
Prostate Cancer Prevention
Prostate Cancer Prevention
Last Section Update: 03/2021
Table of Contents
1 Introduction
Summary and Quick Facts for Prostate Cancer Prevention
- More than 230,000 new cases of prostate cancer are diagnosed each year. Risk of prostate cancer increases considerably with advancing age, so taking preventive steps is important for aging men.
- This protocol reviews how prostate cancer develops and how it is typically diagnosed and treated, along with some intriguing tests and natural therapies targeting prostate cancer prevention. Dietary, lifestyle and supplement approaches that may support prostate health will be summarized as well.
- Various preparations of pomegranate and green tea have been shown in clinical trials to reduce levels of PSA, a marker reflective of prostate cancer progression.
Prostate Cancer Prevention Controversy
By William Faloon
An editorial I generated for the May 2013 issue of Life Extension magazine® received quite a bit of feedback and critique.
Some Life Extension® customers said it should be a mandatory part of physician education. Others raised concerns about the use of the PSA blood test as a screening tool, why I suggest Avodart® for certain men, and why drugs were mentioned since there are nutrients that function via similar mechanisms.
The most impressive critique came from Patrick C. Walsh, MD, who may be the most renowned prostate cancer expert in the world. Dr. Walsh was involved in identifying the genetic characteristic of hereditary prostate cancer and pioneered “nerve-sparing” surgery. I have urged hundreds of prostate cancer patients to travel to Johns Hopkins to have Patrick Walsh perform their surgery, as I consider him the finest in the world.
So when Dr. Walsh writes us, I pay attention, and Life Extension customers should be informed that there are credentialed individuals that are against using drugs in the class of Avodart® for cancer prevention purposes.
Shortly after my 2013 editorial was published, the American Urological Association issued revised guidelines for PSA screening. They now say PSA screening should be mostly considered only for men aged 55-69 (AUA 2013). We vehemently disagree with this new recommendation and chastise this group for not emphasizing the need to devise safer and more efficient ways of performing prostate diagnostics.
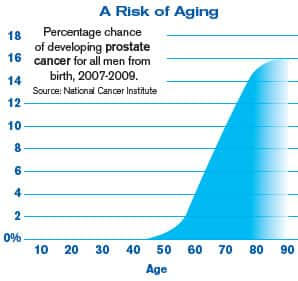
To emphasize the seriousness of all this, the chart titled “A Risk of Aging” shows the spiraling incidence of prostate cancer that occurs as men age. Autopsy results reveal that 85% of men have atypical cells in their prostate glands and 1 in 4 has cancer (Billis 1986). While many men with atypical lesions or even malignant cells in their prostate do not ever progress to clinical disease, aging men cannot ignore this problem.

The public still accepts absurdly short life spans. We at Life Extension do not and that is just one reason why our position on prostate cancer differs from the mainstream.
There is something to be said about attending live lectures as opposed to staying glued to our computer/TV screens. A good speaker can make an impact that you may forever remember.
I’ll never forget a lecture I attended in 1977 at a South Florida condominium social hall. The place was packed with retirees. The lecturer was over age 80 and passionately urged all men to visit a urologist once a year for a digital rectal exam. He began by reading a long list of the names of the many members of his retirement community who had suffered agonizing deaths from metastatic prostate cancer.
The lecturer understood that a digital rectal exam would not detect all prostate cancers, but he knew it could save lives. If the PSA blood test had been available at that time, I can only imagine how feverish this benevolent speaker would have been in advocating PSA tests to his fellow men.
Move forward 35 years and the federal government and some mainstream medical groups are recommending against PSA screening, which is more reliable than digital rectal exams, though both ideally should be done annually (AUA 2013; Moyer 2012).
What Makes Prostate Cancer Different?
Prostate cancer is unusual in that it has a blood marker called prostate-specific antigen (PSA) that can facilitate early detection, thereby enabling therapies to be employed before cancer spreads to regional lymph nodes or distant metastases occur (Catalona 1994).
With the advent and widespread use of PSA screening, an argument can be made based on a large human study that huge numbers of men could be spared agonizing deaths from metastatic prostate cancer (Bokhorst 2013; USDC 2012). The earlier diagnosis of prostate cancer, however, must be put into context of the individual patient to ascertain which men need to be treated and which men are reasonable candidates for active surveillance or “watchful waiting.”
The journal European Urology published a study in 2013 conducted on nearly 35,000 men aged 55-69 (Bokhorst 2013). This data came from the European Randomized Study of Screening for Prostate Cancer, a major, robust study examining the impact of PSA screening over a median period of 13 years on prostate cancer mortality. The eye-opening conclusion was that men who underwent repeated PSA screening were 51% less likely to die from prostate cancer than men who did not undergo screening (Bokhorst 2013). If the statistics from this study are applied to the entire population of men aged 55-69 in the United States, PSA screening could potentially save over 80,000 lives in a 13-year period (USDC 2012).
The United States Preventive Services Task Force (USPSTF) published a report in 2012 recommending that men stop undergoing PSA screening (Moyer 2012).
Life Extension disagreed with the USPSTF recommendation, particularly as it relates to our customers to whom we are steadfastly committed. We know that in the absence of PSA screening, prostate cancer will once again be diagnosed at an advanced stage, when there is painful bulky disease and only a small chance of curative therapy.
The widespread use of PSA testing beginning in 1987 enabled doctors to identify prostate cancer at a greatly reduced stage of disease (NCI 2012). If the dictum of the USPSTF is followed, a major advance in medicine will be erased.
The Staggering Statistics
Here is what the American Cancer Society says about prostate cancer in the United States (ACS 2013d):
- More than 238,000 new cases of prostate cancer are diagnosed yearly (based on 2013 data).
- Over 29,000 men die of prostate cancer yearly (based on 2013 data).
- About 1 man in 6 will be diagnosed with prostate cancer during his lifetime.
- The average age at diagnosis is 67.
- Prostate cancer is the second leading cause of cancer death among American men.
- About 1 man in 36 will die from prostate cancer.
If prostate cancer were an infectious illness, there would be widespread panic. To put this in perspective, HIV infected less than 50,000 Americans in 2011 (CDC 2013a).
In 2013, the United States Preventive Services Task Force urged all Americans to undergo routine HIV screening (USPSTF 2013).

There are valid reasons for HIV screening, but almost five times more Americans are diagnosed with prostate cancer each year compared to HIV (ACS 2013d; CDC 2013a). The same government-funded Task Force that suggests universal HIV screening does not want aging men to benefit from early detection of prostate cancer. They maintain that the treatment is worse than the disease. They confuse the message conveyed by the PSA with the judgment and actions of physicians who too often are programmed toward invasive and expensive therapies.
Do we toss out the baby with the bath water, so to speak, because physicians are not taking the time, or possibly do not have the expertise to advise patients soundly? The actions of the USPSTF and the American Urological Association should be to fix the deficiency of the physician with strict guidelines, just as was done in the 1980s to alter the routine use of the radical mastectomy performed in almost every woman diagnosed with breast cancer (Ghossain 2009).
The United States Preventive Services Task Force (USPSTF) prefers aging men wallow in ignorance concerning their prostate health, which within the next decade will send death rates spiraling upwards. The USPSTF clearly wants aging men to bury their heads in the sand and not concern themselves about prostate cancer.
The hard statistics showing more than 238,000 newly-diagnosed prostate cancer cases annually proves otherwise (ACS 2013d). While the USPSTF recommendations will save government health programs billions of dollars in the short term, there will be catastrophic long term costs to pay when record numbers of men who could have been cured instead develop metastatic disease.
Why Life Extension Customers Are Different
There are factors that influence mainstream recommendations that do not pertain to Life Extension customers. The typical American male over age 60 is remarkably unhealthy, often suffering multiple underlying maladies relating to metabolic syndrome and other pathologies called “co-morbidities” (Ervin 2009). This is indicative of a state of disease in the biologic environment of the patient.
A frank diagnosis (or indication) of prostate cancer should act as an early warning that something is amiss in the patient’s overall health and that further attention is warranted to various systems. Thus a diagnosis of prostate cancer need not be equated with invasive procedures such as radical prostatectomy, radiation therapy, cryosurgery, high-intensity focused ultrasound, or androgen deprivation therapy, but with a call to the patient and physician to be alert to pathologic states that if corrected can stabilize or repair some or all of the systems that are amiss.
One reason the USPSTF believes that PSA screening should be halted is that so many men are already in such poor health they are likely to die of some other cause before prostate cancer becomes clinically relevant (Moyer 2012).
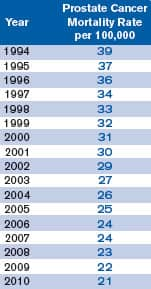
Huge Decline in Prostate Cancer Deaths Since Advent of PSA Screening
This is the opposite of Life Extension customers, who go to extraordinary efforts to slow aging and protect against degenerative disease. It would be irrational for healthy Life Extension customers to stop PSA screening merely because their age group on average is in such poor overall health.
Few doctors today have comprehensive programs designed to reverse multiple underlying factors that lead to clinically-diagnosed prostate cancer. The typical aging person does not know about lifestyle changes, drugs, and nutrients that may keep an indolent cancer confined to the prostate gland.
Life Extension customers have long been armed with this information and have access to Wellness Specialists to help guide them to more effective ways of working with their physician to improve their odds of keeping low-grade prostate cancer, or indications of low-grade prostate cancer (such as rising PSA), under control. This protocol is dedicated to reminding members and alerting the public about these novel approaches to disease prevention.
Most urologists believe when PSA reaches a certain level that their only choice is to perform needle biopsies. They often overlook existing tests, such as testing and properly analyzing blood results of free PSA percentage, PSA density, and PSA velocity, along with other diagnostics such as PCA3 urinary test and advanced non-invasive techniques that can provide additional insight that may reduce the need for invasive procedures (Vessella 2000; Lieberman 1999; Stephan 2005; Loeb, Carter 2013; Hessels 2009). Urology patients are not always made aware of these non-invasive choices, and especially of the importance of measuring the PSA rise over time (PSA velocity) to help ascertain if prostate biopsy is warranted.
What clearly separates Life Extension customers from the general public, however, are the aggressive steps we take to achieve meaningful extensions of our healthy life spans. Those advising against PSA screening are largely “writing-off” men over age 70.
Life Extension male members need to ensure their prostate health is assessed and maintained at an optimal level for the many decades of extended life they expect.
American Urological Association Capitulates
When the United States Preventive Services Task Force suggested that aging men stop PSA screening altogether, the American Urological Association disagreed. About a year later, the American Urological Association issued revised guidelines that will sharply reduce the number of PSA screenings performed (AUA 2013; Pollack 2012; Allard 2012). And other professional groups have issued similar opinions (Qaseem 2013).
The latest recommendation from the American Urological Association (AUA) is for men over age 70 to avoid PSA screening (AUA 2013). The AUA is essentially saying that once you move past age 70, your life span is too short to bother with.
The American Urological Association is also writing off men aged 40-54 for prostate screening because of the relative low incidence of cancer in this group compared to men over 54 (AUA 2013). This is a tragedy as it condemns younger men who do develop prostate cancer to probable death. Earlier diagnosis provides a huge advantage when attempting curative therapy. Just ask Prostate Cancer Foundation Chairman Michael Milken, who insisted on a PSA test at age 46 and discovered he had prostate cancer in time to benefit from curative therapy (Moore 2005).
On the flip side are famous people like Frank Zappa, Telly Savalas, Bill Bixby, and other younger men who likely could have identified their prostate cancer earlier had they undergone PSA screening (PCCNC 2013). These men probably had rising PSA levels long before metastatic disease manifested.
Overlooking More Efficient Procedures
In recommending more limited PSA-screening, the American Urological Association is tacitly admitting that conventional diagnostic and early treatment of prostate cancer is so inadequate, or performed so incompetently, that it’s better to wait for full-blown metastatic disease to manifest. Once advanced stage prostate cancer develops, however, treatments are seldom curative.
Instead of looking at physicians who are diagnosing and treating early stage prostate cancer using less invasive procedures and then emulating these skilled artists, the American Urological Association has apparently caved in to accepting and promoting mediocrity within their profession.
A big problem is that most urologists are not properly assessing PSA results, nor are they efficiently implementing further diagnostic and treatment protocols. And on the other end of the spectrum are the many men who are promptly sent for ultrasound-guided biopsies after one PSA elevation. And again, to add insult to injury, the biopsies are often not ones targeted to abnormalities within the prostate but merely targeting the prostate as a gland.
It is one issue to biopsy an ultrasound lesion that may represent the needle in the haystack, but it’s another issue, and a sad one at that, when it is the haystack that is the target. You know that this is the case when a man has had 2, 3, or 4 prostate biopsies showing no cancer cells, and then he is referred, finally, to a competent physician who uses excellent ultrasound equipment to directly target suspicious lesions within the prostate gland.
In these cases, it seems the diagnosis is magically made; but it’s not magic, it is just an issue of a far higher degree of competence. All men are not equal in talent and all equipment is not of the same quality. The unfortunate outcome is that too many aging men are being subjected to needless and incompetently administered invasive procedures that sometimes result in unnecessary suffering and premature death.
Instead of recommending that medical professionals upgrade their evaluation and treatment protocols to deliver state-of-the-art technology, the United States Preventive Services Task Force suggests that aging men not undergo PSA screening at all, while the American Urological Association limits its recommendation for PSA screening mostly to men aged 55-69 (AUA 2013).
The media treats these authoritarian groups as being virtually infallible.
Prostate Cancer Not an Isolated Disease
A common mistake made by doctors and patients is thinking that prostate cancer manifests in isolation from other pathological events occurring as a person ages. This is not the case.
Research shows that other serious pathological conditions are frequently seen in prostate cancer patients (Post 1999). These factors involved in prostate malignancy can adversely impact other parts of the body (Howcroft 2013).
For example, Life Extension has shown one way prostate cancer and coronary atherosclerosis are related is that they are both influenced by the breakdown of bone (Faloon 2009). As an aging man develops osteoporosis, excess calcium released into the blood contributes to arterial calcification (Faloon 2009). What’s lost in the bone ends up in the coronary arteries and other major vessels of the body (Faloon 2009; Demer 2004).
These atherosclerotic lesions are not vascular “calcifications” but bone growth or osteogenesis (Demer 2004; Demer 2009; Abedin 2004). Bone breakdown also releases growth factors into the blood that promote the proliferation of what may have been indolent prostate cancer cells (Patterson 2010). Therefore, it should come as no surprise that nutrients that prevent bone loss such as vitamin K2 also inhibit vascular calcification (Beulens 2009; Fodor 2010).
PSA screening thus provides an important clue of a man’s overall health, with the advantage of identifying problems early enough to take effective corrective actions. That’s a LOT of benefit for assessing one’s prostate gland once a year utilizing PSA blood testing.
Where’s the Accountability?
The level of medical competency directly affects the quality and quantity of the lives of others, yet there is not enough monitoring of patient outcomes.
When it comes to treating prostate disease, there needs to be a reporting of serious side effects such as incontinence, impotence, and major blood loss or urethral strictures after a urologist performs a radical prostatectomy.
This kind of accountability is relatively non-existent in today’s bureaucratic medical environment, though the Internet may eventually enable patients to assess the degree of medical competency of a physician they entrust their life to.
What Makes Cancer Cells Propagate?
When designing prevention and treatment strategies, Life Extension focuses on underlying mechanisms of disease that are fueled by specific biological factors in the body. This is not perfect science however because you can block one factor involved in tumor development, and cancer cells will use other growth-promoting vehicles to progress.
What we seek to do is stay two steps ahead of the cancer by cutting off its many growth promoters and pathways used to escape eradication. For instance, we know that dihydrotestosterone (DHT) promotes prostate cell growth (proliferation) (Wen 2013). This growth affects both benign prostate cells as well as cancerous ones. In the context of a man with prostate cancer, a serial rise in PSA is circumstantial evidence that the tumor cell population is increasing. Such an increase in PSA is not only of importance insofar as prompting investigations to rule out prostate cancer. Basic research data suggest that PSA can function to break down cellular proteins and help malignant cells invade tissues (Webber 1995; Cummings 2011). This could enable malignant prostate cells to penetrate the prostate capsule and spread to soft tissue, regional lymph nodes and eventually bones.
But suppressing DHT alone is not a total solution. There are other prostate tumor growth promoters such as insulin, estrogen, prolactin, transforming growth factor beta (TGF-1 and TGF-2), and vascular endothelial growth factor (VEGF) that also should be brought under control (Cox 2009; Singh PB 2008; Giton 2008; Dagvadorj 2007; Tu 2003; Ling 2005; Häggström 2000). Fortunately, many of the nutrients Life Extension customers already take can help suppress growth factors used by prostate cancer cells (and other cancers) to proliferate (Meyer 2005; Ripple 1999; Yan 2009; Hussain 2003; Giovannucci 1995; McLarty 2009; Liang 1999; Singh RP 2008; Smith 2008; Xing 2001).
There are other mechanisms involved in the evolution of a prostate tumor such as 5-lipooxygenase (5-LOX) (Gupta S 2001; Matsuyama 2004; Ghosh 1997) and cyclooxygenase-2 (COX-2) (Xu 2008) that can be markedly improved by dietary changes, along with curcumin (Bengmark 2006; Lantz 2005), fish oil (Taccone-Gallucci 2006; Calder 2003; Norrish 1999), boswellia (Safayhi 1995), aspirin (Salinas 2010), Zyflamend® (Bemis 2005; Yang P 2007; Capodice 2009; Huang 2012; Sandur 2007), and prescription COX-2 inhibitors like Celebrex® (Harris 2009; Pruthi 2006).
Genetic factors involved in prostate cancer initiation and promotion may be favorably modulated by taking relatively high doses of vitamin D (Chen L 2009; Flanagan 2006). Hormonal influences like prolactin and insulin can benefit from using prolactin-suppressing drugs like cabergoline (Dostinex®) (Webster 1992) or Lisuride (Bohnet 1979) and the insulin-suppressing drug metformin (Clements 2011; Hitron 2012; Wright 2009).
The overriding goal in reversing any cancer is to induce favorable changes in the genes that regulate cell proliferation and apoptosis (cell destruction). We know that nutrients like curcumin (Teiten 2010; Shishodia, Singh 2007; Reuter 2011), genistein (Chen 2011; Lakshman 2008; Davis 1998; Davis 1999), fish oil (Berquin 2007; Deckelbaum 2006), and vitamin D (Krishnan 2003; Mantell 2000) favorably affect genes involved in carcinogenesis, as do drugs like aspirin (Yoo 2007; Kim 2005), metformin (Jalving 2010; Avci 2013; Isakovic 2007), finasteride (Proscar®) (Luo 2003), and dutasteride (Avodart®) (Schmidt 2009).
THE WHOLISTIC NATURE OF HEALTH IN RELATION TO PROSTATE CANCER
As we learn more about specific health issues we see evidence of the interconnectivity of all key processes involved in mind and body functions. This should come as no surprise since this phenomenon characterizes all living entities, from the atom to the universe.
Importance of Food Choices
What one eats (and doesn’t eat) makes a huge impact on whether prostate cancer ever develops (Miano 2003; Itsiopoulos 2009).
Healthier eating patterns also improve the odds of treatment success (Ornish 2008; Kenfield 2007).
A rising PSA level or prostate cancer diagnosis can be the signal that it’s time to switch what you eat more towards a Mediterranean diet that focuses on fish instead of red meat, whole vegetables instead of glucose-spiking starches/sugars, foods cooked at lower temperatures, and reduced intake of omega-6 fats (Ferris-Tortajada 2012; Kenfield 2013; Sofi 2008).
Those who pioneered aggressive dietary changes to help treat cancer were decades ahead of their time. While it’s unlikely that aggressive dietary alterations will cure clinically diagnosed prostate cancer, there are strong mechanistic values to consuming foods/beverages that suppress prostate cancer proliferation (like cruciferous vegetables [Xiao 2003; Garikapaty 2005; Srivastava 2003] and green tea [Chuu 2009; Thakur 2012]) as opposed to continuing to eat foods that have been related to higher prostate cancer risk such as red meat (Punnen 2011; Michaud 2001; Richman 2011), starches and sugars (Bidoli 2005; Freedland 2009), excess dairy (Michaud 2001; Song 2013; Chan 2001; Gao 2005), and excess omega-6 fats that contribute to a high omega-6:omega-3 ratio (Williams 2011; Masko 2013).
Vitamin D Decreases Gleason Tumor Score
If a needle biopsy of the prostate detects a malignancy, it will be graded with a Gleason score number as follows:
Under 7(low-grade): Slow growing and not likely to be aggressive (SJPHS 2013). Low-grade prostate cancers are seldom the cause of death in men over age 70, especially those that are in poor health (Stangelberger 2008). Low-grade are the majority of prostate tumors found and the ones where “watchful waiting” is often employed in lieu of radical procedures (Stangelberger 2008; Gofrit 2007).
Over 7 (high-grade): Fast growing, aggressive tumors that require intervention such as radical prostatectomy, radiation, androgen ablation, etc (SJPHS 2013). High-grade prostate tumors make up less than 15%* of newly diagnosed prostate cancers (Gofrit 2007).
*Caveat: Errors in the pathology lab can result in lethal mistakes, such as issuing a low Gleason score to a high-grade tumor. These errors are discovered when a radical prostatectomy is performed and it is found to have a Gleason score of 8-10 as opposed to a 6 Gleason score found in the biopsied specimen (Carter 2012).
A study published in 2012 evaluated a group of men with early-stage prostate cancer who received a 4,000 IU vitamin D3 supplement each day for a year (Marshall 2012).
Mean 25-hydroxyvitamin D blood levels at baseline were 32.8 ng/mL and increased to 66.2 ng/mL after vitamin D supplementation (Marshall 2012).
After one year, 55% of the men showed a decrease in tumor-sensitive biopsies or a decrease in the Gleason tumor score. An additional 11% showed no change (meaning the cancer had not progressed).
The study also showed that over time, supplementation with vitamin D3 led to a decrease in the number of positive cores taken during prostate biopsies. This is in stark contrast to the untreated control group who experienced an increase in the number of positive cores on repeat biopsies (Marshall 2012).
Only 34% of men taking vitamin D progressed compared to 63% of the control group. This represents a 46% reduction in the number of men who moved to advancing disease, indicating powerful effects of taking 4,000 IU/day of vitamin D for one year.
The men in this study had not received any other treatment than vitamin D and all were in an active surveillance program that carefully measured disease progression or regression.
This study showed that just one intervention (4,000 IU/day/vitamin D) was able to reverse the clinical course of disease in a significant percentage of these prostate cancer patients.
This study helps validate the importance of PSA screening. Had these men not known they had early-stage prostate cancer, they would not have known to take vitamin D, and their disease would have likely progressed until symptoms such as bone pain manifested.
The section of this protocol titled, “Impact of Diet on Prostate Cancer Risk and Mortality” describes foods that promote prostate cancer and which ones protect against it. We explain how consuming the wrong foods can fuel prostate cancer growth, while following healthy dietary choices can reduce the risk that you will develop clinically diagnosed prostate cancer.
Some men instinctively start eating healthier as they mature, but it took a higher PSA reading (1.4 ng/mL) ten years ago for me to alter my diet in a healthier direction. My diet is not perfect, but it’s a huge improvement over what I consumed in my younger years. My last PSA test came in at 0.4 ng/mL…a 71% decrease in a ten-year period (PSA levels normally rise with age).
If I had not had my PSA checked annually, I may have continued making poor dietary choices and may have developed prostate cancer by now. My father was diagnosed with it around age 75. He consumed a typical diet for his era, with a daily intake of red meat and high glycemic starches like potatoes, while never touching a vegetable or fruit. He set himself up perfectly to encourage prostate cancer growth and mutation.
Even for those who aren’t sure if they are making the proper food choices, laboratory tests like the Omega Check™ test (a fatty acid profile) enable one to evaluate their diet and supplement program and make changes to optimize health. You are what you eat and what you assimilate does have a bearing on your health.
Five Stages of Prostate Cancer Progression

As prostate cancer progresses from Stage I to Stage IV, the cancer cells grow within the prostate, through the outer layer of the prostate into nearby tissue, and then to lymph nodes or other parts of the body.
A More Rational Approach
Most prostate tumors are very sensitive to their internal environment or what we prefer to call their “biological milieu.” We know this because when androgen-deprivation therapy is properly administered, PSA levels can drop to near zero and prostate cancer cells die through the process of programmed cell death, a.k.a. apoptosis (Grossmann 2001; Nishiyama 2011).
However, it is not uncommon for prostate cancers to eventually find other growth factors to fuel their continued proliferation and the anti-proliferative and pro-apoptotic effects of androgen-deprivation therapy wear off, as evidenced by a continuously rising PSA that was once brought down to below 0.05 ng/mL by adequately suppressing testosterone (Cox 2009; Singh PB 2008; Giton 2008; Dagvadorj 2007; Tu 2003; Ling 2005; Häggström 2000; Nishiyama 2011).
When a diagnosis of prostate cancer occurs in the setting of a rising PSA in the lower range (below 4 ng/dL ideally), Life Extension views this as an opportunity for early intervention that might result in one’s body regaining control over tumor expansion.
We know that the drug Avodart® (dutasteride) lowers PSA levels by inhibiting the formation of dihydrotestosterone (DHT) (Arena 2013). Avodart® and its less potent cousin Proscar® (finasteride) are 5ARIs (5-alpha reductase inhibitors) (Knezevich 2013). 5-alpha reductase is the enzyme that converts testosterone to DHT (Knezevich 2013). The effect of DHT on prostate cancer cell growth is five times greater than that of testosterone (UCF 2013). By blocking DHT, drugs like Avodart® and Proscar® provide a unique opportunity to suppress tumor growth. At the same time, comprehensive adjunct protocols can be initiated that are designed to deprive tumor cells of growth factors or fuels, further inhibiting cancer growth and/or invasion.
For example, a recent study found that men taking finasteride for prostate cancer prevention were far more likely to benefit if they had lower estrogen levels prior to initiation of treatment with finasteride (Kristal 2012). This study clearly showed high concentrations of estrogen to be associated with increased cancer risk. So much so that the elevated estrogen neutralized the prostate cancer prevention impact of finasteride. Life Extension has repeatedly warned aging men about the critical need of achieving estrogen balance. One reason was our continued observation of high estrogen levels in newly diagnosed prostate cancer patients. Men can easily suppress elevated estrogen levels with aromatase-inhibiting therapies (Ta 2007).
So in response to a rising PSA and/or other indicators of prostate disease, men have a range of diagnostic options to assess whether there is underlying malignancy and if there is, what may be helping to fuel it (such as elevated DHT or estrogen).
If non-invasive diagnostics indicate malignancy, a color Doppler ultrasound-guided biopsy can indicate whether it may be high-grade (Gleason score over 7 that requires treatment) or low-grade (Gleason score under 7 that may be controlled with comprehensive surveillance/intervention).
Some men choose to attack a rising PSA as if there is already low-grade prostate cancer present, especially if they suffer urinary symptoms relating to benign prostatic hyperplasia (enlargement). In consultation with their doctor, they may choose to take 0.5 mg of Avodart® daily (though it may not need to be taken every day) and simultaneously introduce an arsenal of mechanistic approaches to restrain benign and/or tumor cell propagation and induce benign and/or tumor cell apoptosis.
The use of Avodart® or finasteride can shrink prostate gland volume by 25% thus relieving benign symptoms, improve the accuracy of a needle biopsy if this diagnostic procedure is needed, and deprive tumor cells of one growth promoter, i.e. DHT (Cohen 2007; Nickel 2004).
A comprehensive arsenal of mechanistic approaches might involve healthy eating, high doses of specific nutrients (at least temporarily), hormone adjustment aimed at reducing DHT, insulin, prolactin and estrogen (but maintaining free testosterone [Yavuz 2008] in youthful ranges), and drugs like metformin and aspirin. If prolactin levels are elevated, the drug Dostinex® (carbergoline) can be used to suppress this cancer stimulating pituitary hormone.
Genetic Tests for Men Undergoing Prostate Biopsy
About half of US men diagnosed with prostate cancer are classified as low-risk by use of conventional measures such as Gleason score (a form of tumor grading), the prostate-specific antigen test (PSA), and a physical exam (Genomic Health 2013). Nonetheless, nearly 90% of these low-risk patients will choose to undergo immediate aggressive treatment such as radical prostatectomy or radiation even though there is less than a 3% chance of deadly progression (Genomic Health 2013).
A new test called Oncotype DX is now available to physicians and their patients. It measures the level of expression of 17 genes across four biological pathways to predict prostate cancer aggressiveness (Genomic Health 2013).
Test results are reported as a Genomic Prostate Score (GPS) ranging from 0 to 100; this score is assessed along with other clinical factors to clarify a man’s risk prior to treatment intervention (Genomic Health 2013). This multi-gene test can be used in conjunction with the needle biopsy sample taken before the prostate is removed, thereby providing the opportunity for low risk patients to avoid invasive treatments. According to the principal investigator of the validation study, “Individual biological information from the Oncotype DX prostate cancer test almost tripled the number of patients who can more confidently consider active surveillance and avoid unnecessary treatment and its potential side effects” (Genomic Health).
The advantage of this test for those who choose the comprehensive surveillance program utilized by Life Extension customers (which involves the use of several drugs, targeted nutrients, and adherence to healthy dietary patterns) is to provide greater assurance the right course of action is being followed.
For information about the Oncotype DX test, log on to http://www.oncotypedx.com/
Prolaris® is another genomic test developed to aid physicians in predicting prostate cancer aggressiveness in conjunction with clinical parameters such as Gleason score and PSA (Myriad 2013).
Prolaris® measures prostate cancer tumor biology at the molecular level. By measuring and analyzing the level of expression of genes directly involved with cancer replication, Prolaris® may be able to more accurately predict disease progression (Myriad 2013).
Prolaris® is a tool designed to measure the aggressiveness of a patient’s cancers to better predict and stratify an individual’s relative risk of disease progression within ten years (Myriad 2013). It may enable physicians to better define a treatment/monitoring strategy for their patients.
Prolaris® claims to be significantly more prognostic than currently used variables and provides unique additional information that can be combined with other clinical factors in an attempt to make a more accurate prediction of a patient’s cancer aggressiveness and therefore disease progression (Myriad 2013).
Prolaris® has been shown to predict clinical progression in four different clinical cohorts, in both pre and post-treatment scenarios (Myriad 2013).
In the treatment of prostate cancer, Prolaris® is prognostic at the point of diagnosis and in the post-surgery setting (Myriad 2013).
At diagnosis, Prolaris® can help to identify patients with less aggressive cancer who may be candidates for active surveillance. In addition, Prolaris® can define patients who appear clinically low-risk but have a more aggressive disease that requires more aggressive treatment.
Prolaris® testing is also well suited for use in post-prostatectomy patients that have higher risk features after surgery to better estimate their risk of disease recurrence and therefore adjust the level of monitoring or add additional therapy.
For more information about Prolaris®, log on to the company website: www.myriad.com

How Life Extension Differs From the Mainstream
A common approach to dealing with biopsied-confirmed low-grade prostate cancer is called “watchful waiting.” Under this scenario, PSA tests are performed at reasonable intervals and treatment decisions based on indicators of disease progression (or regression).
In the presence of persistently rising PSA and other markers, the patient and their doctor discuss wide ranges of treatment options ranging from surgical removal of the prostate gland, different forms of radiation, cryoablation, and/or androgen ablation to temporarily reduce PSA and buy more time. All of these treatment modalities have side effects to consider.
Instead of merely “watching” a PSA rise until risky therapies are required, we at Life Extension view a low-grade prostate cancer (or even a biopsy that reveals no cancer) as an opportunity to intervene aggressively with a multitude of non-toxic approaches that benefit one’s overall health. Success or failure is measured by monthly PSA testing, along with other tests to ensure that other growth factors like insulin, estrogen, DHT, and prolactin are being adequately suppressed.
To clarify the point about a no cancer diagnosis, the accuracy of typical initial needle biopsies today is only around 75% (Taira 2010). So if your urologist tells you he has good news, i.e., the biopsy showed no tumor cells in your prostate gland, there may be a 25% chance you do have tumor cells, thus making the kinds of comprehensive intervention that benefits your entire body a rational choice.
So rather than “watchfully wait,” as your underlying disease may progress, we suggest comprehensive intervention. The objective is to take away every route that enables tumor cells to propagate and escape confinement within the prostate gland.
For those who require a prostate biopsy, there are new (and expensive) genetic tests that may more accurately predict which tumors are aggressive and likely to metastasize and those that are so indolent that only minimal changes may be needed to keep control over them. If these genetic tests prove themselves in the clinical setting (outside the bias of company-sponsored clinical trials), intelligently using the results of these tests can spare many men from needless treatments and provide information about genetic mutations to target in prostate cells that may enable better long-term control.
Enhanced Diagnostic Procedures
What patients should understand is the diagnosis of prostate cancer per ultrasound-guided biopsies is also related to the skill of the physician performing the procedure, as well as the nature of the ultrasound (gray-scale versus color Doppler). CDU (color Doppler ultrasound) also indicates the degree of vascularity (angiogenesis) of the cancer, which if present is a factor associated with tumor aggressiveness. The more vascular the cancer the more aggressive it is. Dietary approaches, supplements, and medications to reduce angiogenesis should be considered in the arsenal of how we prevent the emergence or evolution of clinically significant prostate cancer.
An additional emerging area that may allow a better understanding of clinically significant prostate cancer and clarify the issue of risk of high-grade prostate cancer with 5-alpha reductase inhibitor drugs like Avodart® and Proscar® involves replacing the transrectal ultrasound of the prostate (TRUSP) with MRI utilizing parameters such as DWI (diffusion weighted imaging) and the associated grading of DWI using the Apparent Diffusion Coefficient (ADC). Studies indicate a much higher specificity for the diagnosis of prostate cancer than TRUSP when DWI and ADC are used together (Shimamoto 2012; Ibrahiem 2012).
Our Enlarging Prostate Glands
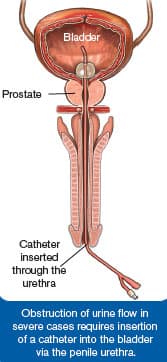
Aging results in a proliferation of prostate cells that is technically referred to as benign prostatic hyperplasia (BPH) (Gharaee-Kermani 2013). The graphic titled “The Development of Benign Prostatic Hyperplasia” depicts an advanced case of BPH with a constricted urethra that would impede or block urine flow.
Symptoms associated with BPH include frequent urination and urinary hesitancy that can be especially troublesome at night (Gharaee-Kermani 2013). In severe cases obstruction of urine flow requires insertion of a catheter into the bladder via the penile urethra.
A major culprit involved in the benign over-proliferation of prostate cells is dihydrotestosterone (DHT) (Clark 2004). Drugs such as Avodart® (dutasteride) or Proscar® (finasteride) reduce DHT levels and shrink the size of an enlarged prostate gland, which reduces BPH symptoms (Schmidt 2011). These drugs also lower PSA levels by almost 50%, which may reflect the mechanism(s) that explain why men taking these drugs have reduced overall prostate cancer risk (Kaplan 2002; Handel 2006; Nelson 2010). In two large studies, men taking Avodart® or Proscar® had about a 24% reduced risk of prostate cancer (Thompson 2003; Andriole 2010).

Men should know that testosterone is not the culprit behind prostate problems. Numerous studies suggest that youthful levels of testosterone do not increase prostate cancer risk (Tan 2004; Agarwal 2005; Gooren 2003; Morgentaler 2007; Rhoden 2008; Raynaud 2006). What happens in the aging man’s body, however, is that testosterone converts to estrogen and DHT, and these two testosterone metabolites have been shown to be involved in benign and malignant prostate disease. Fortunately, there are low-cost methods available to suppress DHT and estrogen in aging men, while maintaining youthful ranges of free testosterone.
Recall that PSA is not just a marker of prostate cancer, but functions as a tumor promoter by degrading barrier structures in the prostate gland that may contain isolated tumor cells.
What troubles Dr. Walsh and some other experts is that some of the men taking Avodart® or finasteride who do contract prostate cancer have been shown in two studies to develop more aggressive forms of the disease. They are so concerned that they warn men not to use these drugs for the purpose of prostate cancer prevention, as does the FDA.
On the flip side are proponents of these drugs who point out that Avodart® as well as Proscar® (finasteride) reduce prostate gland volume by such a degree that the ability to identify high-grade tumors via prostate biopsy is improved. So it does not appear that Avodart® or Proscar® causes more high-grade tumors. Instead, these drugs facilitate earlier detection of such cancers, which is another reason to consider taking them.
A frustration with needle biopsies is that they miss as many as 20-30% of prostate cancers (Taira 2010; Rabbani 1998; Numao 2012). The larger one’s prostate gland, the easier it is to have the biopsy miss those sites that are malignant. The illustration titled “12-core Prostate Needle Biopsy” depicts a 12-core biopsy to show why a larger prostate gland makes it more difficult to detect malignant cells. So an advantage of shrinking one’s prostate gland using drugs like Avodart® or Proscar® is that if a needle biopsy is required, it may more accurately detect underlying malignancy (Kulkarni 2006).
In the December 2013 Life Extension magazine article titled The Avodart®-Proscar® Debate, there is compelling evidence that these drugs may reduce high-grade prostate cancer risk.
Another virtue to using 5-alpha reductase inhibitors (like Avodart® or Proscar®) is that in the presence of prostate cancer, PSA levels don’t decrease as much after these drugs are initiated (Kaplan 2002; Handel 2006; Nelson 2010).
Physicians using 5-alpha reductase inhibitors should take into account the PSA-lowering effect of these agents by doubling the PSA lab value (Andriole 2006). Given that PSA decreases less in the presence of prostate cancer, the doubling of PSA will result in a higher value of PSA and will trigger the need for diagnostic investigations sooner.
What doctors have observed is that drugs like Avodart® or finasteride suppress PSA levels more effectively in men with benign prostate enlargement or low-grade prostate cancer. When PSA levels drop then start raising again, this indicates that the 5-alpha reductase inhibitor is reducing low-grade cells of questionable clinical significance but is not affecting higher grade malignancies (Cohen 2007). This finding is another plus for using a 5-alpha reductase inhibitor as it can increase the sensitivity of the PSA test to reveal which men need aggressive diagnostics such as needle biopsies.
12-core Prostate Needle Biopsy
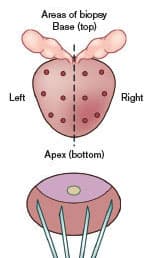
This diagram depicts a 12-core needle biopsy of a prostate gland. Note how many areas of the prostate are missed during biopsy. In the PCPT (Prostate Cancer Prevention Trial) where only 6 core biopsies from 6 regions of the gland were obtained, the effect of Proscar® in reducing gland volume was to increase the ability to detect high-grade prostate cancer (Yavuz 2008; Redman 2008).
Why We Suggest Certain Drugs
When it comes to combatting cancer, Life Extension long ago learned that the initial treatment regimen should be aggressive enough to deprive tumor cells of an opportunity to mutate into forms that are resistant to future therapies. If we know of a relatively side effect-free drug that works via a single or multiple mechanisms to impede tumor survival, we’re going to include it in our comprehensive surveillance program.
Let’s talk first about metformin. It was used in England in 1958 but did not make it into the United States until 1995—37 years later (Dowling 2011)! I am familiar with metformin because the FDA tried to have me incarcerated for recommending it as an anti-aging drug long before it was “approved” to treat type II diabetes.
What’s been happening over the last ten years is an explosion of published studies that consistently show that metformin reduces the risks of certain tumors and may be an effective cancer treatment (Hirsch 2009; Anisimov 2005; Vazquez-Martin 2011; Tomimoto 2008; Gotlieb 2008; Cantrell 2010; Libby 2009; Memmott 2010).
People ask me all the time, how can an anti-diabetic drug work so well against cancer? The encouraging news is that metformin functions via multiple mechanisms to create a less favorable environment for tumor progression (Evans 2005; Currie 2009; Nagi 1993; Choi 2013; Luo 2010; Ben Sahra 2011; Loubière 2013; Zakikhani 2008; Ben Sahara 2008; Ersoy 2008). We know that insulin (and glucose) increase the risk of many tumors (Parekh 2013). This is of particular concern to obese men with prostate tumors. Metformin lowers blood glucose and insulin levels. The sidebar titled “Anti-Cancer Actions of Metformin” reveals the multiple anti-cancer mechanism of metformin.
There are nutrients that can have similar effects such as standardized green coffee extract (Ong 2013). We nonetheless suggest that a man with an elevated or rising PSA should ask his doctor to consider prescribing metformin. The starting dose can be 500 mg of extended release (Metformin ER) taken with breakfast each day. Under the supervision of the patient’s local medical doctor, the dose can be increased to 500 mg ER taken at breakfast and at dinner. (Dose ranges for non-extended release metformin are 250-850 mg taken before no more than three meals a day.) Metformin is an inexpensive generic drug and can be taken along with nutrients (like green coffee extract) that similarly function to reduce glucose/insulin.
Metformin does more than slash tumor-promoting glucose/insulin levels. It also acts directly on cancer cells to induce apoptosis and/or inhibit proliferation (Jalving 2010). Metformin does this conserving the process by which food is converted to energy (Choi 2013; Luo 2010; Ben Sahra 2011; Loubière 2013). Healthy cells react to metformin by adjusting their functions to use less energy. A cancer cell, on the other hand, that is forced to minimize energy consumption is less able to exhibit aggressive metastatic or proliferative behavior (Dunlap 2012). In other scenarios, the energy stress caused by metformin is sufficient to cause cancer cell death.
The National Cancer Institute is sponsoring a clinical study where metformin will be tested to see if it can slow the progression of prostate cancer in men undergoing active surveillance (watchful waiting) with low-grade tumors (Fleshner 2013). We hope the study design includes the measurement of 2-hour post-prandial (2 hours after meals) blood glucose levels as well as glycosylated hemoglobin (HbA1c) to ascertain that optimal dosing of study subjects has been achieved.
