Life Extension Magazine®

Many people think of whey protein as a supplement only used by athletes wanting to increase their muscle mass. But evolving research suggests the branched-chain amino acids (BCAAs) leucine, isoleucine, and valine and other fractions found in whey can mimic the longevity benefits of calorie restriction (CR)!
Searching for CR-mimetics, scientists recently investigated the life span effects of a balanced amino acid mixture high in the branched chain amino acids found in whey.1,2 In keeping with earlier studies on other organisms,3,4 they found branched chain amino acids significantly increased life span in mice.2
This “superfood” provides all 18 amino acids, naturally occurring bioactive components such as immunoglobulins and lactoferrin, and a cysteine concentration over 4-fold higher than other proteins—all in a highly absorbable form.5-8 And whey is one of the greatest foods for maximizing glutathione,9,10 the endogenous antioxidant that quenches oxidative stress11,12 to inhibit diseases of aging.10,13-15
In addition to expanding life span, whey has been found to reduce muscle wasting in the elderly, reduce the risk of diabetes, inhibit cancer development, and defend against cognitive decline.16-19
Given all these benefits, whey has now been substantiated by significant science as a powerful super food in the quest for healthy longevity.
Calorie Restriction Benefits—Without Starving!

It is surprising to learn that about 45% of older individuals in the general population—and 84% to 100% in residential care facilities—are not adequately nourished.1,20 This results from reduced appetite and food intake, impaired nutrient absorption, and age-related medical and social changes.20,21
In particular, an insufficient intake of quality protein can lead to loss of muscle mass, reduced strength, decreased bone mass, low immunity, cognitive impairment, and delayed wound and surgery recovery. Low protein intake is a strong independent predictor of mortality in aging people.1,22 This problem leads to the functional decline known as frailty, a recognized geriatric syndrome.23
A second unfortunate factor is that, although calorie restriction (CR) has been shown to have beneficial, health-and-longevity effects, long-term adherence requires a major commitment of will power—and its potential risks for the elderly have yet to be determined.24,25
These two factors together—reduced protein intake among the aging, and the unlikelihood that people will follow a CR regime—bring us to whey’s CR-mimetic effects. Providing the benefits of CR without a reduction in food intake has now become a hot area of investigation within gerontology. 25,26
Whey is a potential solution. First, whey represents a high-quality protein supplement for aging persons—with a rich source of BCAAs to stimulate protein synthesis and inhibit protein breakdown.27,28
Second, whey delivers CR benefits—playing pivotal roles in hormone secretion and action, intracellular signaling, and regulation of gene transcription and translation.1,2,29 Let’s review some of these longevity studies.
CR Mimetic: Whey Can Add the Equivalent of Nearly 10 Human Years
Scientists investigating calorie restriction mimetic compounds discovered that increasing the availability of the branched chain amino acids abundantly found in whey—leucine, isoleucine, and valine—increased life span in yeast (Saccharomyces cerevisiae).1,3
Similarly, it was found that the most striking distinction common only to long-lived mutant varieties of Caenorhabditis elegans worms was their increased up-regulation of these same branched chain amino acids.1
Then in a breakthrough study, scientists supplemented the diet of mice with a branched chain amino acids-enriched mixture of amino acids, similar to the composition of whey. The test mice received the mixture daily beginning at nine months of age, which is middle-aged for mice. There was no significant change in maximum life span for any of the mice in the study. However, the median life span was 774 days for the untreated controls, and 869 days for the whey supplemented mice—an increase of more than 12% after starting the protocol only in middle age!2
In human terms, based on today’s life expectancy of 78.7 years,30 this median increase—even beginning supplementation only at midlife—translates to an additional 9.4 years of life!
Whey Can Prevent Type II Diabetes—A Major Aging Disease!
Scientists demonstrated that whey can reduce the risk of developing type II diabetes.16 Normally, a high-fat diet produces excessive weight gain, adiposity, and metabolic complications associated with higher risk for type II diabetes and fatty liver disease. Scientists placed mice on a high-fat diet for eleven weeks and gave one group 100 grams of whey protein per liter of drinking water (equivalent to approximately 12 grams for an average 165 pound human).
With no other intervention, the whey-protein mice improved both their glucose tolerance and insulin sensitivity. They also maintained lower weight and greater percentage of lean body mass, compared to control mice consuming the same daily calories but without the added whey. The team concluded that whey protein boosted metabolic rate in the test group and may be beneficial in preventing the development of type II diabetes.16
Another team found that whey remarkably decreases blood sugar without increasing insulin secretion. For non-diabetic individuals, taking protein along with oral glucose would normally decrease the usual rise in blood sugar. Researchers wondered whether whey protein could still lower blood glucose in humans diagnosed with insulin-resistant prediabetes—or whether, instead, their insulin resistance would blunt the hypoglycemic effect. 31
They divided the human subjects into three groups according to level of insulin resistance. For eleven mornings, they gave 0,5, or 30 grams of protein (from whey protein concentrate) and canola oil to all participants along with doses of 50 grams of oral glucose.31
Whey significantly reduced blood glucose levels in all three categories of insulin resistance—yet the rate of insulin secretion was not affected. They concluded that, despite very high levels of insulin resistance in some of the individuals, whey was still able to decrease blood sugar levels.31
What You Need to Know
 |
Whey: The New CR Powerhouse
- Although whey is often viewed simply as a protein source, emerging evidence indicates that the branched-chain amino acids (BCAAs) and other fractions found in whey act as a calorie restriction-mimetic.
