Life Extension Magazine®

If the information you are about to read could be turned into a patented drug, it would be worth billions of dollars of annual sales to whoever owned it.
What’s regrettable is very few doctors provide this lifesaving data to their prostate cancer patients. A staggering number of lives could be spared if the dietary changes discussed in this article are widely implemented.
In the February 2007 issue of Life Extension magazine®, we published an article titled “Eating Your Way to Prostate Cancer.”
In the April 2003 issue, we published an article titled “Eating Food Cooked at High Temperature
Accelerates Aging.”
Since these articles were published, large numbers of confirmative studies have been conducted that substantiate what we warned about.
This article will describe recently published science showing how eating the wrong foods markedly increase one’s risk of developing prostate cancer.
It also reveals data showing that men already diagnosed with prostate cancer who consume the wrong foods progress to advanced disease and death faster.
Cancer cells are present in the prostate glands of many aging men, yet only one in six men are ever diagnosed with prostate cancer.1,2 If one looks at what is required for a single cancer cell to develop into a detectable tumor, it becomes obvious that natural barriers exist to protect men against full-blown cancer.
Unfortunately, dietary choices in the Western world circumvent the body’s protective barriers.3 The end result is that most men unwittingly provide, through their food choices, biological fuel for existing prostate cancer cells to propagate and metastasize.
An understanding of the biological roles of diet and specific nutrients can enable aging men to achieve a considerable amount of control over whether isolated cancer cells in their prostate gland will ever show up as a clinically diagnosed disease.
The impact of the food we ingest on cell growth and death is so pronounced that it can be similar to the effects displayed by anticancer drugs—without the toxicities.
Don’t Eat Overcooked Meat

This Increases Risk of
Aggressive Prostate Cancer
Any meat (including fish) cooked at high temperatures creates dangerous carcinogens.4 Scientists looked at men whose diets included high intake of red meat cooked at high temperatures, pan-fried, or well-done. Their findings published in 2012 showed specific gene expression changes that predisposed these men to advanced prostate cancer.5 These kinds of studies show that one can exert a degree of control over their cell regulatory genes by avoiding overcooked meats.
Aggressive malignancies are those that rapidly propagate, infiltrate and metastasize.6 A 2011 study evaluated almost 1,000 men and found that higher consumption of any ground beef or processed meats was associated with an increased risk of aggressive prostate cancer.7 Men who ate ground beef showed the strongest association with a 130% increased incidence. The association primarily reflected intake of grilled or barbequed meat, with more well-done meat conferring a higher risk of aggressive prostate cancer. In contrast, consumption of rare/medium cooked ground beef was not associated with aggressive prostate cancer.7
A 2011 study looked at dietary patterns of 726 newly diagnosed prostate cancer cases and compared them to 527 controls.8 For advanced prostate cancer (but not localized disease), there was an associated 79% increased risk in men who ate hamburgers, a 57% increased risk with processed meats, a 63% increased risk with grilled red meat, and a 52% increased risk with well-done red meat.8 This study corroborated others associating consumption of processed meat and red meat, especially when cooked at high temperatures, with increased cases of advanced prostate cancer.5,6
Concern About Eggs and Milk
Large-scale studies associate egg consumption with sharply increased cancer risks.9,10
A 2011 study looked at 27,607 men who developed or died from prostate cancer over a 14-year period.9 Men who consumed 2.5 or more eggs per week had an 81% increased risk of lethal prostate cancer compared to those who consumed less than half an egg per week.9 This study showed that consumption of eggs increased the risk of healthy men developing metastatic prostate cancer.
A 2013 evaluation was done using data from the famous Physician’s Health Study to identify the impact of consumption of skim or whole milk on incidence and survival after diagnosis of prostate cancer.11 This analysis involved 21,660 physicians who were followed for 28 years. Skim/low fat milk was associated with increased risk of low grade prostate cancer, whereas whole milk was associated with increased risk of fatal prostate cancers. In these men diagnosed with prostate cancer, consumption of whole milk was associated with a 117% increased risk of progression to fatal disease.11 This finding further substantiates the important role of diet even after prostate cancer is diagnosed.
The take-home lesson so far is if one has an elevated or rising PSA, it is especially prudent to avoid over cooked red meats, processed meat, eggs, and whole cow’s milk.
What You Need to Know
 |
Prostate Cancer: Food For Thought
- Dietary choices in the Western world circumvent the body’s protective barriers to cancer.
- Men who regularly consume certain plant foods have sharply lower rates of prostate cancer.
- High intakes of red meat cooked at high temperatures can result in specific gene expression changes that may predispose men to advanced prostate cancer.
- Large-scale studies associate egg consumption with sharply increased cancer risks.
- Skim/low fat milk was associated with increased risk of low grade prostate cancer, whereas whole milk was associated with increased risk of high-grade prostate cancers.
- Diets high in omega-6 fats and saturated fats are associated with greater prostate cancer risk, whereas increased intake of the type of omega-3 fats found in fish has been shown to confer protection.
- Arachidonic acid, found abundantly in eggs and chicken, is metabolized by the 5-LOX enzyme to 5-hydroxyeicosatetraeonic acid (5-HETE), a potent survival factor that prostate cancer cells use to escape destruction.
- Not only do 5-LOX products directly stimulate cancer cell propagation, but the breakdown products that 5-LOX produces from arachidonic acid (such as leukotriene B4, 5-HETE, and hydroxylated fatty acids) cause tissue destruction, chronic inflammation, and increased resistance of tumor cells to apoptosis (programmed cell destruction).