Anti-Cancer Actions of Metformin
Numerous studies show the anti-diabetic drug metformin can slow growth of existing cancers and decrease risk of developing new cancers. Some studies show metformin may protect against prostate cancer and aid in treatment. Here are some of its anti-cancer mechanisms:
- Metformin reduces levels of glucose, insulin, and insulin-like growth factors that fuel tumor growth (Evans 2005; Currie 2009; Nagi 1993; Choi 2013).
- Metformin activates a powerful molecule called AMPK (adenosine monophosphate-activated protein kinase) that subjects cancer cells to unique metabolic stresses not experienced by healthy tissues. (Activated AMPK promotes death [apoptosis] of malignant cells and prevents their development.) (Choi 2013; Luo 2010)
- Metformin independently inhibits mTOR (mammalian target of rapamycin) that regulates cell growth, energy metabolism, cell motility, cell survival, and protein synthesis (Ben Sahra 2011; Loubière 2013).
- Metformin mimics the benefits of a hormone called adiponectin in activating AMPK-dependent growth inhibition in prostate cancer cells (Zakikhani 2008).
- Metformin blocks cancer cell reproductive cycles by decreasing levels of a growth-promoting protein called cyclin D1 (Ben Sahra 2008).
- Metformin increases production of a protein (p27) that inhibits the cell division cycle (Ben Sahra 2008).
- Metformin suppresses vascular endothelial growth factor (VEGF) thereby cutting off the blood supply to tumors (Ersoy 2008).
At a cancer conference in early 2013, the results of a study were reported of 22 men (median age 64, median PSA 6 ng/mL) with confirmed prostate cancer that were given 500 mg of metformin three times a day 41 days prior to surgery (prostatectomy). In response to metformin the men showed the expected reductions in glucose and insulin growth factor-1 (IGF-1) blood levels, along with abdominal fat loss (Joshua 2012). What got the researchers excited was that compared to biopsied specimens, the surgically removed prostate glands showed a 32% reduction in a marker of cell proliferation (Ki-67) and a favorable alteration in a pathway tumor cells use to proliferate out of control (via mTOR) (Carlson 2012).
Knowledgeable customers point out that curcumin interferes with these tumor growth pathways via similar mechanisms, which we at Life Extension have long been familiar with (Ravindran 2009). My argument for recommending metformin is that it should produce potent additive effects to curcumin. Moreover, we still don’t know what the upper dose limits are for metformin and/or curcumin for cancer treatment, so taking both may have some obvious advantages.
Furthermore, because metformin is a drug, it tends to get more attention from researchers, perhaps because it is easier to obtain funding for drug studies. A European study published this year showed that metformin was effective against advanced castration-resistant prostate cancer. The doctors who conducted this study concluded:
To our knowledge, our results are the first clinical data to indicate that metformin use may improve PSA-recurrence free survival, distant metastasis-free survival, prostate cancer specific mortality, overall survival and reduce the development of castration resistant prostate cancer in prostate cancer patients. Further validation of metformin’s potential benefits is warranted (Spratt 2013).
Interestingly, men who are on androgen deprivation therapy to treat prostate cancer often show rising insulin levels that can stimulate tumor growth (Currie 2009; Hvid 2013). By taking metformin, some of the side effects of androgen deprivation therapy can be mitigated, as was shown in this newly published European study.
So while nutrients like curcumin and green coffee extract and others may share functions that are similar to metformin, we cannot ignore the strong data showing specific benefits to low-cost metformin.
Another hormone that prostate tumors use to escape eradication is prolactin (Dagvadorj 2007), and this can easily be suppressed by taking 0.25 mg to 0.5 mg of cabergoline (Dosintex®) two to three times weekly (Drugs.com 2013).
Aspirin functions in multiple ways to interfere with prostate cancer propagation and metastasis and it may induce genetic changes that facilitate apoptosis (Langley 2011). There is too much data about the potential role of aspirin as an adjuvant cancer treatment for men with rising PSAs not to use it.
Daily Use of Aspirin May Decrease Prostate Risks
Researchers studied 2,447 men over 12 years, examining them every other year. After adjusting for age, diabetes, hypertension, and other factors, they found that men who took a daily aspirin or another NSAID (like ibuprofen) reduced their risk of moderate or severe urinary symptoms by 27% and lowered their risk of an enlarged prostate by 47%. Even more intriguing was the finding that men who consumed aspirin or another NSAID were 48% less likely to have an elevated level of prostate-specific antigen (PSA) (St Sauver 2006).
Aspirin inhibits the cyclooxygenase (COX-1 and COX-2) enzymes, which are also involved in the arachidonic acid inflammatory pathway (Egan 2006; Wu 2003). COX-2 in particular is known to promote the proliferation of prostate cancer cells (Xu 2008).
Treat Yourself As If You Already Have Prostate Cancer
This protocol is supposed to be about prostate cancer prevention, and here I am talking about therapies overlooked by most doctors that may facilitate enhanced treatment outcomes.
The reason we can’t ignore treatments is that aging men should accept the reality that in all likelihood there are malignant cells in their prostate glands now. This makes it easier to consistently follow prevention programs that can reduce the risk that clinically diagnosed disease will ever manifest. It also keeps one on the lookout for non-toxic treatments that may also have preventative benefits.
As I have related in the past, when my PSA reading came back at 1.4 ng/mL in year 2003, I treated it as if I had early stage prostate cancer by adopting healthier dietary choices and taking every nutrient and drug that had shown efficacy in prostate cancer prevention. Ten years later my PSA is 0.4 ng/mL.
I will remain on an aggressive prostate cancer treatment regimen and in the process reduce my risk for virtually every other age-related disease.
The protocol provides comprehensive approaches for the prevention of prostate cancer, including a comprehensive overview demonstrating the prostate cancer prevention benefits in response to Avodart® and finasteride. Men with any type of prostate malignancy may also benefit, as the programs we advocate for prevention may also facilitate better overall treatment.
What if PSA Screen Detects a Potential Problem?
If an annual PSA screen reveals a potential problem, a man has an early opportunity to:
- Review state-of-the-art studies to establish his status regarding the presence of prostate cancer.
- Confirm the diagnosis and get a Gleason score reading by an expert in prostate cancer pathology.
- Utilize published nomograms and neural nets to present the patient probabilities of organ-confined prostate cancer, capsular penetration, or disease progression to seminal vesicles and/or lymph nodes.
- Obtain refined laboratory studies and imaging studies to confirm or refute the above.
- Sit down with a physician that is least biased on a particular procedure and discuss the pros and cons of all therapies, including active surveillance.
- Investigate and discuss all co-related illnesses that might have gone unrecognized but that play a role in stimulating prostate cancer growth.
Don’t Accept Archaic Diagnostics
The highly variable skills of the urologist performing TRUSP (transrectal ultrasound guided needle biopsy of the prostate) is of great concern when a biopsy is needed.
Too often the urologist uses the TRUSP to target the prostate gland per se, rather than abnormal areas within the prostate. Rarely do we see a dedicated TRUSP report that mentions all of the important findings that can and should be related by the urologist e.g., gland volume, PSA density, status of the capsule and seminal vesicles, as well as location of hypoechoic and hyperechoic lesions within the prostate. Using the TRUSP to target the prostate gland, and not the various lesions within the gland is akin to diluting a vintage wine with ice cubes. (For illustration and a description of a model TRUSP report, see Appendix F of the book A Primer on Prostate Cancer by Strum and Pogliano available from Life Extension Media by calling 1-800-544-4440 or logging on to www.lifeextension.com)
The varying quality of the ultrasound device and whether it is a standard gray-scale ultrasound or a color Doppler ultrasound is also significant. Color Doppler ultrasound, for instance, discloses pathologic states of increased blood vessel growth (angiogenesis) that is associated with more clinically aggressive prostate cancer, which is often of a higher Gleason score (Strohmeyer 2001).
MRI (magnetic resonance imaging) using DWI (diffusion weighted imaging) will also add to understanding the risk a particular patient with prostate cancer faces. That’s because when color Doppler ultrasound is combined with MRI-DWI, a predictive value regarding the level of aggressiveness of the prostate cancer may be established (Ibrahiem 2012).
In this manner, selecting only those men whose prostate cancers are most likely to be “bad actors” and who need invasive therapy can be accomplished, while sparing those men with cancers of low grade, which are often amenable to changes in lifestyle, diet, and use of supplements.
2 Prostate Diagnostic and Assessment Tests
There are now a number of diagnostic tests to identify early stage prostate cancer and then monitor the success or failure of a wide range of treatment options.
This section succinctly describes conventional prostate gland diagnostic tests along with those that mainstream medicine often overlooks, to the detriment of their patients. All of these tests, however, are commercially available.
Tests
PSA (Prostate-Specific Antigen)
Perhaps the greatest breakthrough in the detection and management of prostate cancer was the approval of the prostate-specific antigen (PSA) blood test in 1986, but it was only approved for men already diagnosed with prostate cancer (NCI 2012). It wasn’t until 1994 that the FDA approved the PSA test as a prostate cancer screening test for all men (NCI 2012). Prostate-specific antigen is a protein produced by the cells of the prostate gland, including both cancerous cells as well as cells that are benign (NCI 2012). Since very little PSA escapes into the bloodstream from a healthy prostate, an elevated PSA level in the blood indicates an abnormal condition of the prostate gland—which can be either benign or malignant. PSA test results can be used both to detect potential prostate problems and to follow the progress of prostate cancer therapy (NCI 2012).
Because tumor growth is essentially exponential, with one cell dividing into two, two to four, four to eight, and so on, a tumor cell product such as PSA can reflect such exponential growth—measuring the time it takes for PSA to double (PSA doubling time, or PSADT) (Strum 2005). Also, the PSA rate of rise (PSA velocity), although not a more specific marker, may have value in prostate cancer prognosis—because men with prostate cancer whose PSA level increased by more than 2.0 ng/mL during the year before their diagnosis showed a higher risk of death from prostate cancer (D’Amico 2004). Additionally, though not an absolute criteria for or against malignancy, PSA velocity can serve as a gauge regarding the likelihood of a malignant condition (Strum 2005). A rising PSA velocity in excess of .75 ng/mL/year relates to an increased probability of a malignant condition (Strum 2005).
The reference interval provided by most conventional laboratories for the PSA test is 0.00-4.00 nanograms per milliliter (ng/mL) (LabCorp 2013a). Conventional reference ranges suggest that PSA levels under 4.0 ng/mL are normal, but any reading over 2.0 ng/mL can indicate unhealthy activity, such as prostatitis, benign prostate hypertrophy, or prostate cancer (Strum 2005). Furthermore, based upon findings from the Prostate Cancer Prevention Trial (PCPT), a PSA level of 1.1 – 2.0 ng/mL can be viewed as an evidence-based threshold of concern. In the PCPT, among men whose PSA never exceeded 4.0 ng/mL during seven years of follow-up, prostate cancer was detected on biopsy in 17% of those with a PSA level of 1.1 – 2.0 ng/mL. Among men with PSA levels of 2.1 – 3.0 ng/mL, 24% had prostate cancer (Thompson 2004). This evidence suggests that even with PSA levels between 1.1 to 2.0 ng/mL, additional monitoring and/or preventative diet and lifestyle activities may be appropriate. However, it is important to note there is no specific PSA value that is definitively diagnostic nor one that excludes the possibility of prostate cancer.
If PSA readings begin to elevate, there are interventions that can reduce or stabilize the production of PSA, shutting down a mechanism that may be used by cancer cells to escape their confinement within the prostate gland (Webber 1995). PSA readings can increase immediately after ejaculation, returning slowly to baseline levels within 24-48 hours (Tchetgen 1996; Herschman 1997).
Chart of PSA Ranges with Succinct Suggestions
PSA (ng/mL) |
Concise Recommendation |
<1.0 |
Optimal |
1.1-2 |
Threshold of concern - Initiate measures to support prostate health and have digital rectal exam performed |
2-4 |
Moderate concern–assess PSA velocity—have digital rectal exam—consider other tests. |
>4 |
Too high—additional diagnostics recommended |
Free PSA
Free PSA is a newer evaluation for prostate health. Most PSA in the blood is bound to serum proteins, but a small amount is not protein-bound and is called free PSA (Strum 2005; Gion 1997). In men with prostate cancer, the ratio of free (unbound) PSA to total PSA is decreased (Strum 2005). The free PSA test measures the percentage of free PSA relative to the total amount (Abrahamsson 1997). The lower the ratio, the greater the probability of prostate cancer. Measuring free PSA may help eliminate unnecessary biopsies (Gion 1997). Free PSA readings increase immediately after ejaculation, returning slowly to baseline levels within about 24 hours (Tchetgen 1996). Although not used as an initial screening test, a lower percentage of free PSA might mean your doctor needs to do a further workup.
Below are the percentage of PSA ranges and what they represent as far as prostate cancer risk. Note that when the percentage of free PSA is high (over 20%), this means the risk of prostate cancer is low, whereas a low percentage of free PSA (under 11% indicates high risk).
PRoStAte CAnCeR RISk
Free PSA% |
50-64 Years |
65-75 Years |
0.00-10.00% |
56% |
55% |
10.01-15.00% |
24% |
35% |
15.01-20.00% |
17% |
23% |
20.01-25.00% |
10% |
20% |
Over 25% |
5% |
9% |
PCA3 Urine
PCA3 is a molecular diagnostic test performed on urine rather than blood and detects mRNA that is excreted into the urethra via the epithelial cells that line the prostatic ducts (Hessels 2009). Prostate cancer cells tend to produce this compound far more than normal cells do (Hessels 2009). The PCA3 urine test has to be done in a urologist’s, or other doctor’s, office, because it requires a digital rectal massage just prior to collection of the urine (Day 2011).
PCA3 testing is most useful when repeated over a period of time to monitor for changes in the observed value. In general, a PCA3 score of 35 is considered the optimal cut-off. A score of greater than 35 reflects an increased probability of a positive biopsy. A score of less than 35 reflects a decreased probability of a positive biopsy.
25-Hydroxy Vitamin D
Research points to a connection between vitamin D levels and cancer (Qin 2013; van den Bemd 2002). Experimental studies indicate that low levels of vitamin D increase prostate cancer risk (Lou 2004). And further evidence shows that the active form of vitamin D promotes differentiation and inhibits proliferation, invasiveness, and metastasis of human prostate cancer cells (Lou 2004; John 2005). Detecting deficient levels allows you and your physician to implement vitamin D supplementation to help avert illnesses associated with inadequate vitamin D levels. For this nutrient, individualized dosing is of particular importance, and the only way to accomplish this is through vitamin D blood testing. Although conventional laboratory reference ranges list a reference interval of 30-100 ng/mL, Life Extension supports maintaining vitamin D in the 50-80 ng/mL range (LabCorp 2013b).
Prolactin
Prolactin, a peptide hormone largely secreted by the anterior pituitary gland, has typically remained restricted to the fields of lactation and infertility. However, researchers discovered that prolactin plays a major role in the differentiation and development of the prostate gland (Sethi 2012). Both malignant and healthy prostates produce prolactin. Prostatic fluids from patients with cancer also have higher prolactin levels than controls (Sethi 2012). In vitro, prolactin induces proliferation and antagonizes apoptosis in prostate organ culture and in some tumor cell lines (Sethi 2012). Increased levels of prolactin have significant stimulatory action on the prostate and on prostate ductal development and may lead to hyperplastic growth, independent of elevations in circulating androgen levels (Kindblom 2003).
Labcorp normal reference range - Male: 4.0-15.2 ng/mL
Optimal for Prostate Cancer- <5 ng/mL
What You Need to Know
Methods of Diagnosing and Assessing Prostate Cancer
- Prostate cancer remains unique in that there are many tests to identify early stage disease and then monitor the success or failure of a wide range of treatment options.
- Perhaps the greatest breakthrough in the detection and management of prostate cancer was the approval of the prostate-specific antigen (PSA) blood test.
- Experimental studies indicate that low levels of vitamin D increase prostate cancer risk.
- Men can easily be tested for palpable prostate abnormalities with a digital rectal exam (DRE), a simple test that provides a lot of information.
- A comprehensive blood test for specific hormone levels is useful since many hormones have been shown to play a role in the proliferation of prostate cancer.
- Combining imaging tests such as ultrasound, MRI, QCT, Color Doppler, and bone scan can give the most complete picture, allowing for full physical and architectural assessment of tumors, including those that have spread beyond the prostate.
DRE (Digital Rectal Exam)
Men can easily be tested for palpable prostate abnormalities with a digital rectal exam (DRE), a simple test that provides a lot of information (Tisman 2001). It gives the physician a sense of the prostate gland volume. The bigger the prostate, the more PSA the gland is entitled to make without indicating a potential problem. A basic rule is that the prostate gland volume multiplied by the amount of PSA produced per unit of volume in benign prostate tissue is 0.067 ng (Tisman 2001). This means that a 50-year old man with a normal size prostate of 30 grams (or cubic centimeters) would therefore be entitled to make approximately 2 ng of PSA. If such a man has a PSA of 4.0 ng/mL, it would indicate an excess of about 2 ng of PSA and the need for further investigation to rule out prostate cancer.
In addition to estimating prostate gland volume and calculating the benign cellular contribution to the total PSA value, the DRE can also aid in finding hard nodules or other evidence of disease. Palpable (able to be felt) abnormalities of the prostate gland relate to tumor volume, also called tumor burden (Tisman 2001). In the years before routine testing with PSA, most prostate cancers were already palpable via DRE by the time of diagnosis. Today, close to 70% of prostate cancers newly diagnosed in the US are no longer associated with palpable disease (Tisman 2001). This shows the value of PSA screening in allowing an earlier diagnosis of prostate cancer — before the cancer has had a chance to get bulkier and manifest itself as a palpable disease, known as a T2 disease. Most American men when first diagnosed with prostate cancer have non-palpable, or T1, prostate cancer (Tisman 2001).
Blood Hormone Profile
A comprehensive blood test for specific hormone levels is useful. In addition to the free and total testosterone levels covered earlier, a complete blood test should include levels of estradiol, DHT (dihydrotestosterone), pregnenolone, DHEA-S (dehydroepiandrosterone sulfate), FSH (follicle-stimulating hormone), LH (luteinizing hormone), and possibly, IGF-1 (insulin-like growth factor 1). DHT plays a role in the development and exacerbation of benign prostatic hyperplasia, as well as prostate cancer (NCI 2013c). FSH (follicle-stimulating hormone) and LH (luteinizing hormone) regulate the reproductive processes of the body, and in aging men, a rise in FSH and LH can be indicative of andropause (Dandona 2010; Miwa 2006). Studies have shown that increased levels of IGF promote cancer growth and confer resistance to conventional treatments (chemotherapy and radiation) (Arnaldez 2012; Kojima 2009).
PAP (Prostatic Acid Phosphatase) Test
The PAP test is a simple blood test, used to measure the amount of an enzyme—called prostatic acid phosphatase (PAP)—that is produced by prostate epithelial cells and is abundant in seminal fluid (Kong 2013). Higher levels of PAP are associated with prostate cancer (Kong 2013). PAP determination, in conjunction with PSA measurements, is useful in assessing the prognosis of prostate cancer. It is an important test, because it allows identification of prostate cancer patients who have an elevation of PAP, but not of PSA. This helps monitor the course of disease and response to treatment.
Baseline PAP 2 |
Freedom from prostate cancer recurrence at 5 years after prostate cancer surgery defined as psa>0.2 ng/mL |
<0.4 U/liter |
87% |
0.4-0.5 U/liter |
79% |
>0.5 U/liter |
63% |
Circulating Tumor Cells Assay
This test provides a measurement of cancer cells that have separated from a solid tumor site and are circulating in the bloodstream (Ligthart 2013). Detecting the presence of circulating tumor cells in the blood has clinical usefulness in assessing the disease status and prognosis of metastatic prostate cancer, and is predictive of overall survival (Miller 2010). Fasting prior to the blood draw is not required.
Imaging
Transrectal Ultrasound
Transrectal ultrasound creates an image of the organs in the pelvis, and the most common indication is for the evaluation of the prostate gland in men with elevated PSA levels, or with prostatic nodules on a digital rectal exam (UPMC 2013). Ultrasound clarifies the size of the prostate gland and aids in the distinction between benign prostate conditions and prostate cancer (UPMC 2013). This type of imaging can also be used to help guide a biopsy of the prostate (UPMC 2013).

Color Doppler Ultrasound
Color Doppler ultrasound is a medical imaging technique that is used to provide visualization of blood flow, using computer processing to add color to the image to greatly clarify what is happening inside the body (WebMD 2010). An ultrasound transducer is used to beam sound into the area of interest, and it reads the returning sound. When the sound bounces off a moving target such as a blood vessel, the pitch changes as a result of the Doppler effect. The transducer can detect very subtle pitch changes, record them visually, and generate an image showing where blood is flowing, and in what direction. Because a simple grayscale image can be a bit difficult to read, the ultrasound machine assigns different color values, depending on whether blood is moving towards or away from the transducer. In addition to showing the direction of flow, the colors also vary in intensity depending on the velocity of the flow, allowing doctors to also see how quickly the blood is moving (UPMC 2013).
A color Doppler ultrasound of a patient with a suspected tumor will reveal the precise areas where the velocity of blood flow is changing, mapping out the problem in full color (Fleischer 2000). This type of imaging can map out the tumor’s blood supply to clarify exactly how far the growth has spread (PCRI 2011). This can have an impact on what treatments are selected, and how surgery and other measures are approached. While color Doppler ultrasound can be done using a transducer on the outside of the body, it can also be used for transrectal procedures, in which the probe is inserted to get a better view.
MRI (Magnetic Resonance Imaging)
The MRI has been used for over 30 years for prostate cancer detection and evaluation (Gupta RT 2013). In contrast to ultrasound imaging, prostate MRI has superior soft tissue resolution (UCCM 2013). Magnetic fields are used to locate and characterize prostate cancer. To do so, radiologists use multi-parametric MRI, which includes four different types of MRI sequences (Gupta RT 2013). Currently, MRI is used to identify targets for prostate biopsy, and to make a surgical plan for men undergoing robotic prostatectomy. MRI imaging also helps surgeons decide whether to resect or spare the neurovascular bundle and assess surgical difficulty (Gupta RT 2013).
(Nuclear) Bone Scan
Prostate cancer can cause “hot spots” to appear on a bone scan if the cancer has metastasized to the bone (ACS 2013a; Tombal 2012). A bone scan for cancer uses nuclear technology and involves administering a radioactive substance called a tracer to produce gamma radiation that can be picked up by a special camera (ACS 2013a). The tracer consists of radionuclides that bind to the bone and show up as dark or light spots. After the technician injects the tracer, it usually takes between one and four hours for the radioactive substance to move throughout the skeleton. During this time, patients will be asked to drink up to six glasses of water to flush any tracer material not absorbed by bone. Then, the patient must remain still on a padded table while a large camera passes over the body.
A dark spot, also called a cold spot, might indicate lack of absorption of the tracer (ACS 2013a). This may also indicate that cancer has spread to the bone from the prostate gland. A normal scan shows evenly distributed tracer throughout the body. Risks associated with a bone scan are considered low, with a very small level of radiation exposure (ACS 2013a). The radionuclides injected into the bloodstream are excreted through the urine and have a low risk of toxicity (ACS 2013a).
Quantitative Computed Tomography (QCT)
Osteoporosis, or bone thinning, is associated with prostate cancer and can be a side effect of prostate cancer treatment (Tuck 2013). Quantitative computed tomography (QCT) is a highly sensitive test that is better able to determine bone density changes than common methods such as DEXA testing (Smith 2001). Studies have shown that DEXA testing can often read degenerative changes involving bone and joint tissues and calcium deposits within blood vessels as bone density thus not suggesting osteoporosis when in fact bone loss is present (Bolotin 2001; Meirelles 1999; von der Recke 1996; AHRQ 2011). Quantitative computed tomography (QCT) is similar to other forms of computed tomography (CT). As with any CT scan, an X-ray tube and sensor rotate around the body area in a circular or spiral pattern, and a series of pictures are transmitted to a computer (Feretti 1999). The primary difference with QCT is the special analysis performed by special QCT software. While most computed tomography (CT) software produces a composite visual image to detect fractures or other symptoms in the scanned bone or soft tissue, QCT uses the data provided by the scanner to generate numerical values for the volume, mass, and density of bone (Feretti 1999). This allows QCT to distinguish between cortical bone, which lines the outside of the bones, and trabecular bone, the softer tissue that makes up the center of the bone (IDI 2013). Trabecular bone is much more metabolically active than cortical bone—meaning, the two types of bone are replaced at different rates (IDI 2013). As a result, these two bone types can show different rates of change in bone mineral density.
Genetic Testing
About half of US men diagnosed with prostate cancer are classified as low-risk by use of conventional measures such as Gleason Score (a form of tumor grading), the prostate-specific antigen test (PSA), and a physical exam (Genomic Health 2013). Nonetheless, nearly 90% of these low-risk patients will choose to undergo immediate aggressive treatment such as radical prostatectomy or radiation even though there is less than a 3% chance of deadly progression (Genomic Health 2013).
A new test called Oncotype DX is now available to physicians and their patients. It measures the level of expression of 17 genes across four biological pathways to predict prostate cancer aggressiveness (Genomic Health 2013).
Test results are reported as a Genomic Prostate Score (GPS) ranging from 0 to 100; this score is assessed along with other clinical factors to clarify a man’s risk prior to treatment intervention (Genomic Health 2013). This multi-gene test has been validated using the prostate needle biopsy sample taken before the prostate is removed, thereby providing the opportunity for low risk patients to avoid invasive treatments. According to the principle investigator of the validation study, individual biological information from the Oncotype DX prostate cancer test almost tripled the number of patients who can more confidently consider active surveillance and avoid unnecessary treatment and its potential side effects (Genomic Health 2013).
The advantage of this test for those who choose the comprehensive surveillance program utilized by Life Extension customers (which involves the use of several drugs, targeted nutrients, and adherence to healthy dietary patterns) is to provide greater assurance the right course of action is being followed.
For information about the Oncotype DX test, log on to www.genomichealth.com
Prolaris® is another genomic test developed to aid physicians in predicting prostate cancer aggressiveness in conjunction with clinical parameters such as Gleason score and PSA (Myriad 2013).
Prolaris® measures prostate cancer tumor biology at the molecular level. By measuring and analyzing the level of expression of genes directly involved with cancer replication, Prolaris® may be able to more accurately predict disease progression (Myriad 2013).
Prolaris® is a tool designed to measure the aggressiveness of a patient’s cancers to better predict and stratify an individual’s relative risk of disease progression within ten years (Myriad 2013). It may enable physicians to better define a treatment/monitoring strategy for their patients.
Prolaris® claims to be significantly more prognostic than currently used variables and provides unique additional information that can be combined with other clinical factors in an attempt to make a more accurate prediction of a patient’s cancer aggressiveness and therefore disease progression (Myriad 2013).
Prolaris® has been shown to predict clinical progression in four different clinical cohorts, in both pre and post-treatment scenarios.
In the treatment of prostate cancer, Prolaris® is prognostic at the point of diagnosis and in the post-surgery setting (Myriad 2013).
At diagnosis, Prolaris® can help to identify patients with less aggressive cancer who may be candidates for active surveillance. In addition, Prolaris® can define patients who appear clinically low-risk but have a more aggressive disease that requires more aggressive treatment.
Prolaris® testing is also well suited for use in post-prostatectomy patients that have higher risk features after surgery to better estimate their risk of disease recurrence and therefore adjust the level of monitoring or add additional therapy.
For more information about Prolaris®, log on to www.myriad.com
Summary
Prostate cancer is the most common malignancy in US men (excluding non-melanoma skin cancer), afflicting one male in every six (Siegel 2012; PCF 2013b). A significant percentage of men have underlying prostate cancer without even knowing it (Harvei 1999; Billis 1986; Sakr 1995).
These men have access to an arsenal of tools for their doctors to diagnose and then monitor the success or failure of various treatment modalities, including “active surveillance” or “watchful waiting.”
As you’ll learn in this protocol, one does not have to sit back and “watch” their PSA level steadily rise. Nutritional, hormonal, and drug approaches exist to help control early-stage, low-grade prostate tumors. There is data to support the efficacy of some of them as effective adjuvants in men with high-grade tumors as well (Gallardo-Williams 2004; Choi 2010; Dorai 2001; Fleshner 1999; Yang Y 2007; McCann 2008; von Holtz 1998; Zaidman 2007; Thirugnanam 2008).
3 The Avodart®-Proscar® Debate
Elevated levels of dihydrotestosterone (DHT) contribute to benign prostate enlargement (Andriole 2004).
Based on evidence that DHT is also involved in prostate cancer (NCI 2013c), two large studies were conducted in aging men to see if drugs that reduce DHT also lower prostate cancer risk (Thompson 2003; Andriole 2010; FDA 2013).
One study evaluated the drug Avodart® (dutasteride) and the other Proscar® (finasteride) (Thompson 2003; Andriole 2010). Both of these drugs inhibit the 5-alpha reductase enzyme, thus blocking the conversion of testosterone to much more powerful DHT (FDA 2013).
Findings from the two studies showed both drugs reduce prostate cancer risk by about 23-25% (Thompson 2003; Andriole 2010). These drugs also substantially reduce symptoms of benign prostatic hyperplasia (Andriole 2004; FDA 2013).
Some doctors, however, were concerned that in men who did develop prostate cancer, more in the group taking either of these drugs (Avodart® or Proscar®) developed high-grade prostate cancer (Thompson 2003; Andriole 2010). This is a justified concern. High-grade prostate cancer requires aggressive therapeutic intervention and is more challenging to cure (Tewari 2007).
This protocol will reveal overlooked findings showing that Avodart® or Proscar® do not increase high-grade prostate cancer risk and may reduce it.
When prostate cancer is suspected, the typical diagnostic procedure is a needle biopsy of the prostate gland (ACS 2013c). The biopsied specimen is sent to a pathologist to determine the “grade” of any suspicious structures observed from the biopsied specimen (ACS 2013c).
The “grade” is determined by the “Gleason” scoring system (ACS 2013c). It is based on microscopic tumor patterns assessed by a pathologist while interpreting the biopsied specimen. The Gleason “grade” is a fundamental determinate used today to estimate low-grade, intermediate-grade, or high-grade malignancy (ACS 2013c).
The Gleason score is one measure of a patient’s specific risk of dying due to prostate cancer (O’Dowd 2001). Once diagnosis of prostate cancer is made on biopsy, the Gleason score strongly influences decisions regarding options for therapy (O’Dowd 2001). Here is how Gleason numbers are correlated with tumor grade (ACS 2013c):
Gleason score under 7: Low-grade
Gleason score of 7: Intermediate-grade
Gleason score over 7: High-grade
The higher the Gleason score, the more aggressive the tumor is likely to act and the worse the patient’s prognosis (O’Dowd 2001).
Problems With Gleason Testing
Physicians and lay people often overestimate the degree of certainty when it comes to diagnostic testing. This is clearly seen with tests like Gleason scoring, which is open to the interpretation of the individual pathologist examining the biopsied specimen and other variables. Several studies show that significant percentages of Gleason scores are graded too high or too low (Shapiro 2012; Moreira 2009; Berglund 2008; Nayyar 2010).
One way that Gleason scores are found to be sometimes erroneous is to compare tissue obtained from surgical prostatectomy to what was removed during the fine needle biopsy on the same patient (Moreira 2009; Nayyar 2010). There is far more tissue volume to examine from surgically-removed prostate glands compared with the relatively minute amounts obtained from needle biopsies, thus enabling more accurate Gleason score-grading of surgically-removed samples.
As it relates to men taking drugs that dramatically collapse the size of the prostate gland (like Proscar® and Avodart®), pathologists have noted that a consequence may be to induce lower-grade cancer to resemble higher-grade cancer (Andriole 2004; Bostwick 2004; Nelson 2009). They have stated that in men taking drugs like Avodart® or Proscar®, there would be a “grading bias” in which Gleason scores would indicate a worse grade tumor than really exists.
Proscar® Study Subjects Had Lower Rates of High-Grade Tumors
In the study evaluating Proscar® over a 7-year period, there was a 25% reduction in the risk of prostate cancer, but with an apparent increase in the risk of high-grade disease (as measured by Gleason scores) (Redman 2008).
A further analysis of the data, however, revealed the contrary. When examining tissue taken from men who underwent radical prostatectomy (complete removal of prostate gland), there was a 27% reduction in high-grade disease in the Proscar® group as measured by Gleason scoring (Redman 2008).
It turned out that there was a biopsy-sensitivity issue in the Proscar® group that resulted in high-grade Gleason scores being assigned to some men that were really low-grade (Redman 2008).
The startling finding that Proscar® (finasteride) may reduce high-grade prostate cancer was revealed in a study titled “Finasteride Does Not Increase the Risk of High-Grade Prostate Cancer: A Bias-Adjusted Modeling Report” (Redman 2008).
This study was published in the journal Cancer Prevention Research, but has been largely overlooked by the media, the FDA, and many physicians.
Shrinking Prostate Gland Makes High-Grade Cancer Easier To Detect
The impact of Avodart® or Proscar® on the size (volume) of the prostate gland is profound. Either drug reduces prostate gland volume by 17-25% over a relatively brief period of time (Andriole 2010; Nelson 2009).
Avodart® or Proscar® has been shown to reduce PSA more significantly in the presence of no cancer or low-grade prostate cancer (Kaplan 2002; Handel 2006; Nelson 2010). This effect can better enable physicians to identify cases of suspicious high-grade disease, since PSA levels don’t drop as low or stay as low in the presence of high-grade prostate cancer (Nelson 2010; Krejcarek 2007).
By reducing the size of the prostate gland, drugs like Avodart® or Proscar® can improve sensitivity of prostate biopsy and digital rectal exam (Redman 2008). To understand this concept, understand that there were a minimum of 6-core biopsies done in the Proscar® study and 10-core biopsies for the Avodart® trial (Andriole 2010; Redman 2008). These fine needle biopsies only remove a small percentage of tissue from a prostate gland. The larger the size of a prostate gland, the easier it is to miss malignant regions.
By reducing the bulk of the prostate gland, it was far easier to “hit” malignancies with a needle biopsy in men taking Avodart® or Proscar® compared to biopsies performed on the larger glands of those taking placebo (Andriole 2010; Redman 2008).
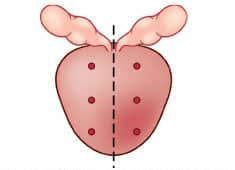
Needle biopsies of the prostate gland are only about 75% accurate to begin with (Taira 2010). So it may be easy to understand why needle biopsies uncovered more high-grade tumors in men taking Avodart® or Proscar® compared to the placebo arm.
Prostate gland marking a 6-core needle biopsy. Note how many areas of the gland may be missed when performing needle biopsies. The larger the gland, the more likely a needle biopsy may miss a tumor mass (Moreira 2009).
What You Need to Know
Prostate Biopsies: Size Matters
- Elevated levels of dihydrotestosterone (DHT) are involved in prostate cancer and contribute to benign prostate enlargement.
- In separate studies evaluating the prostate cancer protective benefit of Proscar® and Avodart®, medications to reduce DHT levels, there was a 22-25% reduction in the risk of prostate cancer, but with an apparent increase in the risk of high-grade disease (as measured by Gleason scores).
- Avodart® or Proscar® have been shown to reduce prostate gland volume by 17-25% over a relatively brief period of time.
- A review of several published reports attribute the higher Gleason scores observed in the groups taking Avodart® or Proscar® to the fact that it was easier to find high-grade cancers in the shrunken glands of men taking these drugs compared to the much larger glands of men in the placebo group.
- By reducing the size of the prostate gland, Avodart® or Proscar® can improve sensitivity of prostate biopsy and digital rectal exam.
How Much Did Prostate Glands Shrink?
As noted, the median prostate volume in men taking finasteride was 25% lower compared to the placebo group, which represents a huge relative reduction in bulky prostate tissues (Cohen 2007).
A group of scientists calculated prostate gland volume differences along with other variables that were omitted when compiling the initial report on finasteride. When adjusting for all the variables, these scientists demonstrated a 45% lower risk of prostate cancer in the finasteride group (Cohen 2007). The scientists noted that, “Adjustment for gland volume and number of cores biopsied (i.e., sampling density) eliminated the differences in high-grade cancer between the two arms” (Cohen 2007).
Their published paper was titled: “Detection Bias Due to the Effect of Finasteride on Prostate Volume: A Modeling Approach for Analysis of the Prostate Cancer Prevention Trial.” According to these scientists, when all co-variables were added in there was a trend toward 12% fewer high-grade prostate cancers amongst men taking finasteride (Cohen 2007).
The conclusions from this analysis published in the Journal of the National Cancer Institute were:
“Although analyses using postrandomization data require cautious interpretation, these results suggest that sampling density bias alone could explain the excess of high-grade cancers among the finasteride-assigned participants in the PCPT” (Cohen 2007).
A review of several published reports attribute the higher Gleason scores observed in the groups taking the 5-alpha reductase inhibitors (Avodart® or Proscar®) to the fact that it was easier to find high-grade cancers in the shrunken glands of men taking these drugs compared to the much larger glands of men in the placebo group (Kulkarni 2006; Lucia 2007).
The fact that there are published reports defending the safety of Proscar® and Avodart® does not mean the matter is settled (Kulkarni 2006; Lucia 2007; Monga 2013). The debate may continue for years or decades. Aging men at risk for prostate cancer, however, do not have the luxury of waiting for uniform consensus.
Conclusion of Independent Analysis of Proscar® (finasteride)
The researchers who conducted the analysis showing that Proscar® (finasteride) may reduce high-grade prostate cancer risk concluded their published paper by stating:
“Men must weigh the established benefits of a 25% reduction in prostate cancer (estimated to be 30% in the present analysis), decreased urinary symptoms, and decreased complications of an enlarged prostate against the established side effects, which include reduced sexual function. We found no evidence that finasteride increased the risk of high-grade prostate cancer in the PCPT. Therefore, we conclude that men 55 years or older have no need to be concerned about an increased risk of high-grade prostate cancer with finasteride” (Redman 2008).
Role of Estrogen
Estrogen is a cell growth promoter that has been implicated in the development of prostate cancer (Nelles 2011; Carruba 2007).
In a published analysis of the Proscar® study, scientists looked at levels of various sex hormones beyond just DHT. They found that men with the highest pre-treatment concentrations of testosterone were associated with a 36% reduced rate of prostate cancer when taking Proscar® (Kristal 2012). These same scientists also noted men with the highest level of estrone (an estrogen) had a 38% higher risk when taking Proscar® (Kristal 2012).
This study supports the theory that when prostate cancer cells are deprived of a primary growth factor like DHT, they may readily adapt to utilizing estrogen to fuel propagation (Kristal 2012). The doctors who conducted this study concluded:
Further research is needed to evaluate whether “low post-treatment serum estrogens may identify men more likely to benefit from the use of finasteride to prevent prostate cancer” (Kristal 2012).
Life Extension’s long-standing position has been for aging men to have their estrogen blood level tested and if it is elevated, initiate aromatase-inhibiting therapy to suppress estrogen to safe ranges. This may be of particular importance for men seeking to impede or reverse the course of prostate cancer. It also helps explain why some men taking Proscar® developed prostate cancer despite suppressing their DHT level.
The Proscar® (Finasteride) Studies
The Prostate Cancer Prevention Trial (PCPT) was a large-scale, long-term randomized, placebo-controlled study designed to evaluate if Proscar® (finasteride) could reduce risk of prostate cancer (Nelson 2009).
Participants in this PCPT study were all aged 55 years or older and had baseline PSA levels less than or equal to 3 ng/mL. One group received 5 mg/day of finasteride and the other a placebo (Thompson 2003).
This study was initiated based on multiple lines of evidence available in the early 1990s that suggested that treatment with finasteride (Proscar®) would reduce a man’s risk of developing prostate cancer. Finasteride functions to inhibit the 5-alpha reductase enzyme, which then lowers dihydrotestosterone (DHT) levels (Kulkarni 2006).
The plan was to evaluate the prevalence of prostate cancer in each group (finasteride and placebo) during the 7-year trial. About 15 months before the trial was scheduled to end, however, it was terminated because it had already met its primary objective (Nelson 2009).
The PCPT study demonstrated a 24.8% reduction in the prevalence of prostate cancer with finasteride treatment. An unanticipated finding, however, was that cancers with a high-grade Gleason score of 7-10 were more common in men treated with finasteride (6.4%) than in men treated with placebo (5.1%) (Cohen 2007).
To put these percentages of high-grade Gleason scores into perspective, they suggest that if all 18,882 men who entered the trial were given the placebo, 963 of them would have been diagnosed with high-grade disease. If all 18,882 men had taken finasteride, 1,208 men would have been diagnosed with high-grade disease. The difference is 245 more men being diagnosed with high-grade disease based on this assumption.
Of these 18,882 men, however, 4,323 would have contracted any grade of prostate cancer if all were given placebo based upon the results of the study, whereas only 3,134 would have contracted any grade of prostate cancer if all were given finasteride (Cohen 2007). That’s a difference of 1,189 men who would have avoided prostate cancer altogether during the study trial period if they all took finasteride.
We at Life Extension are well aware that high-grade prostate cancer grows faster and is more likely to spread beyond the prostate gland (Tewari 2007). Depending on your long-term longevity objectives, however, the data showing 1,189 fewer men developing any form of prostate cancer compared to 245 more men being diagnosed with high-grade disease might favor the finasteride group. And as you’ve read already, it was much easier to detect prostate cancer in men taking finasteride, meaning that the drug itself should not be blamed for the higher rate of diagnosis (Cohen 2007).
As we’ve shown, there appears to be no increase in high-grade disease in men taking finasteride, just higher rates of biopsy detection based on smaller size prostate volume, along with misinterpreted pathologies of biopsies that later showed 27% fewer high-grade cases in men taking finasteride based on examination of surgically-removed prostate glands (Nelson 2009).
