bethesda). 2011 Aug;26(4):214-24.
- Laplante M, Sabatini DM. An emerging role of mTOR in lipid biosynthesis. Curr Biol. 2009 Dec 1;19(22):R1046-52.
- Gauhar R, Hwang SL, Jeong SS, et al. Heat-processed Gynostemma pentaphyllum extract improves obesity in ob/ob mice by activating AMP-activated protein kinase. Biotechnol Lett. 2012 Sep;34(9):1607-16.
- Sanguesa G, Roglans N, Baena M, et al. mTOR is a Key Protein Involved in the Metabolic Effects of Simple Sugars. Int J Mol Sci. 2019 Mar 5;20(5):1117.
- Escobar KA, Cole NH, Mermier CM, et al. Autophagy and aging: Maintaining the proteome through exercise and caloric restriction. Aging Cell. 2019 Feb;18(1):e12876.
- de Cabo R, Mattson MP. Effects of Intermittent Fasting on Health, Aging, and Disease. N Engl J Med. 2019 Dec 26;381(26):2541-51.
- Ding S, Jiang J, Zhang G, et al. Resveratrol and caloric restriction prevent hepatic steatosis by regulating SIRT1-autophagy pathway and alleviating endoplasmic reticulum stress in high-fat diet-fed rats. PLoS One. 2017;12(8):e0183541.
- Park SH, Huh TL, Kim SY, et al. Antiobesity effect of Gynostemma pentaphyllum extract (actiponin): a randomized, double-blind, placebo-controlled trial. Obesity (Silver Spring). 2014 Jan;22(1):63-71.
- Rizza S, Muniyappa R, Iantorno M, et al. Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J Clin Endocrinol Metab. 2011 May;96(5):E782-92.
- Richter EA, Ruderman NB. AMPK and the biochemistry of exercise: implications for human health and disease. Biochem J. 2009 Mar 1;418(2):261-75.
- Kroemer G. Autophagy: a druggable process that is deregulated in aging and human disease. J Clin Invest. 2015 Jan;125(1):1-4.
- Mihaylova MM, Shaw RJ. The AMPK signalling pathway coordinates cell growth, autophagy and metabolism. Nat Cell Biol. 2011 Sep 2;13(9):1016-23.
- Liu X, Hu D, Zeng Z, et al. SRT1720 promotes survival of aged human mesenchymal stem cells via FAIM: a pharmacological strategy to improve stem cell-based therapy for rat myocardial infarction. Cell Death Dis. 2017 Apr 6;8(4):e2731.
- Martins R, Lithgow GJ, Link W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging Cell. 2016 Apr;15(2):196-207.
- Warr MR, Binnewies M, Flach J, et al. FOXO3A directs a protective autophagy program in haematopoietic stem cells. Nature. 2013 Feb 21;494(7437):323-7.
- Signer Robert AJ, Morrison Sean J. Mechanisms that Regulate Stem Cell Aging and Life Span. Cell Stem Cell. 2013 Feb 7;12(2):152-65.
- Brunet A, Sweeney LB, Sturgill JF, et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science. 2004 Mar 26;303(5666):2011-5.
- Mouchiroud L, Houtkooper RH, Moullan N, et al. The NAD(+)/Sirtuin Pathway Modulates Longevity through Activation of Mitochondrial UPR and FOXO Signaling. Cell. 2013 Jul 18;154(2):430-41.
- Ruderman NB, Xu XJ, Nelson L, et al. AMPK and SIRT1: a long-standing partnership? Am J Physiol Endocrinol Metab. 2010 Apr;298(4):E751-60.
- Miyamoto K, Araki KY, Naka K, et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell. 2007 Jun 7;1(1):101-12.
- Tothova Z, Gilliland DG. FoxO transcription factors and stem cell homeostasis: insights from the hematopoietic system. Cell Stem Cell. 2007 Aug 16;1(2):140-52.