Confusion About Omega-6 Fats
Omega-6 fats are essential to life. We are unable to make them in our body and must get them from foods.12
The problem is that Western diets have become so overloaded with omega-6s that our bodies have become poisoned with them. The typical American tends to consume up to 25 times more omega-6 fats than the healthier omega-3 fats.12
One reason we have become so overloaded with omega-6s is that in the rush to switch from red meat and other saturated fats such as lard, we have been gobbling down too many omega-6-rich foods. These include vegetable oils used in all kinds of processed and fried foods, margarine, salad dressing, mayonnaise, certain nuts, peanut butter, and even poultry, a meat with high omega-6 content.13
Commercial food companies deceivingly promote polyunsaturated vegetable oils like corn and safflower as healthy because of early studies showing reduced cardiovascular risk factors in those who consumed vegetable oils compared to animal-based fats such as butter.14
Remember that you require omega-6s to live, but not in the large quantity consumed in the typical American diet. This means you want to lower the percentage of calories in your diet that comprise of omega-6 fats.
The chart below lists foods high in omega-6 fats. Eating any of these foods in moderation is not a problem, but when they comprise a high percentage of your overall diet, your body becomes overloaded with omega-6s, which sets the stage for a wide variety of disorders. We next describe how a high intake of omega-6 fats contributes to prostate cancer.
Table 1
 |
Dietary Sources of Omega-6 Fats13
Avocados |
Peanut Oil |
Almonds |
Sunflower Oil |
Macadamia Nuts |
Safflower Oil |
Peanuts |
Canola Oil |
Brazil Nuts |
Macadamia Nut Oil |
Hazelnuts |
Grapeseed Oil |
Pecans |
Corn Oil |
Pistachios |
Peanut Butter |
Avocado Oil |
Almond Butter |
Almond oil |
Cashew Butter |
Role of Omega-6 Fats in Prostate Cancer
Diets high in omega-6 fats and saturated fats are associated with greater prostate cancer risk, whereas increased intake of the type of omega-3 fats found in fish has been shown to confer protection.15-17
Based on consistent epidemiological findings across a wide range of human populations, scientists have sought to understand why eating the wrong kinds of fat (saturated and omega-6 fats) provokes a stimulatory effect on prostate cancer.15,17
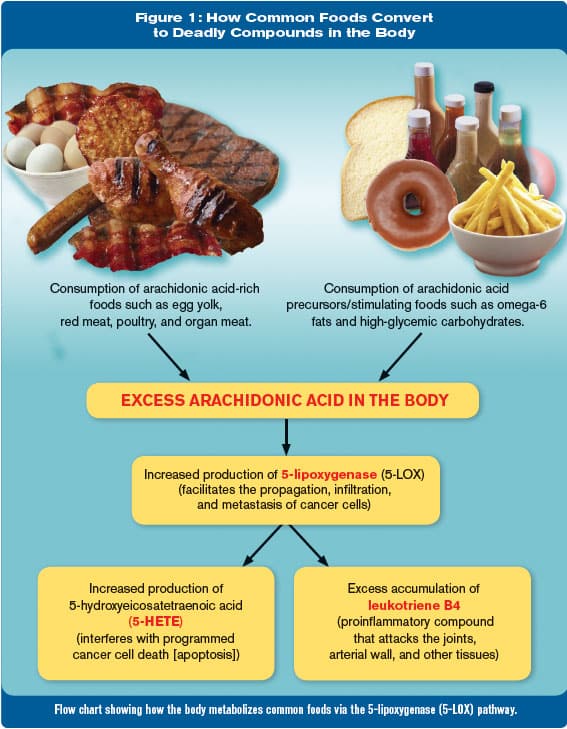
To ascertain what happens after we eat bad fats, all one has to do is look at the metabolic breakdown pathways that these fats follow in the body, as shown in the chart on the next page (Figure 1). For example, let us assume that for dinner, you eat a steak (a source of saturated fat), potato (a high-glycemic starch) and a salad with a typical dressing of soybean and/or safflower oils (omega-6 fats).
As can be seen in the flow chart, omega-6 fats can convert to arachidonic acid in the body. Meat itself contains arachidonic acid.18 One way that the body rids itself of excess arachidonic acid is by provoking a dangerous metabolizing pathway through 5-lipoxygenase (5-LOX).
It is well established that 5-LOX products stimulate prostate cancer cell proliferation via several well-defined mechanisms.19-21 High glycemic foods also promote formation of 5-LOX in the body, via activation of enzymes involved in the formation of arachidonic acid.18
Arachidonic acid, found abundantly in eggs and chicken, is metabolized by the 5-LOX enzyme to 5-hydroxyeicosatetraeonic acid (5-HETE), a potent survival factor that prostate cancer cells use to escape destruction. 18,22,23
The flow chart (Figure 1) clearly demonstrates how consuming a diet rich in arachidonic acid provokes the production of dangerous 5-LOX products, which can promote prostate cancer progression.18-23 In addition to 5-HETE, 5-LOX also metabolizes arachidonic acid to leukotriene B4 and other pro-inflammatory agents that promote cancer.24
The chart (Figure 1) on this page provides a list of foods that are high in arachidonic acid. Just because a food is listed on this chart does not mean you have to avoid it. It is wise, however, to pick which high-arachidonic acid foods are that important compared to ones you may not even realize you’re consuming.
5-LOX Is Over-Expressed in Prostate Cancer
Based on studies showing that consumption of foods rich in omega 6 fatty acids is associated with higher incidences of prostate cancer, scientists sought to determine how much of the 5-LOX enzyme is present in malignant versus benign prostate tissues.25
Using biopsy samples taken from living human patients, the researchers found that 5-LOX mRNA levels were an astounding six-fold greater in malignant prostate tissues compared with benign tissues. This study also found that levels of 5-HETE were 2.2-fold greater in malignant versus benign prostate tissues.26 The scientists concluded this study by stating that selective inhibitors of 5-LOX may be useful in the prevention or treatment of patients with prostate cancer.26
5-LOX Promotes Tumor Growth Factors
As the evidence mounts that ingesting “bad fats” increases prostate cancer risk, scientists are evaluating the effects of 5-LOX on various growth factors involved in the progression, angiogenesis, and metastasis of cancer cells.
