Life Extension Magazine®
Fear of Alzheimer's disease, with its relentless erosion of memory and personality, gnaws at the edges of baby boomers' minds just as surely as they are aging. A little uneasiness is understandable, given that the primary risk factor for developing Alzheimer's disease (AD) is age.1,2 After all, no one wants to lose her mind. Risk of succumbing to dementia is relatively small in early old age, but the odds climb inexorably with each passing year. And there is evidence that some cognitive decline is inevitable in the elderly, even in the absence of other diseases such as Alzheimer's.3 In the developed world, it's estimated that dementia afflicts just 1.5% of the population at 65 years. But dementia increases exponentially: by 80 years of age 30% will be stricken.4-7 Dementia may or may not be associated with concurrent AD. For reasons that remain unclear, dementia associated with AD strikes females more commonly than males, and AD dementia is more common in the developed countries of the West. Vascular dementia is a term used to describe loss of mental function due to pathologies of the cerebrovascular system. Stroke is but one example. All such pathologies involve interruptions of normal blood flow to the brain. Hope on the horizon Fortunately, researchers have discovered some compounds that may slow, halt, or even reverse the otherwise relentless deterioration of brain structures that begins with mild cognitive impairment and often progresses to the full-blown dementia of Alzheimer's disease or vascular dementia. One promising newer compound is an acetylcholine precursor derived from soy: L-alpha glycerylphosphorylcholine (GPC). The brain converts GPC into acetylcholine, an essential neurotransmitter whose decline is centrally implicated in brain aging and neurodegeneration. Available only by prescription in Europe, where it is sold under the trade name, Gliatilin, GPC is currently available to Americans as an over-the-counter dietary supplement. It is a derivative of the natural compound phosphatidylcholine. Interest in GPC among gerontologists, neurologists and other researchers appears to be growing. As the vast baby boom generation continues to age, GPC may well become a popular hedge against the forgetfulness and confusion that inevitably afflict the elderly. Make no mistake: researchers generally agree that even the sharpest (and most fortunate) of minds will eventually experience some decline in normal thought or cognition due to the ravages of old age. Even in the absence of concurrent diseases such as AD or vascular dementia, we can all expect to experience some decline in memory, attention and general cognition as we age, if steps are not take to intervene. 3,11 A call for early intervention Late last year, in an article published in the highly respected British medical journal, The Lancet, authors Alistair Burns M.D. and Michael Zaudig M.D. argued that memory complaints are "an almost universal early symptom of dementia". Furthermore, say the authors, "mild cognitive impairment defines a transitional stage between normal aging and dementia."19,20 They note that 25 million people across the globe are afflicted with dementia. Mild cognitive impairment "represents an opportunity for early intervention," say Burns and Zaudig. They believe that mild cognitive impairment deserves additional attention from the medical community as a target for early dementia-prevention strategies. They suggest that doctors should take complaints of memory loss more seriously. They note that memory difficulties represent an early warning sign identifying patients who may be at high risk of developing dementia. Lucilla Parnetti M.D., PhD, an Italian neuroscience researcher who co-authored a retrospective analysis of GPC research, shares Burns' and Zaudig's faith in early intervention. Dr. Parnetti believes that a firm case can be made for pre-emptive treatment with GPC. She notes that early acetylcholine depletion and cholinergic receptor demise begins approximately in the fourth or fifth decades of life, progressing thereafter. Is it possible that GPC may be capable of preventing these detrimental conditions of aging outright? Dr. Parnetti says, "It might be." Her guarded optimism is understandable. Research into intervention therapy is ongoing, and much work remains to be done. Why it works It's long been established that aging brains are characterized by a deficiency in the neurotransmitter acetylcholine. Aging brains also lose cholinergic receptors; structures within the nerves that receive and propagate the messages transmitted by acetylcholine.10 The resulting erosion of memory may be exaggerated by the presence of other pathological conditions such as Alzheimer's disease, or vascular dementia.3 Acetylcholine is an extremely important messenger molecule that facilitates numerous activities within the brain, particularly in areas associated with memory, learning and attention. In the body, acetylcholine is crucial to muscular control. Among Alzheimer's patients with dementia, acetylcholine