More recent data provided further insight into the benefits of finasteride for prostate cancer prevention. At the 2018 American Urological Association Annual Meeting, Dr. Ian Thompson, a urologist from the University of Texas Health Science Center at San Antonio, and principal investigator of the PCPT, reported that the daily use of finasteride does not increase the long-term risk of prostate cancer. As he reported during an interview, this finding opens the possibility to use this inexpensive and effective drug to prevent prostate cancer (Mulcahy 2018).
The 2003 PCPT study, which first reported that finasteride decreases the risk of prostate cancer, found an effect that was so dramatic that the placebo-controlled arm of the trial was stopped, and the results were published. However, at the same time, the results also revealed an increase in the risk of high-grade prostate cancers as compared to placebo (Thompson 2003). This small, but statistically significant increase in the risk of high-grade prostate cancer made people subsequently avoid finasteride for prostate cancer prevention. However, several pieces of evidence revealed that this is not the case.
Evidence for Benefit and Safety of Finasteride (Proscar) Continues to Grow
Two papers, published in 2018 and 2019, further support the health benefits and safety profile of the 5-alpha reductase inhibitor finasteride.
The first study, which reported on 16 years of follow-up of the PCPT participants, confirmed the study findings: during the 7.5 years after the beginning of the trial, those assigned to the finasteride group had a 29% lower risk of a prostate cancer diagnosis compared with those in the placebo group. And even after the trial ended—meaning the study subjects were no longer taking daily finasteride—there was no significant increase in prostate cancer risk. Over the entire follow-up period, a median of 16 years, those in the finasteride group had a 21% decreased risk of prostate cancer compared with those in the placebo group (Unger 2018).
The second paper, which further analyzed the follow-up results from the PCPT, observed that after a median of 18.4 years, men who received finasteride had a 25% lower risk of dying from prostate cancer. Importantly, there was no increased risk for high-grade prostate cancer in the finasteride group (Goodman 2019).
Overall, the evidence points to a benefit of finasteride in preventing prostate cancer without increasing risk of high-grade disease.
The Avodart® (Dutasteride) Study
A study involving over 8,000 men was initiated to ascertain if Avodart® (dutasteride) could reduce prostate cancer risk over a 4-year period (Andriole 2010). The criteria to participate in this trial were:
- Age between 50 and 75 years
- PSA level of 2.5 to 10 ng/mL
- A single pre-study negative needle biopsy of the prostate
- A prostate volume ≤ 80 mL
Patients were randomized to receive 0.5 mg of Avodart® daily or placebo. All patients received 10-core prostate biopsies at baseline and 10-core prostate biopsies at 2 years and at 4 years after study enrollment (Androle 2010).
Here are summary results of the trial (Andriole 2010):
- The mean age of the men enrolled was 63 years in each arm.
- The mean PSA level of the patients in each arm at baseline was 5.9 ng/mL.
- Avodart® reduced the risk of prostate cancer by 22.8% compared to placebo over the 4 years of the study.
- Avodart® reduced the rate of acute urinary retention by 77.3% compared with placebo.
- The Avodart® group showed a 33% increase in high-grade (8-10) Gleason scores 0.9% for Avodart® compared to 0.6% for placebo.
“If our conclusion that finasteride accelerates the detection of high-grade cancer yet may not promote its development is correct, then the implications regarding the clinical impact of this drug are quite favorable. The occurrence of lower-grade tumors of questionable clinical significance would be reduced, and the early detection of more serious tumors would be enhanced”(Cohen 2007).
The higher number of high-grade Gleason scores in the Avodart® group can be attributed to the same factors identified in the finasteride (PCPT) study, such as higher rates of biopsy detection based on smaller prostate gland size in men taking Avodart®, along with biopsy bias based on collapsing prostate glands that may have made some tumors appear higher-grade than they really were (Andriole 2010).
The FDA’s response, however, was to issue a label change for Avodart® and Proscar® to warn of the increased risk of being diagnosed with a higher-grade prostate cancer while taking these drugs (FDA 2012). This warning provides little benefit to aging men who are routinely prescribed these drugs to treat benign prostatic hyperplasia. It creates confusion as patients query their doctor as to why a drug that the FDA says is potentially dangerous is being prescribed to them.
In 2012, the results of another study (Lancet) were released showing that Avodart® was effective in slowing the progression of low-grade prostate tumors in 38% of men undergoing active surveillance (watchful waiting) (Fleshner 2012). This study of 302 men diagnosed with low-grade prostate cancer used 12-core biopsies obtained at 1.5 and 3 years. This Lancet study showed there to be slightly fewer (14%) higher-grade tumors in the Avodart® group compared to placebo (16%) (Fleshner 2012).
A study published in May 2013 evaluated 82 men with very low-risk prostate cancer who underwent active surveillance (watchful waiting) and were treated with a 5-alpha reductase inhibitor drug over a 3-year period. The results demonstrated the safety of the drugs and noted that at the first re-staging biopsy, 54% of the subjects no longer had prostate cancer (Shelton 2013). This small study helps substantiate the value of Avodart® or Proscar® for low-risk prostate cancer patients, but is not relevant to those with high-grade tumors that often require aggressive treatment.
We Must Avoid A “Tomato Effect”
“The Tomato Effect” was first described in the Journal of the American Medical Association in 1984 (Goodwin 1984). It analogized how doctors historically have ignored or rejected efficacious treatments that did not fit with accepted theories of disease prevention/treatment at the time.
“The Tomato Effect” is named for a period from the 1600s to early 1800s in America where tomatoes were considered poisonous and therefore unsafe to eat. This fear persisted despite the fact that Americans knew Europeans were regularly eating tomatoes with no ill effects.
The perception of the tomato changed in 1820 when a man ate a tomato on the steps of a New Jersey courthouse to prove they are safe (Goodwin 1984). Within a decade, Americans were regularly eating tomatoes that for over 200 years were considered poisonous.
In today’s world, we have an opposite problem that nonetheless can create a lethal “tomato effect” when it comes to rejecting lifesaving therapies. Physicians and patients are overloaded with information and lack the time to analyze data to accurately determine safety and efficacy.
Humans have a strong propensity to remember negative details. This human failing has caused many physicians and patients to reject 5-alpha reductase inhibitors because they recall something about more high-grade prostate tumors in men taking Proscar® or Avodart®. Never mind hard facts showing the opposite. People today want a succinct summary, a curbside explanation, and not too many distracting details.
As we have repeatedly shown in this protocol, what appeared to be more high-grade tumors in two studies of Proscar® and Avodart® were apparently based on a mistaken interpretation of the data.
Sensitivity Analysis Supports Safety of 5-alpha Reductase Inhibitors
Another independent analysis of the prostate-gland shrinking effects of Proscar® was conducted and published in the Journal of the American Statistical Association (Shepherd 2008).
In technical terms, this study is called a “sensitivity analysis,” which in lay language may be explained as allowing a reviewer to assess the impact that changes in certain parameters will have on a study’s conclusions. By way of example, here is one quote from this study:
“Because finasteride shrinks the prostate volume, the 6-core biopsies covered a larger area of the prostate for cases in the finasteride arm and hence were probably more likely to detect high-grade prostate cancer than on the placebo arm“ (Shepherd 2008).
To validate this statement, the researchers noted that the ability to detect high-grade prostate cancer in the placebo group was 21% lower than in the finasteride arm (Shepherd 2008). The reason the researchers knew this is that biopsies of prostate glands surgically removed showed that slightly more men in the placebo arm had high-grade prostate cancer than those taking finasteride, yet the needle biopsies erroneously reflected the opposite. For those who enjoy seeing the math, here is how the researchers calculated this:
“In the placebo group the sensitivity of biopsy for high-grade detection was 45% (55 biopsy high-grades/123 prostatectomy high-grades), compared to 66% on finasteride (76 biopsy high-grades/115 prostatectomy high-grades), suggesting a substantial downward bias in detecting high-grade cancer on placebo relative to finasteride” (Shepherd 2008).
The following chart helps explain the sensitivity analysis described in the preceding paragraph (Shepherd 2008):
|
Placebo |
Finasteride |
Number of biopsy specimens detecting high-grade cancer |
55 |
76 |
Number of surgery specimens detecting high-grade cancer |
123 |
115 |
Percentage of high-grade cancers detected at biopsy |
45% |
66% |
This chart shows finasteride improved detection of high-grade prostate tumors in biopsies, but there were not more high-grade tumors in men taking finasteride when surgical specimens were examined.
In the haste of today’s busy medical practices, we are concerned that huge numbers of men who could benefit enormously from 5-alpha reductase inhibitors will not be prescribed them. We hope Life Extension customers appreciate the effort we have made to analyze the data so that they can make rational choices when confronted with real or potential low-grade prostate cancer.
Worst Case Scenario: Assume We Are Wrong?
We have provided solid evidence that Avodart® (dutasteride) or Proscar® (finasteride) do not increase high-grade prostate cancer risk. But what if we are wrong?
Here is what would happen under such circumstance using the Prostate Cancer Prevention Trial (PCPT) study for reference:
- More than 238,000 men will be diagnosed with prostate cancer in 2013 (PCF 2013a).
- If all these men had taken Proscar®, about 57,120 of them (24%) would avoid it.
- Based upon the results of the PCPT study, if none of these men took Proscar®, 52,598 would have high-grade (Gleason score ≥ 7) disease as opposed to 65,840 who would be diagnosed with high-grade disease (assuming Proscar® somehow causes high-grade disease) (Shepherd 2008).
- So each year, 57,120 men would avoid prostate cancer altogether, but 8,720 more men would be diagnosed with high-grade disease (assuming Proscar® [or Avodart®] really cause it).
- Under this worst case scenario, one could argue there would be greater numbers of beneficial outcomes (as opposed to adverse ones) if all men took Proscar® (or Avodart®).
We at Life Extension® don’t recommend these drugs for all men. They appear effective for reducing risk of low-grade prostate cancer and helping to better diagnose high-grade prostate cancer by shrinking prostate gland volume and better enabling the PSA marker to identify high-grade malignancy.
Our opposition might state that most men over age 69 with low-grade prostate cancer don’t have to be concerned because they are likely to die of something else before their prostate cancer spreads to other parts of the body.
While this may be the case for typical men over age 69, it’s a far cry from the longevity objectives of Life Extension customers. And low-grade prostate cancers do kill some men and are the most prevalent form of prostate cancer diagnosed.
With all due respect to Patrick Walsh, MD, who is advocating that urologists change the medical classification of low-grade prostate malignancy to remove the word “cancer” so as not to create psychological stress in aging men, and to avoid overly aggressive medical procedures, pretending low-grade malignancies are something else will not make them go away.
The documentation presented in this protocol that low-grade prostate cancers may be reversible in some men using a variety of inexpensive drugs, nutrients and dietary changes mandates that aging men have annual PSA blood tests and other diagnostics needed to assess the health of their prostate gland. If high-grade disease is detected, it is curable in its early stages, whereas the more prevalent low-grade prostate cancers are often controllable or reversible without requiring side effect-prone treatments.
New 18-Year Study Confirms Benefits of Finasteride
As we were finalizing the Life Extension magazine article titled The Avodart®-Proscar® Debate, a new study was published in the New England Journal of Medicine that further verified the safety and efficacy of finasteride in the prevention of prostate cancer (Thompson 2013).
This study meticulously followed all the men in the original Prostate Cancer Prevention Trial for up to 18 years (Thompson 2013).
The findings showed that long-term prostate cancer risk was reduced by about 33% in men who had received finasteride compared to the placebo group (Thompson 2013). This approximate 33% reduction in prostate cancer incidence was greater than the original study findings that looked at these same men over a shorter (7-year) time period (Thompson 2003; Thompson 2013).
Of men who did develop prostate cancer, those in the finasteride group had a 17% greater chance of high-grade disease, yet long-term mortality data was virtually identical in both groups (Thompson 2003). This adds a tremendous weight of evidence as to the safety of finasteride since if it really caused an increase in high-grade disease, more men in the finasteride group would be expected to have died sooner.
In addition, the 17% greater chance of high-grade disease seen in this long-term follow up was far lower than the 25.5% seen in the early phase of the Prostate Cancer Prevention Trial (Thompson 2003; Thompson 2013). The authors of this new study emphasized that the reason that more men in the finasteride group were found with high grade disease was “detection bias.” As we stated earlier, 5-alpha reductase inhibitor drugs like finasteride (Proscar®) and dutasteride (Avodart®), markedly shrink prostate gland volume, thus making detection of tumors much more efficient (Nelson 2009). Proscar® or Avodart® do not appear to cause high-grade tumors, they just make finding them much easier, which is of significant importance in obtaining curative treatment before these aggressive cancer cells escape from the confines of the prostate gland.
An editorial accompanying this New England Journal of Medicine study stated:
“For men who choose regular prostate cancer screening, the use of finasteride meaningfully reduces the risk of prostate cancer and thus the morbidity associated with treatment of the disease” (LeFevre 2013).
In maintaining the conventional party line that recommends against PSA screening, the editorial also stated:
“Men who are aware of and understand the benefits, risks, and uncertainties associated with the use of finasteride for prevention may make a rational decision to take the drug to reduce the potential harms of PSA screening. Of course, another way to reduce the harm of screening is to choose not to be screened” (LeFevre 2013).
Said differently, the author of this editorial is stubbornly sticking to irrational conventional dogma that advises men to avoid prostate cancer screening because of side effects that may occur during needle biopsy or treatment. The data the author is reporting, however, clearly shows that by taking just finasteride alone for a relatively short time period, an aging man can reduce the risk he will ever contract prostate cancer (and thus the need for “harmful” diagnostics and treatment) by 33% (Thompson 2013)!
There is evidence to suggest that prostate cancer risk reduction would have been greater had these men continued taking finasteride. As Life Extension reveals in this protocol, there are many other steps men can take to slash low- and high-grade prostate cancer risk, and at the same time, reduce overall incidence of degenerative disease.
Medical Technology Is Regressing!
We are witnessing a lethal regression in the use of technologies that could spare tens of thousands of aging men from prostate cancer deaths each year. Instead of seeking to incorporate proven methods to reduce side effects associated with conventional treatment, doctors are telling patients today to avoid screening. While this will save Medicare and Medicaid big dollars in the short-term, the epidemic of metastatic prostate cancer that will manifest in 5-10 years will extract a horrific toll of human suffering, premature death, and catastrophic costs to government healthcare systems.
Treating metastatic prostate cancer is a prolonged and extremely expensive process. Death can be postponed, but the side effects of treating advanced disease are often harsh.
A Real World Example of What This Nation Faces
When taking on the federal government and medical establishment like Life Extension routinely does, we seek to be meticulously accurate in everything we publish. Our credibility is at stake in every one of these scientific debates.
A friend of mine used to have his blood tested annually using Life Extension’s comprehensive Male Panel that includes PSA. He retired 7 years ago at the age of 60 and received “free” healthcare from his union (and later Medicare). So he stopped using Life Extension’s testing and instead let his doctor prescribe annual blood tests.
Each year he would have his blood tested, and each year his doctor said his results were fine. What my friend did not know is that the doctor stopped testing for PSA. When my friend started developing health problems his wife contacted me and said his doctors could not figure out what was wrong. I suggested he have his blood tested using our comprehensive Male Panel.
His PSA came back at 31. He appears to have metastatic disease and is undergoing aggressive treatment. He wrote me that he was shocked his doctor had not tested for PSA.
What happened to my friend is occurring throughout the United States right now. Doctors are following federal government guidelines and are intentionally omitting PSA screening. This devolution in health care must be reversed.
Startling Statistic Reported by New England Journal of Medicine
In reporting on the long-term data showing that finasteride slashed prostate cancer risk, the authors of the New England Journal of Medicine editorial opened by stating:
“With the advent of prostate-specific antigen (PSA) testing in the 1980s, the rate of diagnosis of prostate cancer rose dramatically… The timing and magnitude of the 44% reduction in prostate cancer mortality after the widespread adoption of PSA testing suggests that both screening and treatment improvements have contributed to this decline” (Thompson 2013).
The authors then go on to list all the side effects of treatment that prompted the federal government to suggest men should avoid PSA screening. Recommending against PSA screening will go down as one of the great travesties in medical history.
Prostate cancer is one of the most prevalent malignancies striking aging men. Technology developed four decades ago has resulted in a steep drop in prostate cancer-related deaths. Yet our federal government proclaimed in 2012 that this technology (PSA screening) should be abandoned.
Don’t be victimized by this nonsense.
4 Impact of Diet on Prostate Cancer Risk and Mortality
If the information you are about to read could be turned into a patented drug, it would be worth billions of dollars of annual sales to whoever owned it.
What’s regrettable is very few doctors provide this lifesaving data to their prostate cancer patients. A staggering number of lives could be spared if the dietary changes discussed in this protocol are widely implemented.
In the February 2007 issue of Life Extension magazine, we published an article titled “Eating Your Way to Prostate Cancer.”
In the April 2003 issue, we published an article titled “Eating Food Cooked at High Temperature Accelerates Aging.”
Since these articles were published, large numbers of confirmative studies have been conducted that substantiate what we warned about.
This section will describe recently published science showing how eating the wrong foods markedly increase one’s risk of developing prostate cancer.
It also reveals data showing that men already diagnosed with prostate cancer who consume the wrong foods progress to advanced disease and death faster.
Cancer cells are present in the prostate glands of many aging men, yet only one in six men are ever diagnosed with prostate cancer (ACS 2013e; Roehrborn 2006). If one looks at what is required for a single cancer cell to develop into a detectable tumor, it becomes obvious that natural barriers exist to protect men against full-blown cancer.
Unfortunately, dietary choices in the Western world circumvent the body’s protective barriers (Erdelyi 2009). The end result is that most men unwittingly provide, through their food choices, biological fuel for existing prostate cancer cells to propagate and metastasize.
An understanding of the biological roles of diet and specific nutrients can enable aging men to achieve a considerable amount of control over whether isolated cancer cells in their prostate gland will ever show up as a clinically diagnosed disease.
The impact of the food we ingest on cell growth and death is so pronounced that it can be similar to the effects displayed by anticancer drugs—without the toxicities.
Don’t Eat Overcooked Meat
Any meat (including fish) cooked at high temperatures creates dangerous carcinogens (NCI 2010). Scientists looked at men whose diets included high intake of red meat cooked at high temperatures, pan-fried, or well-done. Their findings published in 2012 showed specific gene expression changes that predisposed these men to advanced prostate cancer (Joshi 2012). These kinds of studies show that one can exert a degree of control over their cell regulatory genes by avoiding overcooked meats.
Aggressive malignancies are those that rapidly propagate, infiltrate and metastasize (NCI 2013b). A 2011 study evaluated almost 1,000 men and found that higher consumption of any ground beef or processed meats was associated with an increased risk of aggressive prostate cancer (Punnen 2011). Men who ate ground beef showed the strongest association with a 130% increased incidence. The association primarily reflected intake of grilled or barbequed meat, with more well-done meat conferring a higher risk of aggressive prostate cancer. In contrast, consumption of rare/medium cooked ground beef was not associated with aggressive prostate cancer (Punnen 2011).
A 2011 study looked at dietary patterns of 726 newly diagnosed prostate cancer cases and compared them to 527 controls (John 2011). For advanced prostate cancer (but not localized disease), there was an associated 79% increased risk in men who ate hamburgers, a 57% increased risk with processed meats, a 63% increased risk with grilled red meat, and a 52% increased risk with well-done red meat (John 2011). This study corroborated others associating consumption of processed meat and red meat, especially when cooked at high temperatures, with increased cases of advanced prostate cancer (Joshi 2012; NCI 2013b).
Concern About Eggs and Milk
Large-scale studies associate egg consumption with sharply increased cancer risks (Richman 2011; Aune 2009).
A 2011 study looked at 27,607 men who developed or died from prostate cancer over a 14-year period (Richman 2011). Men who consumed 2.5 or more eggs per week had an 81% increased risk of lethal prostate cancer compared to those who consumed less than half an egg per week (Richman 2011). This study showed that consumption of eggs increased the risk of healthy men developing metastatic prostate cancer.
A 2013 evaluation was done using data from the famous Physician’s Health Study to identify the impact of consumption of skim or whole milk on incidence and survival after diagnosis of prostate cancer (Song 2013). This analysis involved 21,660 physicians who were followed for 28 years. Skim/low fat milk was associated with increased risk of low grade prostate cancer, whereas whole milk was associated with increased risk of fatal prostate cancers. In these men diagnosed with prostate cancer, consumption of whole milk was associated with a 117% increased risk of progression to fatal disease (Song 2013). This finding further substantiates the important role of diet even after prostate cancer is diagnosed.
The take-home lesson so far is if one has an elevated or rising PSA, it is especially prudent to avoid over cooked red meats, processed meat, eggs, and whole cow’s milk.
Confusion About Omega-6 Fats
Omega-6 fats are essential to life. We are unable to make them in our body and must get them from foods (UMMC 2013b).
The problem is that Western diets have become so overloaded with omega-6s that our bodies have become poisoned with them. The typical American tends to consume up to 25 times more omega-6 fats than the healthier omega-3 fats (UMMC 2013b).
One reason we have become so overloaded with omega-6s is that in the rush to switch from red meat and other saturated fats such as lard, we have been gobbling down too many omega-6-rich foods. These include vegetable oils used in all kinds of processed and fried foods, margarine, salad dressing, mayonnaise, certain nuts, peanut butter, and even poultry, a meat with high omega-6 content (SELF 2012). `
Commercial food companies deceivingly promote polyunsaturated vegetable oils like corn and safflower as healthy because of early studies showing reduced cardiovascular risk factors in those who consumed vegetable oils compared to animal-based fats such as butter (Mekki 2002).
Remember that you require omega-6s to live, but not in the large quantity consumed in the typical American diet. This means you want to lower the percentage of calories in your diet that comprise of omega-6 fats.
Table 1 lists foods high in omega-6 fats. Eating any of these foods in moderation is not a problem, but when they comprise a high percentage of your overall diet, your body becomes overloaded with omega-6s, which sets the stage for a wide variety of disorders. We next describe how a high intake of omega-6 fats contributes to prostate cancer.
Table 1
Dietary Sources of Omega-6 Fats (SELF 2012)
Avocados |
Peanut Oil |
Almonds |
Sunflower Oil |
Macadamia Nuts |
Safflower Oil |
Peanuts |
Canola Oil |
Brazil Nuts |
Macadamia Nut Oil |
Hazelnuts |
Grapeseed Oil |
Pecans |
Corn Oil |
Pistachios |
Peanut Butter |
Avocado Oil |
Almond Butter |
Almond oil |
Cashew Butter |
What You Need to Know
Prostate Cancer: Food For Thought
- Dietary choices in the Western world circumvent the body’s protective barriers to cancer.
- Men who regularly consume certain plant foods have sharply lower rates of prostate cancer.
- High intakes of red meat cooked at high temperatures can result in specific gene expression changes that may predispose men to advanced prostate cancer.
- Large-scale studies associate egg consumption with sharply increased cancer risks.
- Skim/low fat milk was associated with increased risk of low grade prostate cancer, whereas whole milk was associated with increased risk of high-grade prostate cancers.
- Diets high in omega-6 fats and saturated fats are associated with greater prostate cancer risk, whereas increased intake of the type of omega-3 fats found in fish has been shown to confer protection.
- Arachidonic acid, found abundantly in eggs and chicken, is metabolized by the 5-LOX enzyme to 5-hydroxyeicosatetraeonic acid (5-HETE), a potent survival factor that prostate cancer cells use to escape destruction.
- Not only do 5-LOX products directly stimulate cancer cell propagation, but the breakdown products that 5-LOX produces from arachidonic acid (such as leukotriene B4, 5-HETE, and hydroxylated fatty acids) cause tissue destruction, chronic inflammation, and increased resistance of tumor cells to apoptosis (programmed cell destruction).
Role of Omega-6 Fats in Prostate Cancer
Diets high in omega-6 fats and saturated fats are associated with greater prostate cancer risk, whereas increased intake of the type of omega-3 fats found in fish has been shown to confer protection (Newcomer 2001; Leitzmann 2004; Pelser 2013).
Based on consistent epidemiological findings across a wide range of human populations, scientists have sought to understand why eating the wrong kinds of fat (saturated and omega-6 fats) provokes a stimulatory effect on prostate cancer (Newcomer 2001; Pelser 2013).
To ascertain what happens after we eat bad fats, all one has to do is look at the metabolic breakdown pathways that these fats follow in the body, as shown in Figure 1. For example, let us assume that for dinner, you eat a steak (a source of saturated fat), potato (a high-glycemic starch) and a salad with a typical dressing of soybean and/or safflower oils (omega-6 fats).
As can be seen in the flow chart, omega-6 fats can convert to arachidonic acid in the body. Meat itself contains arachidonic acid (Sears 2011). One way that the body rids itself of excess arachidonic acid is by provoking a dangerous metabolizing pathway through 5-lipoxygenase (5-LOX).
It is well established that 5-LOX products stimulate prostate cancer cell proliferation via several well-defined mechanisms (Hassan 2006; Moreti 2004; Ghosh 1998). High glycemic foods also promote formation of 5-LOX in the body, via activation of enzymes involved in the formation of arachidonic acid (Sears 2011).
Arachidonic acid, found abundantly in eggs and chicken, is metabolized by the 5-LOX enzyme to 5-hydroxyeicosatetraeonic acid (5-HETE), a potent survival factor that prostate cancer cells use to escape destruction (Sears 2011; Ghosh 1998; Sundaram 2006). The flow chart (Figure 1) clearly demonstrates how consuming a diet rich in arachidonic acid provokes the production of dangerous 5-LOX products, which can promote prostate cancer progression (Sears 2011; Hassan 2006; Moretti 2004; Ghosh 1997; Ghosh 1998; Sundaram 2006). In addition to 5-HETE, 5-LOX also metabolizes arachidonic acid to leukotriene B4 and other pro-inflammatory agents that promote cancer (Larré 2008).
The chart (Table 1) provides a long list of foods that are high in arachidonic acid. Just because a food is listed on this chart does not mean you have to avoid it. It is wise, however, to pick which high-arachidonic acid foods are that important compared to ones you may not even realize you’re consuming.

Flow chart showing how the body metabolizes common foods via a 5-lipoxygenase (5-LOX) pathway.
5-LOX Is Over-Expressed in Prostate Cancer
Based on studies showing that consumption of foods rich in omega 6 fatty acids is associated with higher incidences of prostate cancer, scientists sought to determine how much of the 5-LOX enzyme is present in malignant versus benign prostate tissues (Ritch 2007).
Using biopsy samples taken from living human patients, the researchers found that 5-LOX mRNA levels were an astounding six-fold greater in malignant prostate tissues compared with benign tissues. This study also found that levels of 5-HETE were 2.2-fold greater in malignant versus benign prostate tissues (Gupta I 2001). The scientists concluded this study by stating that selective inhibitors of 5-LOX may be useful in the prevention or treatment of patients with prostate cancer (Gupta I 2001).
5-LOX Promotes Tumor Growth Factors
As the evidence mounts that ingesting “bad fats” increases prostate cancer risk, scientists are evaluating the effects of 5-LOX on various growth factors involved in the progression, angiogenesis, and metastasis of cancer cells.
One study found that 5-LOX activity is required to stimulate prostate cancer cell growth by epidermal growth factor (EGF) and other tumor cell proliferating factors produced in the body (Hassan 2006). When 5-LOX levels were reduced, the cancer cell stimulatory effect of EGF and other growth factors was diminished (Hassan 2006).
In a mouse study, an increase in 5-LOX resulted in a corresponding increase in vascular endothelial growth factor (VEGF), a key growth factor that tumor cells use to stimulate new blood vessel formation (angiogenesis) into the tumor. 5-LOX inhibitors have been shown to reduce tumor angiogenesis along with a host of other growth factors (Ye 2004).
In both androgen-dependent and androgen-independent human prostate cancer cell lines, the inhibition of 5-LOX has consistently been shown to halt the growth stimulatory action of 5-LOX and prompt rapid and massive apoptosis (cancer cell destruction) (Moretti 2004; Ghosh 1998; Ghosh 2003; Anderson 1998).
Omega-3 Fatty Acids: A First Line of Defense
One reason that fish oil supplements have become so popular is that their beneficial EPA/DHA fatty acids can help reduce the production of arachidonic acid-derived tumor promoting byproducts in the body (Norris 2012; Barham 2000; Adan 1999; Schwartz 2000). As shown in Figure 2, if arachidonic acid levels are reduced, there would be a corresponding suppression of 5-LOX and its metabolic byproducts 5-HETE and leukotriene B4.
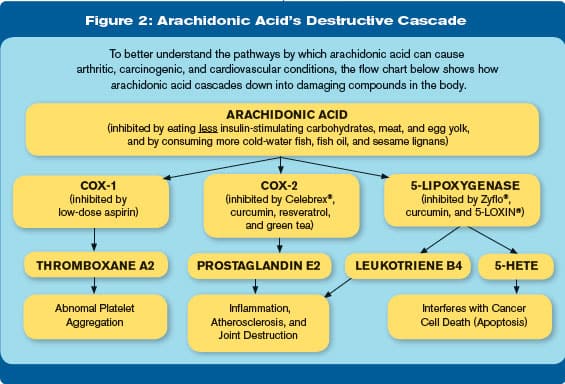
Once one understands the lethal 5-LOX cascades, it is easy to see why people who excessively consume foods rich in arachidonic acid, and those who fail to reduce the production of arachidonic acid metabolites (such as 5-HETE) by ensuring adequate intake of omega-3 fatty acids, are setting themselves up for prostate cancer and a host of inflammatory disorders (including atherosclerosis).
Nutrients That Suppress 5-LOX
Health-conscious people already take nutrients like curcumin and fish oil that help to lower 5-LOX activity in the body (Taccone-Gallucci 2006; Gupta SC 2013). Studies show that lycopene and saw palmetto extract also help to suppress 5-LOX (Hazai 2006; Bonvissuto 2011). The suppression of 5-LOX by these nutrients may partially account for their favorable effects on the prostate gland.
As humans age, however, chronic inflammatory processes can cause the over-expression of 5-LOX in the body (Qu 2000). For maturing males, the result of excess 5-LOX may be the epidemic of prostate cancer observed as men age (Roehrborn 2006).
Based on the cumulative knowledge that 5-LOX itself and its metabolic products can promote the progression and metastasis of prostate cancer cells, it would appear advantageous to take aggressive steps to suppress this lethal enzyme (Hazai 2006).
This can be done by avoiding foods that promote 5-LOX formation in the body and taking supplements that inhibit 5-LOX via differing pathways.
Research Substantiating Boswellia
Specific extracts from the boswellia plant selectively inhibit 5-lipoxygenase (5-LOX), a potent inducer of inflammation and carcinogenic byproducts (Safayhi 1997; Safayhi 1995).
Boswellia extracts have been used for centuries, particularly in India as anti-inflammatory agents (Siddiqui MZ 2011). In several well-controlled human studies, boswellia has been shown to be effective in alleviating various chronic inflammatory disorders (Kimmatkar 2003; Ammon 2002; Gupta I 2001; Gerhardt 2001; Gupta 1998).
Scientists have discovered that the specific constituent in boswellia responsible for suppressing 5-LOX is AKBA (3-O-acetyl-11-keto-B-boswellic acid) (Siddiqui MZ 2011). Boswellia-derived AKBA binds directly to 5-LOX and inhibits its activity (Siddiqui MZ 2011). Other boswellic acids only partially and incompletely inhibit 5-LOX (Siddiqui MZ 2011; Sailer 1996).
Formulas containing high concentrations of AKBA from boswellia have been developed based on its ability to treat inflammatory disorders. Standardized boswellia extracts have long been included in prostate support nutrient formulas for the purpose of suppressing excess 5-LOX.
MULTIPLE DANGERS OF EXCESS ARACHIDONIC ACID
In response to arachidonic acid overload, the body increases its production of enzymes like 5-lipoxygenase (5-LOX) to degrade arachidonic acid (Jiang 2006). Not only do 5-LOX products directly stimulate cancer cells propagation, but the breakdown products that 5-LOX produces from arachidonic acid (such as leukotriene B4, 5-HETE, and hydroxylated fatty acids) cause tissue destruction, chronic inflammation, and increased resistance of tumor cells to apoptosis (programmed cell destruction) (Hassan 2006; Moretti 2004; Ghosh 1997; Jiang 2006; Yoshimura 2004; Zhang 2006; Soumaoro 2006; Hoque 2005; Zhi 2003; Rubinsztajn 2003; Subbarao 2004; Laufer 2003; Hennig 2002; Ding 1999).
It is important to understand that 5-LOX is not the only dangerous enzyme the body produces to break down arachidonic acid. As can be seen in Figure 2, both cyclooxygenase-1 and cyclooxygenase-2 (COX-1 and COX-2) also participate in the degradation of arachidonic acid.
COX-1 causes production of thromboxane A2, which can promote abnormal arterial blood clotting (thrombosis), resulting in heart attack and stroke (de Leval 2004; Cheng 2002; Catella-Lawson 2001; James 2001; Garcia Rodriguez 2001). Thromboxane A2 is also involved in tumor metastasis (Nie 2000; Dassesse 2006). COX-2 is directly involved in cancer cell propagation, while its breakdown product (prostaglandin E2) promotes chronic inflammation (Laufer 2003; Wang 2004; Onguru 2005; Wang 2006). Most health-conscious people already inhibit the COX-1 and COX-2 enzymes by taking low-dose aspirin, curcumin, green tea, and various plant flavonoids such as pomegranate (Catella-Lawson 2001; Feldman 2001; Zhang 1999; Bengmark 2006; Park 2006; Hong 2004; Yoysungnoen 2006; Tunstall 2006; Lee 2005; Hong 2001; Ahmed 2002; Shukla 2008; Rosillo 2012; Hämäläinen 2011; O’Leary 2004).
A more integrative approach to this problem, however, would be to also reduce dietary levels of arachidonic acid, which is the precursor of 5-HETE and leukotriene B4 (Riehl 1992).
Prostate-Protecting Properties of Boswellia
Tumor necrosis factor-alpha (TNF-alpha) is a proinflammatory cytokine that often increases in aging people (Gupta 2003).
From the standpoint of keeping prostate cancer cells in check, boswellia has been shown to prevent the TNF-alpha-induced expression of a proteindegrading enzyme called matrix metalloproteinase (MMP) (Roy 2005). Cancer cells use the MMP enzyme to tear apart natural barriers in the body that would normally encase them (Katiyar 2006). Prostate cancer cells are notorious for inducing the production of this enzyme (TNF-alpha) that causes containment structures within the prostate gland to vanish, thus enabling the cancerous prostate cells to break through healthy prostate tissue and eventually metastasize (Ridriguez-Berriguete 2013; Mizokami 2000).
Prostate cancer cells use adhesion molecules (known as VCAM-1 and ICAM-1) to facilitate their spread throughout the body. Boswellia has been shown to prevent the up-regulation of these adhesion molecules, which are directly involved in inflammatory processes (Roy 2005). Chronic inflammation is tightly linked to the induction of aberrant angiogenesis used by cancer cells to facilitate the growth of new blood vessels (angiogenesis) into tumors (Rajashekhar 2006).
The potent 5-LOX-inhibiting properties of boswellia, and its ability to suppress other inflammatory factors such as TNF-alpha, make it an important nutrient for those concerned with prostate cancer (Siddiqui MZ 2011; Roy 2005).
TEA, SOY, LIGNANS, AND CRUCIFEROUS VEGETABLES
Men who regularly consume certain plant foods have sharply lower rates of prostate cancer (Chan 2009). Studies show that cauliflower, broccoli, flax lignans, and soy isoflavones protect against a host of diseases, including prostate cancer (Heald 2007; Hedelin 2006; Kumar 2004; Lee 2003; McCann 2005; Vij 2004; Sonoda 2004; Clark 2008; Abdull Razis 2013).
A 2013 study reported that soy food consumption, more popular in Asian populations, is associated with a 25 to 30% reduced risk of prostate cancer (Kimura 2012). There is a lot of confusion about soy in the alternative health community. Everyone agrees that limiting intake of soybean oil is important because it is loaded with omega-6 fats (SELF 2012). Other constituents found in soy, however, such as genistein and daidzein have demonstrated anti-prostate cancer mechanisms (Chen 2011; Lakshman 2008; Davis 1998; Davis 1999; Ozasa 2004; Park 2013; Dong 2013).
Polyphenols found in green tea decrease serum levels of PSA, hepatocyte growth factor, and vascular endothelial growth factor (VEGF) in prostate cancer patients (Henning 2006; Bettuzzi 2006; Pandey 2009; McLarty 2009; Siddiqui 2006; Chuu 2009).
The FDA says, however, that there is insufficient evidence to claim that green tea consumption protects against prostate cancer (WebMD 2005). Life Extension issued a rebuttal to the FDA position on green tea and prostate cancer in the November 2005 edition of Life Extension magazine.
Urgent Need to Alter Dietary Patterns
Those consuming Western diets predispose themselves to cancer (Erdelyi 2009). It is encouraging that we know what food groups increase prostate cancer risk and what foods/nutrients reduce it.
All aging men should shift their diet towards foods that protect against prostate cancer. Those with rising or elevated PSA should be especially diligent in avoiding dietary factors that can fuel the growth of prostate tumors (WHF 2013).
The lethal impact of cancer-promoting foods may be mitigated by taking supplements such as green tea, curcumin, fish oil, pomegranate, and boswellia, along with regular inclusion of cruciferous vegetables and other healthy foods in one’s diet.
5 A Natural Arsenal for Prostate Cancer Prevention
A remarkable new study has validated a method to slow prostate cancer progression that was long ago recommended to Life Extension customers.
What made this study even more noteworthy is where it was presented.
The annual gathering of the American Society of Clinical Oncology (ASCO) is considered the world’s most prestigious cancer forum. More than 25,000 oncology experts attend this meeting, and the media eagerly reports on meaningful advances in cancer prevention and treatment.
At the 2013 ASCO meeting, findings from a study were released that underscored how effective certain natural compounds can be as a prostate cancer therapy.
In this placebo-controlled, double-blind trial of treatment-refractory prostate cancer patients, a four-nutrient supplement resulted in a 63.8% median reduction in the increase of PSA levels (Thomas 2013). The PSA marker is used by oncologists to determine progression or regression of prostate cancer, and to evaluate whether treatments are working or failing.
In the study presented at ASCO, patients with a PSA relapse after radiotherapy or surgery for localized prostate cancer took two daily capsules containing pomegranate seed, broccoli, green tea, and turmeric. Over a six-month period, median PSA levels increased only 14.7% in the supplement group—compared to 78.5% in the placebo group (Thomas 2013)! PSA levels remained stable, or below, baseline values for a compelling 46% of the supplement patients—but for only 14% of the placebo patients.
Prostate cancer is the most common malignancy in US men (excluding non-melanoma skin cancer) (Siegel 2012), affecting one male in every six (PCF 2013b). Autopsy findings show a significant percentage of men have underlying prostate cancer without even knowing it (Harvei 1999; Billis 1986; Sakr 1995).
This section will present evidence about the prostate cancer preventing effects of a wide range of nutrients. What makes this topic so compelling are the recent findings presented at ASCO showing that pomegranate seed, green tea, broccoli, and turmeric (source of curcumin) were so effective in prostate cancer patients (Thomas 2013). The implication is that these nutrients may also afford considerable protection against prostate cancer progression.
A comprehensive defense against prostate cancer involves healthy diet, supplemental nutrients, hormone balance, and annual PSA screening. The foods and nutrients described herein have been documented in published studies to target prostate cancer and help prevent or attenuate its development. As a bonus, they also confer huge protection against other age-related disorders.
Since there are overlapping mechanisms of action amongst many of these foods/nutrients, it may not be necessary to take every one of them. Most impressive, however, is the voluminous amount of scientific evidence that substantiates the anti-cancer properties of these nutrients. Yet mainstream medicine remains largely in the dark.
Nutrients for Prostate Cancer Prevention
Flaxseed
Flaxseeds provide a rich supply of lignans and essential fatty acids that promote prostate health. The lignans in flaxseed are believed to offer protection against chronic disease and cancer, including hormone-dependent malignancies (Donaldson 2004; Stark 2002; Demark-Wahnefried 2004).
A large study demonstrated that men with higher enterolactone levels were up to 72% less likely to have prostate cancer than those with the lowest levels (Hedelin 2006). Studies have confirmed that flaxseed supplementation lowers PSA levels, and significantly reduces the proliferation of normal prostate cells and prostate cancer cells (Demark-Wahnefried 2004; Demark-Wahnefried 2008). A pilot study on men who were scheduled to have a repeat prostate biopsy found that supplementation with flaxseeds, as part of a low-fat diet, lowered levels of PSA and prostate cell proliferation (Demark-Wahnefried 2004).
Boron
Research has shown that boron can reduce the risk of prostate cancer (Zhang 2001). In one study, men with the highest boron intake showed a 54% lower risk of prostate cancer compared to those with the least intake (Cui 2004).
In a validated animal model of prostate cancer, researchers found that oral administration of various concentrations of a boron-containing solution led to 25-38% decreases in tumor size, and 86-89% reductions in PSA levels (Gallardo-Williams 2004). The suggestion that supplemental boron may help to shrink prostate tumors while also decreasing levels of PSA is exciting. That’s because PSA—in addition to being an important prostate cancer marker—may itself be a contributor to prostate cancer promotion (Webber 1995).
Boron compounds inhibit the activity of prostate-specific antigen (PSA) (Gallardo-Williams 2004). Higher boron levels in the blood lower the risk of prostate cancer by reducing intracellular calcium signals and storage (Henderson 2009). At normal concentrations, boron operates selectively—inhibiting prostate cancer cell proliferation while allowing normal prostate cells to grow (Barranco 2004).
The typical daily intake range for boron is 1-8 milligrams daily; however, individuals living in boron-rich environments may consume far greater than this amount (Meacham 2010). If lab studies can be replicated in human patients, higher daily dosages may become an effective and low-cost adjuvant therapy. Life Extension® customers already obtain boron (3-6 mg) in their supplements.