One study found that 5-LOX activity is required to stimulate prostate cancer cell growth by epidermal growth factor (EGF) and other tumor cell proliferating factors produced in the body.19 When 5-LOX levels were reduced, the cancer cell stimulatory effect of EGF and other growth factors was diminished.19
In a mouse study, an increase in 5-LOX resulted in a corresponding increase in vascular endothelial growth factor (VEGF), a key growth factor that tumor cells use to stimulate new blood vessel formation (angiogenesis) into the tumor. 5-LOX inhibitors have been shown to reduce tumor angiogenesis along with a host of other growth factors.27
In both androgen-dependent and androgen-independent human prostate cancer cell lines, the inhibition of 5-LOX has consistently been shown to halt the growth stimulatory action of 5-LOX and prompt rapid and massive apoptosis (cancer cell destruction).20,22,28,29

Omega-3 Fatty Acids: A First Line of Defense
One reason that fish oil supplements have become so popular is that their beneficial EPA/DHA fatty acids can help reduce the production of arachidonic acid-derived tumor promoting byproducts in the body.30-33 As shown on the flow chart above (Figure 2), if arachidonic acid levels are reduced, there would be a corresponding suppression of 5-LOX and its metabolic byproducts 5-HETE and leukotriene B4.
Once one understands the lethal 5-LOX cascades, it is easy to see why people who excessively consume foods rich in arachidonic acid, and those who fail to reduce the production of arachidonic acid metabolites (such as 5-HETE) by ensuring adequate intake of omega-3 fatty acids, are setting themselves up for prostate cancer and a host of inflammatory disorders (including atherosclerosis).
Nutrients That Suppress 5-LOX
Health-conscious people already take nutrients like curcumin and fish oil that help to lower 5-LOX activity in the body.34,35 Studies show that lycopene and saw palmetto extract also help to suppress 5-LOX.36,37 The suppression of 5-LOX by these nutrients may partially account for their favorable effects on the prostate gland.
As humans age, however, chronic inflammatory processes can cause the over-expression of 5-LOX in the body.38 For maturing males, the result of excess 5-LOX may be the epidemic of prostate cancer observed as men age.2
Based on the cumulative knowledge that 5-LOX itself and its metabolic products can promote the progression and metastasis of prostate cancer cells, it would appear advantageous to take aggressive steps to suppress this lethal enzyme.36
This can be done by avoiding foods that promote 5-LOX formation in the body and taking supplements that inhibit 5-LOX via differing pathways.
Multiple Dangers of Excess Arachidonic Acid
 |
In response to arachidonic acid overload, the body increases its production of enzymes like 5-lipoxygenase (5-LOX) to degrade arachidonic acid.48 Not only do 5-LOX products directly stimulate cancer cells propagation, but the breakdown products that 5-LOX produces from arachidonic acid (such as leukotriene B4, 5-HETE, and hydroxylated fatty acids) cause tissue destruction, chronic inflammation, and increased resistance of tumor cells to apoptosis (programmed cell destruction).19-21,48-58
It is important to understand that 5-LOX is not the only dangerous enzyme the body produces to break down arachidonic acid. As can be seen in the chart on the previous page, both cyclooxygenase-1 and cyclooxygenase-2 (COX-1 and COX-2) also participate in the degradation of arachidonic acid.
COX-1 causes production of thromboxane A2, which can promote abnormal arterial blood clotting (thrombosis), resulting in heart attack and stroke.59-63 Thromboxane A2 is also involved in tumor metastasis.64,65 COX-2 is directly involved in cancer cell propagation, while its breakdown product (prostaglandin E2) promotes chronic inflammation.56,66-68 Most health-conscious people already inhibit the COX-1 and COX-2 enzymes by taking low-dose aspirin, curcumin, green tea, and various plant flavonoids such as pomegranate.61,69-82
A more integrative approach to this problem, however, would be to also reduce dietary levels of arachidonic acid, which is the precursor of 5-HETE and leukotriene B4.83
Research Substantiating Boswellia
Specific extracts from the boswellia plant selectively inhibit 5-lipoxygenase (5-LOX), a potent inducer of inflammation and carcinogenic byproducts.39,40
Boswellia extracts have been used for centuries, particularly in India as anti-inflammatory agents.41 In several well-controlled human studies, boswellia has been shown to be effective in alleviating various chronic inflammatory disorders.42-46
Scientists have discovered that the specific constituent in boswellia responsible for suppressing 5-LOX is AKBA ( 3-O-acetyl-11-keto-B-boswellic acid).41 Boswellia-derived AKBA binds directly to 5-LOX and inhibits its activity.41 Other boswellic acids only partially and incompletely inhibit 5-LOX.41,47
Formulas containing high concentrations of AKBA from boswellia have been developed based on its ability to treat inflammatory disorders. Standardized boswellia extracts have long been included in prostate support nutrient formulas for the purpose of suppressing excess 5-LOX.