transmission in the hippocampus is severely impaired. Cholinergic neurons die at an increasingly rapid pace, with dementia the inevitable dreaded result. Even in otherwise healthy people, short-term memory loss and declining thought processes may be due to the malfunctioning of this messenger/receptor system. The rationale for GPC therapy goes back to a hypothesis, developed more than three decades ago, that a decline in this important neurotransmitter - and a concurrent decrease in the number of neurons that are its intended target - is responsible for a range of cognitive deficits.10 By artificially bolstering acetylcholine levels in the brain, researchers reasoned, we might be able to reverse those cognitive deficits and brain structure changes.3 Scientists enthusiastically investigated the potential therapeutic benefits of natural acetylcholine precursor compounds such as lecithin and choline, but results were disappointing.11-14 Choline in particular appeared to hold great promise initially. In the body choline serves not only as a precursor to acetylcholine, but also as one of the building blocks for phosphatidylcholine, a phospholipid that is an important component of brain cell membranes. Without it, membranes lose structural integrity and neurons wither. The body partially compensates for acetylcholine deficits by "raiding" existing phosphatidylcholine for conversion to acetylcholine. While it's an intriguing example of the body's remarkable adaptability and economy, this reallocation ultimately serves only to weaken cell membrane integrity by depleting phosphatidylcholine stores. Soon after it became clear that choline and lecithin were not significantly reversing cognitive decline, researchers began looking at GPC. Results were dramatically different. Finally, a precursor with the ability to rejuvenate acetylcholine levels, receptors, and neuronal structural integrity had been discovered. Numerous clinical trials have scrutinized the efficacy and safety of GPC in animal models and in humans.16-18 These studies - large and small, controlled and informal - have universally demonstrated favorable results and an excellent safety and tolerability profile.3 Past studies have looked at everything from changes in learning, memory and brain structure in rats, to stroke-induced cognitive deficits in humans, to induced - and restored - memory function deficits in laboratory animals. By early 2001, a retrospective analysis of published clinical trials involving 4,054 patients found that overall, GPC improved patients' clinical conditions.3 A majority of the ten studies devoted to dementia disorders were controlled trials that compared the efficacy of GPC to either placebo or a reference drug. Lucilla Parnetti, MD, PhD, co-author of the analysis writes, "Administration of [GPC] significantly improved patient clinical condition…results were superior or equivalent to those observed in control groups under active treatment and superior to the results observed in placebo groups." GPC has been directly compared to some popular, alledgedly nootropic (or brain-enhancing) substances; oxiracetam and acetyl-L-carnitine. Cognitive improvement scores among GPC patients were similar to those seen in patients taking oxiracetam.29 Tests comparing the efficacy of GPC versus acetyl-L-carnitine demonstrated that GPC delivers superior cognitive benefits.30 Research on GPC's therapeutic effect in cases of vascular dementia caused by stroke suggests that GPC may well promote functional recovery. These studies, involving 2,484 patients in three trials who had suffered cerebrovascular stroke and/or transient ischemic attacks were uncontrolled, however, so additional research is in order to fully establish GPC's efficacy in these situations.3 |
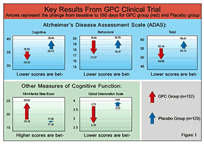
| Promising new research New results from a multi-center clinical trial further bolster the case for GPC supplementation.1 Researchers in Mexico concluded that GPC improves cognitive function significantly among patients with mild to moderate cognitive impairment. The impairment was attributed to probable early Alzheimer's disease. With an enrollment of only 261 patients, the study was fairly small. But the results are nevertheless encouraging. They echo positive findings from previous studies that have examined the efficacy and safety of GPC in humans.3 In the Mexico study, participants received 1200 mg GPC per day, for six months. Subjects ranged in age from 60 to 80 years. All had suffered documented declines in cognitive function before entering the study. Patients were randomly assigned to receive either GPC or dummy placebo pills. Baseline psychometric tests, administered to both patient groups before the double-blind trial began, showed no statistically significant differences. In essence, all subjects entered the study at a more or less comparable level of early cognitive decline. On a variety of scales designed to assess the progression of Alzheimer's disease, GPC patients scored more favorably than patients from the control group, all of