Cruciferous Vegetables
In recently released studies, three phytochemicals derived from cruciferous vegetables (such as broccoli) have shown promise in inhibiting prostate cancer in experimental models (Beaver 2012; Yu 2013). Because their chemical names are challenging—indole-3-carbinol, 3,3’-diindolylmethane, and phenethyl isothiocyanate—they are better known as I3C, DIM, and PEITC, respectively.
I3C has several different actions that help prevent and inhibit prostate cancer. It helps activate detoxification pathways, prevents cancer cell growth, induces apoptosis, regulates gene expression, protects DNA from damage, and modulates a variety of cell signaling pathways (Yu 2013; Sarkar 2004; Fong 1990; Chinni 2001).
DIM has been shown to protect against prostate cancer by inhibiting the phosphorus-transferring enzyme Akt, inhibiting the master DNA-transcription regulator nuclear factor-kappaB (NF-kB)—and blocking the crosstalk between them (Li 2005). This is a novel mechanism through which DIM inhibits cell growth and induces apoptosis in prostate cancer cells, but not in non-tumorigenic prostate epithelial cells (Li 2005). The ability of DIM to target aberrant epigenetic changes coupled with its ability to promote the detoxification of carcinogens, make it an effective chemopreventive agent as it is able to target multiple stages of prostate carcinogenesis (Banerjee 2011).
In a study released in May 2013, PEITC was found to suppress a compound known as PCAF (P300/CBP-associated factor)—which in turn inhibits androgen receptor-regulated transcriptional activity in prostate cancer cells (Beaver 2012). Daily suggested dosages are 14 milligrams for DIM, and 80-160 milligrams for I3C. An I3C dosage of 200-600 milligrams daily is suggested as an adjuvant for prostate cancer therapy. Dosages for PEITC are not well-established.
Vitamin D
As the New England Journal of Medicine clarified, “Cancer results from the accumulation of mutations in genes that regulate cellular proliferation” (Haber 2000). In other words, cancer is essentially caused by the genetic mutations that occur over the lifespan. The fascinating impact of vitamin D is that it protects against cancer by enabling us to regain control over the genes that regulate cell proliferation. Vitamin D affects at least 200 human genes (Holick 2007). These genes are responsible for regulating crucially important aspects of cells: their proliferation, differentiation, and apoptosis.
In recent years, a multitude of studies have shown cancer risk reductions of 50% and greater based on higher vitamin D status (Garland 1989; Garland 1991; Garland 2007; Gorham 2005). People with higher vitamin D levels have lower risks of lethal prostate cancer, as well as reduced risks of other cancers (Holick 2007; Garland 1989; Yousef 2013; Skinner 2006; Polesel 2006; Shui 2012). Individual blood testing is needed to determine individual-appropriate dosages, which typically range from 2,000 to 10,000 international units (IU) daily for prevention. Life Extension suggests an optimal vitamin D blood level of 50-80 nanograms per milliliter (ng/mL).
Soy Isoflavones
Some studies show that the highest intake of soybased foods correlates with a 42-75% lower risk of prostate cancer (Lee 2003; Sonoda 2004; Zhou 1999; Jacobsen 1998). Early animal studies found that this difference is most likely attributable to soy isoflavones inhibiting prostate tumor growth by acting directly against tumor cells and indirectly against tumor neovasculature (growth of new blood cells) (Zhou 1999). Human studies support this evidence.
Japanese scientists took blood samples from over 14,000 men during 1988-1990. Their analysis clearly established that elevated serum levels of all three isoflavones assessed—genistein, daidzein, and equol—imparted strong protective effects against prostate cancer (Ozasa 2004). Men with the highest circulating levels of genistein, daidzein, and equol reduced prostate cancer risk by 62%, 59%, and 66%, respectively. Genistein and daidzein are found in soy, and equol is derived from daidzein by bacterial flora in the intestines (Ozasa 2004; USDA 2013; Wang 2005). Also, genistein was shown to have “potent anti-proliferative effects” against human prostate cells (Shen 2000) and inhibit metastasic potential of sex gland cancers such as prostate cancer (Schleicher 1999). Genistein also blocks an enzyme that destroys an anticancer vitamin D metabolite in cancer cells (Farhan 2002). A suggested dosage of soy isoflavones is 135-270 milligrams daily with food.
Green Tea Extract
Laboratory research with cultures has long suggested that green tea catechins, including epigallocatechin-3 gallate (EGCG), may inhibit the growth of cancer cells. Evidence from human studies now demonstrates that green tea compounds can help prevent prostate cancer. A clinical trial demonstrated that green tea catechins were 90% effective in preventing prostate cancer in men with pre-malignant lesions (Bettuzzi 2006). Researchers recruited 60 men, aged 45-75. Thirty participants received 200 milligrams of green tea catechins (50% EGCG) three times daily, while the other 30 subjects received a placebo. Biopsies were conducted at six and 12 months. Remarkably, only one man in the treatment group was diagnosed with prostate cancer, compared to nine men in the control group who developed the disease. No significant side effects or adverse reactions were reported (Bettuzzi 2006). The lead researcher concluded that “90% of chemoprevention efficacy could be obtained by [green tea catechin] administration in men prone to developing prostate cancer” (Bettuzzi 2006).
Green tea polyphenols have also shown efficacy as an adjunctive therapy. Prostate cancer patients were given 1,300 milligrams of green tea polyphenols, mostly EGCG, prior to the time of radical prostatectomy. They showed significant reductions in PSA and other tumor promoters such as vascular endothelial growth factor (McLarty 2009). Suggested dosages of EGCG are 300-350 milligrams daily, and adjuvant cancer therapy dosages of EGCG range up to 3,000 milligrams daily. The FDA, however, does not believe there is sufficient evidence to say that green tea reduces prostate cancer risk. A federal judge ruled against the FDA’s attempt to suppress claims that green tea may reduce prostate cancer risk (Faloon 2012).
Omega-3 Fatty Acids
In scientific studies, high blood levels of the omega-3 fatty acids DHA and EPA (docosahexaenoic acid and eicosapentaenoic acid, respectively) have been demonstrated to correspond to a lower risk of developing prostate cancer (Norrish 1999). EPA has been shown to suppress the formation of the omega-6 fatty acid arachidonic acid (AA) by inhibiting the enzyme delta-5-desaturase (Dias 1995). EPA has also been found to contribute to the inhibition of uPA—a substance known as urokinase-type plasminogen activator believed to play a role in prostate cancer invasion and metastasis (du Toit 1996).
Although cold water fish such as tuna, sardines, herring, and salmon provide a rich omega-3 source, commercially available pharmaceutical-grade fish oils also deliver large amounts of EPA and DHA (Cleveland Clinic 2009). Suggested dosages are 2-4 grams of fish oil concentrate supplying 700-1,400 milligrams of EPA and 500-1,000 milligrams of DHA, daily with food. For adjuvant cancer therapy, recommended dosages are 4-8 grams of fish oil concentrate supplying up to 2,800 milligrams of EPA and up to 2,000 milligrams of DHA, daily with food.
Curcumin
Curcumin strikes at multiple targets in prostate cancer (Shishodia, Chaturvedi 2007; Kunnumakkara 2008). It induces apoptosis, interferes with the spread of cancer cells, and regulates inflammatory responses through the master regulator nuclear factor-kappaB (NF-kB), a protein complex that controls the transcription of DNA (Plummer 1999; Teiten 2010; Khan 2010; Aggarwal 2007). Natural molecules that inhibit NF-kB can limit inflammatory changes (Gukovsky 2003). Prostate cancer is often dependent on sex hormones for its growth; curcumin reduces expression of sex-hormone receptors (androgen receptors and androgen receptor-related cofactors) in the prostate (Nakamura 2002; Choi 2010). This speeds androgen receptor breakdown and impairs cancer cells’ ability to respond to the effects of testosterone (Tsui 2008; Shi 2009).
Both in vitro and in vivo models demonstrate that curcumin inhibits prostate cancer promotion by blocking metastases of cancer cells in the prostate, and by regulating enzymes required for tissue invasiveness (Hong 2006; Herman 2009). In certain human prostate cancer cell lines, curcumin completely inhibited a type of phosphorus transferring enzyme known as Akt (also known as protein kinase B or PKB), suggesting that curcumin inhibits prostate cancer cell growth through this Akt-inhibiting mechanism (Chaudhary 2003). Curcumin has been shown to inhibit angiogenesis in prostate cancer cells in vivo (Dorai 2001).
Coenzyme Q10
Low blood levels of coenzyme Q10 (CoQ10 or Q10) have been found in patients with a variety of cancer types (Rusciani 2006; Folkers 1997). Several published animal and human studies have demonstrated CoQ10’s remarkable effects against some cancers (Hodges 1999; Folkers 1993; Lockwood 1994; Perumal 2005; Folkers 1996; Portakal 2000; Ren 1997; Jolliet 1998), but research into its potentially protective effects against prostate cancer has been very limited. In 2005, after reviewing anecdotal reports appearing in the peer-reviewed scientific literature, the National Cancer Institute (NCI) reported that coenzyme Q10 has been anecdotally reported to lengthen the survival of patients with cancer of the prostate, as well as several other cancers (NCI 2013a). Despite these findings, the NCI pointed out that the absence of a control group in the human studies and other scientific weaknesses made it impossible to determine whether these beneficial results were directly related to CoQ10 therapy (NCI 2013a).
Later that same year, University of Miami researchers reported research showing that adding coenzyme Q10 in vitro to the most common prostate cancer cell line, PC3, inhibited cell growth by 70% over 48 hours (UMMSM 2013). Evidence suggested that there had been a reduction in the expression of a key, anti-apoptotic gene protein, bcl-2, and through this mechanism, CoQ10 had restored the ability for apoptosis, allowing the cancer cells to kill themselves. “The most amazing part,” said UM research associate Niven Narain, “is that we’ve been able to restore a cancer cell’s ability to kill itself, while not impacting normal cells” (UMMSM 2013). The suggested preventive dosage of coenzyme Q10 is 100 milligrams daily, and a suggested adjuvant dosage is 200-500 milligrams daily, both taken after a meal.
Gamma-Tocopherol Vitamin E
A large study showed that the risk of prostate cancer declines with increasing concentrations of the alpha-tocopherol form of vitamin E, with the highest level corresponding to a 35% lower risk; however, these protective effects were only observed when levels of gamma-tocopherol and levels of selenium were also high (Helzlsouer 2000).
Men with the highest gamma-tocopherol levels, those in the highest fifth of the distribution, were found to have a 5-fold greater reduction in the risk of developing prostate cancer than men in the lowest fifth (Helzsouer 2000). Other research has shown that vitamin E reduces the growth rate of existing prostate cancers that are specifically exacerbated by a high-fat diet—reducing tumor growth rate within a high-fat diet to the same tumor growth rate as in a lower-fat (ideal) diet (Fleshner 1999).
While both alpha- and gamma-tocopherols are potent antioxidants, gamma-tocopherol has a unique function. Because of its different chemical structure, gamma-tocopherol scavenges reactive nitrogen species, which can damage proteins, lipids, and DNA, and promote carcinogenesis (Prins 2008; Stone 1997; Christen 1997; Cooney 1993). The suggested dosage of gamma-tocopherol is 200-250 milligrams daily, and the suggested adjuvant therapy dosage is 400-1,000 milligrams daily, taken with food.
Lycopene
Lycopene is a carotenoid occurring abundantly in tomatoes. The relationship between its ingestion and prostate health is well established (Soares 2013; Rafi 2013; Giovannucci 1995; Wertz 2009; Lowe 2006; Trejo-Solís 2013; Obermuller-Jevic 2003; Kucuk 2001; Gann 1999). One laboratory experiment found that lycopene inhibited the growth of normal human prostate cells (Obermuller-Jevic 2003). Then, a clinical trial conducted on prostate cancer patients demonstrated that lycopene supplementation decreases the growth of prostate cancer (Kucuk 2001). In another compelling study, healthy men with the highest lycopene levels in their blood were shown to have a 60% reduced risk of developing prostate cancer (Gann 1999).
Scientists found that lycopene works by reducing oxidative stress in prostate tissue; lowering inflammatory signaling; preventing DNA damage; modulating expression of endocrine growth factors; and may block cancer cells from growing out of control through enhanced communication between cancer cells at “gap junctions” (Wertz 2009; Trejo-Solís 2013). Lycopene also may slow the new blood vessel growth that prostate cancers need for development (Trejo-Solís 2013). Suggested dosages of 15-30 milligrams daily are for prevention and up to 45 milligrams daily with food for adjuvant support in existing prostate cancer.
Selenium
The body only needs small quantities of selenium (NIH 2013); however, blood levels of this mineral decrease with age, placing middle-aged to older men at high risk for inadequate selenium levels. Lower levels of selenium in the blood can correspond to an increased risk of an enlarged prostate, the condition known as benign prostatic hyperplasia (BPH) (Eichholzer 2012). Low selenium levels were also found to parallel a four- to five-fold higher risk of prostate cancer (Brooks 2001). Remarkably, supplementation with selenium has been demonstrated to produce an up to 63% reduced risk of prostate cancer (Duffield-Lillico 2002; Clark 1998). The mechanism behind this protection appears to be related to an antiproliferative effect, resulting from selenium’s upregulation of cell-cycle regulators (Venkateswaran 2002).
However, confusion arose in 2009 due to publication of a single negative study that substantially contributed to misinformation about the value of selenium against prostate cancer. Known as SELECT—for Selenium and Vitamin E Cancer Prevention Trial—the study appeared to show that selenium, alone or in combination with vitamin E, had no detectable effect on preventing cancers (Klein 2011; Lippman 2009). Many experts have since condemned the trial’s methodology and conclusions (El-Bayoumy 2009)—and for a number of reasons.
One problem with the 2009 study was that it used only a single form of selenium (Klein 2011; Marshall 2011). This selenium compound is just one of several different forms in which selenium is available for nutritional supplementation. Data indicate that three forms of selenium—the two organic forms called L-selenomethionine and selenium-methyl L-selenocysteine, plus the inorganic form known as sodium selenite—have different degrees of action with regard to the effect on any incipient cancer cells that might be developing (El-Sayed 2006; Suzuki 2010; Lunoe 2011). Using one form weakened the potential protective benefits in the study.
More importantly, the highly flawed 2009 SELECT study used only one form of vitamin E, a synthetic form known as dl-alpha tocopheryl acetate. We have known for about 15 years that when alpha tocopherol is taken by itself, it displaces critically important gamma tocopherol—the form of vitamin E that is the most protective against prostate cancer (Christen 1997; Galli 2004; Jiang 2004; Gysin 2002; Helzlsouer 2000; Moyad 1999). By supplementing aging men with only one form of vitamin E, synthetic dl-alpha tocopheryl acetate, scientists in the 2009 SELECT study may have unwittingly increased subjects’ prostate cancer risk by depriving prostate cells of critical gamma tocopherol. Then, a 2011 meta-analysis of nine randomized, controlled clinical trials including 152,538 participants established that selenium supplementation cut risk for all cancers by 24%. The cancer-preventive effect rose to 36% in people with low baseline selenium levels (Lee EH 2011).
Based on research involving non-melanoma skin cancer patients—in which patients received either 200 micrograms daily of selenium or a placebo— researchers concluded that selenium supplementation can slash the risk of dying from any type of cancer by 50% (Clark 1996). Also, selenium’s efficacy could potentially be enhanced: one study observed the protective effects of high selenium levels against prostate cancer only when the concentrations of gamma-tocopherol, an isomer of vitamin E, were also high—suggesting that these two nutrients may work best together (Helzlsouer 2000). It is suggested that selenium be taken at dosages of 200 micrograms daily with food.
Zinc
Evidence suggests that zinc may play an important and direct role in the prostate. For example, studies found that total zinc levels in the prostate are much higher than in other soft tissues in the body, and those with prostate cancer have been shown to have exceedingly low levels of zinc in the prostate (Gómez 2007; Zaichick 1997). Also, in normal prostate cells, zinc is highly concentrated intracellularly in the glandular epithelium—but adenocarcinoma cells taken from prostate tumors have lost their ability to amass zinc (Bataineh 2002; Huang 2006; Liang 1999). Supplementation with 15 milligrams of zinc daily showed a trend toward modestly reduced risk of all invasive prostate cancers, but there was a significant 66% reduction in risk of advanced prostate cancer (Gonzalez 2009). This indicates that zinc supplements may be beneficial in some subgroups of men for the most advanced forms of the disease. There was also a greater reduction in prostate cancer risk from zinc supplementation among men whose vegetable intake was high (Gonzalez 2009). Suggested preventive and adjuvant zinc dosages range between 15 and 50 milligrams a day.
Milk Thistle
Evidence demonstrates that the compounds in milk thistle—isosilybin, silibinin, and silymarin—offer protection against prostate cancer. Both silibinin and silymarin are strong antioxidants and inhibit human carcinoma cell growth and DNA synthesis (Zi 1999). Silibinin was found in animal research to exert cancer-fighting effects against an advanced form of human prostate tumor cells, resulting in a decrease in proliferation and an increase in programmed cancer-cell death (Singh 2004a; Singh 2003). Silymarin may block cancer cell development and growth; it was found to contain one or more constituents that induce cancer cell apoptosis and inhibit mitogenic (cell-division promoting) and survival signaling by prostate cancer cells, showing silymarin’s ability to tackle cancer from a number of different angles (Singh 2004b). Both silymarin and silibinin inhibit the secretion of pro-angiogenic factors from tumor cells, which are necessary for these cells to recruit the blood supply required for their continued growth (Singh 2004a).
In animal research, silibinin was found to exert cancer-fighting effects against an advanced form of human prostate tumor cells, resulting in decreased proliferation and increased cancer-cell apoptosis (Singh 2003). Silibinin has high bioavailability in the prostate after oral administration, and scientists concluded that it has strong potential to be developed as an intervention for hormone-refractory (castration-resistant) human prostate cancer (Zi 1999). Silibinin may also work synergistically with the chemotherapy drug doxorubicin to help kill cancer cells, making it a potential candidate for adjuvant therapy (Singh 2004a).
However, isosilybin B—a lesser known constituent that comprises no more than 5% of silymarin and is absent from silibinin—appears to be more potent against prostate cancer cells than the other milk thistle substances (Davis-Searles 2005). Scientists reported that other compounds may require much higher concentrations to achieve the same anti-cancer effect elicited by a relatively small dose of isosilybin B (Davis-Searles 2005). It is important to note that some preparations sold as milk thistle extract, silymarin, or silibinin may contain little, or even no, isosilybin B. A typical suggested dosage of a quality standardized milk thistle extract is 750 milligrams daily, taken with or without food.
Gamma-Linolenic Acid (GLA)
Gamma-linolenic acid (GLA) is an omega-6 essential fatty acid found mostly in plant-based oils. Not all omega-6 fatty acids behave the same: for example, the omega-6s called linoleic acid and arachidonic acid tend to be unhealthy because they promote inflammation; GLA, on the other hand, may serve to reduce inflammation (UMMC 2013a). Much of the GLA taken as a supplement is converted to a substance called DGLA (dihomogamma- linolenic acid), an omega-6 fatty acid with demonstrated anti-inflammatory effect (UMMC 2013a). Similar to the effect of the omega-3 fatty acid eicosapentaenoic acid (EPA), GLA has been found to inhibit the production of urokinase-type plasminogen activator (uPA), a substance believed to play a role in the invasiveness and metastasis of cancer cells (Dias 1995).
Scientists have also found that GLA metabolites suppress the activity of 5alpha-reductase, an enzyme that converts testosterone to a more potent androgen (5alpha-dihydrotestosterone or DHT) and that is involved in the pathway of prostate cancer (Pham 2002). It is believed that GLA may also increase the effectiveness of some anticancer drug treatments (UMMC 2013a). The suggested GLA dosage for prevention is 300 milligrams daily, or for adjuvant therapy, 700-900 milligrams daily, both with food.
Zeaxanthin
Limited evidence suggests that higher zeaxanthin levels may be protective against prostate cancer (Lu 2001). In a 2001 study, a scientific team analyzed the plasma levels of various substances in a group of participants that included 65 patients with prostate cancer and 132 cancer-free controls. They found that, relative to those in the lowest quartile, those in the highest quartile of plasma zeaxanthin had a 78% reduced risk of prostate cancer (Lu 2001). More study is needed to explore this potential benefit. Appropriate zeaxanthin supplementation amounts for prostate cancer defense have not been determined, but 3.75 milligrams daily is a current suggested dosage.
Pomegranate
Use of pomegranate (Punica granatum L. var. spinosa) juice, peel, and oil has been shown to possess anticancer activities, including interference with tumor cell proliferation, cell cycle, invasiveness, and angiogenesis (Lansky 2007). Apoptosis was implicated as a mechanism for this interference with prostate cancer cell proliferation in a laboratory study in which researchers found that pomegranate extract increases expression of a protein that promotes cancer cell death, while decreasing expression of a protein that inhibits cancer cell death (Malik 2005). Later, in a 2012 study, scientists found that the in vitro cytotoxic activity of an extract of pomegranate against prostate cancer cells was dose-dependent—and they also suggested that this antiproliferative effect followed an apoptosis-dependent pathway (Sineh Sepehr 2012).
Further clarifying pomegranate’s effects against prostate cancer cells, scientists found evidence of induced beneficial gene expression—inhibiting proinflammatory, DNA-related protein nuclear factor kappa B (NF-kB) (Heber 2008) and downregulating production of cancer-stimulating androgen receptors in prostate cells (Hong 2008). The suggested dosage for prostate cancer prevention is 80-120 milligrams daily (of punicalagins), and for adjuvant cancer therapy, 280-375 milligrams daily (of punicalagins), with or without food.
Saw Palmetto
Saw palmetto (Serenoa repens or Sabal serrulata) is now one of the most widely used phytotherapies for BPH (benign prostatic hyperplasia) in the US (Gerber 2002; Wilt 2000), a condition characterized by an enlarged prostate gland. However, evidence has been emerging that saw palmetto also has biological activity in prostate cancer cells and may defend against prostate cancer (Yang Y 2007). For instance, a saw palmetto extract was shown to inhibit the activity of 5alpha-reductase (Pais 2010), an enzyme that converts testosterone to the most potent androgen and that is involved in the pathway of prostate cancer. Saw palmetto also appears to have anti-inflammatory properties and—crucially—a tendency to promote apoptosis in prostate cancer cells (Sirab 2013; Hostanska 2007).
In one study, researchers described how they used saw palmetto extract to slow the growth of prostate cancer cells in vitro. This growth-inhibitory effect was more potent on prostate cancer cells than on other cancer cell lines on which they tested saw palmetto (Goldmann 2001). One new mechanism identified by this group of scientists was the saw palmetto-induced reduction in the expression of cyclooxygenase-2 (COX-2) in prostate cancer cells. Cancer cells often use COX-2 as biological fuel to hyperproliferate, and as the researchers presenting this report concluded, “We hypothesize that COX-2 inhibition induced by saw palmetto berry extract may provide an important basis for potential chemopreventative action” (Goldmann 2001). A typical suggested dose of saw palmetto is 320 milligrams daily.
Resveratrol
By working through over a dozen anticancer mechanisms and selectively targeting cancer cells, resveratrol inhibits prostate cancer at multiple stages of development (Jang 1999). This potent compound, found in grapes and other plants, was first isolated in 1940 and is now viewed as a potential defense against this disease (Jang 1999; Jang 1997; Aggarwal 2004). In a study that examined the effect of various polyphenols on different types of prostate cancer cells, scientists concluded that resveratrol was the most potent against advanced prostate cancer cells (Kampa 2000).
Resveratrol has the ability to modulate the activity of estrogen and testosterone at both the cellular (receptor) and molecular (genetic) levels (Lu 1999; Seeni 2008; Mitchell 1999). In fact, after examining its effects on hormone-responsive genes in prostate cancer cells, researchers concluded that, “Resveratrol may be a useful chemopreventive/chemotherapeutic agent for prostate cancer” (Mitchell 1999). Also, resveratrol reverses increases in PSA in cancer cells (Mitchell 1999; Hsieh 2000). For example, in one study, four days of resveratrol treatment resulted in an 80% reduction in PSA levels in prostate cancer cells (Hsieh 2000). Resveratrol also modulates growth factors, protects DNA, blocks cancer-causing chemicals and radiation, and fights free radicals and inflammation (MSKCC 2013a; Dubuisson 2002). The same anticancer gene activated by non-steroidal anti-inflammatory drugs (NSAIDs) demonstrates enhanced expression by resveratrol (Baek 2002).
Using a DNA microarray—a scientific research tool that simultaneously examines how particular phytocompounds affect thousands of genes—scientists found that resveratrol exerts a striking effect on cancer-related genes. Among other things, resveratrol activates tumor suppressor genes, other genes that destroy cancer cells, and genes that control the cell cycle—while suppressing genes that allow cancer cells to communicate with one another (Narayanan 2003). This ability to get inside cancer cells and activate or deactivate genes is a powerful weapon against cancer growth—especially since resveratrol exerts its effects without toxicity (Lu 1999). Many resveratrol supplements on the market are diluted. For pure resveratrol, the suggested dosage is 20-250 milligrams a day, taken with or without food.
Supplemental Lignans
Many different plant sources provide rich sources of lignans—and this may partially explain why men who eat healthier diets enjoy sharply reduced rates of prostate cancer (Miano 2003; Lamblin 2008). Lignan molecules are involved in plant defense mechanisms (Lamblin 2008). But experimental evidence suggests that dietary lignans also offer humans significant protection against tumors in a variety of organs—including tumors of the prostate (Yokota 2007; Bergman Jungeström 2007; Suzuki 2008; McCann 2008). In fact, researchers found that men with higher blood levels of lignans have the lowest incidence of prostate cancer (Hedelin 2006). Bacteria in the intestines convert dietary and supplemental lignans into mammalian lignan compounds called enterolactones, which enter the bloodstream (Heald 2006).
Findings from human, animal, and in vitro studies indicate that enterolactones protect against hormone-dependent cancers (Pietinen 2001; Piller 2006; Wang 2002). Tyrosine kinases are activated in metastatic prostate cancer cells, and enterolactones help to inhibit the tyrosine kinase enzyme (Chen LH 2009). Enterolactones have been shown to inhibit 5alpha-reductase, an enzyme that converts testosterone to a more potent androgen (Evans 1995). Anti-angiogenesis effects and cancer-cell apoptosis were found to be enhanced by enterolactones in animal models of hormone-related cancers, including prostate cancer (Bergman Jungeström 2007; Chen 2007). Enterolactone also functions via several mechanisms to reduce estrogen input to cells and has been shown in a number of studies to be a factor in the development of benign prostate enlargement and prostate cancer (Wang 2002; Takase 2006; Bonkhoff 2005; Yang 2004; Steiner 2003).
A dosage of 20-50 milligrams daily of lignans is suggested to defend against prostate cancer. For adjuvant prostate cancer support, 75-125 milligrams daily is suggested.
Vitamin K
The anti-tumor potential of vitamin K has been a part of scientific research since 1947 (Lamson 2003). Researchers have observed tumor cell destruction in prostate cancer patients following supplementation with a combination of vitamin C and vitamin K3, the synthetic form of vitamin K (Lasalvia-Prisco 2003). (This same combination was later developed into the prostate cancer drug Apatone®, which has shown similar results [Tareen 2008]).
Subsequently, a study that followed 11,319 men for an average of 8.6 years found that those with the highest intake of vitamin K2 were 63% less likely to develop advanced prostate cancer (Nimptsch 2008). The same research team found no effect on prostate cancer from vitamin K1 supplementation. Optimum prostate cancer prevention dosages for vitamin K2 are not known, but typically suggested daily dosages are 1,000 micrograms for the menaquinone-4 form of K2 (MK-4) and 200 micrograms for the menaquinone-7 (MK-7) form.
Beta-Sitosterol
A plant fat and phytosterol known as beta-sitosterol, used in several European prostate drugs, has been found to block the growth of prostate cancer cells. A study on an androgen-dependent line of prostate cancer cells showed that beta-sitosterol decreased cancer cell growth by 24% and increased apoptosis four-fold (von Holtz 1998). These findings correlated with a 50% increase in production of ceramide (von Holtz 1998), an important cell membrane component believed to induce apopotosis (Duan 2005).
In another study, an androgen-dependent line of human prostate cancer cells (PC-3 cell line) was implanted in mice, and scientists compared both the in vivo and in vitro effects of a 2% mixture of beta-sitosterol with those of a 2% mixture of cholesterol on these cells. Compared to controls, beta-sitosterol, as well as another phytosterol known as campesterol, inhibited growth of the prostate cancer cells by 70% and 14%, respectively (Awad 2001). By contrast, the cholesterol mixture increased cell growth by 18%. Various other parameters were also measured.
For example, the phytosterol mixtures inhibited the invasion of the prostate cancer cells into Matrigel-coated membranes—a measure of cancer invasiveness— by 78%, compared to controls, while the cholesterol mixture increased invasiveness by 43% (Awad 2001). Also, migration of the prostate tumor cells through 8-micron pore membranes—a measure of tumor motility—was reduced by 60-93% when they were in the phytosterol mixtures, but it was increased by 67% when in the cholesterol (Awad 2001). In a measure of adhesiveness and ability to form tumor clumps, phytosterol supplementation reduced the binding of these cancer cells to laminin by 15-38% and to fibronectin by 23%, while cholesterol increased cell-binding to type IV collagen by 36% (Awad 2001). The research team concluded that—indirectly in vivo as a dietary supplement, and directly in vitro in tissue culture media—phytosterols inhibited the growth and metastasis of these (PC-3) prostate cancer cells. Beta-sitosterol, however, was determined to be much more effective than campesterol in offering this protection in most parameters assessed (Awad 2001).
In later research on the mechanism involved, scientists determined that phytosterols such as beta-sitosterol may induce the inhibition of tumor growth by stimulating apoptosis and arresting cells at different locations in the cell cycle, and that this may involve alterations in reactive oxygen species and production of prostaglandin (Awad 2005). A suggested phytosterol dosage is 169 milligrams twice daily with or without food.
Apigenin
In studies on human cancer cells, scientists observed that the vegetable extract apigenin inhibits angiogenesis and cell proliferation (Fang 2005; Fang 2007; Luo 2008). These effects were confirmed in an animal experiment in which scientists transplanted an androgen dependent line of human prostate cancer cells into mice bred to serve as a model for tumor growth conditions (Shukla 2005). A liquid suspension containing either apigenin or placebo was given to the mice daily, via a gastric tube, for eight or ten weeks. Administering apigenin to mice—beginning either two weeks before, or two weeks after, inoculation with the cells—inhibited the volume of prostate cancer cells in a dose-dependent manner by as much as 59% and 53%, respectively (Shukla 2005). Induction of apoptosis in the tumor xenografts was observed. In the same study, exposure of prostate cancer cells to apigenin in a culture for as little as 24 hours appeared to inhibit cell cycle progression by nearly 69% (Shukla 2005).
Scientists believe these effects may result from apigenin’s modulation of the IGF (insulin-like growth factors) axis, which plays signaling roles in cell proliferation and cell death (Shukla 2010). Later research demonstrated that apigenin also inhibits motility and invasiveness of prostate carcinoma cells (Franzen 2009). The importance of supplementation for prostate protection is reflected in the fact that Americans typically consume only 13 milligrams of flavonoids (including flavones like apigenin) daily (Shukla 2010); however, a suggested apigenin preventive dosage is 25-50 milligrams daily, and adjuvant dosage for prostate cancer patients may exceed 100 milligrams daily.
Ginger (Zingiber officinale)
A study reported in 2013 demonstrated that ginger phytochemicals work synergistically to inhibit the proliferation of human prostate cancer cells (PC-3 cell line) (Brahmbhatt 2013). In past research, ginger showed anti-inflammatory, antioxidant, and antiproliferative activities, suggesting a promising role as a chemopreventive agent (Shukla 2007; Karna 2012). Then, a 2012 study became the first report to clearly demonstrate the anticancer activity of orally taken, whole ginger extract for the therapeutic management of prostate cancer (Karna 2012). This breakthrough research found that ginger resulted in growth inhibition, cell-cycle arrest, and induced caspase-dependent intrinsic apoptosis in prostate cancer cells (Karna 2012). In vivo studies by this team showed that—without any detectable toxicity—ginger significantly inhibited tumor growth in xenografts of a line of prostate cancer cells (PC3) subcutaneously implanted in nude mice (Karna 2012).
Specifically, the scientific team orally fed a solution containing ginger extract to the tumor-implanted mice for eight weeks. Daily measurements of tumor volume were performed. Tumors in control mice that received a placebo solution showed unrestricted growth. But tumors in mice that received the ginger extract solution showed a time-dependent inhibition of growth over the eight-week period. Remarkably, the tumor burden in the ginger group was reduced by about 56% after just eight weeks of feeding (Karna 2012). Tumor tissue from ginger extract-treated mice showed a reduced proliferation index and “widespread apoptosis” compared with controls (Karna 2012). Ginger treatment was well tolerated, and the test mice maintained normal weight gain and showed no signs of discomfort during the treatment regimen. Most importantly, orally taken ginger extract did not exert any detectable toxicity in normal, rapidly dividing tissues such as the gut and bone marrow.
Although further research is urgently needed, this study suggests that ginger extract has anticancer effects against human prostate cancer cells. No dosage for this purpose has been determined, but the study team performed allometric scaling calculations to extrapolate the mice dosage to humans. The human equivalent dose of ginger extract was found to be approximately 567 milligrams daily for a 154-pound (70 kilogram) human adult (Karna 2012; Reagan-Shaw 2008). This may be viewed as an adjuvant therapy dosage, and an appropriate preventive dosage would be significantly less.
Inositol Hexaphosphate (IP6)
Inositol hexaphosphate, or IP6, is a phytochemical found in cereals, soy, legumes, and other fiber-rich foods (ACS 2013b). Building on earlier in vitro research showing that IP6 strongly inhibits growth and induces differentiation of human prostate cancer cells (PC-3 cells) (Shamsuddin 1995), scientists designed an animal study. They injected mice with a line of human prostate cancer cells (DU145 cells) and then gave them either normal drinking water or water that included 1% or 2% IP6 for 12 weeks. The hormone-refractory (castration-resistant) prostate cancer growth was reduced 47% in the 1% IP6-solution mice and reduced 66% in the 2% IP6-solution mice, compared to littermates without the IP6-enriched drinking water diet (Singh 2004c).
Then, in 2013, scientists designed an IP6 experiment on TRAMP mice, which are genetically modified to develop metastatic prostate cancer (Raina 2013). For 24 weeks, mice with prostate cancer were given drinking water that was 0%, 1%, 2%, or 4% IP6. The study team periodically conducted magnetic resonance imaging (MRI) tests on each mouse prostate to assess prostate volume and tumor vascularity. The animals that received higher concentrations of IP6 showed a “profound” reduction in prostate tumor size, due in part to the compound’s antiangiogenic effect (the ability of the compound to reduce new blood vessel formation) (Raina 2013). The researchers discovered a decrease in a glucose transporter protein, known as GLUT-4, in the prostates of IP6-treated mice, and observed that IP6 decreased glucose metabolism and membrane phospholipid synthesis—meaning there was substantial energy deprivation with the tumor itself. This demonstrates “a practical and translational potential of IP6 treatment in suppressing growth and progression of prostate cancer in humans” (Raina 2013).
N-Acetylcysteine (NAC)
N-acetylcysteine, or NAC, is a metabolite of the amino acid cysteine, which is found in many protein-containing foods (WebMD 2013). It is used both as a prescription drug and a dietary supplement. As a drug, it is given orally to treat acetaminophen overdose; as a supplement, it is used as an antioxidant and to promote metabolism of glutathione, a potent endogenous antioxidant (MSKCC 2013b). Research now indicates it can inhibit growth and block the metastasis of prostate cancer. In an in vitro study, researchers found that NAC significantly inhibited androgen-independent prostate carcinoma cells (PC-3 cells) in a dose- and time-dependent manner—suggesting a potent antiproliferative effect and the promise that NAC may be of benefit in the management of prostate cancer (Lee YJ 2011).
Scientists then conducted another lab study to assess the effect of NAC on the metastasis of human prostate cancer cells. They found that NAC inhibited the growth, migration, and invasion of two cell lines (DU145 and PC3 cells) (Supabphol 2012). Also, NAC significantly reduced the ability of the prostate cancer cells to attach themselves (to collagen IV-coated surfaces) (Supabphol 2012). Inhibition occurred in both cell lines. The team concluded that NAC has high potential to attenuate migration of human prostate cancer cells and to suppress the growth of primary and secondary tumors—and they suggested NAC may represent an affordable and low-toxicity, adjuvant-therapy option for prostate cancer (Supabphol 2012). Dosages of 600 milligrams daily are typical, but higher dosages may be needed for adjuvant cancer therapy.
Quercetin
Quercetin is a flavonoid found in a broad range of fruits and vegetables (Nair 2004). Lab research has suggested that quercetin inhibits prostate cancer development. Scientists found that quercetin produces a 69% reduction in the growth of highly aggressive prostate cancer cells, a greater than 50% upregulation of tumor-suppressor genes, and a 61-100% downregulation of cancer-promoting oncogenes (Nair 2004). A study suggested that quercetin works partially by blocking the androgen receptors used to sustain growth by prostate cancer cells—potentially preventing these cells from forming tumors (Yuan 2010). Another quercetin anticancer mechanism was revealed in a study on human prostate cancer (PC-3) cells. Quercetin induced the mitochondrial apoptotic signaling pathway and endoplasmic reticulum stress, triggering DNA damage and apoptotic death in these cells (Liu 2012). Other research confirmed that quercetin inhibits the migration and invasiveness of prostate cancer cells (Senthilkumar 2011). A suggested preventive dosage is 500 milligrams daily and an adjuvant prostate cancer dosage is 1,000-3,000 milligrams daily. (The lower dosage of 500 milligrams daily is currently being tested in a double-blind, human clinical trial on the effect of quercetin on the rate of increase in PSA and on the incidence of prostate cancer, but these results are not expected to be available until 2014 [Bischoff 2012]).
Reishi
Constituents called triterpenes in the fungus Ganoderma lucidum, better known as reishi mushroom, provide important anti-inflammatory and antiproliferative effects that play a role in cancer (Dudhgaonkar 2009). These mechanisms, combined with the polysaccharides and other components in reishi, can inhibit cancer—including prostate cancer cells (Zaidman 2007; Zaidman 2008). While reishi has been heavily studied for its ability to enhance immunity, some scientists adopted a novel approach to researching potential effects of fungi against prostate cancer. They evaluated the ability of various fungus extracts to act from within the cell to interfere with the androgen receptor and thus, inhibit prostate cancer growth (Zaidman 2007; Zaidman 2008).
These researchers investigated over 200 fungus extracts for their anti-androgenic activity—and of these, G. lucidum (reishi) was one of two mushrooms selected for further investigation (Zaidman 2008). This extract also blocked cell proliferation and decreased cancer cell viability (Zaidman 2008). Reishi inhibited androgen-sensitive, human prostate adenocarcinoma cells (LNCaP cells) (Zaidman 2007). The published report concluded that, “G. lucidum extracts have profound activity against LNCaP cells that merits further investigation as a potential therapeutic agent for the treatment of prostate cancer” (Zaidman 2007). A suggested preventive dosage of reishi extract is 980 milligrams daily (standardized to contain 13.5% polysaccharides and 6% triterpenes). For adjuvant support in prostate cancer, dosages range from 980 up to 3,000 milligrams daily (standardized to contain 13.5% polysaccharides and 6% triterpenes).
Boswellia serrata extract
Aging humans are at increased risk of health complications and mortality via the upregulation of a proinflammatory enzyme called 5-lipoxygenase, or 5-LOX (Chu 2009). The 5-LOX enzyme generates a cascade of dangerous inflammatory effects throughout the body—which results in increased vulnerability of the organs to disease and functional deficits, particularly as the aging process progresses (Chu 2009; Chinnici 2007). This enzyme stimulates the manufacture of pro-inflammatory molecules called leukotrienes, which are linked in abundant research to numerous age-related diseases—including cancer (Chu 2009; Sampson 2009; Goodman 2011; Angelucci 2008; Faronato 2007). Compounds in the flowering plant genus Boswellia—beta-boswellic acid, keto-beta-boswellic acid, and acetyl-keto-beta-boswellic acid (AKBA)—were shown to induce apoptosis in cancer cells (Liu 2002). But a purified extract of Boswellia has been specifically shown to selectively inhibit the 5-LOX enzyme (Safayhi 1997; Safayhi 1995; Lalithakumari 2006).
This purified extract—5-Loxin®—is standardized for AKBA content and protects against inflammatory diseases, including prostate cancer, through several mechanisms. For example, virtually all human cancer cell lines, including prostate cancer cells, induce production of a protein-degrading enzyme called matrix metalloproteinase (MMP), which cancer cells employ to tear apart containment structures within the prostate gland that would normally encase them. This allows the prostate cancer cells to break through healthy prostate tissue and metastasize (Katiyar 2006). However, 5-Loxin® has been shown to prevent expression of MMP—inhibiting the spread of prostate cancer cells.
Prostate cancer cells also use adhesion molecules called VCAM-1 and ICAM-1—which are directly involved in inflammatory processes—to facilitate their spread throughout the body. 5-Loxin® was shown to prevent the upregulation of these adhesion molecules (Lalithakumari 2006). Also, the process of angiogenesis that feeds blood to developing cancer tumors is tightly linked to chronic inflammation (Rajashekhar 2006). A typical suggested dosage of 5-Loxin® is 70-100 milligrams daily with or without food. Individuals with prostate cancer may consider dosages of 170 to 270 milligrams a day of 5-Loxin®.