- Studies on middle-aged animals demonstrate that supplementing with the branched chain amino acids and other components in whey increase median life span by up to the equivalent of 9.6 years in human terms!
- This effect is modulated along typical CR pathways—boosting mitochondrial biogenesis, reactive oxygen species (ROS) defense, cell signaling, and expression of sirtuin 1 (SIRT1)—the mammalian longevity gene CR.
- Whey constituents increase median, but not maximum, life span—indicating disease prevention at work. And whey’s protection against age-related diseases has been separately confirmed.
- Whey offers an ideal supplement to secure calorie restriction’s life extension benefits—without the rigors of a low calorie regime.
Whey Maintains Youthful Weight and Lean Body Mass
In 2013, scientists reviewed many previous studies and concluded that the evidence supports the use of whey as a therapeutic treatment for obesity.32 Specifically, the study team concluded that:
“Whey protein, via bioactive peptides and amino acids generated during gastrointestinal digestion, enhances the release of several hormones…that lead to reduced food intake and increased satiety.”32
In a host of studies—all with similar conclusions—scientists have investigated pathways through which the proteins, peptides, and minerals in whey boost satiety, influence glucose homeostasis, and optimize lean body mass.33-37
The branched chain amino acids play an essential role in supporting lean muscle mass,especially among older adults.13,38 One trial compared diets high in leucine—abundant in whey—to calorie-equivalent diets high in carbohydrate. After 16 weeks, the leucine group lost 6.8 pounds more in total body weight, 7 pounds more in body fat—but lost 1.5 pounds less in lean body mass!39
What is a Calorie Restriction Mimetic?
 |
A wealth of scientific data spanning more than seven decades shows that calorie restriction (CR), the reduction of caloric intake without malnutrition, has power to boost longevity in nearly all forms of life—as much as 100% in some species.84,85
However, most people cannot submit to a sufficiently restrictive dietary regimen to obtain the benefits. And the effects and risks for elderly individuals have not been well investigated.
The good news is that pioneering researchers have brought to light a novel class of nutrients called calorie restriction mimetics (CR-mimetics). The administration of a CR-mimetic results in some of the same physiological changes seen in CR itself. One way these substances work is by influencing specific genes that ultimately affect either cell repair or cell death.
A handful of natural compounds have been shown to safely simulate some of the beneficial effects of calorie restriction— without the need for stringent dietary measures.
Each of the following CR-mimetics operates in a multi-targeted and complementary fashion—allowing you the opportunity to activate your longevity genes without hunger:99-104
Resveratrol, the most widely known of the CR mimetics, is a polyphenol found most abundantly in red wine grape skins, as well as in many other darkly colored fruits.
Fisetin is a relatively rare flavonoid found only in minute quantities in the plant kingdom that augments the effects of resveratrol.
Pterostilbene is a polyphenol closely related to resveratrol, but with unique attributes.
Grape Seed Extract is a derivative of whole grape seeds; resveratrol is a polyphenol contained in grape seeds.
Quercetin is a plant-derived flavonoid found in fruits, vegetables, leaves, and grains.
Black Tea, a type of tea that is more oxidized than oolong, green, and white teas, contains polyphenols and other constituents.
Whey has newly emerged as a CR mimetic and is gaining acceptance as the first whole food CR-mimetic.
Block Age-Related Muscle Loss and Muscle Wasting
Approximately 30% of people aged 60 or over have sarcopenia, age-related muscle-wasting that increases the risk of falls and disability.40 By sparing and synthesizing muscle, whey blocks this process.41,42 (This mimics the proven sarcopenia-delaying effects of calorie restriction.43)
The most metabolically active branched chain amino acid in whey is leucine, which promotes muscle synthesis by activating a signaling pathway in muscle that modulates the body’s anabolic drive.44-48 Aging muscle becomes resistant to leucine stimulation, but taking leucine with meals (or in leucine-rich whey) can overcome this aging deficit, stimulating muscle synthesis.46-50
Cachexia is a clinical wasting syndrome characterized by progressive loss of weight, muscle, and strength.51 Increased calorie intake doesn’t help.51 Cachexia can occur with cancer, AIDS, rheumatoid arthritis, and various chronic or malignant diseases—and is a major cause of death.
