Life Extension Magazine®
|
Roy L. Walford, M.D. The only scientific proof of slowing aging is extending maximum life span, which calorie restriction can do in lab animals. Here's why it may do the same in humans. Scientists have discovered an almost certain method to achieve a very substantial retardation of the aging process, to delay aging of the brain, skin and muscle, and to live longer, healthier and younger. It involves calorie restriction, the so-called CR diet, one of the most extensively studied of all gerontologic phenomena, and we now have good clues as to how it works. First, a little background. Each species exhibits a characteristic maximum life span. For humans, it's 110 years, except for very occasional people like the recently deceased French woman, Jeanne Calmet, who was 122 at the time of her death last year. But generally, out of millions and millions of people, 110 has always been a fixed point for our species. By contrast, average life span (the age at which half of the population has died off, but half still remains alive) can vary a lot. In ancient Rome it was 22 years; in today's Western countries it's about 75 years. The increase is due to better hygiene and medicine, etc., but it's not due to any retardation of aging. The measure of aging in a population is not the average life span, but rather the maximum life span, and that hasn't changed. Can we extend maximum life span? Yes, by nutritional manipulation in a wide variety of species, and almost certainly in humans. With a naturally long-lived strain of mice, half were kept on a normal diet and half on a diet restricted in calories but adequate in everything else (sometimes referred to as a CRAN diet, for "calorie restriction with adequate nutrition"). The maximum life span of the mice on the normal diet was 41 months-about 110 human years. However, for the calorie-restricted mice, maximum life span was pushed out to 56 months-150 human years! Similar studies in mice, rats, fish and other species have been done in numerous university laboratories during the past 50 or more years, and they all agree. The observation is solid: A CR diet increases the maximum life span characteristic of the species. It also, by the way, increases the population's average life span, so the two together translate into longer and healthier life. For years I have emphasized this species-specific maximum life span as a criterion in claims for anti-aging therapies. There are strains of mice that have a maximum life span much shorter than 41 months. That's generally because they experience a high incidence of one or several diseases. Let's say the maximum life span of one such strain is 25 months, but when you give them "treatment X," the maximum life span goes out to 36 months. So you can legitimately claim that the treated mice lived 44 percent longer than the controls. But that may simply be prevention or cure of disease, not necessarily anti-aging. Of course, if you happen to be that particular strain of mouse (or of human), 36 is better than 25. Therefore, "treatment X" may be worthwhile , but it's not necessarily anti-aging. Before accepting "treatment X" as true anti-aging, I want to see it produce a 45 to 50-month-old mouse, and so far the CR diet is the only method shown to produce such mice. This fact, plus the gathering evidence that it will work in humans, is why the CR diet is now one of the major areas of research in academic gerontology. Indeed, five of the 10 members of the Life Extension Foundation's Scientific Advisory Board have worked or are working partly in this area. And it is receiving major emphasis from the National Institute on Aging. What is the calorie restriction regimen? Simply that giving animals fewer calories than they would consume by choice makes them live longer and with fewer diseases. But these fewer calories can't come from a mouse equivalent of McDonald's or Burger King. Not good enough in quality! While reducing the calories, you must increase the quality of the diet so that essential nutrients like vitamins, minerals, amino acids, and so forth are not reduced. The animals must not be malnourished. This explains the reason the semi-starved populations in parts of the world don't live longer-they are calorie restricted and malnourished. The "adequate nutrition" side of CR is essential. Given that, the result is that the fewer the calories, the longer the life (down to about 50 percent restriction), with other beneficial effects following proportionately. Interestingly, CR is not an all-or-nothing phenomenon. Even 10 percent restriction has a very measurable beneficial effect. Of course, there is a lower limit. Much below 50 percent takes you into actual calorie starvation, and the death rate increases. What is the mechanism behind all these effects? If we knew the mechanism, perhaps we could achieve the same results by an easier method than restricting food intake. But here we have an embarrassment of possibilities. The mechanism of CR can be interpreted according to most of the current theories of aging. CR increases DNA repair, definitely decreases oxidative damage, and probably (there is some debate here) increases the body's own antioxidant defense systems, increases the heat shock protein response, is associated with less glycation than seen in controls, and delays age-related immunological decline as shown by virtually all immune functional tests. Further, it's the strongest cancer-preventive technique known, although disease prevention is a side effect of the age-retardation. On a very practical level, in the one rigorous human study conducted inside Biosphere 2 [the sealed-in-glass experimental earth environment in Arizona where the author was chief of medical operations from 1990-1994], CR sharply lowered blood cholesterol by up to 35 percent, blood sugar and blood insulin by 15 to 20 percent, blood pressure by 20 percent or more, and induced other changes paralleling those seen in CR