Watercress Extract
Epidemiological evidence suggests that increased intake of cruciferous vegetables reduces the risk of prostate cancer, prompting scientists to identify the specific compounds responsible for this cancer-preventive effect. They found that a metabolite of phenethyl isothiocyanate (PEITC) that is abundant in watercress inhibits the proliferation of prostate cancer cells and their ability to form tumors (Chiao 2004). And watercress is the richest source of a glucosinolate known as nasturtiin—which is transformed into PEITC in the digestive tract (Palaniswamy 2003).
A delicate balance of estrogens is crucially important for men’s health as well as women’s. In a study that examined the ratio of estrogen metabolites relative to prostate cancer risk, elevated levels of the more active metabolite, 16-hydroxyestrone, were linked with an increased risk of prostate cancer (Muti 2002).
Cruciferous vegetables such as watercress are very rich in the compounds indole-3-carbinol (I3C) and 3,3’-diindolylmethane (DIM), which beneficially modulate estrogen metabolism—correlating with a reduced risk of prostate cancer (Li 2005; Kristal 2002; Heath 2010).
The constituents in watercress were also found to induce phase I and phase II liver enzymes, providing detoxification support that could explain their ability to inhibit the cancer-provoking effects of a variety of chemical compounds (Lhoste 2004). The suggested dosage for watercress extract is 50-100 milligrams daily, taken with or without food.
Grapeseed
Grapeseed extract contains a mixture of phenolic compounds including flavonoids, anthocyanins, and stilbene compounds such as resveratrol (Nassiri-Asl 2009). Emerging research suggests it may be a chemopreventive agent (Kaur 2009; Brasky 2011). Several investigators reported a reduction or delay of prostate tumor incidence when male animals were fed grapeseed extract (Raina 2007). Also, grapeseed proanthocyanidins inhibited human prostate carcinoma cells in lab culture (Vayalil 2004). However, it wasn’t until 2011 that scientists investigated the association of long-term grapeseed supplementation with prostate cancer risk in human males (Brasky 2011).
In a 2011 prostate cancer study of more than 35,000 men aged 50 to 76, researchers found that, compared to non-users, men who supplemented with any amount of grapeseed extract reduced their risk of prostate cancer by 41% (Brasky 2011). However, men with a high 10-year average use of grapeseed supplements experienced a remarkable 62% reduction in prostate cancer risk (Brasky 2011).
Studies on consumption of wine—which contains grapeseed phenols—found no association with prostate cancer risk (Albertsen 2002; Chao 2010; Sutcliffe 2007). Also, two large studies on food-based intake of flavonoids, flavonols, and flavones found no association with prostate cancer risk (Hirvonen 2001; Mursu 2008). Scientists reporting the compelling beneficial results of grapeseed extract supplementation on prostate cancer risk in the 2011 study (above) suggested that, “One explanation for the discrepancy…is that users of grapeseed supplements may be exposed to higher doses of these phenolic compounds than they would from their regular diet” (Brasky 2011). The suggested preventive dosage is 50-100 milligrams daily, and the suggested adjuvant therapeutic dosage is 300 milligrams daily.
Glycyrrhizin
Glycyrrhizin, a triterpene compound isolated from the roots of licorice has been found to exhibit potent in vitro cytotoxic activity against both hormone-dependent (LNCaP), and hormone-independent (DU- 145), lines of human prostate cancer (Thirugnanam 2008). In one study, glycyrrhizin inhibited cell proliferation in these cell lines in a time- and dose-dependent manner (Thirugnanam 2008). The decreased viability was found to be due to apoptosis. Glycyrrhizin also caused DNA damage in these cell lines in a time-dependent manner (Thirugnanam 2008). This suggests that this licorice compound has therapeutic potential against prostate cancer, although a recommended dosage has not been determined.
Modified Citrus Pectin
Pectin is a highly complex, branched polysaccharide fiber that is present in most plants and is particularly abundant in citrus fruits like oranges, lemons, and grapefruit (Niture 2013). Citrus pectin, in its original form, has a limited solubility in water and therefore limited bioavailability to humans (Niture 2013). But in its modified form after hydrolysis, a special formulation of modified citrus pectin becomes a unique water-soluble fiber (Niture 2013; Glinsky 2009). This modified form has been shown to bind to the important galectin molecules on the surface of cells (Glinsky 2009). Scientists believe that this ability of the modified citrus pectin to adhere to molecules—specifically to the galectin-3 molecule—is responsible for its demonstrated ability to inhibit cancer cells (Inohara 1994; Nangia-Makker 2002; Guess 2003). This preventive effect was shown in animal research. For example, oral administration of modified citrus pectin inhibited the spontaneous extraprostatic colonization of injected cells from a prostate cancer cell line and in a dose-dependent fashion (Pienta 1995).
Cancer cells must communicate with one another to invade, colonize, and proliferate in healthy tissue; but this proprietary citrus pectin appears to disrupt this inter-cellular communication, slowing metastasis. The American Cancer Society suggests that modified citrus pectin may “be useful for preventing or slowing the growth of metastatic tumors in very early stages of development” (ACS 2008). For instance, 70% of prostate cancer patients treated orally for 12 months with a modified citrus pectin preparation experienced a slow-down in the rise of prostate-specific antigen, or PSA, concentrations in the blood—without side effects (Guess 2003). A suggested dosage is 5-15 grams daily, taken without food.
Four-Nutrient Supplement – Pomegranate, Broccoli, Green Tea, and Turmeric
As discussed, inhibiting effects against prostate cancer have been reported in published studies for a number of individual nutrients, including pomegranate extract (Malik 2005; Sineh 2012), broccoli compounds (I3C, DIM, and PEITC) (Banerjee 2011; Beaver 2012; Yu 2013), green tea extract (Bettuzzi 2006; McLarty 2009), and curcumin (a key compound in turmeric) (Shishodia, Chaturvedi 2007; Hong 2006; Herman 2009). A recent, double-blind study documented the potency—and possible synergism—of a supplement that combines powders from all four of these food sources (Thomas 2013).
Patients with a PSA relapse after radiotherapy or surgery for localized prostate cancer were randomized to receive capsules of either placebo or the four-nutrient supplement, three times daily. After six months, the median increase in PSA levels in the supplemented group was only 14.7%, while the median PSA increase in the placebo group was 78.5% (Thomas 2013). A striking 46% of the supplemented subjects showed PSA levels that were at or below baseline values, compared to only 14% of the placebo subjects. Among supplemented patients, 92.6% were able to continue on active surveillance, compared to just 74% of the placebo patients (Thomas 2013). There were no statistically significant side effects (Thomas 2013). This identical formula is now commercially available, though it’s likely that many Life Extension® customers have already been taking comparable potencies in supplements that contain these specific nutrients.
What You Need to Know
Prostate Cancer Prevention
- Prostate cancer afflicts one male in every six, and a significant percentage of men have underlying prostate cancer without even knowing it.
- New research reveals the effectiveness of a number of compounds in preventing and inhibiting this disease. We present here a comprehensive arsenal of tools available to prevent, monitor, and attenuate this disease.
- Aging men seeking to live a long and healthy life must be serious about avoiding the development of prostate cancer and serious about reversing its progression.
- These men—and their support network—now have, in one place, the latest scientific information they need to start a broadly effective, multi-action defense program today.
Summary
This section described a huge number of nutrients that have been shown in published scientific studies to help reduce prostate cancer risk.
These nutrients function via multiple mechanisms to inhibit the development and progression of prostate cancer and/or induce cancer cell apoptosis (cell destruction).
The latest research—including a remarkable, controlled clinical trial (Thomas 2013)—reveals the dramatic effectiveness of combining some of these nutrients in men who failed initial treatment for prostate cancer. This is the kind of controlled study that mainstream doctors look to when assessing the efficacy of a particular therapy.
Aging men have an incredible opportunity to reduce their risk of prostate cancer, and while doing so, protect against most other degenerative diseases as well.
Long-time supporters of Life Extension® should appreciate this voluminous data as they have been taking many of these nutrients over a multi-decade time period.
6 New Study Reveals Four Nutrients that Lower PSA and Slow Cancer Progression
Prostate cancer is a leading cause of cancer death among men. Yet, only about 15% of new prostate cancer diagnoses require immediate and aggressive treatment (Bastian 2012; CDC 2013b).
The majority of newly diagnosed prostate cancer cases have low- or intermediate-risk malignancies. For men with low risk malignancies, oncologists sometimes practice “watchful waiting” or “active surveillance,” monitoring parameters such as prostate-specific antigen (PSA) to evaluate tumor progression (Loeb, Berglund 2013; Bul 2012). This approach can delay the need for aggressive treatment, and in many cases is turning out to reduce or eliminate the need for surgery, chemo-, or radiation therapy (Loeb, Berglund 2013).
During this period of watchful waiting, there is an additional option that has been shown to lower PSA. A landmark study from the United Kingdom has demonstrated that a combination of four foods—a fruit (pomegranate), an herb (green tea), a spice (turmeric), and a vegetable (broccoli)—concentrated into a pill, dramatically slowed markers of prostate cancer growth by a median of nearly 64% (Thomas 2013).
Working closely with the National Cancer Research Network, this formula was developed based on extensive documentation showing how certain foods function to slow prostate cancer growth. We begin this report with a critical review of this groundbreaking study conducted on human prostate cancer patients.
Why PSA Matters in Prostate Cancer
Prostate Cancer: Food For Thought
PSA and PSA kinetics are the primary markers to follow disease progress in men with known cancer of the prostate gland (Ilic 2013; Van Neste 2012). But PSA is more than just a marker; we now realize that it is an enzyme that possibly degrades the matrix proteins holding cells together (Pezzato 2004). That is one way the cancer invades and spreads.
A tumor that produces a rapidly rising PSA, therefore, is one at risk for breaking out of the prostate gland itself and spreading either into local tissue or forming distant metastases, both of which place the patient at high risk of death (Benchikh 2008).
That’s why physicians and patients should follow PSA levels carefully once a prostate tumor has been discovered. And that’s why therapies that lower PSA are not just producing an encouraging marker, they are in fact demonstrably slowing disease progression and lowering the patient’s risk of dying.
Landmark UK Study: Food Pill Slows Evidence Of Prostate Cancer Growth
In June of 2013, the American Society of Clinical Oncology included in its program a report on a “food pill” that had a dramatic impact on men with prostate cancer (Thomas 2013). For those who don’t know, the annual conference of the American Society of Clinical Oncology (ASCO) is where many cancer treatment breakthroughs are announced to the world.
The study reported at the ASCO conference was an exploration of the role of four polyphenol-rich foods with known anti-cancer properties (Thomas 2013). The trial development team worked in partnership with the UK government’s National Cancer Research Network, which ensured the highest scientific credibility and quality assurance. They extensively scrutinized the clinical and laboratory data for foods that have a high chance of an anti-cancer effect. They came up with a specific blend of four cancer-fighting foods concentrated into a capsule designed to be taken twice daily. They then set out to test its effect in the most rigorous of scientific trials—a double-blind placebo-controlled randomized trial within which they examined its effect on prostate-specific antigen, or PSA.
The researchers recruited 203 men aged 53 to 89 years (average age 74 years) with prostate cancer proven by biopsy (Thomas 2013). Fifty-nine percent of the men had not yet undergone any treatment and were being followed closely with periodic PSA measurements, while 41% had already had a radical intervention (surgery, chemotherapy, or radiation) but had relapsed with climbing PSA levels.
The subjects were then randomly assigned to receive either a twice-daily oral capsule containing a blend of pomegranate seed, green tea, turmeric, and broccoli, or an identical placebo for 6 months. At baseline, there were no significant differences between the two groups, except that the placebo group was on average 4 years older than the treatment group. Neither the doctors supervising the trial nor the men knew whether they were taking a placebo or the test product.
The men in the study had their PSA levels measured at baseline, at 3 months, and at 6 months, to determine the rate of rise. The results were remarkable.
In the placebo group, PSA levels rose by a median of 78.5% over the 6-month period, while in the supplemented group, PSA rose by a median of only 14.7%, a statistically significant 63.8% difference (Thomas 2013).
In addition, and importantly, 46% of men in the supplemented group had a stable or lower PSA by the end of the study, compared with just 14% of the placebo group; again, this was a significant difference, and suggested that in nearly half of the treated men, their cancers had stopped growing or had even regressed.
In another remarkable measure, just 7.4% of supplemented men being monitored by active surveillance or watchful waiting required a change in management plan, while 26% of those in the placebo group required a change in their management plan (Thomas 2013). In other words, the supplement directly supported the decision to defer care and avoid painful, costly, and invasive procedures in this group of men.
Following the success of this trial, the research team is designing a range of new scientific trials involving this unique fruit and vegetable blend collaborating with academic cancer centers across the world. These include men already taking androgen deprivation therapies, or those in PSA remission following successful primary treatments such as surgery, brachytherapy, or radiotherapy. They are also partnering with clinicians outside the urology cancer field to determine its effect on osteoarthritis, chronic breast pain, hot flushes, and even tinnitus, and hopefully the results of these trials will be available by early 2015.
Let’s now look more closely at each of the ingredients in this new prostate-cancer-fighting food pill, to see what each one brings uniquely to the formula and how each reinforces the other to reduce the risk of prostate cancer progression.
What You Need to Know
Functional Foods as Powerful Combatants Against Prostate Cancer
- Prostate cancer becomes a killer when it invades or metastasizes; prior to those events it can be detected and successfully treated.
- But too many men with prostate cancer undergo needless and invasive surgery, chemo-, or radiation therapy.
- A new pill containing concentrated forms of four functional foods has now been shown to significantly slow the rise of PSA, the major marker of prostate cancer progression.
- The components, pomegranate, green tea, turmeric, and broccoli, have all independently been shown to have protective effects on prostate tissue; a new study demonstrates that they can work together in concert to slow the disease in men who already have prostate cancer.
- Each food component works by different, but powerful, epigenetic mechanisms to modify the way prostate cells regulate their growth and development.
- Using all four in one simple pill optimizes both prevention and treatment of prostate cancer, without significant side effects.
Pomegranate
Pomegranate compounds suppress enzymes in the intestine and liver that convert certain molecules (procarcinogens) into cancer-causing agents (Faria 2007; Saruwatari 2008). As it relates to those with prostate cancer, the active constituents in pomegranate have proven to be potent inducers of malignant cell death through apoptosis (Vicinanza 2013; Sineh Sepehr 2012; Lee ST 2012; Gasmi 2010; Adhami 2009; Pantuck 2006; Malik 2006; Malik 2005; Albrecht 2004).
During the development of androgen independence, prostate cancer cells are known to increase testosterone synthesis inside their own cells, which maintains cancer cell growth in the absence of significant amounts of circulating testosterone. Overexpression of the androgen receptor occurs in androgen-independent prostate cancer and has been proposed as another mechanism promoting the development of androgen independence. Pomegranate has been shown to inhibit expression of the androgen receptor and androgen synthesizing genes in prostate cells, which helps block an important survival mechanism utilized by prostate cancer cells to escape eradication (Hong 2008).
Multiple basic laboratory and human studies have demonstrated that pomegranate treatment, specifically various active compounds, slows PSA doubling time and reduces production of PSA in malignant prostate cells (Adhami 2009; Malik 2006; Paller 2013; Seeram 2007).
In one recent study, pomegranate juice treatment in men with rising PSA after surgery or radiotherapy resulted in a significant delay in PSA doubling time (the time it takes PSA levels to rise) from a mean of 15 months before treatment to 54 months following supplementation (Malik 2006). Another study found a more modest, but still significant delay in doubling time, from 11.9 months to 18.8 months (Paller 2013).
Animal studies demonstrate additional anti-cancer activity in pomegranates. In a specialized mouse model of prostate cancer, 100% of untreated mice developed palpable tumors within 20 weeks, compared to as low as 20% in the group treated with pomegranate extract; the treated animals lived for up to a median of 92 weeks, more than twice as long as the 43 weeks survived by untreated mice (Adhami 2012).
These remarkable results are observed in part because naturally occurring pomegranate polyphenols are concentrated in prostate tissue, facilitating their protective effects (Seeram 2007). Once in the prostate, these polyphenols selectively inhibit cancer cell proliferation, leaving healthy prostate tissue relatively unaffected (Sineh Sepehr 2012; Hong 2008). This is a potential “epigenetic” effect: pomegranate polyphenols decrease the expression of proteins that cancer cells use to support their rapid rate of replication (Gasmi 2010; Malik 2005; Albrecht 2004; Selvi 2010).
Added prostate cancer-fighting benefits of pomegranate include reduction of the inflammation that drives cancer progression, suppression of new blood vessel growth within a forming prostate tumor, and increased expression of genes that keep cells clumped together normally, thereby inhibiting the invasive potential of prostate cancer (Pantuck 2006; Sartippour 2008; Wang 2012; Pitchakarn 2013).
Green Tea
Green tea makes a unique contribution to the prostate-cancer-fighting pill as a result of a special combination of naturally occurring polyphenols called catechins (Connors 2012; McCarthy 2007; O’Sullivan 2008; Pezzato 2004).
Studies show that one of green tea’s catechins, EGCG, accumulates specifically in prostate tissue, where it selectively kills cancer cells (leaving healthy cells unaffected) and reduces serum PSA levels (Henning 2006; Bettuzzi 2006; Pandey 2009; McLarty 2009; Siddiqui 2006; Chuu 2009).
In a further demonstration of the cancer-suppressing role of green tea, when researchers studied men with a pre-cancerous condition called prostate intraepithelial neoplasia, they found only one tumor after one year in the 30 men given green tea polyphenols, while the 30 placebo recipients developed nine cancers (Bettuzzi 2006). The treatment was safe, and as an extra bonus was found to reduce other lower urinary tract symptoms as well.
Green tea is already acknowledged as a cancer preventive in Japan because of epidemiological studies documenting prostate cancer risk reduction of up to 86% in men who drink the most green tea (Pandey 2009; Fujiki 2012; Jian 2007; Kurahashi 2008).
Laboratory studies point to still other anti-cancer effects from green tea. Its components reduce genetic expression and activity of androgen receptors that most prostate cancers need to survive (Chuu 2009; Harper 2007; Siddiqui IA 2011; Lee YH 2012). Green tea also induces human cancer cell death by apoptosis through a variety of epigenetic mechanisms (Pitchakarn 2013; Gupta 2012; Hagen 2013). And recent studies reveal polyphenols in brewed green tea shut off new blood vessel growth, important in slowing cancer development (McCarthy 2007).
Turmeric
Turmeric’s unique contribution to the prostate-cancer-fighting pill is its extraordinary anti-inflammatory properties, provided chiefly by its natural primary component, curcumin (Dorai 2001; Gupta SC 2013). Reducing inflammation with curcumin reduces the metastases that ultimately kill prostate cancer patients (Rao 2012; Cheng 2013; Killian 2012; Sundram 2012). Curcumin also down-regulates genes involved in adhesion, motility, and invasiveness that prostate cancer cells need to invade and spread (Herman 2009).
Curcumin specifically inhibits prostate cancer cell production of PSA by blocking its genetic expression (Yang 2005; Chung 2011). At the same time, it also reduces activation of the androgen receptors on cancer cells that trigger increased production of PSA (Tsui 2008; Choi 2010).
But the whole turmeric root also contains important oils and other substances that enhance curcumin’s absorption and have health benefits of their own, including anti-cancer actions (Aggarwal 2013).
Turmeric’s components also inhibit cancer cell proliferation, restore cancer cells’ ability to die normally by apoptosis, and decrease the density of blood vessels needed for tumor expansion (Dorai 2001). By modulating cell signaling mechanisms, curcumin arrests the out-of-control cell replication cycle typical in cancer (Teiten 2011; Guo 2013).
Curcumin also sensitizes cancer cells to chemo- and radiation therapy, as well as to the intrinsic “death factor” called TRAIL (TNF-related apoptosis-inducing ligand), one of the body’s natural cancer-suppressing mechanisms (Shankar 2008; Chendil 2004; Goel 2010). Remarkably, these sensitizing effects are not found on normal, healthy cells, so they remain protected during treatment (Goel 2010).
The compound has also been found to block growth factors and androgen receptors used by cancer cells to support themselves (Hung 2012; Shah 2012; Teiten 2012).
Broccoli
Broccoli’s unique contribution to the prostate-cancer-fighting pill is its ability to up-regulate phase II detoxifying enzymes in gut and liver tissue, enabling the body to render harmless thousands of potentially carcinogenic molecules in our diet (Clarke 2011; Rogan 2006; Joseph 2004; Abdul Razis 2013). In addition, the naturally occurring sulfur-rich broccoli constituents sulforaphane, indole-3-carbinol (I3C), and others have now been identified as potent epigenetic regulators (Clarke 2011; Dashwood 2007; Myzak 2007; Myzak 2006).
These broccoli compounds control enzymes called histone deacetylases (HDAC) that regulate the genes encoded in DNA—including those responsible for promoting or suppressing cancer formation (Clarke 2011; Dashwood 2007; Myzak 2007; Myzak 2006). Known collectively as histone deacetylase (HDAC) inhibitors, such molecules are prime objectives of Big Pharma (Gryder 2013; Stettner 2012).
Men with high consumption of broccoli and other cruciferous vegetables have a 40% lower risk of invasive prostate cancer (Kirsh 2007). And in animal studies, broccoli feeding reduced prostate tumor weight by 42% in prostate cancer-prone mice and suppressed growth of implanted human prostate cancer cells by 40% (Myzak 2007; Canene-Adams 2007).
Broccoli compounds reduce PSA production as a result of slowing prostate cancer cell replication in laboratory cell culture models (Zhang 2003; Chiao 2002; Han 2007). They appear to inhibit expression of the androgen receptors that prostate cancer needs to survive (Chiao 2002).
Broccoli’s other prostate cancer-fighting properties include inhibition of growth and transcription factors that are activated in malignancies, restoration of normal tumor suppressor genes, and increased production of apoptosis-inducing proteins (Traka 2010; Melchini 2013; Hahm 2010; Choi 2005; Ho 2009; Traka 2008).
Summary
Prostate cancer is a paradox: Its typically slow growth rate makes it possible to treat if discovered early, but once it has metastasized, it is often lethal.
The combination of four widely-recognized cancer-fighting foods, pomegranate, green tea, turmeric, and broccoli, in a single twice-daily pill has now been shown to significantly reduce the rate of rise of PSA, the tumor marker that indicates progression and invasion of prostate cancer (Thomas 2013).
This new pill appears to work by providing cancer-suppressing actions at a wide variety of targets. All of this pill’s components have the capacity to cause favorable epigenetic changes, reversing the gene damage that leads to cancer development and restoring normal cancer suppression mechanisms (Gasmi 2010; Malik 2005; Albrecht 2004; Connors 2012; Gupta 2012; Hagen 2013; Herman 2009; Clarke 2011; Dashwood 2007; Myzak 2007; Myzak 2006).
In a tightly controlled clinical trial, putting them together in a single pill was shown to be effective at slowing the growth of existing prostate cancers and preventing surgical and other side effect-prone procedures.
If you or someone you know suffers from prostate cancer, or is interested in preventing it, this new functional food pill, or its individual constituents, should be part of their daily program.
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
Abdull Razis AF, Noor NM. Cruciferous vegetables: dietary phytochemicals for cancer prevention. Asian Pac J Cancer Prev. 2013;14(3):1565-70.
Abedin M, Tintut Y, Demer LL. Vascular calcification: mechanisms and clinical ramifications. Arterioscler Thromb Vasc Biol. 2004 Jul;24(7):1161-70.
Abrahamsson PA, Lilja H, Oesterling JE. Molecular forms of serum prostate-specific antigen. The clinical value of percent free prostate-specific antigen. Urol Clin North Am. 1997 May;24(2):353-65.
ACS. American Cancer Society. Imaging (Radiology) Tests. Nuclear Scans. Available at: http://www.cancer.org/treatment/understandingyourdiagnosis/examsandtestdescriptions/imagingradiologytests/imaging-radiology-tests-nuc-scan. Last updated 8/9/2013a. Accessed 10/25/2013.
ACS. American Cancer Society. Inositol Hexaphosphate. Find Support & Treatment page. Available at: http://www.cancer.org/treatment/treatmentsandsideeffects/complementaryandalternativemedicine/dietandnutrition/inositol-hexaphosphate. Last updated 8/5/2013b. Accessed 10/21/2013.
ACS. American Cancer Society. Learn About Cancer page. Prostate Cancer. Early Detection, Diagnosis, and Staging Topics. How is prostate cancer diagnosed? http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-diagnosis. Last updated 8/26/2013c. Accessed 10/24/2013.
ACS. American Cancer Society. Modified Citrus Pectin. Find Support & Treatment page. Available at: http://www.cancer.org/treatment/treatmentsandsideeffects/complementaryandalternativemedicine/dietandnutrition/modified-citrus-pectin. Last updated 11/1/2008. Accessed 10/21/2013.
ACS. American Cancer Society. Prostate Cancer. What are the key statistics about prostate cancer? Available at: http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-key-statistics. Last updated 8/26/2013d. Accessed 10/28/2013.
ACS. American Cancer Society. Prostate Cancer. What are the key statistics about prostate cancer? Available at: http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-key-statistics. Last updated 8/26/2013e. Accessed 10/18/2013.
Adan Y, Shibata K, Sato M, Ikeda I, Imaizumi K. Effects of docosahexaenoic and eicosapentaenoic acid on lipid metabolism, eicosanoid production, platelet aggregation and atherosclerosis in hypercholesterolemic rats. Biosci Biotechnol Biochem. 1999 Jan;63(1):111-9.
Adhami VM, Khan N, Mukhtar H. Cancer chemoprevention by pomegranate: laboratory and clinical evidence. Nutr Cancer. 2009;61(6):811-5.
Adhami VM, Siddiqui IA, Syed DN, Lall RK, Mukhtar H. Oral infusion of pomegranate fruit extract inhibits prostate carcinogenesis in the TRAMP model. Carcinogenesis. 2012 Mar;33(3):644-51.
Agarwal PK, Oefelein MG. Testosterone replacement therapy after primary treatment for prostate cancer. J Urol. 2005 Feb;173(2):533-6.
Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: preclinical and clinical studies. Anticancer Res. 2004 Sep- Oct;24(5A):2783-840.
Aggarwal BB, Sundaram C, Malani N, Ichikawa H. Curcumin: the Indian solid gold. Adv Exp Med Biol. 2007;595:1-75.
Aggarwal BB, Yuan W, Li S, Gupta SC. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: Identification of novel components of turmeric. Mol Nutr Food Res. 2013 Jul 12.
Ahmed S, Rahman A, Hasnain A, et al. Green tea polyphenol epigallocatechin-3-gallate inhibits the IL-1 beta-induced activity and expression of cyclooxygenase-2 and nitric oxide synthase-2 in human chondrocytes. Free Radic Biol Med. 2002 Oct 15;33(8):1097-105.
AHRQ. Agency for Healthcare Research and Quality. Screening and Risk Assessment for Osteoporosis in Postmenopausal Women. Comparison of Recommendations: Measurement of BMD: Modality and Frequency. Available at: http://www.medscape.com/viewarticle/738733_5. 3/16/2011. Accessed 10/25/2013.
Albertsen K, Gronbaek M. Does amount or type of alcohol influence the risk of prostate cancer? Prostate. 2002;52:297-304.
Albrecht M, Jiang W, Kumi-Diaka J, et al. Pomegranate extracts potently suppress proliferation, xenograft growth, and invasion of human prostate cancer cells. J Med Food. 2004 Fall;7(3):274-83.
Allard CB, Dason S, Lusis J, Kapoor A. Prostate cancer screening: Attitudes and practices of family physicians in Ontario. Can Urol Assoc J. 2012 6(3):188-93.
Ammon HP. Boswellic acids (components of frankincense) as the active principle in treatment of chronic inflammatory diseases. Wien Med Wochenschr. 2002 152(15-16):373-8.
Anderson KM, Seed T, Vos M et al. 5-Lipoxygenase inhibitors reduce PC-3 cell proliferation and initiate nonnecrotic cell death. Prostate. 1998 Nov 1;37(3):161-73.
Andriole G, Bruchovsky N, Chung LW, et al. Dihydrotestosterone and the prostate: the scientific rationale for 5alpha-reductase inhibitors in the treatment of benign prostatic hyperplasia. J Urol. 2004 Oct;172(4 Pt 1):1399-403.
Andriole GL, Bostwick DG, Brawley OW, et al; REDUCE Study Group. Effect of dutasteride on the risk of prostate cancer. N Engl J Med. 2010 Apr 1;362(13):1192-202.
Andriole GL, Marberger M, Roehrborn CG. Clinical usefulness of serum prostate specific antigen for the detection of prostate cancer is preserved in men receiving the dual 5alpha-reductase inhibitor dutasteride. J Urol. 2006 May;175(5):1657-62.
Angelucci A, Garofalo S, Speca S, et al. Arachidonic acid modulates the crosstalk between prostate carcinoma and bone stromal cells. Endocr Relat Cancer. 2008 Mar;15(1):91-100.
Anisimov VN, Berstein LM, Egormin PA, et al. Effect of metformin on life span and on the development of spontaneous mammary tumors in HER-2/neu transgenic mice. Exp Gerontol. 2005 Aug-Sep;40(8-9):685-93.
Arena F. Specific antigen prostatic changes during treatment with finasteride or dutasteride for benign prostatic hyperplasia. Minerva Urol Nefrol. 2013 Sep;65(3):211-216.
Arnaldez FI, Helman LJ. Targeting the insulin growth factor receptor 1. Hematol Oncol Clin North Am. 2012 Jun;26(3):527- 42,vii-viii.
AUA. American Urological Association. AUA Releases New Clinical Guideline on Prostate Cancer Screening. Available at: http://www.auanet.org/advnews/press_releases/article.cfm?articleNo=290. 5/3/2013. Accessed 10/28/2013.
Aune D, De Stefani E, Ronco AL, et al. Egg consumption and the risk of cancer: a multisite case-control study in Uruguay. Asian Pac J Cancer Prev. 2009;10(5):869-76.
Avci CB, Harman E, Dodurga Y, Susluer SY, Gunduz C. Therapeutic potential of an anti-diabetic drug, metformin: alteration of miRNA expression in prostate cancer cells. Asian Pac J Cancer Prev. 2013;14(2):765-8.
Awad AB, Burr AT, Fink CS. Effect of resveratrol and beta-sitosterol in combination on reactive oxygen species and prostaglandin release by PC-3 cells. Prostaglandins Leukot Essent Fatty Acids. 2005 Mar;72(3):219-26.
Awad AB, Fink CS, Williams H, Kim U. In vitro and in vivo (SCID mice) effects of phytosterols on the growth and dissemination of human prostate cancer PC-3 cells. Eur J Cancer Prev. 2001 Dec;10(6):507-13.
Baek SJ, Wilson LC, Eling TE. Resveratrol enhances the expression of nonsteroidal anti-inflammatory drug-activated gene (NAG-1) by increasing the expression of p53. Carcinogenesis. 2002 Mar;23(3):425-34.
Banerjee S, Kong D, Wang Z, et al. ATTENUATION OF MULTI-TARGETED PROLIFERATION-LINKED SIGNALING BY 3,3’-DIINDOLYLMETHANE (DIM: FROM BENCH TO CLINIC. Mutat Res. 2011;728(0):47-66.
Barham JB, Edens MB, Fonteh AN, et al. Addition of eicosapentaenoic acid to gamma-linolenic acid-supplemented diets prevents serum arachidonic acid accumulation in humans. J Nutr. 2000 Aug;130(8):1925-31.
Barranco WT, Eckhert CD. Boric acid inhibits human prostate cancer cell proliferation. Cancer Lett. 2004 Dec 8;216(1):21-9.
Barve A, Khor TO, Nair S, et al. Gamma-tocopherol-enriched mixed tocopherol diet inhibits prostate carcinogenesis in TRAMP mice. Int J Cancer. 2009 Apr 1;124(7):1693-9.
Bastian PJ, Boorjian SA, Bossi A, et al. High-risk prostate cancer: from definition to contemporary management. Eur Urol. 2012 Jun;61(6):1096-106.
Bataineh ZM, Hani IH Bani, Al-Alami JR. Zinc in normal and pathological human prostate gland. Saudi Med Jour. 2002;23(2):218-20.
Beaver LM, Yu TW, Sokolowski EI, Williams DE, Dashwood RH, Ho E. 3,3’-Diindolylmethane, but not indole-3-carbinol, inhibits histone deacetylase activity in prostate cancer cells. Toxicol Appl Pharmacol. 2012 Sep 15;263(3):345-51.
Bemis DL, Capodice JL, Anastasiadis AG, Katz AE, Buttyan R. Zyflamend, a unique herbal preparation with nonselective COX inhibitory activity, induces apoptosis of prostate cancer cells that lack COX-2 expression. Nutr Cancer. 2005 52(2):202-12.
Ben Sahra I, Laurent K, Loubat A, et al. The antidiabetic drug metformin exerts an antitumoral effect in vitro and in vivo through a decrease of cyclin D1 level. Oncogene. 2008 Jun 5;27(25):3576-86.
Ben Sahra I, Regazzetti C, Robert G, et al. Metformin, independent of AMPK, induces mTOR inhibition and cell-cycle arrest through REDD1. Cancer Res. 2011 Jul 1;71(13):4366-72.
Benchikh El Fegoun A, Villers A, Moreau JL, Richaud P, Rebillard X, Beuzeboc P. PSA and follow-up after treatment of prostate cancer. Prog Urol. 2008 Mar;18(3):137-44.
Bengmark S. Curcumin, an atoxic antioxidant and natural NFkappaB, cyclooxygenase-2, lipooxygenase, and inducible nitric oxide synthase inhibitor: a shield against acute and chronic diseases. JPEN J Parenter Enteral Nutr. 2006 Jan;30(1):45-51.
Berglund RK, Masterson TA, Vora KC, Eggener SE, Eastham JA, Guillonneau BD. Pathological upgrading and up staging with immediate repeat biopsy in patients eligible for active surveillance. J Urol. 2008 Nov;180(5):1964-7.
Bergman Jungeström M, Thompson LU, Dabrosin C. Flaxseed and its lignans inhibit estradiol-induced growth, angiogenesis, and secretion of vascular endothelial growth factor in human breast cancer xenografts in vivo. Clin Cancer Res. 2007 Feb 1;13(3):1061-7.
Berquin IM, Min Y, Wu R, et al. Modulation of prostate cancer genetic risk by omega-3 and omega-6 fatty acids. J Clin Invest. 2007 Jul;117(7):1866-75.
Bettuzzi S, Brausi M, Rizzi F, Castagnetti G, Peracchia G, Corti A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: a preliminary report from a one-year proof-of-principle study. Cancer Res. 2006 Jan 15;66(2):1234-40.
Beulens JW, Bots ML, Atsma F, et al. High dietary menaquinone intake is associated with reduced coronary calcification. Atherosclerosis. 2009 Apr;203(2):489-93.
Bidoli E, Talamini R, Bosetti C, et al. Macronutrients, fatty acids, cholesterol and prostate cancer risk. Ann Oncol. 2005 Jan;16(1):152-7.
Billis A. Latent carcinoma and atypical lesions of prostate. An autopsy study. Urology. 1986 Oct;28(4):324-9.
Biri H, Ozturk HS, Kacmaz M, et al. Activities of DNA turnover and free radical metabolizing enzymes in cancerous human prostate tissue. Cancer Invest. 1999;17(5):314-9.
Bischoff SC. University of Hohenheim. Clinical Trial on the Effectiveness of the Flavonoids Genistein and Quercetin in Men With Rising Prostate-specific Antigen. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2012 [cited 2013 Oct 21]. Available at: http://clinicaltrials.gov/show/NCT01538316. NLM Identifier: NCT01538316.
Bohnet HG, Hanker JP, Horowski R, Wickings EJ, Schneider HP. Suppression of prolactin secretion by lisuride throughout the menstrual cycle and in hyperprolactinaemic menstrual disorders. Acta Endocrinol (Copenh). 1979 Sep;92(1):8-19.
Bokhorst LP, Bangma CH, van Leenders GJ, et al. Prostate-specific antigen-based prostate cancer screening: Reduction of prostate cancer mortality after correction for nonattendance and contamination in the Rotterdam Section of the European Randomized Study of Screening for Prostate Cancer. Eur Urol. Aug 11 2013.
Bolotin HH. Inaccuracies inherent in dual-energy X-ray absorptiometry in vivo bone mineral densitometry may flaw osteopenic/osteoporotic interpretations and mislead assessment of antiresorptive therapy effectiveness. Bone. 2001;28:548–55.
Bonkhoff H, Fixemer T. Implications of estrogens and their receptors for the development and progression of prostate cancer. Pathologe. 2005 Nov;26(6):461-8.
Bonvissuto G, Minutoli L, Morgia G, et al. Effect of Serenoa repens, lycopene, and selenium on proinflammatory phenotype activation: an in vitro and in vivo comparison study. Urology. 2011 Jan;77(1):248.e9-16.
Bosland MC. Sex steroids and prostate carcinogenesis: integrated, multifactorial working hypothesis. Ann NY Acad Sci. 2006 Nov;1089:168-76.
Bostwick DG, Qian J, Civantos F, Roehrborn CG, Montironi R. Does finasteride alter the pathology of the prostate and cancer grading? Clin Prostate Cancer. 2004 Mar;2(4):228-35.
Brahmbhatt M, Gundala SR, Asif G, Shamsi SA, Aneja R. Ginger phytochemicals exhibit synergy to inhibit prostate cancer cell proliferation. Nutr Cancer. 2013 Feb;65(2):263-72.
Brasky TM, Kristal AR, Navarro SL, et al. Specialty supplements and prostate cancer risk in the VITamins and Lifestyle (VITAL) cohort. Nutr Cancer. 2011;63(4):573-82.
Brooks JD, Metter EJ, Chan DW, et al. Plasma selenium level before diagnosis and the risk of prostate cancer development. J Urol. 2001 Dec;166(6):2034-8.
Bul M, van den Bergh RC, Zhu X, et al. Outcomes of initially expectantly managed patients with low or intermediate risk screen-detected localized prostate cancer. BJU Int. 2012 Dec;110(11):1672-7.
Calder PC. N-3 polyunsaturated fatty acids and inflammation: from molecular biology to the clinic. Lipids. 2003 Apr;38(4):343-52.
Canene-Adams K, Lindshield BL, Wang S, Jeffery EH, Clinton SK, Erdman JW, Jr. Combinations of tomato and broccoli enhance antitumor activity in dunning r3327-h prostate adenocarcinomas. Cancer Res. 2007 Jan 15;67(2):836-43.
Cantrell LA, Zhou C, Mendivil A, Malloy KM, Gehrig PA, Bae-Jump VL. Metformin is a potent inhibitor of endometrial cancer cell proliferation--implications for a novel treatment strategy. Gynecol Oncol. 2010 Jan;116(1):92-8.
Capodice JL, Gorroochurn P, Cammack AS, et al. Zyflamend in men with high-grade prostatic intraepithelial neoplasia: results of a phase I clinical trial. J Soc Integr Oncol. 2009 7(2):43-51.
Carlson RH. Prostate Cancer, Diabetes Link Points to Potential Treatment with Metformin. Oncology Times web page. Available at: http://journals.lww.com/oncology-times/blog/onlinefirst/pages/post.aspx?Postid=451. 4/22/2012. Accessed 10/29/2013.
Carruba G. Estrogen and prostate cancer: an eclipsed truth in an androgen-dominated scenario. J Cell Biochem. 2007 Nov 1;102(4):899-911.
Carter HB, Partin AW, Walsh PC, et al. Gleason score 6 adenocarcinoma: should it be labeled as cancer? J Clin Oncol. 2012 Dec 10;30(35):4294-6.
Catalona WJ, Richie JP, Ahmann FR, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men. J Urol. 1994 May;151(5):1283-90.
Catella-Lawson F. Vascular biology of thrombosis: platelet-vessel wall interactions and aspirin effects. Neurology. 2001 57(5 Suppl 2):S5-S7.
Cavalieri E, Rogan E. Catechol quinones of estrogens in the initiation of breast, prostate, and other human cancers: keynote lecture. Ann NY Acad Sci. 2006 Nov;1089:286-301.
CDC. Centers for Disease Control and Prevention. HIV in the United States: At A Glance. Available at: http://www.cdc.gov/hiv/statistics/basics/ataglance.html. Last updated 4/23/2013a. Accessed 10/28/2013.
CDC. Centers for Disease Control and Prevention. Prostate Cancer Statistics. Available at: http://www.cdc.gov/cancer/prostate/statistics/. Last updated 10/23/2013b. Accessed 12/10/2013.
Chan JM, Stampfer MJ, Ma J, Gann PH, Gaziano JM, Giovannucci E. Dairy products, calcium, and prostate cancer risk in the Physicians’ Health Study. Am J Clin Nutr. 2001 74(4):549-554.
Chan R, Lok K, Woo J. Prostate cancer and vegetable consumption. Mol Nutr Food Res. 2009 Feb;53(2):201-16.
Chao C, Haque R, Van Den Eeden SK, Caan BJ, Poon KY, et al. Red wine consumption and risk of prostate cancer: the California men’s health study. Int J Cancer. 2010;126:171-9.
Chaudhary LR, Hruska KA. Inhibition of cell survival signal protein kinase B/Akt by curcumin in human prostate cancer cells. J Cell Biochem. 2003;89(1):1-5.
Chen L, Davey Smith G, Evans DM, et al. Genetic variants in the vitamin d receptor are associated with advanced prostate cancer at diagnosis: findings from the prostate testing for cancer and treatment study and a systematic review. Cancer Epidemiol Biomarkers Prev. 2009 Nov;18(11):2874-81.
Chen LH, Fang J, Li H, Demark-Wahnefried W, Lin X. Enterolactone induces apoptosis in human prostate carcinoma LNCaP cells via a mitochondrial-mediated, caspase-dependent pathway. Mol Cancer Ther. 2007 Sep;6(9):2581-90.
Chen LH, Fang J, Sun Z, Li H, Wu Y, Demark-Wahnefried W, Lin X. Enterolactone inhibits insulin-like growth factor-1 receptor signaling in human prostatic carcinoma PC-3 cells. J Nutr. 2009 Apr;139(4):653-9.