Because whey has among the highest biological values—meaning absorption, use, and retention—of any other protein source, it attenuates catabolic wasting by boosting muscle mass.5-7,52,53
One study involving HIV-positive men given whey found dramatic increases in glutathione, with most subjects reaching ideal body weight.54
Aging individuals may also benefit from whey’s ability to promote bone formation and suppress bone resorption.55
Whey’s Multiple Anti-Aging Benefits
 |
This highest-quality of all protein sources delivers the wide spectrum of anti-aging effects provided by a calorie restriction (CR) diet, without the need for the willpower of long-term starvation. (See figure 2)
Among the many anti-aging benefits of whey are:
- Mitochondrial biogenesis2
- Bone growth55
- Muscle support13,38
- Hypoglycemic effect16,31
- Anticancer defense56-58
- Antioxidant enhancement10
- Anti-inflammatory protection69
- Anti-hypertensive effect60
- Anti-microbial activity8
- Cholesterol reduction60
- Mood balance66
- Cognitive function67
- Reduced sarcopenia41,42
Whey Protein Inhibits Development of Cancer Cells
Animal studies have demonstrated that whey is superior to other dietary proteins for suppression of cancer development.56-58
This benefit is attributed to whey’s high content of cystine/cysteine and gamma-glutamylcysteine dipeptides—used for synthesis of glutathione—giving whey its rare ability to raise levels of glutathione.9,10 Glutathione is well-known to destroy reactive oxygen species—but it also detoxifies carcinogens and ensures a competent immune system!56
Studies show that whey’s tumor prevention is accompanied by enhanced glutathione levels, spleen lymphocyte proliferation, phagocytosis, and activity of natural killer, T helper, and cytotoxic T cells.56
And the whey component lactoferrin powerfully inhibits tumors by various pathways, including inducing apoptosis, blocking angiogenesis, modulating carcinogen-metabolizing enzymes, and possibly by scavenging iron.56
Whey Component Uniquely Modulates Iron for Broad Health Benefits
 |
Lactoferrin is a minor fraction of whey (see figure 1). Its unique ability to modulate iron—up or down as needed—in different tissues provides a remarkably broad range of health benefits that contribute to whey’s life extension effects.
Extensive scientific research demonstrates that, through its wide network of modulatory effects, lactoferrin:
- Kills dangerous microbes86,87
- Nurtures beneficial bacteria88
- Binds iron to prevent free radicals89,90
- Optimizes blood-iron levels86,91
- Inhibits cancer cells86,92
- Enhances intestinal absorptive cells93 and maturation of dendritic cells94
- Boosts immunity94
- Enhances gene transcription95,96
- Reduces inflammation86,97
- Combats obesity98
It’s no wonder scientists have described the whey protein lactoferrin as “multifunctional!”86
Unique Protein Subfractions Modulate the Origins of Cardiovascular Disease

Figure 1: Whey Proteins are the Healthy Constituent of Milk.
The protein component of milk is composed of 20% whey and 80% casein. The whey fraction is made up of various protein components that each have their own unique benefits.6
Cardiovascular disease is the leading cause of mortality in Western society, and the two main contributing factors are hypertension (high blood pressure) and dyslipidemia (elevated LDL/reduced HDL).59
Recent research demonstrates that whey-based peptides reduce both of these risk factors through various pathways.60,61 And food-derived peptides are considered far safer than anti-hypertension drugs.
Several whey peptides—including alpha-lactalbumin and beta-lactoglobulin—exhibit opioid-like activity. Endogenous opioid peptides have shown promise as blood-pressure modulators.60
And limited evidence suggests whey’s lactoferrin helps modulate platelet binding.60
Casein protein raises cholesterol—but whey protein shows the opposite effect!60,61 Two-generation feeding of rats with sphingolipids—a type of phospholipid found in whey—decreased blood cholesterol levels by about 30%!60 How? Scientists speculate whey inhibits synthesis of cholesterol in the liver.61
Research also demonstrated a marked lowering of LDL cholesterol in rats given whey, which may be mediated through a decrease in VLDL (very low-density lipoprotein) fractions.60
Extremely interesting was the fact that reduction of LDL-cholesterol was not seen in animals fed amino acid mixtures that were identical in profile to whey protein—showing the benefits of whey are specific to its unique protein subfractions as opposed to its amino acid profile alone.61
Clearly, compounds in whey have cardioprotective effects that go beyond those of its amino acid profile!