whom received placebo. A favorable score on the Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-Cog) was considered the primary objective indicator of the oral supplement's effectiveness. At the end of the six-month trial, GPC patients' average scores had decreased by 3.2 points, while placebo patients' scores increased by 2.9 points in the same period. A lower score on the ADAS-Cog is highly favorable, indicating less cognitive impairment. In other words, the mental status of patients on GPC therapy improved, while those receiving no therapy (placebo) worsened. These findings are comparable to the results obtained with the use of the prescription drugs Aricept™(donepezil HCl) Exelon™ (rivastigmine tartrate) and Reminyl™ (galantamine hydrochloride) in the treatment of AD patients, and with fewer side effects. Other efficacy indicators were similarly favorable (see Figure 1, below). GPC patients' scores on all parameters of the Alzheimer's Disease Assessment Scale (ADAS-Total), Mini-Mental State Examination (MMSE), Global Deterioration Scale (GDS), and Clinical Global Impression (CGI) scale consistently improved versus baseline scores after three and six months of therapy, while placebo-group patients' scores either remained unchanged or worsened. After six months, improvement among GPC patients was also documented using the Alzheimer's Disease Assessment Scale-Behavioral Subscale (ADAS-Behav), and the Global Improvement Scale (GIS). Encouraging results These results are heartening, given that the trial was tightly controlled. To rule out any interference due to expectations, researchers and participants alike were "blinded"; no one involved knew whether a given patient was receiving GPC or placebo, until after all data had been gathered. To further rule out confounding factors, potential participants were assessed for depression, alcoholism, anemia, systemic diseases (such as AIDS or cancer), stroke in the previous six months, neurological disorders (other than mild to moderate Alzheimer's disease) or certain vitamin deficiencies. Patients suffering from any of these additional conditions were excluded from participation. Tolerability data were similarly encouraging. In most patients, drug-related adverse events were mild; no patients were withdrawn due to adverse events. The most common complaints among GPC patients included constipation and nervousness. In all, only 11 out of 132 patients complained of adverse events. In the placebo group, three out of 129 patients registered mild complaints, including nausea, dizziness, hostility and headache. Additional benefits No one has suggested that brain aging is a perfectly elucidated, simple process. On the contrary, it's likely that numerous complex interrelated processes contribute to mental decline as we age. For example, researchers at the University of Utah School of Medicine recently made headlines when they released data indicating that declining levels of another neurotransmitter, gamma-aminobutyric acid (GABA), may also play a role in eroding brain function. They found, in essence, that boosting GABA in elderly test animals increased their ability to focus dramatically. Interestingly, past research has shown that GPC stimulates release of GABA, raising the amount available to the brain.24 Some of the dementias, mood disorders and psychoses of both Huntington's and Alzheimer's diseases have been linked to depleted GABA.25 Besides playing a crucial role in memory, attention and general cognition, acetylcholine is also vital to the maintenance of normal sleep patterns. Insomnia is a troubling and all too common symptom of advancing age. Once-sound sleepers find themselves transformed into light sleepers by the encroaching years. This inability to screen out extraneous stimuli is a direct result of acetylcholine depletion. Acetylcholine drives the so-called stimulus barrier, a faculty of the brain that allows us to screen out distractions while concentrating on a task or problem, and to ignore intrusive sounds when sleeping. Insomnia often becomes severe in advanced Alzheimer's disease. Although clinical trials have largely ignored this quality-of-life parameter, it stands to reason that restoring acetylcholine to normal, youthful levels will also restore healthy sleep. Other intriguing research has shown that when GPC is co-administered with growth hormone releasing hormone (GHRH) higher levels of human growth hormone (hGH) are released than is the case when GHRH is given alone. GPC evidently acts to potentiate the effects of GHRH.26 Although the effect was demonstrated in young and old alike, the increase was more dramatic in elderly subjects. Yet another study alludes to further potential benefits of GPC supplementation. Rats that received GPC for six months experienced increased expression of nerve growth factor receptors in the cerebellar cortex. Nerve growth factor is crucial to the regulation of acetylcholine receptor function. It influences receptor growth and maturation and is involved in receptor repair, maintenance and regeneration. Like