Prostate-Protecting Properties of Boswellia
Tumor necrosis factor-alpha (TNF-alpha) is a pro-inflammatory cytokine that often increases in aging people.84
From the standpoint of keeping prostate cancer cells in check, boswellia has been shown to prevent the TNF-alpha-induced expression of a protein-degrading enzyme called matrix metalloproteinase (MMP).85 Cancer cells use the MMP enzyme to tear apart natural barriers in the body that would normally encase them.86 Prostate cancer cells are notorious for inducing the production of this enzyme (TNF-alpha) that causes containment structures within the prostate gland to vanish, thus enabling the cancerous prostate cells to break through healthy prostate tissue and eventually metastasize.87,88
Prostate cancer cells use adhesion molecules (known as VCAM-1 and ICAM-1) to facilitate their spread throughout the body. Boswellia has been shown to prevent the up-regulation of these adhesion molecules, which are directly involved in inflammatory processes.85 Chronic inflammation is tightly linked to the induction of aberrant angiogenesis used by cancer cells to facilitate the growth of new blood vessels (angiogenesis) into tumors.89
The potent 5-LOX-inhibiting properties of boswellia, and its ability to suppress other inflammatory factors such as TNF-alpha, make it an important nutrient for those concerned with prostate cancer.41,85
Tea, Soy, Lignans, and Cruciferous Vegetables
 |
Men who regularly consume certain plant foods have sharply lower rates of prostate cancer.90 Studies show that cauliflower, broccoli, flax lignans, and soy isoflavones protect against a host of diseases, including prostate cancer.91-99
A 2013 study reported that soy food consumption, more popular in Asian populations, is associated with a 25 to 30% reduced risk of prostate cancer.100 There is a lot of confusion about soy in the alternative health community. Everyone agrees that limiting intake of soybean oil is important because it is loaded with omega-6 fats.13 Other constituents found in soy, however, such as genistein and daidzein have demonstrated anti-prostate cancer mechanisms.101-107
Polyphenols found in green tea decrease serum levels of PSA, hepatocyte growth factor, and vascular endothelial growth factor (VEGF) in prostate cancer patients.108-113
The FDA says, however, that there is insufficient evidence to claim that green tea consumption protects against prostate cancer.114 Life Extension issued a rebuttal to the FDA position on green tea and prostate cancer in the November 2005 edition of Life Extension magazine®.
Urgent Need to Alter Dietary Patterns
Those consuming Western diets predispose themselves to cancer.3 It is encouraging that we know what food groups increase prostate cancer risk and what foods/nutrients reduce it.
All aging men should shift their diet towards foods that protect against prostate cancer. Those with rising or elevated PSA should be especially diligent in avoiding dietary factors that can fuel the growth of prostate tumors.115
The lethal impact of cancer-promoting foods may be mitigated by taking supplements such as green tea, curcumin, fish oil, pomegranate, and boswellia, along with regular inclusion of cruciferous vegetables and other healthy foods in one’s diet.
If you have any questions on the scientific content of this article, please call a Life Extension® Health Advisor at 1-866-864-3027.
References
- Available at: http://www.cancer.org/cancer/prostatecancer/detailed guide/prostate-cancer-key-statistics. Accessed August 26, 2013.
- Available at: http://eu-acme.org/europeanurology/upload_articles/roehrborn.pdf. Accessed October 2, 2013.
- Erdelyi I, Levenkova N, Lin EY, et al. Western-style diets induce oxidative stress and dysregulate immune responses in the colon in a mouse model of sporadic colon cancer. J Nutr. 2009 Nov;139(11):2072-8.
- Available at: http://www.cancer.gov/cancertopics/factsheet/risk/cooked-meats. Accessed October 7, 2013.
- Joshi AD, Corral R, Catsburg C, et al. Red meat and poultry, cooking practices, genetic susceptibility and risk of prostate cancer: results from a multiethnic case-control study. Carcinogenesis. 2012 Nov;33(11):2108-18.
- Available at: http://www.cancer.gov/dictionary?cdrid=46053. Accessed October 2, 2013.
- Punnen S, Hardin J, Cheng I, Klein EA, Witte JS. Impact of meat consumption, preparation, and mutagens on aggressive prostate cancer. PLoS One. 2011 6(11):e27711.
- John EM, Stern MC, Sinha R, Koo J. Meat consumption, cooking practices, meat mutagens, and risk of prostate cancer. Nutr Cancer. 2011 63(4):525-37.
- Richman EL, Kenfield SA, Stampfer MJ, Giovannucci EL, Chan JM. Egg, red meat, and poultry intake and risk of lethal prostate cancer in the prostate-specific antigen-era: incidence and survival. Cancer Prev Res (Phila). 2011 Dec;4(12):2110-21.
- Aune D, De Stefani E, Ronco AL, et al. Egg consumption and the risk of cancer: a multisite case-control study in Uruguay. Asian Pac J Cancer Prev. 2009;10(5):869-76.
- Song Y, Chavarro JE, Cao Y, et al. Whole milk intake is associated with prostate cancer-specific mortality among U.S. male physicians. J Nutr. 2013 Feb;143(2):189-96.
- Available at: http://umm.edu/health/medical/altmed/supplement/omega6-fatty-acids. Accessed October 3, 2013.
- Available at: http://nutritiondata.self.com/foods-000141000000000000000-w.html. Accessed October 3, 2013.
- Mekki N, Charbonnier M, Borel P, et al. Butter differs from olive oil and sunflower oil in its effects on postprandial lipemia and triacylglycerol-rich lipoproteins after single mixed meals in healthy young men. J Nutr. 2002 Dec;132(12):3642-9.
- Newcomer LM, King IB, Wicklund KG, Stanford JL. The association of fatty acids with prostate cancer risk. Prostate. 2001 Jun 1;47(4):262-8.
- Leitzmann MF, Stampfer MJ, Michaud DS, et al. Dietary intake of n-3 and n-6 fatty acids and the risk of prostate cancer. Am J Clin Nutr. 2004 Jul;80(1):204-16.