Whey Defends Against Neurocognitive Decline During Aging
Serotonin levels in the body can decline with age.62,63 The rate of the brain’s serotonin synthesis normally depends on its concentration oftryptophan, serotonin’s essential amino acid precursor.64,65 Reinforcing this, dietary intake of tryptophan has been found to relieve depression and stress.66
A double-blind, placebo-controlled study indicated that whey’s alpha-lactalbumin increases the plasma ratio of tryptophan. And in subjects assessed as highly vulnerable to stress, this whey fraction raised brain serotonin activity and coping ability and improved mood under stress.66
Memory performance can decline under chronic stress, believed to result partially from reduced brain serotonin. In a double-blind, placebo-controlled study, whey significantly improved memory-test performance in stress-vulnerable subjects.67
Also, because it boosts glutathione levels,9,10 whey may protect against various age-related diseases—including neurocognitive disorders.13-19, 66-68
A 2012 review suggested that the peptides and alpha-lactalbumin found in whey may help protect against age-related cognitive decline and dementia during aging.68
Whey Components Protect Gastrointestinal Tissue and Boost Beneficial Bacteria
Because whey raises glutathione levels, it protects tissue against damage caused by inflammation.69,70 Lower levels of antioxidants—including glutathione—are associated with inflammatory bowel disease (IBD).71
Also, glutamine is a preferred fuel source for support of epithelial cells lining the small intestine. It is rapidly utilized by tissues during catabolic states, such as gastrointestinal dysfunction among children.72
Dietary components that act as prebiotics can improve balance and persistence of beneficial colonic bacteria by providing substrates (growth surfaces). Whey lactose can be converted to galactooligosaccharides—indigestible ingredients that create a substrate that specifically enhances growth of bifidobacteria.73 These and other bacteria protect the intestine by secreting antimicrobial substances, modulating immune response, and influencing metabolic activities.74,75
In rats, the whey component alpha-lactalbumin showed dose-dependent protection against gastric injury caused by alcohol or stress.76 Researchers concluded that whey may help prevent gastric mucosal injury, ulcers, and other gastrointestinal pathologies—avoiding the need for antiulcer drugs, and their risks.76
Whey Proteins Rejuvenate and Optimize Immune Function!
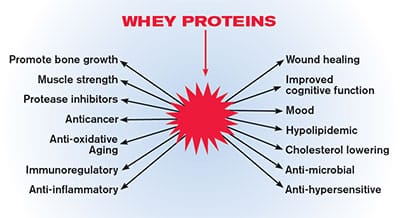
Figure 2: Functional Properties of Whey.
Whey proteins provide a diverse array of functional properties that produce beneficial effects on many different biological processes and organ systems.6
Whey uniquely fortifies the overall immune system—so broadly that it’s remarkable!8
Major and minor whey fractions are established immune-enhancing constituents that modulate a full range of immune functions, including destruction of pathogens and elimination of toxins.8,77,78
Whey supplementation significantly improves primary and secondary antibody responses to a variety of vaccines.79
A key immunity pillar is the cell-mediated, adaptive immune system, which—through clonal expansion (proliferation)—builds up a pool of antigen-reactive bodies.8,77 Whey was found in an array of studies to be superior to other commercially available protein sources in improving adaptive immune reactivity.14,80-82
Mice supplemented with whey produced higher levels of white blood cells, lymphocytes, and cytokines—resulting in greater immune responsiveness and reduced infection severity.8,82
The antibody-mediated, humoral immune system needs to quickly produce antibodies in response to foreign microorganisms. Several studies found that, compared to other protein sources—soy, casein, wheat, corn, egg white, fish, beef, or spirulina maxima—whey proteins much more substantially boost production of both humoral immune response and antibodies.14,82,83 In fact, in some instances, the response was nearly five times greater than with other protein sources!14
Glutathione is the centerpiece of immune protection against endogenously created oxidative stress as well as that from exogenous sources such as pollution, toxins, exercise, and ultraviolet light exposure.11 And as mentioned, whey can greatly increase glutathione levels.9,10
An example of a condition characterized by glutathione deficiency—and greater oxidative stress—is HIV infection. Fortunately, whey supplements have been shown to dramatically increase glutathione levels in those with HIV!54
Summary
Whey is often viewed simply as a high-quality protein source for athletes. However, emerging evidence indicates that the branched-chain amino acids (BCAAs) and other fractions found in whey mimic many of the longevity benefits of calorie restriction (CR).
Studies on various organisms, including middle-aged mice, demonstrate that supplementing with the BCAAs and other components in whey can increase median life span by up to the equivalent of 9.6 years in human terms!
This effect was modulated along typical CR pathways, including increases in mitochondrial biogenesis, reactive oxygen species (ROS) defense, cell signaling, and expression of sirtuin 1 (SIRT1)—the mammalian longevity gene induced by CR!
Whey offers a method to secure some of the anti-aging effects of calorie restriction without extreme food deprivation.
If you have any questions on the scientific content of this article, please call a Life Extension® Health Advisor at 1-866-864-3027.
Editor's Note
Science continues to evolve, and new research is published daily. As such, we have a more recent article on this topic: Whey's Longevity Benefits
References
- Valerio A, D’Antona G, Nisoli E. Branched-chain amino acids, mitochondrial biogenesis, and healthspan: an evolutionary perspective. Aging (Albany NY). 2011 May;3(5):464-78.
- D’Antona G, Ragni M, Cardile A, et al. Branched-chain amino acid supplementation promotes survival and supports cardiac and skeletal muscle mitochondrial biogenesis in middle-aged mice. Cell Metab. 2010 October;12(4):362-72.
- Alvers AL, Fishwick LK, Wood MS, Hu D, Chung HS, Dunn WA, and Aris JP. Autophagy and amino acid homeostasis are required for chronological longevity in Saccharomyces cerevisiae. Aging Cell. 2009;8:353-69.