Chen Y, Zaman MS, Deng G, et al. MicroRNAs 221/222 and genistein-mediated regulation of ARHI tumor suppressor gene in prostate cancer. Cancer Prev Res (Phila). 2011 Jan;4(1):76-86.
Chendil D, Ranga RS, Meigooni D, Sathishkumar S, Ahmed MM. Curcumin confers radiosensitizing effect in prostate cancer cell line PC-3. Oncogene. 2004 Feb 26;23(8):1599-607.
Cheng TS, Chen WC, Lin YY, et al. Curcumin-targeting pericellular serine protease matriptase role in suppression of prostate cancer cell invasion, tumor growth, and metastasis. Cancer Prev Res (Phila). 2013 May;6(5):495-505.
Cheng Y, Austin SC, Rocca B, et al. Role of prostacyclin in the cardiovascular response to thromboxane A2. Science. 2002 Apr 19;296(5567):539-41.
Chiao JW, Chung FL, Kancherla R, Ahmed T, Mittelman A, Conaway CC. Sulforaphane and its metabolite mediate growth arrest and apoptosis in human prostate cancer cells. Int J Oncol. 2002 Mar;20(3):631-6.
Chiao JW, Wu H, Ramaswamy G, et al. Ingestion of an isothiocyanate metabolite from cruciferous vegetables inhibits growth of human prostate cancer cell xenografts by apoptosis and cell cycle arrest. Carcinogenesis. 2004 Aug;25(8):1403-8.
Chinni SR, Li Y, Upadhyay S, Koppolu PK, Sarkar FH. Indole- 3-carbinol (I3C) induced cell growth inhibition, G1 cell cycle arrest and apoptosis in prostate cancer cells. Oncogene. 2001 May 24;20(23):2927-36.
Chinnici CM, Yao Y, Pratico D. The 5-lipoxygenase enzymatic pathway in the mouse brain: young versus old. Neurobiol Aging. 2007 Sep;28(9):1457-62.
Choi HY, Lim JE, Hong JH. Curcumin interrupts the interaction between the androgen receptor and Wnt/beta-catenin signaling pathway in LNCaP prostate cancer cells. Prostate Cancer Prostatic Dis. 2010 Dec;13(4):343-9.
Choi S, Singh SV. Bax and Bak are required for apoptosis induction by sulforaphane, a cruciferous vegetable-derived cancer chemopreventive agent. Cancer Res. 2005 Mar 1;65(5):2035-43.
Choi YK, Park KG. Metabolic roles of AMPK and metformin in cancer cells. Mol Cells. 2013 Jun 19.
Christen S, Woodall AA, Shigenaga MK, Southwell-Keely PT, Duncan MW, Ames BN. Gamma-tocopherol traps mutagenic electrophiles such as NO(X) and complements alpha-tocopherol: physiological implications. Proc Natl Acad Sci U S A. 1997 Apr 1;94(7):3217-22.
Chu J, Pratico D. The 5-lipoxygenase as a common pathway for pathological brain and vascular aging. Cardiovasc Psychiatry Neurol. 2009;2009:174657.
Chung LC, Tsui KH, Feng TH, Lee SL, Chang PL, Juang HH. Curcumin provides potential protection against the activation of hypoxia and prolyl 4-hydroxylase inhibitors on prostate-specific antigen expression in human prostate carcinoma cells. Mol Nutr Food Res. 2011 Nov;55(11):1666-76.
Chuu CP, Chen RY, Kokontis JM, Hiipakka RA, Liao S. Suppression of androgen receptor signaling and prostate specific antigen expression by (-)-epigallocatechin- 3-gallate in different progression stages of LNCaP prostate cancer cells. Cancer Lett. 2009 Mar 8;275(1):86-92.
Clark LC, Combs GF, Jr., Turnbull BW. et al. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. Nutritional Prevention of Cancer Study Group. JAMA. 1996;276:1957-63.
Clark LC, Dalkin B, Krongrad A, et al. Decreased incidence of prostate cancer with selenium supplementation: results of a double- blind cancer prevention trial. Br J Urol. 1998 May;81(5):730-4.
Clark RV, Hermann DJ, Cunningham GR, Wilson TH, Morrill BB, Hobbs S. Marked suppression of dihydrotestosterone in men with benign prostatic hyperplasia by dutasteride, a dual 5alpha-reductase inhibitor. J Clin Endocrinol Metab. 2004 May;89(5):2179-84.
Clarke JD, Dashwood RH, Ho E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008 Oct 8;269(2):291-304.
Clarke JD, Hsu A, Yu Z, Dashwood RH, Ho E. Differential effects of sulforaphane on histone deacetylases, cell cycle arrest and apoptosis in normal prostate cells versus hyperplastic and cancerous prostate cells. Mol Nutr Food Res. 2011 Jul;55(7):999-1009.
Clements A, Gao B, Yeap SH, Wong MK, Ali SS, Gurney H. Metformin in prostate cancer: two for the price of one. Ann Oncol. 2011 Dec;22(12):2556-60.
Cleveland Clinic. Heart and Vascular Health & Prevention. Cleveland Clinic web page. Available at: http://my.clevelandclinic.org/heart/prevention/nutrition/omega3.aspx. 9/2009. Accessed 10/21/2013.
Cohen YC, Liu KS, Heyden NL, et al. Detection bias due to the effect of finasteride on prostate volume: a modeling approach for analysis of the Prostate Cancer Prevention Trial. J Natl Cancer Inst. 2007 Sep 19;99(18):1366-74.
Connors SK, Chornokur G, Kumar NB. New insights into the mechanisms of green tea catechins in the chemoprevention of prostate cancer. Nutr Cancer. 2012;64(1):4-22.
Cooney RV, Franke AA, Harwood PJ, Hatch-Pigott V, Custer LJ, Mordan LJ. γ-Tocopherol detoxification of nitrogen dioxide: Superiority to α-tocopherol. Proc Natl Acad Sci USA. 1993;90(5):1771– 1775.
Cox ME, Gleave ME, Zakikhani M, et al. Insulin receptor expression by human prostate cancers. Prostate. 2009 Jan 1;69(1):33-40.
Cui Y, Winton MI, Zhang ZF, et al. Dietary boron intake and prostate cancer risk. Oncol Rep. 2004 Apr;11(4):887-92.
Cumming AP, et al. Psa Affects Prostate Cancer Cell invasion in Vitro and Induces an Osteoblastic Phenotype in Bone in Vivo. Prostate Cancer and Prostatic Diseases. 2011;14(4):286-94. Print.
Currie CJ, Poole CD, Gale EA. The influence of glucose-lowering therapies on cancer risk in type 2 diabetes. Diabetologia. 2009 Sep;52(9):1766-77.
D’Amico A, Chen M, Roehl K, Catalona W. Preoperative PSA velocity and the risk of death from prostate cancer after radical prostatectomy. N Engl J Med. 2004;351(2):125-35.
Dagvadorj A, Collins S, Jomain JB, et al. Autocrine prolactin promotes prostate cancer cell growth via Janus kinase-2-signal transducer and activator of transcription- 5a/b signaling pathway. Endocrinology. 2007 Jul;148(7):3089-101.
Dandona P, Rosenberg MT. A practical guide to male hypogonadism in the primary care setting. Int J Clin Pract. 2010 May;64(6):682-96.
Dashwood RH, Ho E. Dietary histone deacetylase inhibitors: from cells to mice to man. Semin Cancer Biol. 2007 Oct;17 (5):363-9.
Dassesse T, de Leval X, de Leval L, Pirotte B, Castronovo V, Waltregny D. Activation of the thromboxane A2 pathway in human prostate cancer correlates with tumor Gleason score and pathologic stage. Eur Urol. 2006 Nov;50(5):1021-31.
Davis JN, Kucuk O, Sarkar FH. Genistein inhibits NF-kappa B activation in prostate cancer cells. Nutr Cancer. 1999 35(2):167-74.
Davis JN, Singh B, Bhuiyan M, Sarkar FH. Genistein-induced upregulation of p21WAF1, downregulation of cyclin B, and induction of apoptosis in prostate cancer cells. Nutr Cancer. 1998 32(3):123-31.
Davis-Searles PR, Nakanishi Y, Kim NC, et al. Milk thistle and prostate cancer: differential effects of pure flavonolignans from Silybum marianum on antiproliferative end points in human prostate carcinoma cells. Cancer Res. 2005 May 15;65(10):4448-57.
Day JR, Jost M, Reynolds MA, Groskopf J, Rittenhouse H. PCA3: from basic molecular science to the clinical lab. Cancer Lett. 2011 Feb 1;301(1):1-6.
de Leval X, Hanson J, David JL, et al. New developments on thromboxane and prostacyclin modulators part II: prostacyclin modulators. Curr Med Chem. 2004 May;11(10):1243-52.
Deckelbaum RJ, Worgall TS, Seo T. n-3 fatty acids and gene expression. Am J Clin Nutr. 2006 Jun;83(6 Suppl):1520S-1525S.
Demark-Wahnefried W, Polascik TJ, et al. Flaxseed supplementation (not dietary fat restriction) reduces prostate cancer proliferation rates in men presurgery. Cancer Epidemiol Biomarkers Prev. 2008 Dec;17(12):3577-87.
Demark-Wahnefried W, Robertson CN, Walther PJ, Polascik TJ, Paulson DF, Vollmer RT. Pilot study to explore effects of low-fat, flaxseed-supplemented diet on proliferation of benign prostatic epithelium and prostate-specific antigen. Urology. 2004 May;63(5):900-4.
Demer LL and Abedin M. Skeleton key to vascular disease. J Am Coll Cardiol. 2004;44(10):1977-1979.
Demer LL and Tintut Y. Osteopontin: Between a Rock and Hard Plaque. Circulation Research. 1999;84:250-252.
Dias VC, Parsons HG. Modulation in delta 9, delta 6, and delta 5 fatty acid desaturase activity in the human intestinal CaCo-2 cell line. J Lipid Res. 1995 Mar;36(3):552-63.
Ding XZ, Iversen P, Cluck MW, Knezetic JA, Adrian TE. Lipoxygenase inhibitors abolish proliferation of human pancreatic cancer cells. Biochem Biophys Res Commun. 1999 Jul 22;261(1):218-23.
Donaldson MS. Nutrition and cancer: a review of the evidence for an anti-cancer diet. Nutr J. 2004 Oct 20;3:19.
Dong X, Xu W, Sikes RA, Wu C. Combination of low dose of genistein and daidzein has synergistic preventive effects on isogenic human prostate cancer cells when compared with individual soy isoflavone. Food Chem. 2013 Dec 1;141(3):1923-33.
Dorai T, Cao YC, Dorai B, Buttyan R, Katz AE. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate. 2001;47(4):293-303.
Dowling RJ, Goodwin PJ, Stambolic V. Understanding the benefit of metformin use in cancer treatment. BMC Med. 2011 Apr 6;9:33.
Drugs.com [Internet]. Cabergoline. Copyright © 2013 Wolters Kluwer Health, Inc. Available at: http://www.drugs.com/cdi/cabergoline.html. 10/2/2013. Accessed 10/29/2013.
du Toit PJ, van Aswegen CH, du Plessis DJ. The effect of essential fatty acids on growth and urokinase-type plasminogen activator production in human prostate DU-145 cells. Prostaglandins Leukot Essent Fatty Acids. 1996;55:173-7.
Duan RD. Anticancer compounds and sphingolipid metabolism in the colon. In Vivo. 2005 Jan-Feb;19(1):293-300.
Dubuisson JG, Dyess DL, Gaubatz JW. Resveratrol modulates human mammary epithelial cell O-acetyltransferase, sulfo transferase, and kinase activation of the het- erocyclic amine carcinogen N-hydroxy-PhIP. Cancer Lett. 2002 Aug 8;182(1):27-32.
Dudhgaonkar S, Thyagarajan A, Sliva D. Suppression of the inflammatory response by triterpenes isolated from the mushroom Ganoderma lucidum. Int Immunopharmacol. 2009 Oct;9(11):1272-80.
Duffield-Lillico, AJ, Reid, ME, Turnbull, BW, et al. Baseline characteristics and the effect of selenium supplementation on cancer incidence in a randomized clinical trial: a summary report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiol. Biomarkers Prev. 2002;11:630-9.
Dunlap SM, Chiao LJ, Nogueira L, et al. Dietary energy balance modulates epithelial-to-mesenchymal transition and tumor progression in murine claudin-low and basal-like mammary tumor models. Cancer Prev Res (Phila). 2012 Jul;5(7):930-42.
Egan K, FitzGerald GA. Eicosanoids and the vascular endothelium. Handb Exp Pharmacol. 2006 176 (Pt 1):189-211.
Eichholzer M, Steinbrecher A, Kaaks R, et al. Effects of selenium status, dietary glucosinolate intake and serum glutathione s-transferase a activity on the risk of benign prostatic hyperplasia. BJU Int. 2012 Dec;110(11 Pt C):E879-85.
El-Bayoumy K. The negative results of the SELECT study do not necessarily discredit the selenium-cancer prevention hypothesis. Nutr Cancer. 2009;61(3):285-6.
Ellem SJ, Risbridger GP. Aromatase and prostate cancer. Minerva Endocrinol. 2006 Mar;31(1):1-12.
El-Sayed WM, Aboul-Fadl T, Lamb JG, Roberts JC, Franklin MR. Effect of selenium-containing compounds on hepatic chemoprotective enzymes in mice. Toxicology. 2006 Mar 15;220(2-3):179-88.
Erdelyi I, Levenkova N, Lin EY, et al. Western-style diets induce oxidative stress and dysregulate immune responses in the colon in a mouse model of sporadic colon cancer. J Nutr. 2009 Nov;139(11):2072-8.
Ersoy C, Kiyici S, Budak F, et al. The effect of metformin treatment on VEGF and PAI-1 levels in obese type 2 diabetic patients. Diabetes Res Clin Pract. 2008 Jul;81(1):56-60.
Ervin RB. Prevalence of metabolic syndrome among adults 20 years of age and over, by sex, age, race and ethnicity, and body mass index: United States, 2003- 2006. Natl Health Stat Report. 2009 May 5;(13):1-7.
Evans BA, Griffiths K, Morton MS. Inhibition of 5 alpha-reductase in genital skin fibroblasts and prostate tissue by dietary lignans and isoflavonoids. J Endocrinol. 1995 Nov;147(2):295-302.
Evans JM, Donnelly LA, Emslie-Smith AM, Alessi DR, Morris AD. Metformin and reduced risk of cancer in diabetic patients. BMJ. 2005 Jun 4;330(7503):1304-5
Faloon W. Another Victory Against FDA Censorship. Life Extension Magazine. July 2012.
Faloon W. Protection Against Arterial Calcification, Bone Loss, Cancer, and Aging! Life Extension Magazine. January 2009.
Fang J, Xia C, Cao Z, Zheng JZ, Reed E, Jiang BH. Apigenin inhibits VEGF and HIF-1 expression via PI3K/AKT/p70S6K1 and HDM2/p53 pathways. FASEB J. 2005 Mar;19(3):342-53.
Fang J, Zhou Q, Liu LZ, et al. Apigenin inhibits tumor angiogenesis through decreasing HIF-1alpha and VEGF expression. Carcinogenesis. 2007 Apr;28(4):858-64.
Farhan H, Wahala K, Adlercreutz H, et al. Isoflavonoids inhibit catabolism of vitamin D in prostate cancer cells. J Chromatogr B Analyt Technol Biomed Life Sci. 2002 Sep 25;777(1-2):261-8.
Faria A, Monteiro R, Azevedo I, Calhau C. Pomegranate juice effects on cytochrome P450S expression: in vivo studies. J Med Food. 2007 Dec;10(4):643-9.
Faronato M, Muzzonigro G, Milanese G, et al. Increased expression of 5-lipoxygenase is common in clear cell renal cell carcinoma. Histol Histopathol. 2007 Oct;22(10):1109-18.
FDA. U.S. Food and Drug Administration. Drugs. 5-Alpha Reductase Inhibitor Information. Available at: http://www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/ucm258424.htm. Last updated 4/11/2013. Accessed 10/24/2013.
FDA. U.S. Food and Drug Administration. Drugs. FDA Drug Safety Communication: 5-alpha reductase inhibitors (5-ARIs) may increase the risk of a more serious form of prostate cancer. Available at: http://www.fda.gov/drugs/drugsafety/ucm258314.htm. Last updated 7/20/2012. Accessed 10/24/2013.
Feldman M, Cryer B, Rushin K, Betancourt J. A comparison of every-third-day versus daily low-dose aspirin therapy on serum thromboxane concentrations in healthy men and women. Clin Appl Thromb Hemost. 2001 Jan;7(1):53-7.
Ferretti JL. Peripheral Quantitative Computed Tomography (pQCT) For Evaluating Structural and Mechanical Properties of Small Bone. In: Practical Guide for Mechanical Testing of Bone (Ch. 25).” CRC Press, Boca Raton, FL; 1999.
Ferris-Tortajada J, Berbel-Tornero O, Garcia-Castell J, Ortega-Garcia JA, Lopez-Andreu JA. Dietetic factors associated with prostate cancer: protective effects of Mediterranean diet. Actas urologicas espanolas. 2012 Apr;36(4):239-245.
Flanagan JN, Young MV, Persons KS, et al. Vitamin D metabolism in human prostate cells: implications for prostate cancer chemoprevention by vitamin D. Anticancer Res. 2006,Jul-Aug;26(4A):2567-72.
Fleischer AC. Sonographic depiction of tumor vascularity and flow: from in vivo models to clinical applications. J Ultrasound Med. 2000 Jan;19(1):55-61.
Fleshner N and Joshua A. University Health Network, Toronto. A Randomized, Double-Blind, Placebo-Controlled Trial of Metformin in Reducing Progression Among Men on Expectant Management for Lower Risk Prostate Cancer: The MAST (Metformin Active Surveillance Trial) Study. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2013 [cited 2013 Oct 29]. Available at: http://www.clinicaltrials.gov/ct2/show/NCT01864096. NLM Identifier: NCT01864096.
Fleshner NE, Lucia MS, Egerdie B, et al. Dutasteride in localised prostate cancer management: the REDEEM randomised, double-blind, placebo-controlled trial. Lancet. 2012 Mar 24;379(9821):1103-11.
Fleshner, N., Fair, W.R., Huryk, R. et al. Vitamin E inhibits the high-fat diet promoted growth of established human prostate LNCaP tumors in nude mice. J Urol. 1999;161:1651-4.
Fodor D, Albu A, Poantă L, Porojan M. Vitamin K and vascular calcifications. Acta Physiol Hung. 2010 Sep;97(3):256-66.
Folkers K, Brown R, Judy WV, Morita M. Survival of cancer patients on therapy with coenzyme Q10. Biochem Biophys Res Commun. 1993 Apr 15;192(1):241-5.
Folkers K, Osterborg A, Nylander M, Morita M, Mellstedt H. Activities of vitamin Q10 in animal models and a serious deficiency in patients with cancer. Biochem Biophys Res Commun. 1997 May 19;234(2):296-9.
Folkers K. Relevance of the biosynthesis of coenzyme Q10 and of the four bases of DNA as a rationale for the molecular causes of cancer and a therapy. Biochem Biophys Res Commun. 1996 Jul 16;224(2):358-61.
Fong AT, Swanson HI, Dashwood RH, Williams DE, Hendricks JD, Bailey GS. Mechanisms of anti-carcinogenesis by indole-3-carbinol. Studies of enzyme induction, eletrophile-scavenging, and inhibition of aflatoxin B1 activation. Biochem Pharmacol. 1990 Jan 1;39(1):19-26.
Franzen CA, Amargo E, Todorović V, et al. The chemopreventive bioflavonoid apigenin inhibits prostate cancer cell motility through the focal adhesion kinase/Src signaling mechanism. Cancer Prev Res (Phila Pa). 2009 Sep;2(9):830-41.
Freedland SJ, Aronson WJ. Dietary intervention strategies to modulate prostate cancer risk and prognosis. Curr Opin Urol. 2009 May;19(3):263-7.
Fujiki H, Suganuma M. Green tea: an effective synergist with anticancer drugs for tertiary cancer prevention. Cancer Lett. 2012 Nov 28;324(2):119-25.
Gallardo-Williams MT, Chapin RE, King PE, et al. Boron supplementation inhibits the growth and local expression of IGF-1 in human prostate adenocarcinoma (LNCaP) tumors in nude mice. Toxicol Pathol. 2004 Jan-Feb;32(1):73-8.
Galli F, Stabile AM, Betti M, et al. The effect of alpha- and gamma- tocopherol and their carboxyethyl hydroxychroman metabolites on prostate cancer cell proliferation. Arch Biochem Biophys. 2004 Mar 1;423(1):97-102.
Gann PH, Ma J, Giovannucci E, et al. Lower prostate cancer risk in men with elevated plasma lycopene levels: results of a prospective analysis. Cancer Res. 1999;59:1225-30.
Gao X, LaValley MP, Tucker KL. Prospective studies of dairy product and calcium intakes and prostate cancer risk: a meta-analysis. J Natl Cancer Inst. 2005 Dec 7;97(23):1768-77.
Garcia Rodriguez LA. The effect of NSAIDs on the risk of coronary heart disease: fusion of clinical pharmacology and pharmacoepidemiologic data. Clin Exp Rheumatol. 2001 Nov;19(6 Suppl 25):S41-4.
Garikapaty VP, Ashok BT, Chen YG, et al. Anti-carcinogenic and anti-metastatic properties of indole-3-carbinol in prostate cancer. Oncol Rep. 2005 Jan;13(1):89-93.
Garland CF, Comstock GW, Garland FC, et al. Serum 25-hydroxyvitamin D and colon cancer: eight-year prospective study. Lancet. 1989 Nov 18;2(8673):1176-8.
Garland CF, Garland FC, Gorham ED. Can colon cancer incidence and death rates be reduced with calcium and vitamin D? Am J Clin Nutr. 1991 Jul;54(1 Suppl):193S-201S.
Garland CF, Gorham ED, Mohr SB, et al. Vitamin D and prevention of breast cancer: pooled analysis. J Steroid Biochem Mol Biol. 2007 Mar;103(3-5):708-11.
Gasmi J, Sanderson JT. Growth inhibitory, antiandrogenic, and pro-apoptotic effects of punicic acid in LNCaP human prostate cancer cells. J Agric Food Chem. 2010 Nov 10.
Genomic Health. Genomic Health Announces Availability of Oncotype DX® Prostate Cancer Test with Potential to Triple the Number of Patients Who Can Confidently Choose Active Surveillance and Avoid Side Effects of Invasive Treatments. Available at: http://files.shareholder.com/downloads/GHDX/2097290524x0x661617/85f38a0e-ccd5-47de-a348-8d0c2b5c03bd/GHDX_News_2013_5_8_General.pdf. 5/8/2013. Accessed 10/29/2013.
Gerber GS. Phytotherapy for benign prostatic hyperplasia. Curr Urol Rep. 2002 Aug;3(4):285-91.
Gerhardt H, Seifert F, Buvari P, Vogelsang H, Repges R. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol. 2001 Jan;39(1):11-7.
Gharaee-Kermani M, Macoska JA. Promising molecular targets and biomarkers for male BPH and LUTS. Curr Urol Rep. 2013 Aug 3.
Ghosh J, Myers CE. Arachidonic acid stimulates prostate cancer cell growth: critical role of 5-lipoxygenase. Biochem Biophys Res Commun. 1997 Jun 18;235(2):418-23.
Ghosh J, Myers CE. Inhibition of arachidonate 5-lipoxygenase triggers massive apoptosis in human prostate cancer cells. Proc Natl Acad Sci U S A. 1998 Oct 27;95(22):13182-7.
Ghosh J. Inhibition of arachidonate 5-lipoxygenase triggers prostate cancer cell death through rapid activation of c-Jun N-terminal kinase. Biochem Biophys Res Commun. 2003 Jul 25;307(2):342-9.
Ghossain A, Ghossain MA. History of mastectomy before and after Halsted. J Med Liban. 2009 Apr-Jun;57(2):65-71.
Gion M, Mione R, Barioli P, et al. Percent free prostate-specific antigen in assessing the probability of prostate cancer under optimal analytical conditions. Clin Chem. 1998 Dec;44(12):2462-70.
Giovannucci E, Ascherio A, Rimm EB, et al. Intake of carotenoids and retinol in relation to risk of prostate cancer. J Natl Cancer Inst. 1995;87:1767-76.
Giton F, de la Taille A, Allory Y, et al. Estrone sulfate (E1S), a prognosis marker for tumor aggressiveness in prostate cancer (PCa). J Steroid Biochem Mol Biol. 2008 Mar;109(1-2):158-67.
Glinsky VV, Raz A. Modified citrus pectin anti-metastatic properties: one bullet, multiple targets. Carbohydr Res. 2009 Sep 28;344(14):1788-91.
Goel A, Aggarwal BB. Curcumin, the golden spice from Indian saffron, is a chemosensitizer and radiosensitizer for tumors and chemoprotector and radioprotector for normal organs. Nutr Cancer. 2010;62(7):919-30.
Gofrit ON, Zorn KC, Taxy JB, et al. Predicting the risk of patients with biopsy Gleason score 6 to harbor a higher grade cancer. J Urol. 2007 Nov;178(5):1925-8.
Goldmann WH, Sharma AL, Currier SJ, Johnston PD, Rana A, Sharma CP. Saw palmetto berry extract inhibits cell growth and Cox-2 expression in prostatic cancer cells. Cell Biol Int. 2001;25(11):1117-24.
Gomez Y, Arocha F, Espinoza F, Fernandez D, Vasquez A, Granadillo V. Zinc levels in prostatic fluid of patients with prostate pathologies. Invest Clin. 2007 Sep;48(3):287-94.
Gonzalez A, Peters U, Lampe JW, White E. Zinc intake from supplements and diet and prostate cancer. Nutr Cancer. 2009;61(2):206-15.
Goodman LA, Jarrett CL, Krunkosky TM, et al. 5-Lipoxygenase expression in benign and malignant canine prostate tissues. Vet Comp Oncol. 2011 Jun;9(2):149-57.
Goodman PJ, Tangen CM, Darke AK, et al. Long-Term Effects of Finasteride on Prostate Cancer Mortality. New England Journal of Medicine. 2019;380(4):393-394.
Goodwin JS, Goodwin JM. The tomato effect. Rejection of highly efficacious therapies. JAMA. 1984 May 11;251(18):2387-90.
Gooren L. Androgen deficiency in the aging male: benefits and risks of androgen supplementation. J Steroid Biochem Mol Biol. 2003 Jun;85(2-5):349-55.
Gorham ED, Garland CF, Garland FC, et al. Vitamin D and prevention of colorectal cancer. J Steroid Biochem Mol Biol. 2005 Oct;97(1-2):179-94.
Gotlieb WH, Saumet J, Beauchamp MC, Gu J, Lau S, Pollak MN, Bruchim I. In vitro metformin anti-neoplastic activity in epithelial ovarian cancer. Gynecol Oncol. 2008 Aug;110(2):246-50.
Grossmann ME, Huang H, Tindall DJ. Androgen receptor signaling in androgen-refractory prostate cancer. J Natl Cancer Inst. 2001 Nov 21;93(22):1687-97.
Gryder BE, Rood MK, Johnson KA, et al. Histone deacetylase inhibitors equipped with estrogen receptor modulation activity. J Med Chem. 2013 Jul 3.
Guess BW, Scholz MC, Strum SB, Lam RY, Johnson HJ, Jennrich RI. Modified citrus pectin (MCP) increases the prostate-specific antigen doubling time in men with prostate cancer: a phase II pilot study. Prostate Cancer Prostatic Dis. 2003;6(4):301-4.
Gukovsky I, Reyes CN, Vaquero EC, Gukovskaya AS, Pandol SJ. Curcumin ameliorates ethanol and nonethanol experimental pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2003 Jan;284(1):G85-95.
Guo H, Xu YM, Ye ZQ, Yu JH, Hu XY. Curcumin induces cell cycle arrest and apoptosis of prostate cancer cells by regulating the expression of IkappaBalpha, c-Jun and androgen receptor. Pharmazie. 2013 Jun;68(6):431-4.
Gupta I, Gupta V, Parihar A, et al. Effects of Boswellia serrata gum resin in patients with bronchial asthma: results of a doubleblind, placebo-controlled, 6-week clinical study. Eur J Med Res. 1998 Nov 17;3(11):511-4.
Gupta I, Parihar A, Malhotra P, et al. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med. 2001 Jul;67(5):391-5.
Gupta K, Thakur VS, Bhaskaran N, et al. Green tea polyphenols induce p53-dependent and p53-independent apoptosis in prostate cancer cells through two distinct mechanisms. PLoS One. 2012;7(12):e52572.
Gupta RT, Kauffman CR, Polascik TJ, Taneja SS, Rosenkrantz AB. The state of prostate MRI in 2013. Oncology (Williston Park). 2013 Apr;27(4):262-70.
Gupta S, Chiplunkar S, Kim C, Yel L, Gollapudi S. Effect of age on molecular signaling of TNF-alpha-induced apoptosis in human lymphocytes. Mech Ageing Dev. 2003 Apr;124(4):503-9.
Gupta S, Srivastava M, Ahmad N, et al. Lipoxygenase-5 is overexpressed in prostate adenocarcinoma. Cancer. 2001 Feb 15;91(4):737-43.
Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013 Jan;15(1):195-218.
Gysin R, Azzi A, Visarius T. Gamma-tocopherol inhibits human cancer cell cycle progression and cell proliferation by down-regulation of cyclins. FASEB J. 2002 Dec;16(14):1952-4.
Haber D. Roads leading to breast cancer. N Engl J Med. 2000 Nov 23;343(21):1566-8.
Hagen RM, Chedea VS, Mintoff CP, Bowler E, Morse HR, Ladomery MR. Epigallocatechin-3-gallate promotes apoptosis and expression of the caspase 9a splice variant in PC3 prostate cancer cells. Int J Oncol. 2013 Jul;43(1):194-200.
Häggström S, Bergh A, Damber JE. Vascular endothelial growth factor content in metastasizing and nonmetastasizing Dunning prostatic adenocarcinoma. Prostate. 2000 Sep 15;45(1):42-50.
Hahm ER, Singh SV. Sulforaphane inhibits constitutive and interleukin-6-induced activation of signal transducer and activator of transcription 3 in prostate cancer cells. Cancer Prev Res (Phila). 2010 Apr;3(4):484-94.
Hamalainen M, Nieminen R, Asmawi MZ, Vuorela P, Vapaatalo H, Moilanen E. Effects of flavonoids on prostaglandin E2 production and on COX-2 and mPGES-1 expressions in activated macrophages. Planta Med. 2011 Sep;77(13):1504-11.
Han HY, Shan S, Zhang X, Wang NL, Lu XP, Yao XS. Down-regulation of prostate specific antigen in LNCaP cells by flavonoids from the pollen of Brassica napus L. Phytomedicine. 2007 May;14(5):338-43.
Handel LN, Agarwal S, Schiff SF, Kelty PJ, Cohen SI. Can effect of finasteride on prostate-specific antigen be used to decrease repeat prostate biopsy? Urology. 2006 Dec;68(6):1220-3.
Harper CE, Patel BB, Wang J, Eltoum IA, Lamartiniere CA. Epigallocatechin-3-Gallate suppresses early stage, but not late stage prostate cancer in TRAMP mice: mechanisms of action. Prostate. 2007 Oct 1;67(14):1576-89.
Harris RE. Cyclooxygenase-2 (cox-2) blockade in the chemoprevention of cancers of the colon, breast, prostate, and lung. Inflammopharmacology. 2009 Apr;17(2):55-67.
Harvei S. Epidemiology of prostatic cancer. Tidsskr Nor Laegeforen 1999 Oct 10;119(24):3589-94.
Haseltine F, Paulsen C, Wang C, eds. Reproductive Issues and the Aging Male. Washington, D.C: American Association for the Advancement of Science; 1993:15-34.
Hassan S, Carraway RE. Involvement of arachidonic acid metabolism and EGF receptor in neurotensin-induced prostate cancer PC3 cell growth. Regul Pept. 2006 Jan 15;133(1-3):105-14.
Hazai E, Bikadi Z, Zsila F, Lockwood SF. Molecular modeling of the non-covalent binding of the dietary tomato carotenoids lycopene and lycophyll, and selected oxidative metabolites with 5-lipoxygenase. Bioorg Med Chem. 2006 Oct 15;14(20):6859-67.
Heald CL, Bolton-Smith C, Ritchie MR, Morton MS, Alexander FE. Phyto-oestrogen intake in Scottish men: use of serum to validate a self-administered food-frequency questionnaire in older men. Eur J Clin Nutr. 2006 Jan;60(1):129-35.
Heald CL, Ritchie MR, Bolton-Smith C, Morton MS, Alexander FE. Phyto-oestrogens and risk of prostate cancer in Scottish men. Br J Nutr. 2007 Aug;98(2):388-96.
Heath EI, Heilbrun LK, Li J, et al. A phase I dose-escalation study of oral BR-DIM (BioResponse 3,3’- Diindolylmethane) in castrateresistant, non-metastatic prostate cancer. Am J Transl Res. 2010 Jul 23;2(4):402-11.
Heber D. Multitargeted therapy of cancer by ellagitannins. Cancer Lett. 2008 Oct 8;269(2):262-8.
Hedelin M, Klint A, Chang ET, et al. Dietary phytoestrogen, serum enterolactone and risk of prostate cancer: the cancer prostate Sweden study (Sweden). Cancer Causes Control. 2006 Mar;17(2):169-80.
Helzlsouer KJ, Huang, HY, Alberg AJ, et al. Association between alpha-tocopherol, gamma-tocopherol, selenium, and subsequent prostate cancer. J Natl Cancer Inst. 2000;92(24):2018-23.
Henderson BE, Bernstein L, Ross RK, Depue RH, Judd HL. The early in utero oestrogen and testosterone environment of blacks and whites: potential effects on male offspring. Br J Cancer. 1988 Feb;57(2):216-8.
Henderson K, Stella SL, Kobylewski S, Eckhert CD. Receptor activated Ca(2+) release is inhibited by boric acid in prostate cancer cells. PLoS One. 2009;4(6):e6009.
Hennig R, Ding XZ, Tong WG, et al. 5-Lipoxygenase and leukotriene B(4) receptor are expressed in human pancreatic cancers but not in pancreatic ducts in normal tissue. Am J Pathol. 2002 Aug;161(2):421-8.
Henning SM, Aronson W, Niu Y, et al. Tea polyphenols and theaflavins are present in prostate tissue of humans and mice after green and black tea consumption. J Nutr. 2006 Jul;136(7):1839-43.
Herman JG, Stadelman HL, Roselli CE. Curcumin blocks CCL2- induced adhesion, motility and invasion, in part, through downregulation of CCL2 expression and proteolytic activity. Int J Oncol. 2009 May;34(5):1319-27.
Herschman JD, Smith DS, Catalona WJ. Effect of ejaculation on serum total and free prostate-specific antigen concentrations. Urology. 1997 Aug;50(2):239-43.
Hessels D, Schalken JA. The use of PCA3 in the diagnosis of prostate cancer. Nat Rev Urol. 2009 May;6(5):255-61.
Hirsch HA, Iliopoulos D, Tsichlis PN, Struhl K. Metformin selectively targets cancer stem cells, and acts together with chemotherapy to block tumor growth and prolong remission. Cancer Res. 2009 Oct 1;69(19):7507-11.
Hirvonen T, Virtamo J, Korhonen P, Albanes D, Pietinen P. Flavonol and flavone intake and the risk of cancer in male smokers (Finland). Cancer Causes Control. 2001;12:789-96.
Hitron A, Adams V, Talbert J, Steinke D. The influence of antidiabetic medications on the development and progression of prostate cancer. Cancer Epidemiol. 2012 Aug;36(4):e243-50.
Ho E, Clarke JD, Dashwood RH. Dietary sulforaphane, a histone deacetylase inhibitor for cancer prevention. J Nutr. 2009 Dec;139(12):2393-6.
Ho SM, Tang WY, Belmonte de Frausto J, Prins GS. Developmental exposure to estradiol and bisphenol A increases susceptibility to prostate carcinogenesis and epigenetically regulates phosphodiesterase type 4 variant 4. Cancer Res. 2006 Jun 1;66(11):5624-32.
Hodges S, Hertz N, Lockwood K, Lister R. CoQ10: could it have a role in cancer management? Biofactors. 1999;9(2-4):365-70.
Holick MF. Vitamin D deficiency. N Engl J Med. 2007 Jul 19;357(3):266-81.
Hong J, Bose M, Ju J, et al. Modulation of arachidonic acid metabolism by curcumin and related beta-diketone derivatives: effects on cytosolic phospholipase A(2), cyclooxygenases and 5-lipoxygenase. Carcinogenesis. 2004 Sep;25(9):1671-9.
Hong J, Smith TJ, Ho CT, August DA, Yang CS. Effects of purified green and black tea polyphenols on cyclooxygenase- and lipoxygenase-dependent metabolism of arachidonic acid in human colon mucosa and colon tumor tissues. Biochem Pharmacol. 2001 Nov 1;62(9):1175-83.
Hong JH, Ahn KS, Bae E, Jeon SS, Choi HY. The effects of curcumin on the invasiveness of prostate cancer in vitro and in vivo. Prostate Cancer Prostatic Dis. 2006;9(2):147-52.
Hong MY, Seeram NP, Heber D. Pomegranate polyphenols downregulate expression of androgen-synthesizing genes in human prostate cancer cells overexpressing the androgen receptor. J Nutr Biochem. 2008 Dec;19(12):848-55.
Hoque A, Lippman SM, Wu TT, et al. Increased 5-lipoxygenase expression and induction of apoptosis by its inhibitors in esophageal cancer: a potential target for prevention. Carcinogenesis. 2005 Apr;26(4):785-91.
Hostanska K, Suter A, Melzer J, Saller R. Evaluation of cell death caused by an ethanolic extract of Serenoae repentis fructus (Prostasan) on human carcinoma cell lines. Anticancer Res. 2007 Mar-Apr;27(2):873-81.
Howcroft TK, Campisi J, Louis GB, et al. The role of inflammation in age-related disease. Aging (Albany NY). 2013 Jan;5(1):84-93.
Hsieh TC, Wu JM. Grape-derived chemo preventive agent resveratrol decreases prostate-specific antigen (PSA) expression in LNCaP cells by an androgen receptor (AR)-independent mechanism. Anticancer Res. 2000 Jan-Feb;20(1A):225-8.
Huang EC, McEntee MF, Whelan J. Zyflamend, a combination of herbal extracts, attenuates tumor growth in murine xenograft models of prostate cancer. Nutr Cancer. 2012 64(5):749-60.
Huang L, Kirschke CP, Zhang Y. Decreased intracellular zinc in human tumorigenic prostate epithelial cells: a possible role in prostate cancer progression. Cancer Cell Int. 2006;6:10.
Hung CM, Su YH, Lin HY, et al. Demethoxycurcumin modulates prostate cancer cell proliferation via AMPK-induced down-regulation of HSP70 and EGFR. J Agric Food Chem. 2012 Aug 16.
Hussain M, Banerjee M, Sarkar FH, et al. Soy isoflavones in the treatment of prostate cancer. Nutr Cancer. 2003 47(2):111-7.
Hvid T, Winding K, Rinnov A, et al. Endurance training improves insulin sensitivity and body composition in prostate cancer patients treated with androgen deprivation therapy. Endocr Relat Cancer. 2013 Aug 19;20(5):621-32.
Ibrahiem EI, Mohsen T, Nabeeh AM, Osman Y, Hekal IA, Abou El-Ghar M. DWI-MRI: single, informative, and noninvasive technique for prostate cancer diagnosis. ScientificWorldJournal. 2012;2012:973450.
IDI. Institute of Diagnostic Imaging. Bone Densitometry. Available at: http://www.idifwb.com/services/exam%20descriptions/bonedensity.htm. Accessed 10/25/2013.
Ilic D, Neuberger MM, Djulbegovic M, Dahm P. Screening for prostate cancer. Cochrane Database Syst Rev. 2013;1:Cd004720.
Inohara H, Raz A. Effects of natural complex carbohydrate (citrus pectin) on murine melanoma cell properties related to galectin-3 functions. Glycoconj J. 1994 Dec;11(6):527-32.
Isakovic A, Harhaji L, Stevanovic D, et al. Dual antiglioma action of metformin: cell cycle arrest and mitochondria-dependent apoptosis. Cell Mol Life Sci. 2007 May;64(10):1290-302.
Itsiopoulos C, Hodge A, Kaimakamis M. Can the Mediterranean diet prevent prostate cancer? Mol Nutr Food Res. 2009 Feb;53(2):227-39.
Jacobsen BK, Knutsen SF, Fraser GE. Does high soy milk intake reduce prostate cancer incidence? The Adventist Health Study (United States). Cancer Causes Controls. 1998 Dec;9(6):553-7.
Jalving M, Gietema JA, Lefrandt JD, et al. Metformin: taking away the candy for cancer? Eur J Cancer. 2010 Sep;46(13):2369-80.
James MJ, Penglis PS, Caughey GE, Demasi M, Cleland LG. Eicosanoid production by human monocytes: does COX-2 contribute to a self-limiting inflammatory response? Inflamm Res. 2001 May;50(5):249-53.
Jang M, Cai L, Udeani GO, et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science. 1997 Jan 10;275(5297):218-20.
Jang M, Pezzuto JM. Cancer chemopreventive activity of resveratrol. Drugs Exp Clin Res. 1999;25(2-3):65-77.
Jankowska EA, Rozentryt P, Ponikowska B. Circulating estradiol and mortality in men with systolic chronic heart failure. JAMA. 2009 May 13;301(18):1892-901.
Jeong NH, Song ES, Lee JM, et al. Plasma carotenoids, retinol and tocopherol levels and the risk of ovarian cancer. Acta Obstet Gynecol Scand. 2009;88(4):457-62.