- Pelser C, Mondul AM, Hollenbeck AR, Park Y. Dietary fat, fatty acids, and risk of prostate cancer in the NIH-AARP diet and health study. Cancer Epidemiol Biomarkers Prev. 2013 Apr;22(4):697-707
- Sears B, Ricordi C. Anti-inflammatory nutrition as a pharmacological approach to treat obesity. J Obes. 2011;2011.
- Hassan S, Carraway RE. Involvement of arachidonic acid metabolism and EGF receptor in neurotensin-induced prostate cancer PC3 cell growth. Regul Pept. 2006 Jan 15;133(1-3):105-14.
- Moretti RM, Montagnani MM, Sala A, Motta M, Limonta P. Activation of the orphan nuclear receptor RORalpha counteracts the proliferative effect of fatty acids on prostate cancer cells: crucial role of 5-lipoxygenase. Int J Cancer. 2004 Oct 20;112(1):87-93.
- Ghosh J, Myers CE. Arachidonic acid stimulates prostate cancer cell growth: critical role of 5-lipoxygenase. Biochem Biophys Res Commun. 1997 Jun 18;235(2):418-23.
- Ghosh J, Myers CE. Inhibition of arachidonate 5-lipoxygenase triggers massive apoptosis in human prostate cancer cells. Proc Natl Acad Sci U S A. 1998 Oct 27;95(22):13182-7.
- Sundaram S, Ghosh J. Expression of 5-oxoETE receptor in prostate cancer cells: critical role in survival. Biochem BiophysRes Commun. 2006 Jan 6;339(1):93-8.
- Larré S, Tran N, Fan C, et al. PGE2 and LTB4 tissue levels in benign and cancerous prostates. Prostaglandins Other Lipid Mediat. 2008 Dec;87(1-4):14-9.
- Ritch CR, Wan RL, Stephens LB, et al. Dietary fatty acids correlate with prostate cancer biopsy grade and volume in Jamaican men. J Urol. 2007 Jan;177(1):97-101; discussion 101.
- Gupta S, Srivastava M, Ahmad N, Sakamoto K, Bostwick DG, Mukhtar H. Lipoxygenase-5 is overexpressed in prostate adenocarcinoma. Cancer. 2001 Feb 15;91(4):737-43.
- Ye YN, Liu ES, Shin VY, Wu WK, Cho CH. Contributory role of 5-lipoxygenase and its association with angiogenesis in the promotion of inflammation-associated colonic tumorigenesis by cigarette smoking. Toxicology. 2004 Oct 15;203(1-3):179-88.
- Ghosh J. Inhibition of arachidonate 5-lipoxygenase triggers prostate cancer cell death through rapid activation of c-Jun N-terminal kinase. Biochem Biophys Res Commun. 2003 Jul 25;307(2):342-9.
- Anderson KM, Seed T, Vos M et al. 5-Lipoxygenase inhibitors reduce PC-3 cell proliferation and initiate nonnecrotic cell death. Prostate. 1998 Nov 1;37(3):161-73.
- Norris PC, Dennis EA. Omega-3 fatty acids cause dramatic changes in TLR4 and purinergic eicosanoid signaling. Proc Natl Acad Sci U S A. 2012 May 29;109(22):8517-22.
- Barham JB, Edens MB, Fonteh AN, et al. Addition of eicosapentaenoic acid to gamma-linolenic acid-supplemented diets prevents serum arachidonic acid accumulation in humans. J Nutr. 2000 Aug;130(8):1925-31.
- Adan Y, Shibata K, Sato M, Ikeda I, Imaizumi K. Effects of docosahexaenoic and eicosapentaenoic acid on lipid metabolism, eicosanoid production, platelet aggregation and atherosclerosis in hypercholesterolemic rats. Biosci Biotechnol Biochem. 1999 Jan;63(1):111-9.
- Schwartz J. Role of polyunsaturated fatty acids in lung disease. Am J Clin Nutr. 2000 Jan;71(1 Suppl):393S-6S.
- Taccone-Gallucci M, Manca-di-Villahermosa S, Battistini L, et al. N-3 PUFAs reduce oxidative stress in ESRD patients on maintenance HD by inhibiting 5-lipoxygenase activity. Kidney Int. 2006 Apr;69(8):1450-4.
- Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013 Jan;15(1):195-218.
- Hazai E, Bikadi Z, Zsila F, Lockwood SF. Molecular modeling of the non-covalent binding of the dietary tomato carotenoids lycopene and lycophyll, and selected oxidative metabolites with 5-lipoxygenase. Bioorg Med Chem. 2006 Oct 15;14(20):6859-67.
- Bonvissuto G, Minutoli L, Morgia G, et al. Effect of Serenoa repens, lycopene, and selenium on proinflammatory phenotype activation: an in vitro and in vivo comparison study. Urology. 2011 Jan;77(1):248.e9-16.
- Qu T, Uz T, Manev H. Inflammatory 5-LOX mRNA and protein are increased in brain of aging rats. Neurobiol Aging. 2000 Sep-Oct;21(5):647-52.
- Safayhi H, Rall B, Sailer ER, Ammon HP. Inhibition by boswellic acids of human leukocyte elastase. J Pharmacol Exp Ther. 1997 Apr;281(1):460-3.
- Safayhi H, Sailer ER, Ammon HP. Mechanism of 5-lipoxygenase inhibition by acetyl-11-keto-beta-boswellic acid. Mol Pharmacol. 1995 Jun;47(6):1212-6.
- Siddiqui MZ. Boswellia serrata, a potential antiinflammatory agent: an overview. Indian J Pharm Sci. 2011 May;73(3):255-61.