- Fuchs S, Bundy JG, Davies SK, Viney JM, Swire JS, and Leroi AM. A metabolic signature of long life in Caenorhabditis elegans. BMC Biol. 2010;8:14.
- Yalçin AS. Emerging therapeutic potential of whey proteins and peptides. Curr Pharm Des. 2006;12(13):1637-43.
- Krissansen GW. Emerging health properties of whey proteins and their clinical implications. J Am Coll Nutr. Dec. 2007;26(6):713S-23S.
- Vegarud GE, Langsrud T, Svenning C. Mineral-binding milk proteins and peptides; occurrence, biochemical and technological characteristics. Br J Nutr. 2000;84(Suppl 1):S91-8.
- Available at: http://www.innovatewithdairy.com/sitecollectiondocuments/mono_immunity_0304.pdf. Accessed June 12, 2013.
- Kent KD, Harper WJ, Bomser JA. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol In Vitro. 2003 Feb;17(1):27-33.
- Bounous G, Gervais F, Amer V, Batist G, Gold P. The influence of dietary whey protein on tissue glutathione and the diseases of aging. Clin Invest Med. 1989;12:343-9.
- Townsend DM, Tew KD, Tapiero H. The importance of glutathione in human disease. Biomed Pharmacother. 2003 May-Jun;57(3-4):145-55.
- Wu G, Fang YZ, Yang S, Lupton JR, Turner ND. Glutathione metabolism and its implications for health. J Nutr. 2004 Mar;134(3):489-92.
- Katsanos CS, Chinkes DL, Paddon-Jones D, Zhang XJ, Aarsland A, Wolfe RR. Whey protein ingestion in elderly persons results in greater muscle protein accrual than ingestion of its constituent essential amino acid content. Nutr Res. 2008 Oct;28(10):651-8.
- Bounous G, Batist G, Gold P. Immunoenhancing property of dietary whey protein in mice: role of glutathione. Clin Invest Med. 1989 Jun;12(3):154-61.
- Currais A, Maher P. Functional consequences of age-dependent changes in glutathione status in the brain. Antioxid Redox Signal. 2013 Feb 5. [Epub ahead of print]
- Shertzer HG, Woods SE, Krishan M, Genter MB, Pearson KJ. Dietary whey protein lowers the risk for metabolic disease in mice fed a high-fat diet. J Nutr. 2011 Apr 1;141(4):582-7.
- Paddon-Jones D, Short KR, Campbell WW, Volpi E, Wolfe RR. Role of dietary protein in the sarcopenia of aging. Am J Clin Nutr. 2008 May;87(5):1562S-6S.
- Pepe G, Tenore GC, Mastrocinque R, Stusio P, Campiglia P. Potential anticarcinogenic peptides from bovine milk. J Amino Acids. 2013;2013:939804.
- Markus CR, Olivier B, de Haan EH. Whey protein rich in alpha-lactalbumin increases the ratio of plasma tryptophan to the sum of the other large neutral amino acids and improves cognitive performance in stress-vulnerable subjects. Am J Clin Nutr. 2002 Jun;75(6):1051-6.
- Visvanathan R, and Chapman IM. Undernutrition and anorexia in the older person. Gastroenterol Clin North Am. 2009;38:393-409.
- Ahmed T, and Haboubi N. Assessment and management of nutrition in older people and its importance to health. Clin Interv Aging. 2010;5:207-16.
- Chapman IM. Nutritional disorders in the elderly. Med Clin North Am. 2006;90:887-907.
- Ahmed N, Mandel R, and Fain MJ. Frailty: An emerging geriatric syndrome. Am J Med. 2007;120:748-53.
- Heilbronn LK, de Jonge L, Frisard MI, et al. Effect of 6-month calorie restriction on biomarkers of longevity, metabolic adaptation, and oxidative stress in overweight individuals: A randomized controlled trial. JAMA. 2006;295:1539-48.
- Ingram DK, Roth GS. Glycolytic inhibition as a strategy for developing calorie restriction mimetics. Exp Gerontol. 2011;46:148-54.
- Ingram DK, Zhu M, Mamczarz J, et al. Calorie restriction mimetics: an emerging research field. Aging Cell. 2006 Apr;5(2):97-108.
- Solerte SB, Gazzaruso C, Bonacasa R, et al. Nutritional supplements with oral amino acid mixtures increases whole-body lean mass and insulin sensitivity in elderly subjects with sarcopenia. Am J Cardiol. 2008;101:69E-77E.
- Available at: http://www.encognitive.com/files/Potential%20benefits%20of%20improved% 20protein%20intake%20in%20older%20people.pdf. Accessed June 12, 2013.
- Nair KS, Short KR. Hormonal and signaling role of branched-chain amino acids. J Nutr. 2005 Jun;135(6 Suppl):1547S-52S.
- Available at: http://www.cdc.gov/nchs/fastats/lifexpec.htm. Accessed June 12, 2013.