Jian L, Lee AH, Binns CW. Tea and lycopene protect against prostate cancer. Asia Pac J Clin Nutr. 2007;16 Suppl 1:453- 7.
Jiang Q, Wong J, Ames BN. Gamma-tocopherol induces apoptosis in androgen-responsive LNCaP prostate cancer cells via caspasedependent and independent mechanisms. Ann NY Acad Sci. 2004 Dec;1031:399-400.
Jiang WG, Douglas-Jones AG, Mansel RE. Aberrant expression of 5-lipoxygenase-activating protein (5-LOXAP) has prognostic and survival significance in patients with breast cancer. Prostaglandins Leukot Essent Fatty Acids. 2006 Feb;74(2):125-34.
Johansson C, Rytter E, Nygren J, Vessby B, Basu S, Möller L. Down-regulation of oxidative DNA lesions in human mononuclear cells after antioxidant supplementation correlates to increase of gamma-tocopherol. Int J Vitam Nutr Res. 2008 Jul-Sep;78(4-5):183-94.
John EM, Schwartz GG, Koo J, Van Den BD, Ingles SA. Sun exposure, vitamin D receptor gene polymorphisms, and risk of advanced prostate cancer. Cancer Res. 2005 Jun 15;65(12):5470-9.
John EM, Stern MC, Sinha R, Koo J. Meat consumption, cooking practices, meat mutagens, and risk of prostate cancer. Nutr Cancer. 2011 63(4):525-37.
Jolliet P, Simon N, Barre J, et al. Plasma coenzyme Q10 concentrations in breast cancer: prognosis and therapeutic consequences. Int J Clin Pharmacol Ther. 1998 Sep;36(9):506-9.
Joseph MA, Moysich KB, Freudenheim JL, et al. Cruciferous vegetables, genetic polymorphisms in glutathione Stransferases M1 and T1, and prostate cancer risk. Nutr Cancer. 2004;50(2):206-13.
Joshi AD, Corral R, Catsburg C, et al. Red meat and poultry, cooking practices, genetic susceptibility and risk of prostate cancer: results from a multiethnic case-control study. Carcinogenesis. 2012 Nov;33(11):2108-18.
Joshua AM, Zannella V, Bowes B, et al. A phase II study of neoadjuvant metformin in prostatic carcinoma [Abstract]. In: Proceedings of the 103rd Annual Meeting of the American Association for Cancer Research; 2012 Mar 31-Apr 4; Chicago, Illinois. Philadelphia (PA): AACR; 2012. {Abstract CT-04}
Kampa M, Hatzoglou A, Notas G, et al. Wine antioxidant polyphenols inhibit the proliferation of human prostate cancer cell lines. Nutr Cancer. 2000;37(2):223-33.
Kaplan SA, Ghafar MA, Volpe MA, Lam JS, Fromer D, Te AE. PSA response to finasteride challenge in men with a serum PSA greater than 4 ng/ml and previous negative prostate biopsy: preliminary study. Urology. 2002 Sep;60(3):464-8.
Karna P, Chagani S, Gundala SR, et al. Benefits of whole ginger extract in prostate cancer. Br J Nutr. 2012 February;107(4):473-84.
Katiyar SK. Matrix metalloproteinases in cancer metastasis: molecular targets for prostate cancer prevention by green tea polyphenols and grape seed proanthocyanidins. Endocr Metab Immune Disord Drug Targets. 2006 Mar;6(1):17-24.
Kaur M, Agarwal C, Agarwal R. Anticancer and cancer chemopreventive potential of grape seed extract and other grape-based products. J Nutr. 2009 Sep;139(9):1806S-12S
Kenfield SA, Chang ST, Chan JM. Diet and lifestyle interventions in active surveillance patients with favorable-risk prostate cancer. Curr Treat Options Oncol. 2007 Jun;8(3):173-96.
Kenfield SA, Dupre N, Richman EL, et al. Mediterranean diet and prostate cancer risk and mortality in the Health Professionals Follow-up Study. Eur Urol. 2013 Aug 13.
Khan N, Adhami VM, Mukhtar H. Apoptosis by dietary agents for prevention and treatment of prostate cancer. Endocr Relat Cancer. 2010 Mar;17(1):R39-52.
Killian PH, Kronski E, Michalik KM, et al. Curcumin inhibits prostate cancer metastasis in vivo by targeting the inflammatory cytokines CXCL1 and -2. Carcinogenesis. 2012 Dec;33(12):2507-19.
Kim KM, Song JJ, An JY, Kwon YT, Lee YJ. Pretreatment of acetylsalicylic acid promotes tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by down-regulating BCL-2 gene expression. J Biol Chem. 2005 Dec 9;280(49):41047-56.
Kimmatkar N, Thawani V, Hingorani L, Khiyani R. Efficacy and tolerability of Boswellia serrata extract in treatment of osteoarthritis of knee--a randomized double blind placebo controlled trial. Phytomedicine. 2003 Jan;10(1):3-7.
Kimura T. East meets West: ethnic differences in prostate cancer epidemiology between East Asians and Caucasians. Chin J Cancer. 2012 Sep;31(9):421-9.
Kindblom J. 2003. Role of Prolactin in the Prostate Gland – Studies in Transgenic Mouse Models. (Doctoral thesis). Available at: http://www.neurophys.gu.se/digitalAssets/1278/1278430_AVHANDLINGSRAM__Jon_Kindblom.pdf. Accessed 10/24/2013.
Kirsh VA, Peters U, Mayne ST, et al. Prospective study of fruit and vegetable intake and risk of prostate cancer. J Natl Cancer Inst. 2007 Aug 1;99(15):1200-9.
Klein EA, Thompson IM, Jr., Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011 Oct 12;306(14):1549-56.
Knezevich EL, Knezevick JT, Spangler ML. Benign Prostatic Hyperplasia and the Medication Management of Associated Lower Urinary Tract Symptoms. 5-alpha-reductase Inhibitors. Available at: http://www.medscape.com/viewarticle/745454_4. Accessed 10/29/2013.
Kojima S, Inahara M, Suzuki H, Ichikawa T, Furuya Y. Implications of insulin-like growth factor-I for prostate cancer therapies. Int J Urol. 2009 Feb;16(2):161-7.
Kong HY, Byun J. Emerging roles of Human prostatic acid phosphatase. Biomol Ther (Seoul). 2013 Jan;21(1):10-20.
Krejcarek SC, Chen MH, Renshaw AA, Loffredo M, Sussman B, D’Amico AV. Prediagnostic prostate-specific antigen velocity and probability of detecting high-grade prostate cancer. Urology. 2007 Mar;69(3):515-9.
Krishnan AV, Peehl DM, Feldman D. Inhibition of prostate cancer growth by vitamin D: Regulation of target gene expression. J Cell Biochem. 2003 Feb 1;88(2):363-71.
Kristal AR, Lampe JW. Brassica vegetables and prostate cancer risk: a review of the epidemiological evidence. Nutr Cancer. 2002;42(1):1-9.
Kristal AR, Till C, Tangen CM, et al. Associations of serum sex steroid hormone and 5α-androstane-3α,17β-diol glucuronide concentrations with prostate cancer risk among men treated with finasteride. Cancer Epidemiol Biomarkers Prev. 2012 Oct;21(10):1823-32.
Kucuk O, Sarkar FH, Sakr W, et al. Phase II randomized clinical trial of lycopene supplementation before radical prostatectomy. Cancer Epidemiol Biomarkers Prev. 2001;10:861-8.
Kulkarni GS, Al-Azab R, Lockwood G, et al. Evidence for a biopsy derived grade artifact among larger prostate glands. J Urol. 2006 Feb;175(2):505-9
Kumar NB, Cantor A, Allen K, et al. The specific role of isoflavones in reducing prostate cancer risk. Prostate. 2004 May 1;59(2):141-7.
Kunnumakkara AB, Anand P, Aggarwal BB. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008 Oct 8;269(2):199-225.
Kurahashi N, Sasazuki S, Iwasaki M, Inoue M, Tsugane S. Green tea consumption and prostate cancer risk in Japanese men: a prospective study. Am J Epidemiol. 2008 Jan 1;167(1):71-7.
LabCorp. Test Menu. Prostate-specific Antigen (PSA), Serum. LabCorp web page.Copyright © 2013 Laboratory Corporation of America. Accessed 10/24/2013a.
LabCorp. Test Menu. Vitamin D, 25-Hydroxy. Copyright © 2013 Laboratory Corporation of America. Accessed 10/24/2013b.
Lakshman M, Xu L, Ananthanarayanan V, et al. Dietary genistein inhibits metastasis of human prostate cancer in mice. Cancer Res. 2008 Mar 15;68(6):2024-32.
Lalithakumari K, Krishnaraju AV, Sengupta K, Subbaraju GV, Chatterjee A. Safety and toxicological evaluation of a novel, standardized 3-O-acetyl-11-keto-beta-boswellic acid (AKBA)-enriched Boswellia serrata extract (5-Loxin®(R)). Toxicol Mech Methods. 2006;16(4):199-226.
Lamblin F, Hano C, Fliniaux O, Mesnard F, Fliniaux MA, Laine E. Interest of lignans in prevention and treatment of cancers. Med Sci (Paris). 2008 May;24(5):511-9.
Lamson DW and Plaza SM. The Anticancer Effects of Vitamin K. Alt Med Rev. 2003;8(3): 303-318.
Langley RE, Burdett S, Tierney JF, Cafferty F, Parmar MK, Venning G. Aspirin and cancer: has aspirin been overlooked as an adjuvant therapy? Br J Cancer. 2011 Oct 11;105(8):1107-13.
Lansky EP, Newman RA. Punica granatum (pomegranate) and its potential for prevention and treatment of inflammation and cancer. J Ethnopharmacol. 2007;109(2):177-206.
Lantz RC, Chen GJ, Solyom AM, Jolad SD, Timmermann BN. The effect of turmeric extracts on inflammatory mediator production. Phytomedicine. 2005 Jun;12(6- 7):445-52.
Larre S, Tran N, Fan C, et al. PGE2 and LTB4 tissue levels in benign and cancerous prostates. Prostaglandins Other Lipid Mediat. 2008 Dec;87(1-4):14-9.
Lasalvia-Prisco E, Cucchi S, Vazquez J, Lasalvia-Galante E, Golomar W, Gordon W. Serum markers variation consistent with autoschizis induced by ascorbic acid-menadione in patients with prostate cancer. Med Oncol. 2003;20(1):45-52.
Laufer S. Role of eicosanoids in structural degradation in osteoarthritis. Curr Opin Rheumatol. 2003 Sep;15(5):623-7.
Lee EH, Myung SK, Jeon YJ, et al. Effects of selenium supplements on cancer prevention: Meta-analysis of randomized controlled trials. Nutr Cancer. 2011 Nov;63(8):1185-95.
Lee J, Im YH, Jung HH, et al. Curcumin inhibits interferon-alpha induced NF-kappaB and COX-2 in human A549 non-small cell lung cancer cells. Biochem Biophys Res Commun. 2005 Aug 26;334(2):313-8.
Lee MM, Gomez SL, Chang JS, et al. Soy and isoflavone consumption in relation to prostate cancer risk in China. Cancer Epidemiol Biomarkers Prev. 2003 Jul;12(7):665-8.
Lee ST, Wu YL, Chien LH, Chen ST, Tzeng YK, Wu TF. Proteomic exploration of the impacts of pomegranate fruit juice on the global gene expression of prostate cancer cell. Proteomics. 2012 Nov;12(21):3251-62.
Lee YH, Kwak J, Choi HK, et al. EGCG suppresses prostate cancer cell growth modulating acetylation of androgen receptor by anti-histone acetyltransferase activity. Int J Mol Med. 2012 Jul;30(1):69-74.
Lee YJ, Lee DM, Lee CH, et al. Suppression of human prostate cancer PC-3 cell growth by N-acetylcysteine involves over-expression of Cyr61. Toxicol In Vitro. 2011 Feb;25(1):199-205.
LeFevre M. A Role for Finasteride in the Prevention of Prostate Cancer? NEJM. 2013; 369:670-671.
Leitzmann MF, Stampfer MJ, Michaud DS, et al. Dietary intake of n-3 and n-6 fatty acids and the risk of prostate cancer. Am J Clin Nutr. 2004 Jul;80(1):204-16.
Lhoste EF, Gloux K, De W, I, et al. The activities of several detoxication enzymes are differentially induced by juices of garden cress, water cress and mustard in human HepG2 cells. Chem Biol Interact. 2004 Dec 7;150(3):211-9.
Li Y, Chinni SR, Sarkar FH. Selective growth regulatory and pro-apoptotic effects of DIM is mediated by AKT and NF-kappaB pathways in prostate cancer cells. Front Biosci. 2005 Jan 1;10:236- 43.
Liang JY, Liu YY, Zou J, Franklin RB, Costello LC, Feng P. Inhibitory effect of zinc on human prostatic carcinoma cell growth. Prostate. 1999 Aug 1;40(3):200-7.
Libby G, Donnelly LA, Donnan PT, Alessi DR, Morris AD, Evans JM. New users of metformin are at low risk of incident cancer: a cohort study among people with type 2 diabetes. Diabetes Care. 2009 Sep;32(9):1620-5.
Lieberman S. Can percent free prostate-specific antigen reduce the need for prostate biopsy? Eff Clin Pract. 1999 Nov- Dec;2(6):266-71.
Ligthart ST, Coumans FA, Bidard FC, et al. Circulating tumor cells count and morphological features in breast, colorectal and prostate cancer. PLoS One. 2013 Jun 27;8(6):e67148.
Ling MT, Lau TC, Zhou C, et al. Overexpression of Id-1 in prostate cancer cells promotes angiogenesis through the activation of vascular endothelial growth factor (VEGF). Carcinogenesis. 2005 Oct;26(10):1668-76.
Lippman SM, Klein EA, Goodman PJ, et al. Effect of selenium and vitamin E on risk of prostate cancer and other cancers: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2009 Jan 7;301(1):39-51.
Liu J-J, Nilsson A, Oredsson S, Badmaev V, Zhao W, Duan R. Boswellic acids trigger apoptosis via a pathway dependent on caspase- 8 activation but independent on Fas/Fas ligand interaction in colon cancer HT-29 cells. Carcinogenesis. Dec 2002;23(12):2087-93.
Liu KC, Yen CY, Wu RS, et al. The roles of endoplasmic reticulum stress and mitochondrial apoptotic signaling pathway in quercetin-mediated cell death of human prostate cancer PC-3 cells. Environ Toxicol. 2012. doi: 10.1002/tox.21769. Epub March 20, 2012.
Lockwood K, Moesgaard S, Hanioka T, Folkers K. Apparent partial remission of breast cancer in ‘high risk’ patients supplemented with nutritional antioxidants, essential fatty acids and coenzyme Q10. Mol Aspects Med. 1994;15 Suppls231-s40.
Loeb S, Berglund A, Stattin P. Population based study of use and determinants of active surveillance and watchful waiting for low and intermediate risk prostate cancer. J Urol. 2013 May 30.
Loeb S, Carter HB. Point: Impact of prostate-specific antigen velocity on management decisions and recommendations. J Natl Compr Canc Netw. 2013 Mar 1;11(3):281-5.
Lou YR, Qiao S, Talonpoika R, Syvala H, Tuohimaa P. The role of Vitamin D3 metabolism in prostate cancer. J Steroid Biochem Mol Biol. 2004 Nov;92(4):317-25.
Loubière C, Dirat B, Tanti JF, Bost F. New perspectives for metformin in cancer therapy. Ann Endocrinol (Paris). 2013 May;74(2):130-6.
Lowe JF, Frazee LA. Update on prostate cancer chemoprevention. Pharmacotherapy. 2006 Mar;26(3):353-9.
Lu QY, Hung JC, Heber D, et al. Inverse associations between plasma lycopene and other carotenoids and prostate cancer. Cancer Epidemiol Biomarkers Prev. 2001 Jul;10(7):749-56.
Lu R, Serrero G. Resveratrol, a natural product derived from grape, exhibits anti-estrogenic activity and inhibits the growth of human breast cancer cells. J Cell Physiol. 1999 Jun;179(3):297-304.
Lucia MS, Epstein JI, Goodman PJ, et al. Finasteride and high-grade prostate cancer in the Prostate Cancer Prevention Trial. J Natl Cancer Inst. 2007 Sep 19;99(18):1375-83.
Lunoe K, Gabel-Jensen C, Sturup S, Andresen L, Skov S, Gammelgaard B. Investigation of the selenium metabolism in cancer cell lines. Metallomics. 2011 Feb;3(2):162-8.
Luo H, Jiang BH, King SM, Chen YC. Inhibition of cell growth and VEGF expression in ovarian cancer cells by flavonoids. Nutr Cancer. 2008;60(6):800-9.
Luo J, Dunn TA, Ewing CM, Walsh PC, Isaacs WB. Decreased gene expression of steroid 5 alpha-reductase 2 in human prostate cancer: implications for finasteride therapy of prostate carcinoma. Prostate. 2003 Oct 1;57(2):134-9.
Luo Z, Zang M, Guo W. AMPK as a metabolic tumor suppressor: control of metabolism and cell growth. Future Oncol. 2010 Mar;6(3):457-70.
Malik A, Afaq F, Sarfaraz S, Adhami VM, Syed DN, Mukhtar H. Pomegranate fruit juice for chemoprevention and chemotherapy of prostate cancer. Proc Natl Acad Sci USA. 2005 Oct 11;102(41):14813-8.
Malik A, Mukhtar H. Prostate cancer prevention through pomegranate fruit. Cell Cycle. 2006 Feb;5(4):371-3.
Mantell DJ, Owens PE, Bundred NJ, Mawer EB, Canfield AE. 1 alpha,25-dihydroxyvitamin D(3) inhibits angiogenesis in vitro and in vivo. Circ Res. 2000 Aug 4;87(3):214-20.
Marshall DT, Savage SJ, Garrett-Mayer E, et al. Vitamin D3 supplementation at 4000 international units per day for one year results in a decrease of positive cores at repeat biopsy in subjects with low-risk prostate cancer under active surveillance. J Clin Endocrinol Metab. 2012 Jul;97(7):2315-24.
Marshall JR, Ip C, Romano K, et al. Methyl Selenocysteine: singledose pharmacokinetics in men. Cancer Prev Res (Phila). 2011 Nov;4(11):1938-44.
Masko EM, Allott EH, Freedland SJ. The relationship between nutrition and prostate cancer: is more always better? Eur Urol. 2013 May;63(5):810-20.
Matsuyama M, Yoshimura R, Mitsuhashi M, et al. Expression of lipoxygenase in human prostate cancer and growth reduction by its inhibitors. Int J Oncol. 2004 Apr;24(4):821-7.
McCann MJ, Gill CI, Linton T, Berrar D, McGlynn H, Rowland IR. Enterolactone restricts the proliferation of the LNCaP human prostate cancer cell line in vitro. Mol Nutr Food Res. 2008 May;52(5):567-80.
McCann SE, Ambrosone CB, Moysich KB, et al. Intakes of selected nutrients, foods, and phytochemicals and prostate cancer risk in western New York. Nutr Cancer. 2005 53(1):33-41.
McCarthy S, Caporali A, Enkemann S, et al. Green tea catechins suppress the DNA synthesis marker MCM7 in the TRAMP model of prostate cancer. Mol Oncol. 2007 Sep;1(2):196-204.
McLarty J, Bigelow RL, Smith M, Elmajian D, Ankem M, Cardelli JA. Tea polyphenols decrease serum levels of prostate-specific antigen, hepatocyte growth factor, and vascular endothelial growth factor in prostate cancer patients and inhibit production of hepatocyte growth factor and vascular endothelial growth factor in vitro. Cancer Prev Res (Phila). 2009 Jul;2(7):673-82.
Meacham S, Karakas S, Wallace, A, et al. Boron in Human Health: Evidence for Dietary Recommendations and Public Policies. The Open Mineral Processing Journal. 2010;3:36-53.
Meirelles ES, Borelli A, Camargo OP. Influence of disease activity and chronicity on ankylosing spondylitis bone mass loss. Clin. Rheumatol. 1999;18:364–8.
Mekki N, Charbonnier M, Borel P, et al. Butter differs from olive oil and sunflower oil in its effects on postprandial lipemia and triacylglycerol-rich lipoproteins after single mixed meals in healthy young men. J Nutr. 2002 Dec;132(12):3642-9.
Melchini A, Traka MH, Catania S, et al. Antiproliferative activity of the dietary isothiocyanate erucin, a bioactive compound from cruciferous vegetables, on human prostate cancer cells. Nutr Cancer. 2013;65(1):132-8.
Memmott RM, Mercado JR, Maier CR, Kawabata S, Fox SD, Dennis PA. Metformin prevents tobacco carcinogen—induced lung tumorigenesis. Cancer Prev Res (Phila). 2010 Sep;3(9):1066-76.
Meyer F, Galan P, Douville P, et al. Antioxidant vitamin and mineral supplementation and prostate cancer prevention in the SU.VI.MAX trial. Int J Cancer. 2005 Aug 20;116(2):182-6.
Miano L. Mediterranean diet, micronutrients and prostate carcinoma: a rationale approach to primary prevention of prostate cancer. Arch Ital Urol Androl. 2003 Sep;75(3):166-78.
Michaud DS, Augustsson K, Rimm EB, Stampfer MJ, Willet WC, Giovannucci E. A prospective study on intake of animal products and risk of prostate cancer. Cancer Causes Control. 2001 Aug;12(6):557-67.
Miller MC, Doyle GV, Terstappen LW. Significance of circulating tumor cells detected by the CellSearch system in patients with metastatic breast colorectal and prostate cancer. J Oncol. 2010;2010:617421.
Mitchell SH, Zhu W, Young CY. Resveratrol inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells. Cancer Res. 1999 Dec 1;59(23):5892-5.
Miwa Y, Kaneda T, Yokoyama O. Correlation between the Aging Males’ Symptoms Scale and sex steroids, gonadotropins, dehydroepiandrosterone sulfate, and growth hormone levels in ambulatory men. J Sex Med. 2006 Jul;3(4):723-6.
Mizokami A, Gotoh A, Yamada H, Keller ET, Matsumoto T. Tumor necrosis factor-alpha represses androgen sensitivity in the LNCaP prostate cancer cell line. J Urol. 2000 Sep;164(3 Pt 1):800-5.
Monga N, Sayani A, Rubinger DA, Wilson TH, Su Z. The effect of dutasteride on the detection of prostate cancer: A set of meta-analyses. Can Urol Assoc J. 2013 Mar-Apr;7(3-4):E161-7.
Moore G. A Call to Action: The story of the Prostate Cancer Foundation. Santa Monica, CA. Copyright © 2005: 13-18.
Moreira Leite KR, Camara-Lopes LH, Dall’Oglio MF, et al. Upgrading the Gleason score in extended prostate biopsy: implications for treatment choice. Int J Radiat Oncol Biol Phys. 2009 Feb 1;73(2):353-6.
Moretti RM, Montagnani MM, Sala A, Motta M, Limonta P. Activation of the orphan nuclear receptor RORalpha counteracts the proliferative effect of fatty acids on prostate cancer cells: crucial role of 5-lipoxygenase. Int J Cancer. 2004 Oct 20;112(1):87-93.
Morgentaler A. Testosterone replacement therapy and prostate cancer. Urol Clin North Am. 2007 Nov;34(4):555-63.
Moyad MA, Brumfield SK, Pienta KJ. Vitamin E, alpha- and gamma-tocopherol, and prostate cancer. Semin Urol Oncol. 1999 May;17(2):85-90.
Moyer VA, U.S. Preventive Services Task Force. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012 Jul 17;157(2):120-34.
MSKCC. Memorial Sloan-Kettering Cancer Center. Integrative Medicine. About Herbs, Botanicals & Other Products. Resveratrol. Available at: http://www.mskcc.org/cancer-care/herb/resveratrol. Last updated 3/19/2013a. Accessed 10/21/2013.
MSKCC. Memorial Sloan-Kettering Cancer Center. Integrative Medicine. About Herbs, Botanicals & Other Prodeucts. N-Acetylcysteine. Available at: http://www.mskcc.org/cancer-care/herb/n-acetylcysteine. Last updated 4/9/2013b. Accessed 10/21/2013.
Mulcahy N. New Data: Finasteride Safely Prevents Prostate Cancer. Medscape Infectious Diseases. 2018 (Available at: https://www.medscape.com/viewarticle/896942#vp_1. Last accessed May 22, 2018.).
Mursu J, Nurmi T, Tuomainen TP, Salonen JT, Pukkala E, Voutilainen S. Intake of flavonoids and risk of cancer in Finnish men: The Kuopio Ischaemic Heart Disease Risk Factor Study. Int J Cancer. 2008;123:660-3.
Muti P, Westerlind K, Wu T, et al. Urinary estrogen metabolites and prostate cancer: a case-control study in the United States. Cancer Causes Control. 2002 Dec;13(10):947-55.
Myriad. Prolaris®: A Prognostic Medicine Product for Prostate Cancer. Myriad Products web page. Available at: http://www.myriad.com/products/prolaris/. Accessed 10/29/2013.
Myzak MC, Dashwood WM, Orner GA, Ho E, Dashwood RH. Sulforaphane inhibits histone deacetylase in vivo and suppresses tumorigenesis in Apc-minus mice. Faseb j. 2006 Mar;20(3):506-8.
Myzak MC, Tong P, Dashwood WM, Dashwood RH, Ho E. Sulforaphane retards the growth of human PC-3 xenografts and inhibits HDAC activity in human subjects. Exp Biol Med (Maywood). 2007 Feb;232(2):227-34.
Nagi DK, Yudkin JS. Effects of metformin on insulin resistance, risk factors for cardiovascular disease, and plasminogen activator inhibitor in NIDDM subjects. A study of two ethnic groups. Diabetes Care. 1993 16(4):621-29.
Nair HK, Rao KV, Aalinkeel R, Mahajan S, Chawda R, Schwartz SA. Inhibition of prostate cancer cell colony formation by the flavonoid quercetin correlates with modulation of specific regulatory genes. Clin Diagn Lab Immunol. 2004 Jan;11(1):63-9.
Nakamura K, Yasunaga Y, Segawa T, et al. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int J Oncol. 2002;21(4):825-30.
Nangia-Makker P, Hogan V, Honjo Y, et al. Inhibition of human cancer cell growth and metastasis in nude mice by oral intake of modified citrus pectin. J Natl Cancer Inst. 2002 Dec 18;94(24):1854-62.
Narayanan BA, Narayanan NK, Re GG, Nixon DW. Differential expression of genes induced by resveratrol in LNCaP cells: P53- mediated molecular targets. Int J Cancer. 2003 Mar 20;104(2):204- 12.
Nassiri-Asl M, Hosseinzadeh H. Review of the pharmacological effects of Vitis vinifera (Grape) and its bioactive compounds. Phytother Res. 2009;23:1197-1204.
Nayyar R, Singh P, Gupta NP, et al. Upgrading of Gleason score on radical prostatectomy specimen compared to the pre-operative needle core biopsy: an Indian experience. Indian J Urol. 2010 Jan- Mar;26(1):56-9.
NCI. National Cancer Institute. Coenzyme Q10 (PDQ®): Questions and Answers About Coenzyme Q10. Accessed 10/21/2013a.
NCI. National Cancer Institute. National Cancer Institute FactSheet. Chemicals in Meat Cooked at High Temperatures and Cancer Risk. Available at: http://www.cancer.gov/cancertopics/factsheet/Risk/cooked-meats. Last reviewed 10/15/2010. Accessed 10/18/2013.
NCI. National Cancer Institute. NCI Dictionary of Cancer Terms. Aggressive. Available at: http://www.cancer.gov/dictionary?cdrid=46053. Accessed 10/18/2013b.
NCI. National Cancer Institute. Prostate Cancer Prevention Trial (PCPT). News page. Available at: http://www.cancer.gov/newscenter/qa/2008/PCPTQandA. Last updated 8/14/2013c. Accessed 10/14/2013.
NCI. National Cancer Institute. Prostate-Specific Antigen (PSA) Test. Available at: http://www.cancer.gov/cancertopics/factsheet/detection/PSA. Last updated 7/24/2012. Accessed 10/24/2013.
NCI. National Cancer Institute. Surveillance, Epidemiology, and End Results Program. Cancer Statistics page. Fast Stats. http://seer.cancer.gov/faststats/selections.php?run=runit&output=2&data=2&statistic=1&year=201302&race=1&sex=2&age=1&series=cancer&cancer=66#Output. 4/2013d. Accessed 10/29/2013.
Nelles JL, Hu WY, Prins GS. Estrogen action and prostate cancer. Expert Rev Endocrinol Metab. 2011 May;6(3):437-451.
Nelson R. Dutasteride Enhances Predictive Usefulness of PSA Test. Available at: http://www.medscape.com/viewarticle/734687. 12/21/2010. Accessed 10/29/2013.
Nelson R. Finasteride Does Not Increase Risk for High-Grade Prostate Cancer. Clin Cancer Res. 2009; 15:4694-4699.
Nelson WG, DeMarzo AM, DeWeese TL, et al. The role of inflammation in the pathogenesis of prostate cancer. J Urol. 2004 Nov;172(5 Pt 2):S6-11.
Newcomer LM, King IB, Wicklund KG, Stanford JL. The association of fatty acids with prostate cancer risk. Prostate. 2001 Jun 1;47(4):262-8.
Nickel JC. Comparison of clinical trials with finasteride and dutasteride. Rev Urol. 2004;6 Suppl 9:S31-9.
Nie D, Lamberti M, Zacharek A, et al. Thromboxane A(2) regulation of endothelial cell migration, angiogenesis, and tumor metastasis. Biochem Biophys Res Commun. 2000 Jan 7;267(1):245-51.
NIH. National Institutes of Health: Office of Dietary Supplements. Dietary Supplement Fact Sheet: Selenium. Health Information page. Available at: http://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/. 7/2/2013. Accessed 10/21/2013.
Nimptsch K, Rohrmann S, Linseisen J. Dietary intake of vitamin K and risk of prostate cancer in the Heidelberg cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Heidelberg). Am J Clin Nutr. 2008 Apr;87(4):985-92.
Nishiyama T. (2011). Paradigm Shift in the Concept of Hormonal Milieu of Prostate Cancer. In: Prostate Cancer - From Bench to Bedside, Dr. Philippe E. Spiess (Ed.), ISBN: 978-953-307-331-6, InTech, Available at: http://cdn.intechopen.com/pdfs/24233/Inech-Paradigm_shift_in_the_concept_of_hormonal_milieu_of_prostate_cancer.pdf.
Niture SK, Refai L. Plant pectin: A potential source of cancer suppression. Amer J Pharmacol Toxicol, 2013;8(1):9-19.
Norris PC, Dennis EA. Omega-3 fatty acids cause dramatic changes in TLR4 and purinergic eicosanoid signaling. Proc Natl Acad Sci U S A. 2012 May 29;109(22):8517-22.
Norrish AE, Skeaff CM, Arribas GL, Sharpe SJ, Jackson RT. Prostate cancer risk and consumption of fish oils: a dietary biomarker-based case-control study. Br J Cancer. 1999 Dec;81(7):1238-42.
Numao N, Kawakami S, Sakura M, et al. Characteristics and clinical significance of prostate cancers missed by initial transrectal 12-core biopsy. BJU Int. 2012 Mar;109(5):665-71.
O’Dowd GJ, Veltri RW, Miller MC, et al. The Gleason Score: A Significant Biologic Manifestation of Prostate Cancer Aggressiveness On Biopsy. Prostate Cancer Research Institute (PCRI) web page. Available at: http://prostate-cancer.org/the-gleason-score-a-significant-biologic-manifestation-of-prostate-cancer-aggressiveness-on-biopsy/. Reprinted from PCRI Insights January 2001; v4.1. Accessed 10/24/2013.
O’Leary KA, de Pascual-Tereasa S, Needs PW, Bao YP, O’Brien NM, Williamson G. Effect of flavonoids and vitamin E on cyclooxygenase-2 (COX-2) transcription. Mutat Res. 2004 Jul 13;551(1-2):245-54.
O’Sullivan J, Sheridan J, Mulcahy H, Tenniswood M, Morrissey C. The effect of green tea on oxidative damage and tumour formation in Lobund-Wistar rats. Eur J Cancer Prev. 2008 Nov;17(6):489-501.
Obermuller-Jevic UC, Olano-Martin E, Corbacho AM, et al. Lycopene inhibits the growth of normal human prostate epithelial cells in vitro. J Nutr. 2003;133:3356-60.
Ong KW, Hsu A, Tan BK. Anti-diabetic and anti-lipidemic effects of chlorogenic acid are mediated by ampk activation. Biochem Pharmacol. 2013 May 1;85(9):1341-51.
Onguru O, Casey MB, Kajita S, Nakamura N, Lloyd RV. Cyclooxygenase- 2 and thromboxane synthase in non-endocrine and endocrine tumors: a review. Endocr Pathol. 2005 16(4):253-77.
Ornish D, Magbanua MJ, Weidner G, et al. Changes in prostate gene expression in men undergoing an intensive nutrition and lifestyle intervention. Proc Natl Acad Sci U S A. 2008 Jun 17;105(24):8369-74.
Ozasa K, Nakao M, Watanabe Y, et al. Serum phytoestrogens and prostate cancer risk in a nested case-control study among Japanese men. Cancer Sci. 2004 Jan;95(1):65-71.
Pais P. Potency of a novel saw palmetto ethanol extract, SPET- 085, for inhibition of 5alpha-reductase II. Adv Ther. 2010 Aug;27(8):555-63.
Palaniswamy UR, McAvoy RJ, Bible BB, Stuart JD. Ontogenic variations of ascorbic acid and phenethyl isothiocyanate concentrations in watercress (Nasturtium officinale R.Br.) leaves. J Agric Food Chem. 2003 Aug 27;51(18):5504-9.
Paller CJ, Ye X, Wozniak PJ, et al. A randomized phase II study of pomegranate extract for men with rising PSA following initial therapy for localized prostate cancer. Prostate Cancer Prostatic Dis. 2013 Mar;16(1):50-5.
Pandey M, Gupta S. Green tea and prostate cancer: from bench to clinic. Front Biosci (Elite Ed). 2009;1:13-25.
Pantuck AJ, Leppert JT, Zomorodian N, et al. Phase II study of pomegranate juice for men with rising prostate-specific antigen following surgery or radiation for prostate cancer. Clin Cancer Res. 2006 Jul 1;12(13):4018-26.
Parekh N, Lin Y, Vadiveloo M, Hayes RB, Lu-Yao GL. Metabolic dysregulation of the insulin-glucose axis and risk of obesity-related cancers in the Framingham Heart Study-Offspring Cohort (1971-2008). Cancer Epidemiol Biomarkers Prev. 2013 Sep 24.
Park C, Kim GY, Kim GD, et al. Induction of G2/M arrest and inhibition of cyclooxygenase-2 activity by curcumin in human bladder cancer T24 cells. Oncol Rep. 2006 May;15(5):1225-31.
Park HJ, Jeon YK, You DH, Nam MJ. Daidzein causes cytochrome c-mediated apoptosis via the Bcl-2 family in human hepatic cancer cells. Food Chem Toxicol. 2013 Oct;60:542-9.
Patterson K. A Patient’s Guide to Bone Health. Published in CURE. Available at: http://media.curetoday.com/downloads/documents/BoneHealth_PG_rev_b.pdf. Fall 2010.
PCCNC. Prostate Cancer Coalition of North Carolina. Prostate Cancer Awareness. Available at: http://www.pccnc.org/awareness/. Accessed 10/28/2013.
PCF. Prostate Cancer Foundation. Prostate Cancer FAQs. Available at: http://www.pcf.org/site/c.leJRIROrEpH/b.5800851/k.645A/Prostate_Cancer_FAQs.htm. 2013a. Accessed 10/24/2013.
PCF. Prostate Cancer Foundation. Understanding Prostate Cancer: Prostate Cancer Risk Factors. Available at: http://www.pcf.org/site/c.leJRIROrEpH/b.5802027/k.D271/Prostate_Cancer_Risk_Factors.htm. Copyright © 2013b. Accessed 10/25/2013.
PCRI. Prostate Cancer Research Institute. Diagnostic Imaging. Available at: http://prostate-cancer.org/decision-aide/understanding-your-diagnosis/diagnostic-imaging/. Last updated 8/1/2011. Accessed 10/24/2013.
Pelser C, Mondul AM, Hollenbeck AR, Park Y. Dietary fat, fatty acids, and risk of prostate cancer in the NIH-AARP diet and health study. Cancer Epidemiol Biomarkers Prev. 2013 Apr;22(4):697-707
Perumal SS, Shanthi P, Sachdanandam P. Energy-modulating vitamins—a new combinatorial therapy prevents cancer cachexia in rat mammary carcinoma. Br J Nutr. 2005 Jun;93(6):901-9.
Pezzato E, Sartor L, Dell’Aica I, et al. Prostate carcinoma and green tea: PSA-triggered basement membrane degradation and MMP-2 activation are inhibited by (-) epigallocatechin-3-gallate. Int J Cancer. 2004 Dec 10;112(5):787-92.
Pham H, Ziboh VA. 5a-Reductase-catalyzed conversion of testosterone to dihydrotestosterone is increased in prostatic adenocarcinoma cells: suppression by 15-lipoxygenase metabolites of gamma-linolenic and eicosapentaenoic acids. Steroid Biochem Mol Biol. 2002;82:393-400.
Pienta KJ, Naik H, Akhtar A, et al. Inhibition of spontaneous metastasis in a rat prostate cancer model by oral administration of modified citrus pectin. J Natl Cancer Inst. 1995;87:348-53.
Pietinen P, Stumpf K, Mannisto S, Kataja V, Uusitupa M, Adlercreutz H. Serum enterolactone and risk of breast cancer: a casecontrol study in eastern Finland. Cancer Epidemiol Biomarkers Prev. 2001 Apr;10(4):339-44.
Piller R, Chang-Claude J, Linseisen J. Plasma enterolactone and genistein and the risk of premenopausal breast cancer. Eur J Cancer Prev. 2006 Jun;15(3):225-32.
Pitchakarn P, Chewonarin T, Ogawa K, et al. Ellagic Acid inhibits migration and invasion by prostate cancer cell lines. Asian Pac J Cancer Prev. 2013;14(5):2859-63.
Plummer SM, Holloway KA, Manson MM, et al. Inhibition of cyclo-oxygenase 2 expression in colon cells by the chemopreventive agent curcumin involves inhibition of NF-kappaB activation via the NIK/IKK signalling complex. Oncogene. 1999 Oct 28;18(44):6013-20.
Polesel J, Talamini R, Montella M, et al. Linoleic acid, vitamin D and other nutrient intakes in the risk of non-Hodgkin lymphoma: an Italian case-control study. Ann Oncol. 2006 Apr;17(4):713-8.
Pollack CE, Platz EA, Bhavsar NA, et al. Primary care providers’ perspectives on discontinuing prostate cancer screening. Cancer. 2012 Nov 15;118(22):5518-24.
Portakal O, Ozkaya O, Erden IM, et al. Coenzyme Q10 concentrations and antioxidant status in tissues of breast cancer patients. Clin Biochem. 2000 Jun;33(4): 279-84.
Post PN, Kil PJ, Hendrikx AJ, Janssen- Heijnen ML, Crommelin MA, Coebergh JW. Comorbidity in patients with prostate cancer and its relevance to treatment choice. BJU Int. 1999 Oct;84(6):652-6.
Prins GS, Birch L, Couse JF, et al. Estrogen imprinting of the developing prostate gland is mediated through stromal estrogen receptor alpha: studies with alphaERKO and betaERKO mice. Cancer Res. 2001 Aug 15;61(16):6089-97.
Prins GS, Birch L, Tang WY, Ho SM. Developmental estrogen exposures predispose to prostate carcinogenesis with aging. Reprod Toxicol. 2007 Apr;23(3):374-82.
Prins GS, Tang WY, Belmonte J, Ho SM. Perinatal exposure to oestradiol and bisphenol A alters the prostate epigenome and increases susceptibility to carcinogenesis. Basic Clin Pharmacol Toxicol. 2008 Feb;102(2):134-8.
Pruthi RS, Derksen JE, Moore D, et al. Phase II trial of celecoxib in prostate-specific antigen recurrent prostate cancer after definitive radiation therapy or radical prostatectomy. Clin Cancer Res. 2006 Apr 1;12(7 Pt 1):2172-7.
Punnen S, Hardin J, Cheng I, Klein EA, Witte JS. Impact of meat consumption, preparation, and mutagens on aggressive prostate cancer. PLoS One. 2011 6(11):e27711.
Qaseem A, Barry MJ, Denberg TD, et al. Screening for prostate cancer: a guidance statement from the Clinical Guidelines Committee of the American College of Physicians. Ann Intern Med. 2013 May 21;158(10):761-9.
Qin W, Smith C, Jensen M, Holick MF, Sauter ER. Vitamin d favorably alters the cancer promoting prostaglandin cascade. Anticancer Res. 2013 Sep;33(9):3861-6.
Qu T, Uz T, Manev H. Inflammatory 5-LOX mRNA and protein are increased in brain of aging rats. Neurobiol Aging. 2000 Sep- Oct;21(5):647-52.
Rabbani F, Stroumbakis N, Kava BR, Cookson MS, Fair WR. Incidence and clinical significance of false-negative sextant prostate biopsies. J Urol. 1998 Apr;159(4):1247-50.
Rafi MM, Kanakasabai S, Reyes MD, Bright JJ. Lycopene modulates growth and survival associated genes in prostate cancer. J Nutr Biochem. 2013 Oct;24(10):1724-34.
Raina K, Ravichandran K, Rajamanickam S, Huber KM, Serkova NJ, Agarwal R. Inositol hexaphosphate inhibits tumor growth, vascularity, and metabolism in TRAMP mice: a multiparametric magnetic resonance study. Cancer Prev Res. January 2013;6:40-50.