- Kimmatkar N, Thawani V, Hingorani L, Khiyani R. Efficacy and tolerability of Boswellia serrata extract in treatment of osteoarthritis of knee--a randomized double blind placebo controlled trial. Phytomedicine. 2003 Jan;10(1):3-7.
- Ammon HP. Boswellic acids (components of frankincense) as the active principle in treatment of chronic inflammatory diseases. Wien Med Wochenschr. 2002 152(15-16):373-8.
- Gupta I, Parihar A, Malhotra P, et al. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Med. 2001 Jul;67(5):391-5.
- Gerhardt H, Seifert F, Buvari P, Vogelsang H, Repges R. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol. 2001 Jan;39(1):11-7.
- Gupta I, Gupta V, Parihar A, et al. Effects of Boswellia serrata gum resin in patients with bronchial asthma: results of a double-blind, placebo-controlled, 6-week clinical study. Eur J Med Res. 1998 Nov 17;3(11):511-4.
- Sailer ER, Subramanian LR, Rall B, et al. Acetyl-11-keto-beta-boswellic acid (AKBA): structure requirements for binding and 5-lipoxygenase inhibitory activity. Br J Pharmacol. 1996 Feb;117(4):615-8.
- Jiang WG, Douglas-Jones AG, Mansel RE. Aberrant expression of 5-lipoxygenase-activating protein (5-LOXAP) has prognostic and survival significance in patients with breast cancer. Prostaglandins Leukot Essent Fatty Acids. 2006 Feb;74(2):125-34.
- Yoshimura R, Matsuyama M, Mitsuhashi M, et al. Relationship between lipoxygenase and human testicular cancer. Int J Mol Med. 2004 Mar;13(3):389-93.
- Zhang L, Zhang WP, Hu H, et al. Expression patterns of 5-lipoxygenase in human brain with traumatic injury and astrocytoma. Neuropathology. 2006 Apr;26(2):99-106.
- Soumaoro LT, Iida S, Uetake H, et al. Expression of 5-lipoxygenase in human colorectal cancer. World J Gastroenterol. 2006 Oct 21;12(39):6355-60.
- Hoque A, Lippman SM, Wu TT, et al. Increased 5-lipoxygenase expression and induction of apoptosis by its inhibitors in esophageal cancer: a potential target for prevention. Carcinogenesis. 2005 Apr;26(4):785-91.
- Zhi H, Zhang J, Hu G, et al. The deregulation of arachidonic acid metabolism-related genes in human esophageal squamous cell carcinoma. Int J Cancer. 2003 Sep 1;106(3):327-33.
- Rubinsztajn R, Wronska J, Chazan R. Urinary leukotriene E4 concentration in patients with bronchial asthma and intolerance of non-steroids anti-inflammatory drugs before and after oral aspirin challenge. Pol Arch Med Wewn. 2003 Aug;110(2):849-54.
- Subbarao K, Jala VR, Mathis S, et al. Role of leukotriene B4 receptors in the development of atherosclerosis: potential mechanisms. Arterioscler Thromb Vasc Biol. 2004 Feb;24(2):369-75.
- Laufer S. Role of eicosanoids in structural degradation in osteoarthritis. Curr Opin Rheumatol. 2003 Sep;15(5):623-7.
- Hennig R, Ding XZ, Tong WG, et al. 5-Lipoxygenase and leukotriene B(4) receptor are expressed in human pancreatic cancers but not in pancreatic ducts in normal tissue. Am J Pathol. 2002 Aug;161(2):421-8.
- Ding XZ, Iversen P, Cluck MW, Knezetic JA, Adrian TE. Lipoxygenase inhibitors abolish proliferation of human pancreatic cancer cells. Biochem Biophys Res Commun. 1999 Jul 22;261(1):218-23.
- de Leval X, Hanson J, David JL, et al. New developments on thromboxane and prostacyclin modulators part II: prostacyclin modulators. Curr Med Chem . 2004 May;11(10):1243-52.
- Cheng Y, Austin SC, Rocca B, et al. Role of prostacyclin in the cardiovascular response to thromboxane A2. Science. 2002 Apr 19;296(5567):539-41.
- Catella-Lawson F. Vascular biology of thrombosis: platelet-vessel wall interactions and aspirin effects. Neurology. 2001 57(5 Suppl 2):S5-S7.
- James MJ, Penglis PS, Caughey GE, Demasi M, Cleland LG. Eicosanoid production by human monocytes: does COX-2 contribute to a self-limiting inflammatory response? Inflamm Res. 2001 May;50(5):249-53.
- Garcia Rodriguez LA. The effect of NSAIDs on the risk of coronary heart disease: fusion of clinical pharmacology and pharmacoepidemiologic data. Clin Exp Rheumatol. 2001 Nov;19(6 Suppl 25):S41-4.
- Nie D, Lamberti M, Zacharek A, et al. Thromboxane A(2) regulation of endothelial cell migration, angiogenesis, and tumor metastasis. Biochem Biophys Res Commun. 2000 Jan 7;267(1):245-51.
- Dassesse T, de Leval X, de Leval L, Pirotte B, Castronovo V, Waltregny D. Activation of the thromboxane A2 pathway in human prostate cancer correlates with tumor Gleason score and pathologic stage. Eur Urol. 2006 Nov;50(5):1021-31.
- Wang D, Wang H, Shi Q, et al. Prostaglandin E(2) promotes colorectal adenoma growth via transactivation of the nuclear peroxisome proliferator-activated receptor delta. Cancer Cell. 2004 Sep;6(3):285-95.
- Onguru O, Casey MB, Kajita S, Nakamura N, Lloyd RV. Cyclooxygenase-2 and thromboxane synthase in non-endocrine and endocrine tumors: a review. Endocr Pathol. 2005 16(4):253-77.