- Lan-Pidhainy X, Wolever TM. The hypoglycemic effect of fat and protein is not attenuated by insulin resistance. Am J Clin Nutr. 2010 Jan;91(1):98-105.
- Jakubowicz D, Froy O. Biochemical and metabolic mechanisms \by which dietary whey protein may combat obesity and type 2 diabetes. J Nutr Biochem. 2013 Jan;24(1):1-5.
- Hall WL, Millward DJ, Long SJ, Morgan LM. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. Br J Nutr. 2003;89:239-48.
- Pal S, Ellis V. The acute effects of four protein meals on insulin, glucose, appetite and energy intake in lean men. Br J Nutr. 2010;104:1241-8.
- Veldhorst MA, Nieuwenhuizen AG, Hochstenbach-Waelen A, et al. Dose-dependent satiating effect of whey relative to casein or soy. Physiol Behav. 2009;96:675-82.
- Bowen J, Noakes M, Trenerry C, Clifton PM. Energy intake, ghrelin, and cholecystokinin after different carbohydrate and protein preloads in overweight men. J Clin Endocrinol Metab. 2006;91:1477-83.
- Baer DJ, Stote KS, Paul DR, Harris GK, Rumpler WV, Clevidence BA. Whey protein but not soy protein supplementation alters body weight and composition in free-living overweight and obese adults. J Nutr. 2011;141:1489-94.
- Dangin M, Boirie Y, Guillet C, Beaufrere B. Influence of the protein digestion rate on protein turnover in young and elderly subjects. J Nutr. 2002 Oct;132(10):3228S-33S.
- Available at: http://www.usdec.org/files/pdfs/2008monographs/weighweightmanagement_english.pdf . Accessed June 13, 2013.
- Doherty TJ. Invited review: Aging and sarcopenia. J Appl Physiol. 2003. 95(4):1717-27.
- Graf S, Egert S, Heer M. Effects of whey protein supplements on metabolism: evidence from human intervention studies. Curr Opin Clin Nutr Metab Care. 2011 Nov;14(6):569-80.
- Paddon-Jones D, Sheffield-Moore M, Katsanos CS, Zhang XJ, Wolfe RR. Differential stimulation of muscle protein synthesis in elderly humans following isocaloric ingestion of amino acids or whey protein. Exp Gerontol. 2006 Feb;41(2):215-9.
- McKiernan SH, Colman RJ, Lopez M, et al. Caloric restriction delays aging-induced cellular phenotypes in rhesus monkey skeletal muscle. Exp Gerontol. 2011 January;46(1):23-9.
- Paddon-Jones D, Short KR, Campbell WW, Volpi E, Wolfe RR. Role of dietary protein in the sarcopenia of aging. Am J Clin Nutr. 2008 May;87(5):1562S-6S.
- Koopman R, Verdijk L, Manders RJ, et al. Co-ingestion of protein and leucine stimulates muscle protein synthesis rates to the same extent in young and elderly lean men. Am J Clin Nutr. 2006 Sep;84(3):623-32.
- Dardevet D, Sornet C, Balage M, Grizard J. Stimulation of in vitro rat muscle protein synthesis by leucine decreases with age. J Nutr. 2000 Nov;130(11):2630-5.
- Katsanos CS, Kobayashi H, Sheffield-Moore M, Aarsland A, Wolfe RR. A high proportion of leucine is required for optimal stimulation of the rate of muscle protein synthesis by essential amino acids in the elderly. Am J Physiol Endocrinol Metab. 2006 Aug;291(2):E381-7.
- Fujita S, Dreyer HC, Drummond MJ, et al. Nutrient signalling in the regulation of human muscle protein synthesis. J Physiol. 2007 Jul 15;582(Pt 2):813-23.
- Dreyer HC, Drummond MJ, Pennings B, et al. Leucine-enriched essential amino acid and carbohydrate ingestion following resistance exercise enhances mTOR signaling and protein synthesis in human muscle. Am J Physiol Endocrinol Metab. 2008 Feb;294(2):E392-400.
- Fujita S, Volpi E. Amino acids and muscle loss with aging. J Nutr. 2006 Jan;136(1 Suppl):277S-80S.
- Morley JE, Thomas DR, Wilson MM. Cachexia: pathophysiology and clinical relevance. Am J Clin Nutr. 2006 Apr;83(4):735-43.
- Hack V, Schmid D, Breitkreutz R, et al. Cystine levels, cystine flux, and protein catabolism in cancer cachexia, HIV/SIV infection, and senescence. FASEB J. 1997 Jan;11(1):84-92.
- Dillon EL, Basra G, Horstman AM, et al. Cancer cachexia and anabolic interventions: a case report. J Cachexia Sarcopenia Muscle. 2012 Dec;3(4):253-63.
- Bounous G, Baruchel S, Falutz J, Gold P. Whey proteins as a food supplement in HIV-seropositive individuals. Clin Invest Med. 1993 Jun;16(3):204-9.
- Aoe S, Toba Y, Yamamura J, et al. Controlled trial of the effects of milk basic protein (MBP) supplementation on bone metabolism in healthy adult women. Biosci Biotechnol Biochem. 2001;65:913-8.