Raina K, Singh RP, Agarwal R, Agarwal C. Oral grape seed extract inhibits prostate tumor growth and progression in TRAMP mice. Cancer Res. 2007;67:5976-82.
Rajashekhar G, Willuweit A, Patterson CE, et al. Continuous endothelial cell activation increases angiogenesis: evidence for the direct role of endothelium linking angiogenesis and inflammation. J Vasc Res. 2006 43(2):193-204.
Rao KV, Samikkannu T, Dakshayani KB, et al. Chemopreventive potential of an ethyl acetate fraction from Curcuma longa is associated with upregulation of p57(kip2) and Rad9 in the PC-3M prostate cancer cell line. Asian Pac J Cancer Prev. 2012;13(3): 1031-8.
Ravindran J, Prasad S, Aggarwal BB. Curcumin and cancer cells: how many ways can curry kill tumor cells selectively? AAPS J. 2009 Sep;11(3):495-510.
Raynaud JP. Prostate cancer risk in testosterone-treated men. J Steroid Biochem Mol Biol. 2006 Dec;102(1-5):261-6.
Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008;22:659-61.
Redman MW, Tangen CM, Goodman PJ, et al. Finasteride Does Not Increase the Risk of High-Grade Prostate Cancer: A Bias-Adjusted Modeling Approach. Cancer Prev Res. 2008; 1:174-181.
Ren S, Lien EJ. Natural products and their derivatives as cancer chemopreventive agents. Prog Drug Res. 1997;48:147-71.
Reuter S, Gupta SC, Park B, Goel A, Aggarwal BB. Epigenetic changes induced by curcumin and other natural compounds. Genes Nutr. 2011 May;6(2):93-108.
Rhoden EL, Averbeck MA, Teloken PE. Androgen replacement in men under-going treatment for prostate cancer. J Sex Med. 2008 Sep;5(9):2202-8.
Richman EL, Kenfield SA, Stampfer MJ, Giovannucci EL, Chan JM. Egg, red meat, and poultry intake and risk of lethal prostate cancer in the prostate-specific antigen-era: incidence and survival. Cancer Prev Res (Phila). 2011 Dec;4(12):2110-21.
Riehl TE, Turk J, and Stetson WF. Metabolism of oxygenated derivatives of arachidonic acid by Caco-2 cells. Journal of Lipid Research. 1992:33: 323-331.
Ripple MO, Henry WF, Schwarze SR, Wilding G, Weindruch R. Effect of antioxidants on androgen-induced AP-1 and NF-kappaB DNA-binding activity in prostate carcinoma cells. J Natl Cancer Inst. 1999 Jul 21;91(14):1227-32.
Ritch CR, Wan RL, Stephens LB, et al. Dietary fatty acids correlate with prostate cancer biopsy grade and volume in Jamaican men. J Urol. 2007 Jan;177(1):97-101; discussion 101.
Roddam AW, Allen NE, Appleby P, Key TJ. Endogenous sex hormones and prostate cancer: a collaborative analysis of 18 prospective studies. J Natl Cancer Inst. 2008 Feb 6;100(3):170-83.
Rodriguez-Berriguete G, Sanchez-Espiridion B, Cansino JR, et al. Clinical significance of both tumor and stromal expression of components of the IL-1 and TNF-α signaling pathways in prostate cancer. Cytokine. 2013 Sep 21.
Roehrborn C. Insights into the Relationships between Prostatic Disorders and Their Potential Impact on Future Urologic Practice. European Urology Supplements 5 (2006) 698-703.
Rogan EG. The natural chemopreventive compound indole-3-carbinol: state of the science. In Vivo. 2006 Mar-Apr;20 (2):221-8.
Rosillo MA, Sanchez-Hidalgo M, Cardeno A, et al. Dietary supplementation of an ellagic acid-enriched pomegranate extract attenuates chronic colonic inflammation in rats. Pharmacol Res. 2012 Sep;66(3):235-42.
Roy S, Khanna S, Shah H, et al. Human genome screen to identify the genetic basis of the anti-inflammatory effects of Boswellia in microvascular endothelial cells. DNA Cell Biol. 2005 Apr;24(4):244-55.
Rubinsztajn R, Wronska J, Chazan R. Urinary leukotriene E4 concentration in patients with bronchial asthma and intolerance of non-steroids anti-inflammatory drugs before and after oral aspirin challenge. Pol Arch Med Wewn. 2003 Aug;110(2):849-54.
Rusciani L, Proietti I, Rusciani A, et al. Low plasma coenzyme Q10 levels as an independent prognostic factor for melanoma progression. J Am Acad Dermatol. 2006 Feb;54(2):234-41.
Safayhi H, Rall B, Sailer ER, Ammon HP. Inhibition by boswellic acids of human leukocyte elastase. J Pharmacol Exp Ther. 1997 Apr;281(1):460-3.
Safayhi H, Sailer ER, Ammon HP. Mechanism of 5-lipoxygenase inhibition by acetyl-11-keto-beta-boswellic acid. Mol Pharmacol. 1995 Jun;47(6):1212-6.
Sailer ER, Subramanian LR, Rall B, et al. Acetyl-11-keto-betaboswellic acid (AKBA): structure requirements for binding and 5-lipoxygenase inhibitory activity. Br J Pharmacol. 1996 Feb;117(4):615-8.
Sakr WA, Grignon DJ, Haas GP, et al. Epidemiology of high grade prostatic intraepithelial neoplasia. Pathol Res Pract. 1995 Sep;191(9):838-41.
Salinas CA, Kwon EM, FitzGerald LM, et al. Use of aspirin and other nonsteroidal antiinflammatory medications in relation to prostate cancer risk. Am J Epidemiol. 2010 Sep 1;172(5):578-90.
Sampson AP. FLAP inhibitors for the treatment of inflammatory diseases. Curr Opin Investig Drugs. 2009 Nov;10(11):1163-72.
Sandur SK, Ahn KS, Ichikawa H, et al. Zyflamend, a polyherbal preparation, inhibits invasion, suppresses osteoclastogenesis, and potentiates apoptosis through down-regulation of NF-kappa B activation and NF-kappa B-regulated gene products. Nutr Cancer. 2007 57(1):78-87.
Sarkar FH, Li Y. Indole-3-carbinol and prostate cancer. J Nutr. 2004 Dec;134(12 Suppl):3493S-3498S.
Sartippour MR, Seeram NP, Rao JY, et al. Ellagitannin-rich pomegranate extract inhibits angiogenesis in prostate cancer in vitro and in vivo. Int J Oncol. 2008 Feb;32(2):475-80.
Saruwatari A, Okamura S, Nakajima Y, Narukawa Y, Takeda T, Tamura H. Pomegranate juice inhibits sulfoconjugation in Caco-2 human colon carcinoma cells. J Med Food. 2008 Dec;11(4):623-8.
Schleicher RL, Lamartiniere CA, Zheng M, et al. The inhibitory effect of genistein on the growth and metastasis of a transplantable rat accessory sex gland carcinoma. Cancer Lett. 1999 Mar 1;136(2):195-201.
Schmidt LJ, Regan KM, Anderson SK, Sun Z, Ballman KV, Tindall DJ. Effects of the 5 alpha-reductase inhibitor dutasteride on gene expression in prostate cancer xenografts. Prostate. 2009 Dec 1;69(16):1730-43.
Schmidt LJ, Tindall DJ. Steroid 5 α-reductase inhibitors targeting BPH and prostate cancer. J Steroid Biochem Mol Biol. 2011 May;125(1-2):32-8.
Schwartz J. Role of polyunsaturated fatty acids in lung disease. Am J Clin Nutr. 2000 Jan;71(1 Suppl):393S-6S.
Sears B, Ricordi C. Anti-inflammatory nutrition as a pharmacological approach to treat obesity. J Obes. 2011;2011.
Seeni A, Takahashi S, Takeshita K, et al. Suppression of prostate cancer growth by resveratrol in the transgenic rat for adenocarcinoma of prostate (TRAP) model. Asian Pac J Cancer Prev. 2008 Jan-Mar;9(1):7-14.
Seeram NP, Aronson WJ, Zhang Y, et al. Pomegranate ellagitannin-derived metabolites inhibit prostate cancer growth and localize to the mouse prostate gland. J Agric Food Chem. 2007 Sep 19;55(19):7732-7.
SELF. SELF Nutrition Data web page. 2012. Foods highest in Total Omega-6 fatty acids. Available at: http://nutritiondata.self.com/foods-000141000000000000000-w.html. Accessed 10/18/2013.
Selvi BR, Batta K, Kishore AH, et al. Identification of a novel inhibitor of coactivator-associated arginine methyltransferase 1 (CARM1)-mediated methylation of histone H3 Arg-17. J Biol Chem. 2010 Mar 5;285(10):7143-52.
Senthilkumar K, Arunkumar R, Elumalai P, et al. Quercetin inhibits invasion, migration and signalling molecules involved in cell survival and proliferation of prostate cancer cell line (PC-3). Cell Biochem Funct. 2011 Mar;29(2):87-95.
Sethi BK, Chanukya GV, Nagesh VS. Prolactin and cancer: Has the orphan finally found a home? Indian J Endocrinol Metab. 2012 Dec;16(Suppl 2):S195-8.
Shah S, Prasad S, Knudsen KE. Targeting pioneering factor and hormone receptor cooperative pathways to suppress tumor progression. Cancer Res. 2012 Mar 1;72(5):1248-59.
Shamsuddin AM, Yang GY. Inositol hexaphosphate inhibits growth and induces differentiation of PC-3 human prostate cancer cells. Carcinogenesis. 1995 Aug;16(8):1975-9.
Shankar S, Ganapathy S, Chen Q, Srivastava RK. Curcumin sensitizes TRAIL-resistant xenografts: molecular mechanisms of apoptosis, metastasis and angiogenesis. Mol Cancer. 2008;7:16.
Shapiro RH, Johnstone PA. Risk of Gleason grade inaccuracies in prostate cancer patients eligible for active surveillance. Urology. 2012 Sep;80(3):661-6.
Shelton PQ, Ivanowicz AN, Wakeman CM, et al. Active surveillance of very-low-risk prostate cancer in the setting of active treatment of benign prostatic hyperplasia with 5α-reductase inhibitors. Urology. 2013 May;81(5):979-84.
Shen JC, Klein RD, Wei Q, et al. Low-dose genistein induces cyclin-dependent kinase inhibitors and G(1) cell-cycle arrest in human prostate cancer cells. Mol Carcinog. 2000 Oct;29(2):92-102.
Shepherd BE, Redman MW, Ankerst DP. Does finasteride affect the severity of prostate cancer? A causal sensitivity analysis. J Am Stat Assoc. 2008 Dec 1;103(484):1392-404.
Shi Q, Shih CC, Lee KH. Novel anti-prostate cancer curcumin analogues that enhance androgen receptor degradation activity. Anticancer Agents Med Chem. 2009 Oct;9(8):904-12.
Shimamoto T, Ashida S, Yamasaki I, et al. The clinical value of 3 tesla diffusion-weighted magnetic resonance imaging in the diagnosis of prostate cancer. Hinyokika Kiyo. 2012 Mar;58(3):143-8.
Shishodia S, Chaturvedi MM, Aggarwal BB. Role of curcumin in cancer therapy. Curr Probl Cancer. 2007 Jul-Aug;31(4):243-305.
Shishodia S, Singh T, Chaturvedi MM. Modulation of transcription factors by curcumin. Adv Exp Med Biol. 2007 595:127-48.
Shui IM, Mucci LA, Kraft P, et al. Vitamin D-related genetic variation, plasma vitamin D, and risk of lethal prostate cancer: a prospective nested case-control study. J Natl Cancer Inst. 2012 May 2;104(9):690-9.
Shukla M, Gupta K, Rasheed Z, Khan KA, Haqqi TM. Bioavailable constituents/metabolites of pomegranate (Punica granatum L) preferentially inhibit COX2 activity ex vivo and IL-1betainduced PGE2 production in human chondrocytes in vitro. J Inflamm (Lond). 2008 Jun 13;5:9.
Shukla S, Gupta S. Apigenin: A promising molecule for cancer prevention. Pharm Res. 2010 June;27(6):962-78.
Shukla S, Mishra A, Fu P, Maclennan GT, Resnick MI, Gupta S. Up-regulation of insulin-like growth factor binding protein-3 by apigenin leads to growth inhibition and apoptosis of 22Rv1 xenograft in athymic nude mice. FASEB J. 2005 Dec;19(14):2042-44.
Shukla Y, Singh M. Cancer preventive properties of ginger: a brief review. Food Chem Toxicol. 2007 May;45(5):683-90.
Siddiqui IA, Asim M, Hafeez BB, Adhami VM, Tarapore RS, Mukhtar H. Green tea polyphenol EGCG blunts androgen receptor function in prostate cancer. Faseb j. 2011 Apr;25(4):1198-207.
Siddiqui IA, Zaman N, Aziz MH, et al. Inhibition of CWR22Rnu1 tumor growth and PSA secretion in athymic nude mice by green and black teas. Carcinogenesis. 2006 Apr;27(4):833-9.
Siddiqui MZ. Boswellia serrata, a potential antiinflammatory agent: an overview. Indian J Pharm Sci. 2011 May;73(3):255-61.
Siegel R, Naishadham D, Jemal A. Cancer statistics 2012. CA Cancer J Clin. 2012;62:10-29.
Sineh Sepehr K, Baradaran B, Mazandarani M, Khori V, Shahneh FZ. Studies on the cytotoxic activities of Punica granatum L. var. spinosa (Apple Punice) extract on prostate cell line by induction of apoptosis. ISRN Pharm. 2012;2012:547942.
Singh PB, Matanhelia SS, Martin FL. A potential paradox in prostate adenocarcinoma progression: oestrogen as the initiating driver. Eur J Cancer. 2008 May;44(7):928-36.
Singh RP, Agarwal R. A cancer chemopreventive agent silibinin, targets mitogenic and survival signaling in prostate cancer. Mutat Res. 2004b Nov 2;555(1-2):21-32.
Singh RP, Agarwal R. Prostate cancer prevention by silibinin. Curr Cancer Drug Targets. 2004a Feb;4(1):1-11.
Singh RP, Raina K, Sharma G, Agarwal, R. Silibinin inhibits established prostate tumor growth, progression, invasion, and metastasis and suppresses tumor angiogenesis and epithelial-mesenchymal transition in transgenic adenocarcinoma of the mouse prostate model mice. Clin Cancer Res. 2008 Dec 1;14(23):7773-80.
Singh RP, Sharma G, Dhanalakshmi S, Agarwal C, Agarwal R. Suppression of advanced human prostate tumor growth in athymic mice by silibinin feeding is associated with reduced cell proliferation, increased apopotosis, and inhibition of angiogenesis. Cancer Epidemiol Biomarkers Prev. 2003 Sept;12(9):933-9.
Singh RP, Sharma G, Mallikarjuna GU, Dhanalakshmi S, Agarwal C, Agarwal R. In vivo suppression of hormone-refractory prostate cancer growth by inositol hexaphosphate: induction of insulinlike growth factor binding protein-3 and inhibition of vascular endothelial growth factor. Clin Cancer Res. 2004c Jan 1;10(1 Pt 1):244-50.
Sirab N, Robert G, Fasolo V, et al. Lipidosterolic extract of serenoa repens modulates the expression of inflammation relatedgenes in benign prostatic hyperplasia epithelial and stromal cells. Int J Mol Sci. 2013 Jul 10;14(7):14301-20.
SJPHS. St. John Providence Health System. Tumor Factors Defining the Extent and Severity of Cancer. Providence Prostate Cancer Evaluation page. Available at: http://www.stjohnprovidence.org/InnerPage.aspx?PageID=1446. Copyright © 2013. Accessed 10/29/2013.
Skinner HG, Michaud DS, Giovannucci E, et al. Vitamin D intake and the risk for pancreatic cancer in two cohort studies. Cancer Epidemiol Biomarkers Prev. 2006 Sep;15(9):1688-95.
Smith S, Sepkovic D, Bradlow HL, Auborn KJ. 3,3’-Diindolylmethane and genistein decrease the adverse effects of estrogen in LNCaP and PC-3 prostate cancer cells. J Nutr. 2008 Dec;138(12):2379-85.
Smith, M.R., McGovern, F.J., Fallon, M.A. et al. Low bone mineral density in hormone-naive men with prostate carcinoma. Cancer. 2001;91:2238–45.
Soares ND, Teodoro AJ, Oliveira FL, et al. Influence of lycopene on cell viability, cell cycle, and apoptosis of human prostate cancer and benign hyperplastic cells. Nutr Cancer. 2013 Sep 20.
Sofi F, Cesari F, Abbate R, Gensini GF, Casini A. Adherence to Mediterranean diet and health status: meta-analysis. BMJ. 2008 Sep 11;337:a1344.
Song Y, Chavarro JE, Cao Y, et al. Whole milk intake is associated with prostate cancer-specific mortality among U.S. male physicians. J Nutr. 2013 Feb;143(2):189-96.
Sonoda T, Nagata Y, Mori M, et al. A case-control study of diet and prostate cancer in Japan: possible protective effect of traditional Japanese diet. Cancer Sci. 2004 Mar;95(3):238-42.
Soumaoro LT, Iida S, Uetake H, et al. Expression of 5-lipoxygenase in human colorectal cancer. World J Gastroenterol. 2006 Oct 21;12(39):6355-60.
Spratt DE, Zhang C, Zumsteg ZS, Pei X, Zhang Z, Zelefsky MJ. Metformin and prostate cancer: reduced development of castration-resistant disease and prostate cancer mortality. Eur Urol. 2013 Apr;63(4):709-16.
Srivastava SK, Xiao D, Lew KL, et al. Allyl isothiocyanate, a constituent of cruciferous vegetables, inhibits growth of PC-3 human prostate cancer xenografts in vivo. Carcinogenesis. 2003 Oct;24(10):1665-70.
St Sauver JL, Jacobson DJ, McGree ME, Lieber MM, Jacobsen SJ. Protective association between nonsteroidal antiinflammatory drug use and measures of benign prostatic hyperplasia. Am J Epidemiol. 2006 Oct 15;164(8):760-8.
Stangelberger A, Waldert M, Djavan B. Prostate cancer in elderly men. Rev Urol. 2008 Spring;10(2):111-9.
Stark A, Madar Z. Phytoestrogens: a review of recent findings. J Pediatr Endocrinol Metab. 2002 May;15(5):561-72.
Steiner MS, Raghow S. Antiestrogens and selective estrogen receptor modulators reduce prostate cancer risk. World J Urol. 2003 May;21(1):31-6.
Stephan C, Stroebel G, Heinau M, et al. The ratio of prostate-specific antigen (PSA) to prostate volume (PSA density) as a parameter to improve the detection of prostate carcinoma in PSA values in the range of < 4 ng/mL. Cancer. 2005 Sep 1;104(5):993-1003.
Stettner M, Kramer G, Strauss A, et al. Long-term antiepileptic treatment with histone deacetylase inhibitors may reduce the risk of prostate cancer. Eur J Cancer Prev. 2012 Jan;21(1):55-64.
Stone WL, Papas AM. Tocopherols and the etiology of colon cancer. J Natl Cancer Inst. 1997 Jul 16;89(14):1006-14.
Strohmeyer D, Frauscher F, Klauser A, et al. Contrast-enhanced transrectal color doppler ultrasonography (TRCDUS) for assessment of angiogenesis in prostate cancer. Anticancer Res. 2001 Jul- Aug;21(4B):2907-13.
Strum SB, Pogliano D. A Primer on Prostate Cancer: An Empowered Patient’s Guide. Hollywood, FL: The Life Extension Foundation; 2005.
Subbarao K, Jala VR, Mathis S, et al. Role of leukotriene B4 receptors in the development of atherosclerosis: potential mechanisms. Arterioscler Thromb Vasc Biol. 2004 Feb;24(2):369-75.
Sundaram S, Ghosh J. Expression of 5-oxoETE receptor in prostate cancer cells: critical role in survival. Biochem BiophysRes Commun. 2006 Jan 6;339(1):93-8.
Sundram V, Chauhan SC, Ebeling M, Jaggi M. Curcumin attenuates beta-catenin signaling in prostate cancer cells through activation of protein kinase D1. PLoS One. 2012;7(4):e35368.
Supabphol A, Supabphol R. Antimetastatic potential of N-acetylcysteine on human prostate cancer cells. J Med Assoc Thai. 2012 Dec;95 Suppl 12:S56-62.
Sutcliffe S, Giovannucci E, Leitzmann MF, Rimm EB, Stampfer MJ, et al. A prospective cohort study of red wine consumption and risk of prostate cancer. Int J Cancer. 2007;120:1529-35.
Suzuki M, Endo M, Shinohara F, Echigo S, Rikiishi H. Differential apoptotic response of human cancer cells to organoselenium compounds. Cancer Chemother Pharmacol. 2010 Aug;66(3):475-84.
Suzuki R, Rylander-Rudqvist T, Saji S, Bergkvist L, Adlercreutz H, Wolk A. Dietary lignans and postmenopausal breast cancer risk by oestrogen receptor status: a prospective cohort study of Swedish women. Br J Cancer. 2008 Feb 12;98(3):636-40.
Ta N, Walle T. Aromatase inhibition by bioavailable methylated flavones. J Steroid Biochem Mol Biol. 2007 Oct;107(1-2):127-9.
Taccone-Gallucci M, Manca-di-Villahermosa S, Battistini L, et al. N-3 PUFAs reduce oxidative stress in ESRD patients on maintenance HD by inhibiting 5-lipoxygenase activity. Kidney Int. 2006 Apr;69(8):1450-4.
Taira AV, Merrick GS, Galbreath RW, et al. Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis. 2010 Mar;13(1):71-7.
Takase Y, Levesque MH, Luu-The V, et al. Expression of enzymes involved in estrogen metabolism in human prostate. J Histochem Cytochem. 2006 Aug;54(8):911-21.
Tan RS, Salazar JA. Risks of testosterone replacement therapy in ageing men. Expert Opin Drug Saf. 2004 Nov;3(6):599-606.
Tareen B, Summers JL, Jamison JM, et al. A 12 week, open label, phase I/IIa study using apatone for the treatment of prostate cancer patients who have failed standard therapy. Int J Med Sci. 2008;5(2):62-7.
Tchetgen MB, Song JT, Strawderman M, Jacobsen SJ, Oesterling JE. Ejaculation increases the serum prostate-specific antigen concentration. Urology. 1996 Apr;47(4):511-6.
Teiten MH, Gaascht F, Cronauer M, Henry E, Dicato M, Diederich M. Anti-proliferative potential of curcumin in androgendependent prostate cancer cells occurs through modulation of the Wingless signaling pathway. Int J Oncol. 2011 Mar;38 (3):603-11.
Teiten MH, Gaascht F, Eifes S, Dicato M, Diederich M. Chemopreventive potential of curcumin in prostate cancer. Genes Nutr. 2010 Mar;5(1):61-74.
Teiten MH, Gaigneaux A, Chateauvieux S, et al. Identification of differentially expressed proteins in curcumin-treated prostate cancer cell lines. Omics. 2012 Jun;16(6):289-300.
Tewari A, Divine G, Chang P, et al. Long-term survival in men with high grade prostate cancer: a comparison between conservative treatment, radiation therapy and radical prostatectomy--a propensity scoring approach. J Urol. 2007 Mar;177(3):911-5.
Thakur VS, Gupta K, Gupta S. Green tea polyphenols causes cell cycle arrest and apoptosis in prostate cancer cells by suppressing class I histone deacetylases. Carcinogenesis. 2012 Feb;33(2):377-84.
Thirugnanam S, Xu L, Ramaswamy K, Gnanasekar M. Glycyrrhizin induces apoptosis in prostate cancer cell lines DU-145 and LNCaP. Oncol Rep. 2008 Dec;20(6):1387-92.
Thomas RJ, Williams MMA, Sharma H, et al. A double-blind, placebo RCT evaluating the effect of a polyphenol-rich whole food supplement on PSA progression in men with prostate cancer: The U.K. National Cancer Research Network (NCRN) Pomi-T study. J Clin Oncol. 2013;31(suppl):abstract 5008.
Thompson IM Jr, Goodman PJ, Tangen CM, et al. Long-term survival of participants in the prostate cancer prevention trial. N Engl J Med. 2013 Aug 15;369(7):603-10.
Thompson IM, Goodman PJ, Tangen CM, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003 Jul 17;349(3):215-24.
Thompson IM, Pauler DK, Goodman PJ, et al. Prevalence of Prostate Cancer among Men with a Prostate-Specific Antigen Level ≤ 4.0 ng per Milliliter. N. Engl. J Med. 2004;350:2239-2246.
Tisman G and Strum SB. The Prostate Cancer Tools II (PCT II) Software Program as an Aid in Prostate Cancer Patient Counseling. PCR Insights. Published by Prostate Cancer Research Institute. Available at: http://www.prostate-cancer.org/PDFs/Is4-2.pdf. 2001 May; 4(2): 1-20.
Tombal B, Lecouvet F. Modern detection of prostate cancer’s bone metastasis: Is the bone scan era over? Adv Urol. 2012;2012:893193.
Tomimoto A, Endo H, Sugiyama M, et al. Metformin suppresses intestinal polyp growth in ApcMin/+ mice. Cancer Sci. 2008 Nov;99(11):2136-41.
Traka M, Gasper AV, Melchini A, et al. Broccoli consumption interacts with GSTM1 to perturb oncogenic signalling pathways in the prostate. PLoS One. 2008;3(7):e2568.
Traka MH, Spinks CA, Doleman JF, et al. The dietary isothiocyanate sulforaphane modulates gene expression and alternative gene splicing in a PTEN null preclinical murine model of prostate cancer. Mol Cancer. 2010;9:189.
Trejo-Solis C, Pedraza-Chaverri J, Torres-Ramos M, et al. Multiple molecular and cellular mechanisms of action of lycopene in cancer inhibition. Evid Based Complement Alternat Med. 2013;2013:705121.
Tsui KH, Feng TH, Lin CM, Chang PL, Juang HH. Curcumin blocks the activation of androgen and interlukin-6 on prostatespecific antigen expression in human prostatic carcinoma cells. J Androl. 2008 Nov-Dec;29(6):661-8.
Tu WH, Thomas TZ, Masumori N, et al. The loss of TGF-beta signaling promotes prostate cancer metastasis. Neoplasia. 2003 May-Jun;5(3):267-77.
Tuck SP, Hanusch B, Walker J, Datta HK. Prostate cancer and osteoporosis. Curr Osteoporos Rep. 2013 Mar;11(1):11-20.
Tunstall RG, Sharma RA, Perkins S, et al. Cyclooxygenase-2 expression and oxidative DNA adducts in murine intestinal adenomas: modification by dietary curcumin and implications for clinical trials. Eur J Cancer. 2006 Feb;42(3):415-21.
UCCM. University of Cincinnati (UC) College of Medicine. Radiology. Prostate MRI Information: Hot It is Done. UC College of Medicine web page. Available at: http://www.med.uc.edu/radiology/research/prostateimaging/mriprocedure.aspx. Copyright © 2013. Accessed 10/25/2013.
UCF. Urology Care Foundation. Advanced Prostate Cancer. Available at: http://www.urologyhealth.org/urology/index.cfm?article=149. Last updated 3/2013. Accessed 10/29/2013.
UMMC. University of Maryland Medical Center. Gamma-linolenic acid. Available at: http://umm.edu/health/medical/altmed/supplement/gammalinolenic-acid. Last updated 6/24/2013a. Accessed 10/21/2013.
UMMC. University of Maryland Medical Center. Omega-6 fatty acids. Available at: http://umm.edu/health/medical/altmed/supplement/omega6-fatty-acids. Last updated 6/20/2013b. Accessed 10/18/2013.
UMMSM. University of Miami Leonard M. Miller School of Medicine. UM Researchers Present Dramatic Cancer Findings. University of Miami Miller School of Medicine web page. Available at: http://www.med.miami.edu/news/view.asp?id=403. Accessed 10/21/2013.
Unger JM, Hershman DL, Till C, Tangen CM, Barlow WE, Ramsey SD, . . . Thompson IM, Jr. Using Medicare Claims to Examine Long-term Prostate Cancer Risk of Finasteride in the Prostate Cancer Prevention Trial. Journal of the National Cancer Institute. Mar 9 2018.
UPMC. UPMC CancerCenter. Prostate Cancer. Transrectal Ultrasound. Available at: http://www.upmccancercenter.com/cancer/prostate/biopsyultrasound.cfm. 2013. Accessed 10/24/2013.
USDA. U.S. Department of Agricuture. Isoflavones contents of food. Taken from USDA Database: Sources of isoflavones. Available at: http://www.isoflavones.info/isoflavones-content.php. 2013. Accessed 10/21/2013.
USDC. U.S. Department of Commerce. United States Census Bureau. Age and Sex. Age and Sex Composition in the United States: 2011. Available at: http://www.census.gov/population/age/data/2011comp.html. Last updated 11/28/2012. Accessed 10/28/2013.
USPSTF. U.S. Preventive Services Task Force. Summaries for patients. Screening for HIV: U.S. Preventive Services Task Force Recommendation Statement. Available at: http://www.uspreventiveservicestaskforce.org/uspstf13/hiv/hivfinalrs.htm. (First published in Ann Intern Med, 2013;159(1) [No authors listed]).
van den Bemd GJ, Chang GT. Vitamin D and vitamin D analogs in cancer treatment. Curr Drug Targets. 2002 Feb;3(1):85-94.
Van Neste L, Herman JG, Otto G, Bigley JW, Epstein JI, Van Criekinge W. The epigenetic promise for prostate cancer diagnosis. Prostate. 2012 Aug 1;72(11):1248-61.
Vayalil PK, Mittal A, Katiyar SK. Proanthocyanidins from grape seeds inhibit expression of matrix metalloproteinases in human prostate carcinoma cells, which is associated with the inhibition of activation of MAPK and NF kappa B. Carcinogenesis. 2004;25:987-95.
Vazquez-Martin A, Oliveras-Ferraros C, Del Barco S, Martin-Castillo B, Menendez JA. The anti-diabetic drug metformin suppresses self-renewal and proliferation of trastuzumab-resistant tumor-initiating breast cancer stem cells. Breast Cancer Res Treat. 2011 Apr;126(2):355-64.
Venkateswaran, V, Klotz, LH, Fleshner, NE. Selenium modulation of cell proliferation and cell cycle biomarkers in human prostate carcinoma cell lines. Cancer Res. 2002;62:2540-5.
Vessella RL, Lange PH, Partin AW, et al. Probability of prostate cancer detection based on results of a multicenter study using the AxSYM free PSA and total PSA assays. Urology. 2000 Jun;55(6):909-14.
Vicinanza R, Zhang Y, Henning SM, Heber D. Pomegranate juice metabolites, ellagic acid and urolithin A, synergistically inhibit androgen-independent prostate cancer cell growth via distinct effects on cell cycle control and apoptosis. Evid Based Complement Alternat Med. 2013;2013:247504.
Vij U, Kumar A. Phyto-oestrogens and prostatic growth. Natl Med J India. 2004 Jan;17(1):22-6.
von der Recke P, Hansen MA, Overgaard K. et al. The impact of degenerative conditions in the spine on bone mineral density and fracture risk prediction. Osteoporos Int. 1996;6:43–9.
von Holtz RL, Fink CS, Awad AB. Beta-Sitosterol activates the sphingomyelin cycle and induces apoptosis in LNCaP human prostate cancer cells. Nutr Cancer. 1998;32(1):8-12.
Wang D, Dubois RN. Prostaglandins and cancer. Gut. 2006 Jan;55(1):115-22.
Wang D, Wang H, Shi Q, et al. Prostaglandin E(2) promotes colorectal adenoma growth via transactivation of the nuclear peroxisome proliferator-activated receptor delta. Cancer Cell. 2004 Sep;6(3):285-95.
Wang L, Ho J, Glackin C, Martins-Green M. Specific pomegranate juice components as potential inhibitors of prostate cancer metastasis. Transl Oncol. 2012 Oct;5(5):344-55.
Wang LQ. Mammalian phytoestrogens: enterodiol and enterolactone. J Chromatogr B Analyt Technol Biomed Life Sci. 2002 Sep 25;777(1-2):289-309.
Wang XL, Hur HG, Lee JH, Kim KT, Kim SI. Enantioselective synthesis of S-equol from dihydrodaidzein by a newly isolated anaerobic human intestinal bacterium. Appl Environ Microbiol. Jan 2005;71(1):214-9.
Webber MM, Waghray A, Bello D. Prostate-specific antigen, a serine protease, facilitates human prostate cancer cell invasion. Clin Cancer Res. 1995 Oct;1(10):1089-94.
WebMD. Deep Vein Thrombosis Health Center. Doppler Ultrasound. Available at: http://www.webmd.com/dvt/doppler-ultrasound. Last updated 12/4/2010. Accessed 10/24/2013.
WebMD. FDA Rejects Green Tea Cancer Claims. 6/7/2005. Published on FOX NEWS web page. Available at: http://www.foxnews.com/story/2005/07/06/fda-rejects-green-tea-cancer-claims/. Accessed 10/18/2013.
WebMD. Find a Vitamin or Supplement. N-ACETYL CYSTEINE. Overview. Available at: http://www.webmd.com/vitamins-supplements/ingredientmono-1018-N-ACETYL%20CYSTEINE.aspx?activeIngredientId=1018&activeIngredientName=N-ACETYL%20CYSTEINE. Accessed 10/21/2013.
Webster J, Piscitelli G, Polli A, et al. Dose-dependent suppression of serum prolactin by cabergoline in hyperprolactinaemia: a placebo controlled, double blind, multicentre study. Clin Endocrinol (Oxf). 1992 Dec;37(6):534-41.
Wen J, Zhao Y, Li J, et al. Suppression of DHT-induced paracrine stimulation of endothelial cell growth by estrogens via prostate cancer cells. Prostate. 2013 Jul;73(10):1069-81.
Wertz K. Lycopene effects contributing to prostate health. Nutr Cancer. 2009;61(6):775-83.
WHF. The world’s healthiest foods. Omega-3 fatty acids. Nutrient Rating Chart. Copyright © 2001-2013 The George Mateljan Foundation. Available at: http://www.whfoods.com/genpage.php?tname=nutrient&dbid=84#foodchart. Accessed 10/18/2013.
Williams CD, Whitley BM, Hoyo C, et al. A high ratio of dietary n-6/n-3 polyunsaturated fatty acids is associated with increased risk of prostate cancer. Nutr Res. 2011 Jan 31(1):1-8.
Wilt TJ, Ishani A, Rutks I, MacDonald R. Phytotherapy for benign prostatic hyperplasia. Public Health Nutr. 2000 Dec;3(4A):459-72.
Wright JL, Stanford JL. Metformin use and prostate cancer in Caucasian men: results from a population-based case-control study. Cancer Causes Control. 2009 Nov;20(9):1617-22.
Wu KK. Aspirin and other cyclooxygenase inhibitors: new therapeutic insights. Semin Vasc Med. 2003 May;3(2):107-12.
Xiao D, Srivastava SK, Lew KL, et al. Allyl isothiocyanate, a constituent of cruciferous vegetables, inhibits proliferation of human prostate cancer cells by causing G2/M arrest and inducing apoptosis. Carcinogenesis. 2003 May;24(5):891-7.
Xing N, Chen Y, Mitchell SH, Young CY. Quercetin inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells. Carcinogenesis. 2001 Mar;22(3):409-14.
Xu S, Gao JP, Zhou WQ. Cyclooxygenase-2 and cyclooxygenase-2 inhibitors in prostate cancer. Zhonghua Nan Ke Xue. 2008 Nov;14(11):1031-4.
Yan L, Spitznagel EL. Soy consumption and prostate cancer risk in men: a revisit of a meta-analysis. Am J Clin Nutr. 2009 Apr;89(4):1155-63.
Yang GS, Chen ZD. Comparative studies of the expression of estrogen receptor-alpha and estrogen receptor-beta in prostatic carcinoma. Zhonghua Wai Ke Za Zhi. 2004 Sep 22;42(18):1111-5.
Yang L, Zhang LY, Chen WW, et al. Inhibition of the expression of prostate specific antigen by curcumin. Yao Xue Xue Bao. 2005 Sep;40(9):800-3.
Yang P, Cartwright C, Chan D, Vijjeswarapu M, Ding J, Newman RA. Zyflamend-mediated inhibition of human prostate cancer PC3 cell proliferation: effects on 12-LOX and Rb protein phosphorylation. Cancer Biol Ther. 2007 6(2):228-36.
Yang Y, Ikezoe T, Zheng Z, Taguchi H, Koeffler HP, Zhu WG. Saw Palmetto induces growth arrest and apoptosis of androgen-dependent prostate cancer LNCaP cells via inactivation of STAT 3 and androgen receptor signaling. Int J Oncol. 2007 Sep;31(3):593-600.
Yavuz BB, Ozkayar N, Halil M, et al. Free testosterone levels and implications on clinical outcomes in elderly men. Aging Clin Exp Res. 2008 Jun;20(3):201-6
Ye YN, Liu ES, Shin VY, Wu WK, Cho CH. Contributory role of 5-lipoxygenase and its association with angiogenesis in the promotion of inflammation-associated colonic tumorigenesis by cigarette smoking. Toxicology. 2004 Oct 15;203(1-3):179-88.
Yokota T, Matsuzaki Y, Koyama M, et al. Sesamin, a lignan of sesame, down-regulates cyclin D1 protein expression in human tumor cells. Cancer Sci. 2007 Sep;98(9):1447-53.
Yoo J, Lee YJ. Aspirin enhances tumor necrosis factor-related apoptosis-inducing ligand-mediated apoptosis in hormone-refractory prostate cancer cells through survivin down-regulation. Mol Pharmacol. 2007 Dec;72(6):1586-92.
Yoshimura R, Matsuyama M, Mitsuhashi M, et al. Relationship between lipoxygenase and human testicular cancer. Int J Mol Med. 2004 Mar;13(3):389-93.
Yousef FM, Jacobs ET, Kang PT, et al. Vitamin D status and breast cancer in Saudi Arabian women: case-control study. Am J Clin Nutr. 2013 Jul;98(1):105-10.
Yoysungnoen P, Wirachwong P, Bhattarakosol P, Niimi H, Patumraj S. Effects of curcumin on tumor angiogenesis and biomarkers, COX-2 and VEGF, in hepatocellular carcinoma cell-implanted nude mice. Clin Hemorheol Microcirc. 2006 34(1-2):109-15.
Yu C, Gong AY, Chen D, Solelo Leon D, Young CY, Chen XM. Phenethyl isothiocyanate inhibits androgen receptor-regulated transcriptional activity in prostate cancer cells through suppressing PCAF. Mol Nutr Food Res. 2013.
Yu W, Park SK, Jia L, et al. RRR-gamma-tocopherol induces human breast cancer cells to undergo apoptosis via death receptor 5 (DR5)-mediated apoptotic signaling. Cancer Lett. 2008 Feb 8;259(2):165-76.
Yuan H, Young CY, Tian Y, Liu Z, Zhang M, Lou H. Suppression of the androgen receptor function by quercetin through proteinprotein interactions of Sp1, c-Jun, and the androgen receptor in human prostate cancer cells. Mol Cell Biochem. 2010 Jun;339(1- 2):253-62.
Zaichick V, Sviridova TV, Zaichick SV. Zinc in the human prostate gland: normal, hyperplastic and cancerous. Int Urol Nephrol. 1997;29(5):565-74.
Zaidman BZ, Wasser SP, Nevo E, Mahajna J. Androgen receptor-dependent and -independent mechanisms mediate Ganoderma lucidum activities in LNCaP prostate cancer cells. Int J Oncol. 2007 Oct;31(4):959-67.
Zaidman BZ, Wasser SP, Nevo E, Mahajna J. Coprinus comatus and Ganoderma lucidum interfere with androgen receptor function in LNCaP prostate cancer cells. Mol Biol Rep. 2008 Jun;35(2):107-17.
Zakikhani M, Dowling RJ, Sonenberg N, Pollak MN. The effects of adiponectin and metformin on prostate and colon neoplasia involve activation of AMP-activated protein kinase. Cancer Prev Res (Phila Pa). 2008 Oct;1(5):369-75.
Zhang F, Altorki NK, Mestre JR, Subbaramaiah K, Dannenberg AJ. Curcumin inhibits cyclooxygenase-2 transcription in bile acidand phorbol ester-treated human gastrointestinal epithelial cells. Carcinogenesis. 1999 Mar;20(3):445-51.
Zhang J, Hsu BAJ, Kinseth BAM, Bjeldanes LF, Firestone GL. Indole-3-carbinol induces a G1 cell cycle arrest and inhibits prostate-specific antigen production in human LNCaP prostate carcinoma cells. Cancer. 2003 Dec 1;98(11):2511- 20.
Zhang L, Zhang WP, Hu H, et al. Expression patterns of 5-lipoxygenase in human brain with traumatic injury and astrocytoma. Neuropathology. 2006 Apr;26(2):99-106.
Zhang Z-F, Winton MI, Rainey C, et al. Boron is associated with decreased risk of human prostate cancer. FASEB J. 2001;15:A1089.
Zhi H, Zhang J, Hu G, et al. The deregulation of arachidonic acid metabolism-related genes in human esophageal squamous cell carcinoma. Int J Cancer. 2003 Sep 1;106(3):327-33.
Zhou JR, Gugger ET, Tanaka T, Guo Y, Blackburn GL, Clinton SK. Soybean phyto- chemicals inhibit the growth of trans-plantable human prostate carcinoma and tumor angiogenesis in mice. J Nutr. 1999 Sep;129(9):1628-35.
Zi X, Agarwal R. Silibinin decreases prostate-specific antigen with cell growth inhibition via G1 arrest, leading to differentiation of prostate carcinoma cells: implications for prostate cancer intervention. Proc Natl Acad Sci USA. 1999 Jun 22;96(13):7490-5.