- Wang D, Dubois RN. Prostaglandins and cancer. Gut. 2006 Jan;55(1):115-22.
- Feldman M, Cryer B, Rushin K, Betancourt J. A comparison of every-third-day versus daily low-dose aspirin therapy on serum thromboxane concentrations in healthy men and women. Clin Appl Thromb Hemost. 2001 Jan;7(1):53-7.
- Zhang F, Altorki NK, Mestre JR, Subbaramaiah K, Dannenberg AJ. Curcumin inhibits cyclooxygenase-2 transcription in bile acid- and phorbol ester-treated human gastrointestinal epithelial cells. Carcinogenesis. 1999 Mar;20(3):445-51.
- Bengmark S. Curcumin, an atoxic antioxidant and natural NFkappaB, cyclooxygenase-2, lipooxygenase, and inducible nitric oxide synthase inhibitor: a shield against acute and chronic diseases. JPEN J Parenter Enteral Nutr. 2006 Jan;30(1):45-51.
- Park C, Kim GY, Kim GD, et al. Induction of G2/M arrest and inhibition of cyclooxygenase-2 activity by curcumin in human bladder cancer T24 cells. Oncol Rep. 2006 May;15(5):1225-31.
- Hong J, Bose M, Ju J, et al. Modulation of arachidonic acid metabolism by curcumin and related beta-diketone derivatives: effects on cytosolic phospholipase A(2), cyclooxygenases and 5-lipoxygenase. Carcinogenesis. 2004 Sep;25(9):1671-9.
- Yoysungnoen P, Wirachwong P, Bhattarakosol P, Niimi H, Patumraj S. Effects of curcumin on tumor angiogenesis and biomarkers, COX-2 and VEGF, in hepatocellular carcinoma cell-implanted nude mice. Clin Hemorheol Microcirc. 2006 34(1-2):109-15.
- Tunstall RG, Sharma RA, Perkins S, et al. Cyclooxygenase-2 expression and oxidative DNA adducts in murine intestinal adenomas: modification by dietary curcumin and implications for clinical trials. Eur J Cancer. 2006 Feb;42(3):415-21.
- Lee J, Im YH, Jung HH, et al. Curcumin inhibits interferon-alpha induced NF-kappaB and COX-2 in human A549 non-small cell lung cancer cells. Biochem Biophys Res Commun. 2005 Aug 26;334(2):313-8.
- Hong J, Smith TJ, Ho CT, August DA, Yang CS. Effects of purified green and black tea polyphenols on cyclooxygenase- and lipoxygenase-dependent metabolism of arachidonic acid in human colon mucosa and colon tumor tissues. Biochem Pharmacol. 2001 Nov 1;62(9):1175-83.
- Ahmed S, Rahman A, Hasnain A, et al. Green tea polyphenol epigallocatechin-3-gallate inhibits the IL-1 beta-induced activity and expression of cyclooxygenase-2 and nitric oxide synthase-2 in human chondrocytes. Free Radic Biol Med. 2002 Oct 15;33(8):1097-105.
- Shukla M, Gupta K, Rasheed Z, Khan KA, Haqqi TM. Bioavailable constituents/metabolites of pomegranate (Punica granatum L) preferentially inhibit COX2 activity ex vivo and IL-1beta-induced PGE2 production in human chondrocytes in vitro. J Inflamm (Lond). 2008 Jun 13;5:9.
- Rosillo MA, Sánchez-Hidalgo M, Cárdeno A, et al. Dietary supplementation of an ellagic acid-enriched pomegranate extract attenuates chronic colonic inflammation in rats. Pharmacol Res. 2012 Sep;66(3):235-42.
- Hämäläinen M, Nieminen R, Asmawi MZ, Vuorela P, Vapaatalo H, Moilanen E. Effects of flavonoids on prostaglandin E2 production and on COX-2 and mPGES-1 expressions in activated macrophages. Planta Med. 2011 Sep;77(13):1504-11.
- O’Leary KA, de Pascual-Tereasa S, Needs PW, Bao YP, O’Brien NM, Williamson G. Effect of flavonoids and vitamin E on cyclooxygenase-2 (COX-2) transcription. Mutat Res. 2004 Jul 13;551(1-2):245-54.
- Available at: http://www.jlr.org/content/33/3/323.full.pdf. Accessed October 4, 2013.
- Gupta S, Chiplunkar S, Kim C, Yel L, Gollapudi S. Effect of age on molecular signaling of TNF-alpha-induced apoptosis in human lymphocytes. Mech Ageing Dev. 2003 Apr;124(4):503-9.
- Roy S, Khanna S, Shah H, et al. Human genome screen to identify the genetic basis of the anti-inflammatory effects of Boswellia in microvascular endothelial cells. DNA Cell Biol. 2005 Apr;24(4):244-55.
- Katiyar SK. Matrix metalloproteinases in cancer metastasis: molecular targets for prostate cancer prevention by green tea polyphenols and grape seed proanthocyanidins. Endocr Metab Immune Disord Drug Targets. 2006 Mar;6(1):17-24.
- Rodríguez-Berriguete G, Sánchez-Espiridión B, Cansino JR, et al. Clinical significance of both tumor and stromal expression of components of the IL-1 and TNF-α signaling pathways in prostate cancer. Cytokine. 2013 Sep 21.
- Mizokami A, Gotoh A, Yamada H, Keller ET, Matsumoto T. Tumor necrosis factor-alpha represses androgen sensitivity in the LNCaP prostate cancer cell line. J Urol. 2000 Sep;164(3 Pt 1):800-5.