- Parodi PW. A role for milk proteins and their peptides in cancer prevention. Curr Pharm Des. 2007;13(8):813-28.
- McIntosh GH, Regester GO, Le Leu RK, Royle PJ, Smithers GW. Dairy proteins protect against dimethylhydrazine-induced intestinal cancers in rats. J Nutr. 1995 Apr;125(4):809-16.
- Tsuda H, Sekine K, Ushida Y, et al. Milk and dairy products in cancer prevention: focus on bovine lactoferrin. Mutat Res. 2000 Apr;462(2-3):227-33.
- Available at: https://www.cdc.gov/heartdisease/risk_factors.htm. Accessed June 14, 2013.
- Available at: http://usdec.files.cms-plus.com/publications/cardiohealth_english.pdf. Accessed June 14, 2013.
- Zhang X, Beynen AC. Lowering effect of dietary milk-whey protein v. casein on plasma and liver cholesterol concentrations in rats. Br J Nutr. 1993 Jul;70(1):139-46.
- Meltzer CC, Smith G, DeKosky ST, et al. Serotonin in aging, late-life depression, and Alzheimer’s disease: the emerging role of functional imaging. Neuropsychopharmacology. 1998 Jun;18(6):407-30.
- Meltzer CC, Price JC, Mathis CA, et al. Serotonin 1A receptor binding and treatment response in late-life depression. Neuropsychopharmacology. 2004 Dec;29(12):2258-65.
- Fernstrom JD, Wurtman RJ. Brain serotonin content: physiological dependence on plasma tryptophan levels. Science. 1971;173:149-52.
- Fernstrom JD, Wurtman RJ. Brain serotonin content: physiological regulation by plasma neutral amino acids. Science. 1972;178:414-6.
- Markus CR, Olivier B, Panhuysen GE, et al. The bovine protein alpha-lactalbumin increases the plasma ratio of tryptophan to the other large neutral amino acids, and in vulnerable subjects raises brain serotonin activity, reduces cortisol concentration, and improves mood under stress. Am J Clin Nutr. 2000 Jun;71(6): 1536-44.
- Markus CR, Olivier B, de Haan EH. Whey protein rich in alpha-lactalbumin increases the ratio of plasma tryptophan to the sum of the other large neutral amino acids and improves cognitive performance in stress-vulnerable subjects. Am J Clin Nutr. 2002 Jun;75(6):1051-6.
- Camfield DA, Owen L, Scholey AB, Pipingas A, Stough C. Dairy constituents and neurocognitive health in ageing. Br J Nutr. 2011 Jul;106(2):159-74.
- Chatterton DE, Nguyen DN, Bering SB, Sangild PT. Anti-inflammatory mechanisms of bioactive milk proteins in the intestine of newborns. Int J Biochem Cell Biol. 2013 May 6.
- Rahman I, MacNee W. Oxidative stress and regulation of glutathione in lung inflammation. Eur Respir J. 2000 Sep;16(3):534-54.
- Buffinton GD, Doe WF. Depleted mucosal antioxidant defences in inflammatory bowel disease. Free Radic Biol Med. 1995;19:911-8.
- Duggan C, Stark AR, Auestad N, et al. S. Collier. 2004. Glutamine supplementation in infants with gastrointestinal disease: a randomized, placebo-controlled pilot trial. Nutrition.2004;20:752-6.
- Walzem RL, Dillard CJ, German JB. Whey components: millennia of evolution create functionalities for mammalian nutrition: What we know and what we may be overlooking. Crit Rev Food Sci Nutr. 2002;42:353-75.
- Fioramonti J, Theodorou V, Bueno L. Probiotics: what are they? What are their effects on gut physiology? Best Pract Res Clin Gastroenterol. 2003;17:711-24.
- Teitelbaum JE, Walker WA. Nutritional impact of pre- and probiotics as protective gastrointestinal organisms. Annu Rev Nutr 2002;22:107-38.
- Matsumoto H, Shimokawa Y, Ushida Y, Toida T, Hayasawa H. New biological function of bovine alpha-lactalbumin: Protective effect against ethanol- and stress-induced gastric mucosal injury in rats. Biosci Biotechnol Biochem. 2001 May;65(5):1104-11.
- Cross M L, Gill HS. Immunomodulatory properties of milk. Brit J Nutr. 2000;84:S81-9.
- Clare DA, Swaisgood HE. Bioactive milk peptides: A prospectus. J Dairy Sci. 2000;83:1187-95.
- Low PPL, Rutherford KJ, Gill HS, Cross ML. Effect of dietary whey protein concentrate on primary and secondary antibody responses in immunized BALB/C mice. Int Immunopharmacol. 2003;3:393-401.
- Bounous G, Kongshavn PA, Gold P. The immunoenhancing property of dietary whey protein concentrate. Clin Invest Med. 1988 August;11(4):271-8.