other components of the body, nerve growth factor receptors are incapacitated with advancing age. GPC - in the rat model, at least - appears to further reverse the effects of aging by reversing this deterioration.27 Preventive medicine One thing is clear. GPC therapy is already proven beneficial in humans, and it's available for approximately one third the cost of some popular prescription drugs that are currently used for the treatment of Alzheimer's symptoms, such as the acetylcholinesterase inhibitors Aricept™(donepezil HCl) and Exelon™ (rivastigmine tartrate). If researchers such as Dr. Lucilla Parnetti are right, and early intervention is our greatest hope for nipping cognitive decline in the bud, GPC may well prove to be a valuable new weapon in the age-fighting arsenal. References 1 .Moreno M et al. Cognitive impairment in mild to moderate Alzheimer's dementia after treatment with the acetylcholine precursor choline alphoscerate: a multi-center, double-blind, randomized, placebo-controlled trial. Clin Therap 2001; Oct: 178 193. 2. Larson EB et al. Cognitive impairment: Dementia and Alzheimer's disease. Annu Rev Public Health 1992; 13:431-449. 3. Parnetti L et al. Choline alphoscerate in cognitive decline and in acute cerebrovascular disease: an analysis of published clinical data. Mech Ageing Dev 2001 Nov: 122(16): 2041-2055. 4. Hofman A et al. The prevalence of dementia in Europe: a collaborative study of 1980-1990 findings. Int J Epidemiol 1991; 20: 736-748. 5. Jorm AF et al. The prevalence of dementia: a quantitative integration of the literature. Acta Psychiatr Scand 1987; 76: 465-479. 6. Ritchie K et al. Is senile dementia age-related or ageing-related? Evidence from a meta-analysis of dementia prevalence in the oldest old. Lancet 1995; 346: 931-934. 7. Ritchie K, Lovestone S. The dementias. Lancet 2002; 360: 1759-1766. 8. Chandra et al. Prevalence of Alzheimer's disease in and other dementias in rural India: the Indo-US study. Neurology 1998; 51: 1000 1008. 9. Zhang M et al. The prevalence of dementia and Alzheimer's disease in Shangai, China: impact of age, gender and education. Ann Neurol 1990; 27: 428-437. 10. Bartus RT et al. The cholinergic hypothesis of geriatric memory dysfunction. Science 1982; 217: 408-414. 11. Etienne P et al. Alzheimer's disease: lack of effect of lecithin treatment for 3 months. Neurology 1981; 31: 1552-1554. 12. Pomara N et al. Failure of single-dose lecithin to alter aspects of central cholinergic activity in Alzheimer's disease. J Clin Psych 1983; 44: 293-295. 13. Smith RC et al. Comparison of therapeutic response to long-term treatment with lecithin versus piracetam plus lecithin in patients with Alzheimer's disease. Psychopharmacol Bul 1984; 20: 542-545. 14. Thal LJ et al. Choline chloride fails to improve cognition of Alzheimer's disease. Neurobiol Aging 1981; 2: 205-258. 15. Barbagallo SG et al. Glycerylphosphorylcholine in the mental recovery of cerebral ischemic attacks. An Italian multi-center clinical trial. Ann N Y Acad Sci 16. Ricci et al. Oral choline alphoscerate counteracts age-dependent loss of mossy fibers in the rat hippocampus. Mech Ageing Dev 1992; 66 (1): 88-91. 17. Amenta F et al. Long term choline alphoscerate treatment counters age-dependant microanatomical changes in rat brain. Prog Neuropsychopharmacol Biol Psychiatry 1994 Sep: 18(5): 915-924. 18. Amenta F et al. Cholinergic neurotransmission in the hippocampus of aged rats: influence of alpha-L-glycerylphosphorylcholine treatment. Ann N Y Acad Sci 1993Sept: 24: 695: 311-313. 19. Burns A, Zaudig M. Mild cognitive impairment in older people. Lancet 2002; 360: 1963-1965. 20. Petersen RC et al. Current concepts in mild cognitive impairment. Arch Neurol 2001; 58: 1985-1992. 21. Ritchie K et al. Classification criteria for mild cognitive impairment: a population-based validation study. Neurology 2001; 56: 27-42. 22. Bozoki A et al. Mild cognitive impairments predict dementia in non-demented elderly patients with memory loss. Arch Neurol 2000; 58:411-416. 23. Zaudig M. Mild cognitive impairment in the elderly. Curr Opin Psychiatry 2002; 15: 387-393. 24. Ferraro L et al. Evidence for an in vivo and in vitro modulation of endogenous cortical GABA release by alpha-glycerylphosphorylcholine. Neurochem Res 1996 May; 21 (5): 547-552. 25. Cummings JL et al. Neurobiological basis of behavior. IN: Coffey CE, Cummings JL eds. Textbook of Geriatric Neuropsychiatry. American Psychiatric Press; 1994: 72-96. 26. Ceda GP et al. Alpha-glycerylphosphorylcholine administration increases the GH responses to GHRH in young and elderly subjects. Horm Metab Res 1992 Mar; 24(3): 119-121. 27. Vega JA et al. Nerve growth factor receptor immunoreactivity in the cerebellar cortex of aged rats: effect of choline alphoscerate treatment. Mech Ageing Dev 1993 Jun;69(1-2): 119-127. 28. Govoni S et al. PKC translocation in vivo and in vitro by alpha-glycerylphosphorylcholine, a cognition-enhancing drug. Ann N Y Acad Sci 1993 Sep; 24; 695: 307-310. 29. Abatti et al. Nootropic therapy of cerebral aging. Adv Ther 1991;8: 268-276. |