- Rajashekhar G, Willuweit A, Patterson CE, et al. Continuous endothelial cell activation increases angiogenesis: evidence for the direct role of endothelium linking angiogenesis and inflammation. J Vasc Res. 2006 43(2):193-204.
- Chan R, Lok K, Woo J. Prostate cancer and vegetable consumption. Mol Nutr Food Res. 2009 Feb;53(2):201-16.
- Heald CL, Ritchie MR, Bolton-Smith C, Morton MS, Alexander FE. Phyto-oestrogens and risk of prostate cancer in Scottish men. Br J Nutr. 2007 Aug;98(2):388-96.
- Hedelin M, Klint A, Chang ET, et al. Dietary phytoestrogen, serum enterolactone and risk of prostate cancer: the cancer prostate Sweden study (Sweden). Cancer Causes Control. 2006 Mar;17(2):169-80.
- Kumar NB, Cantor A, Allen K, et al. The specific role of isoflavones in reducing prostate cancer risk. Prostate. 2004 May 1;59(2):141-7.
- Lee MM, Gomez SL, Chang JS, et al. Soy and isoflavone consumption in relation to prostate cancer risk in China. Cancer Epidemiol Biomarkers Prev. 2003 Jul;12(7):665-8.
- McCann SE, Ambrosone CB, Moysich KB, et al. Intakes of selected nutrients, foods, and phytochemicals and prostate cancer risk in western New York. Nutr Cancer. 2005 53(1):33-41.
- Vij U, Kumar A. Phyto-oestrogens and prostatic growth. Natl Med J India. 2004 Jan;17(1):22-6.
- Sonoda T, Nagata Y, Mori M, et al. A case-control study of diet and prostate cancer in Japan: possible protective effect of traditional Japanese diet. Cancer Sci. 2004 Mar;95(3):238-42.
- Clarke JD, Dashwood RH, Ho E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008 Oct 8;269(2):291-304.
- Abdull Razis AF, Noor NM. Cruciferous vegetables: dietary phytochemicals for cancer prevention. Asian Pac J Cancer Prev. 2013;14(3):1565-70.
- Kimura T. East meets West: ethnic differences in prostate cancer epidemiology between East Asians and Caucasians. Chin J Cancer. 2012 Sep;31(9):421-9.
- Chen Y, Zaman MS, Deng G, et al. MicroRNAs 221/222 and genistein-mediated regulation of ARHI tumor suppressor gene in prostate cancer. Cancer Prev Res (Phila). 2011 Jan;4(1):76-86.
- Lakshman M, Xu L, Ananthanarayanan V, et al. Dietary genistein inhibits metastasis of human prostate cancer in mice. Cancer Res. 2008 Mar 15;68(6):2024-32.
- Davis JN, Singh B, Bhuiyan M, Sarkar FH. Genistein-induced upregulation of p21WAF1, downregulation of cyclin B, and induction of apoptosis in prostate cancer cells. Nutr Cancer. 1998 32(3):123-31.
- Davis JN, Kucuk O, Sarkar FH. Genistein inhibits NF-kappa B activation in prostate cancer cells. Nutr Cancer. 1999 35(2):167-74.
- Ozasa K, Nakao M, Watanabe Y, et al. Serum phytoestrogens and prostate cancer risk in a nested case-control study among Japanese men. Cancer Sci. 2004 Jan;95(1):65-71.
- Park HJ, Jeon YK, You DH, Nam MJ. Daidzein causes cytochrome c-mediated apoptosis via the Bcl-2 family in human hepatic cancer cells. Food Chem Toxicol. 2013 Oct;60:542-9.
- Dong X, Xu W, Sikes RA, Wu C. Combination of low dose of genistein and daidzein has synergistic preventive effects on isogenic human prostate cancer cells when compared with individual soy isoflavone. Food Chem. 2013 Dec 1;141(3):1923-33.
- Henning SM, Aronson W, Niu Y, et al. Tea polyphenols and theaflavins are present in prostate tissue of humans and mice after green and black tea consumption. J Nutr. 2006 Jul;136(7):1839-43.
- Bettuzzi S, Brausi M, Rizzi F, Castagnetti G, Peracchia G, Corti A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: a preliminary report from a one-year proof-of-principle study. Cancer Res. 2006 Jan 15;66(2):1234-40.
- Pandey M, Gupta S. Green tea and prostate cancer: from bench to clinic. Front Biosci (Elite Ed). 2009;1:13-25.
- McLarty J, Bigelow RL, Smith M, Elmajian D, Ankem M, Cardelli JA. Tea polyphenols decrease serum levels of prostate-specific antigen, hepatocyte growth factor, and vascular endothelial growth factor in prostate cancer patients and inhibit production of hepatocyte growth factor and vascular endothelial growth factor in vitro. Cancer Prev Res (Phila). 2009 Jul;2(7):673-82.
- Siddiqui IA, Zaman N, Aziz MH, et al. Inhibition of CWR22Rnu1 tumor growth and PSA secretion in athymic nude mice by green and black teas. Carcinogenesis. 2006 Apr;27(4):833-9.
- Chuu CP, Chen RY, Kokontis JM, Hiipakka RA, Liao S. Suppression of androgen receptor signaling and prostate specific antigen expression by (-)-epigallocatechin-3-gallate in different progression stages of LNCaP prostate cancer cells. Cancer Lett. 2009 Mar 8;275(1):86-92.
- Available at: http://www.foxnews.com/story/2005/07/06/fda-rejects-green-tea-cancer-claims. Accessed October 4, 2013.
- Available at: http://www.whfoods.com/genpage.php?tname=nutrient&dbid=84#foodchart. Accessed October 4, 2013.