- Bounous G, Papenburg R, Kongshavn PA, Gold P, Fleiszer D. Dietary whey protein inhibits the development of dimethylhydrazine induced malignancy. Clin Invest Med. 1988;11(3)213-7.
- Ford JT, Wong CW, Colditz IG. Effects of dietary protein types on immune responses and levels of infection with Eimeria vermiformis in mice. Immunol Cell Biol 2001;79(1):23-8.
- Bounous G, Kongshavn PA. Differential effect of dietary protein type on the B-cell and T-cell immune responses in mice. J Nutr. 1985;115(11):1403-8.
- McCay CM, Crowel MF. Prolonging the life span. Scientific Monthly. 1934 Nov;39(5):405-14.
- McCay CM, Crowell MF, Maynard LA. The effect of retarded growth upon the length of life span and upon the ultimate body size. Nutrition. 1935;5:155-71.
- Ward PP, Paz E, Conneely OM. Multifunctional roles of lactoferrin: a critical overview. Cell Molecul Life Sci. 2005;62,2540-8.
- Wakabayashi H, Yamauchi K, Takase M. Inhibitory effects of bovine lactoferrin and lactoferricin B on Enterobacter sakazakii. Biocontrol Sci. 2008;13:29-32.
- Liepke C, Adermann K, Raida M, et al. Human milk provides peptides highly stimulating the growth of bifidobacteria. Eur J Biochem. 2002;269:712-8.
- Okazaki Y, Kono I, Kuriki TJ, et al. Bovine lactoferrin ameliorates ferric nitrilotriacetate-induced renal oxidative damage in rats. Clin Biochem Nutr. 2012 Sep;51(2):84-90.
- Raghuveer TS, McGuire EM, Martin SM, et al. Lactoferrin in the preterm infants’ diet attenuates iron-induced oxidation products. Pediatr Res. 2002;52:964-72.
- Shoji H, Oguchi S, Shinohara K, et al. Effects of iron-unsaturated human lactoferrin on hydrogen peroxide-induced oxidative damage in intestinal epithelial cells. Pediatr Res. 2007;61:89-92.
- Iigo M, Shimamura M, Matsuda E, et al. Orally administered bovine lactoferrin induces caspase-1 and interleukin-18 in the mouse intestinal mucosa: a possible explanation for inhibition of carcinogenesis and metastasis. Cytokine. 2004;25:36-44.
- Oguchi S, Walker WA, Sanderson IR. Iron saturation alters the effect of lactoferrin on the proliferation and differentiation of human enterocytes (Caco-2 cells). Biol Neonate. 1995;67:330-9.
- Spadaro M, Caorsi C, Ceruti P, et al. Lactoferrin, a major defense protein of innate immunity, is a novel maturation factor for human dendritic cells. FASEB J. 2008;22:2747-57.
- Ashida K, Sasaki H, Suzuki YA, Lonnerdal B. Cellular internalization of lactoferrin in intestinal epithelial cells. Biometals. 2004;17:311-5.
- Garre C, Bianchi-Scarra G, Sirito M, et al. Lactoferrin binding sites and nuclear localization in K562(S) cells. J Cell Physiol. 1992;153:477-82.
- Tung YT, Chen HL, Yen CC, et al. Bovine lactoferrin inhibits lung cancer growth through suppression of both inflammation and expression of vascular endothelial growth factor. J Dairy Sci. 2013 Apr;96(4):2095-106.
- Ono T, Murakoshi M, Suzuki N, et al. Potent anti-obesity effect of enteric-coated lactoferrin: decrease in visceral fat accumulation in Japanese men and women with abdominal obesity after 8-week administration of enteric-coated lactoferrin tablets. Br J Nutr. 2010 Dec;104(11):1688-95.
- Maher P. Modulation of multiple pathways involved in the maintenance of neuronal function during aging by fisetin. Genes Nutr. 2009 Sep 10.
- Sadruddin S, Arora R. Resveratrol: biologic and therapeutic implications. J Cardiometab Syndr. 2009 Spring;4(2):102-6.
- Joseph JA, Fisher DR, Cheng V, Rimando AM, Shukitt-Hale B. Cellular and behavioral effects of stilbene resveratrol analogues: implications for reducing the deleterious effects of aging. J Agric Food Chem. 2008 Nov 26;56(22):10544-51.
- Salas A, Subirada F, Perez-Enciso M, et al. Plant polyphenol intake alters gene expression in canine leukocytes. J Nutrigenet Nutrigenomics. 2009;2(1):43-52.
- Ruiz PA, Braune A, Holzlwimmer G, Quintanilla-Fend L, Haller D. Quercetin inhibits TNF-induced NF-kappaB transcription factor recruitment to proinflammatory gene promoters in murine intestinal epithelial cells. J Nutr. 2007 May;137(5):1208-15.
- Widlansky ME, Duffy SJ, Hamburg NM, et al. Effects of black tea consumption on plasma catechins and markers of oxidative stress and inflammation in patients with coronary artery disease. Free Radic Biol Med. 2005 Feb 15;38(4):499-506.