animals. Dr. Richard Weindruch and I postulated some years ago that the mechanism is related to an increase in "metabolic efficiency." This can be thought of as referring to less "friction" in the body's generation of energy. Others have referred to this concept as "improved glucose fuel use." From the standpoint of evolutionary theory-another approach to the same question-it has been proposed that CR kicks into play an "adaptive response" whereby animals, faced by episodic periods of food shortage in the wild, shift their allocation of metabolic energy from growth and reproduction to maintenance and repair and so survive the period of deprivation. Along these same lines, Dr. Steve Spindler and I have presented evidence that this "adaptive response" to CR shows certain features of hibernation . . . we mean biochemical and metabolic features, not the enormous drop in body temperature that characterizes hibernation in some (but not all) species, although there is in fact a drop of 1 to 2 degrees in calorie-restricted rodents, monkeys and humans. Recent studies from a rather unexpected quarter, studies on the tiny vinegar worm, C. elegans, may allow us to tie the above hodgepodge of fact and speculation together. When properly manipulated, a particular gene in the vinegar worm, a gene called daf-2 (never mind the origin of the quaint terminology) increases the worm's life span by up to 300 percent (equal to 330 years in human terms). What does this gene do? Well, it has recently been found by scientists at Harvard Medical School that daf-2 plays a key role in the insulin-glucose metabolic pathway. We know that insulin and glucose levels are substantially reduced in calorie restricted animals, including humans. In their benchmark article, the Harvard scientists comment, "Life span regulation by insulin- induced metabolic control [i.e., their work on the worm] is analogous to mammalian longevity enhancement induced by caloric restriction, suggesting a general link between metabolism, diapause (hibernation in higher animals), and longevity." I will add here that daf 2-like genes exist in mammals, and are involved in similar metabolic pathways. So what, finally, is CR's mechanism? My best guess, weighing all the above evidence, is that it's an evolutionarily evolved adaptive response, with overtones of the hibernation response, that operates through genes involved in the insulin/glucose metabolic cycle. Of course, the full cycle of interacting units is going to be complicated to unravel, but hopefully we now know where to look and (roughly) what to look for. That's a big step forward. Will CR retard aging in primates, including humans, and do all the other wonderful things it does in lower animals? It almost certainly will. I say "almost" because it has not been applied in either monkey or human studies long enough to demonstrate a change in maximum life span. Monkey studies will answer this first, and they are ongoing in three different laboratories (University of Wisconsin, University of Maryland, and the National Institute on Aging), although it may be 10 to 15 years before we have unequivocal results. So much for the "almost." The "certainly" in my answer is because, 1) as so far tested, CR works across the whole spectrum of the animal kingdom, so it would indeed be surprising if it did not work in humans; and 2) studies on monkeys in the above three laboratories, and by me on the humans secluded for two years inside Biosphere 2, show quite clearly that most or all of the physiologic and biochemical changes seen in CR rodents are also found in CR primates. If this continues to be the case, but if CR does not retard aging in primates, then one could argue that neither free radical damage, nor DNA damage, nor glycation, nor immune dysfunction and decline, nor pertinent endocrine shifts, nor metabolic efficiency as reflected in insulin/glucose metabolism, nor the evolutionary adaptive response play significant roles in the genesis of aging. And it just doesn't seem very likely that everything modern scientists have postulated about the cause of aging is wrong. Therefore I do not hesitate to say, "Almost certainly it will work in humans." Perhaps the most curious observation in the whole field of CR research is this: while its effect on life span and disease has been documented repeatedly since 1935, while well over 500 articles in peer-review scientific journals of the highest repute have focused on the various aspects of this remarkable phenomenon, and while CR has been for some time a major interest of the National Institute on Aging, you will not to my knowledge find it even mentioned in any of those weighty, modern textbooks on nutrition. On this issue the academic nutrition community is-and never was catch-phrase more on target-"out to lunch." About the author Roy Walford, M.D., professor of pathology at UCLA, is the originator of the immunologic theory of aging. His research has examined the impact on aging of calorie restriction, body temperature reduction and the Major Histocompatibility Gene Complex. He is a Life Extension Foundation advisory board member. Further reading
Walford, R.L., and Spindler, S.R.: "The response to caloric restriction in mammals shows features also common to hibernation: a cross-adaptation hypothesis." J. Gerontology 52A:B pages 179-183, 1997. Weindruch, R: "Caloric restriction and aging." Scientific American 274:46-52, 1996. Weindruch, R., and Walford, R.L.: "The Retardation of Aging and Disease by Dietary Restriction." Charles C. Thomas, Springfield, 1988 Roush, W.: "Worm longevity gene cloned." Science 277:897-898, 1997. End article boxed words
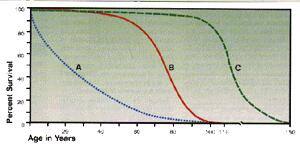
A naturally long-lived strain of mouse was divided into two groups. The maximum life span of the control group, fed a normal diet, (line B) was about 41 months, while the calorie restricted group (line C) enjoyed a maximum life span of 56 months-the equivalent of 150 human years.
|