Life Extension Magazine®

The first surgical attempt to cure pancreatic cancer was demonstrated in Germany in 1909.1 In 1935, a doctor named Allen Whipple devised a more effective way to remove the pancreas and adjacent body parts.2
Dr. Whipple’s technique involves the removal of the head of the pancreas, along with portions of the stomach, small intestine, gall bladder, and common bile duct.
The surgical impact on the body is severe. There is a higher death rate from this procedure than many other hospital operations.3 Sometimes the rearranged internal organs do not hold together and infection spreads inside the patient. This leads to follow-up surgery where the remainder of the pancreas and the spleen are removed to correct problems caused by the first operation.4

1881-1963.
Some patients do not heal well and leak pancreatic juice from where body parts are sewn together. This happens so frequently that the surgeon leaves in drainage catheters for fluids to exit so they don’t accumulate inside the patient.4,5
Another complication is paralysis of the stomach that can take over a month to heal. During this time a feeding tube is surgically placed into the small intestine to provide nourishment.6
Some patients develop type I diabetes because the insulin-producing areas of their pancreas is removed, requiring life-long insulin injections.7
Despite these horrific surgical side effects, most patients who survive the painful hospital ordeal die from metastatic pancreatic cancer. Few are cured.
The name of this surgery is the “Whipple Procedure.” While it’s been refined since Dr. Whipple’s work in 1935, pancreatic cancer still kills the vast majority of its victims—79 years later!8

Pancreatic Cancer Victim
The snail’s pace of progress against malignancies like pancreatic cancer should provoke societal outrage against the establishment. Yet like lambs standing in line awaiting slaughter, the public tolerates mediocre medicine that is inflicting horrific suffering and massive numbers of needless deaths.
We view these bureaucratic lags as intolerable delays that will be ridiculed by future medical historians. This article describes a drug long ago approved by the FDA that can improve outcomes in pancreatic and other cancer cases. This treatment, however, is not being incorporated into conventional practice.
Steve Jobs was criticized for delaying a Whipple Procedure for nine months after being diagnosed with pancreatic cancer.9 The initial approaches Jobs tried (acupuncture, vegan diet, herbs, spiritualists) had no chance of eradicating his primary pancreatic tumor.
It’s hard to blame the then 49-year-old co-founder of Apple, however, for not wanting his body cut up via a Whipple Procedure. Steve Jobs eventually died at age 56 after undergoing multiple aggressive treatments, including a liver transplant.10-12
How many technologies developed in the early 1900s do consumers still use today? Even the stethoscope (invented in 1819) remains state-of-the-art in today’s archaic world of medical practice.
If one is diagnosed with pancreatic cancer at a relatively early stage, the Whipple Procedure is still the best treatment option. Overlooked are a myriad of adjuvant therapies that can markedly improve long-term survival and reduce the horrific complications inherent to the Whipple surgical procedure.
The cancer treatment I describe next is not new. It has long been recommended to Life Extension® members.
Interleukin-2 Versus Placebo
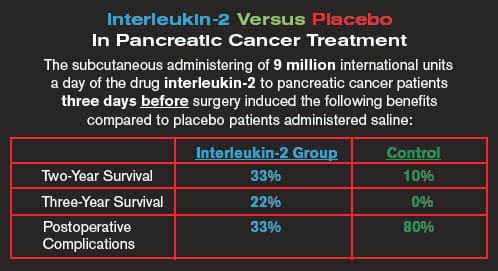
This study should have made headline news. Instead it was buried in a 2006 edition of the journal Hepato-Gastroenterology.46
Life Extension has been recommending moderate dose interleukin-2 as an adjuvant cancer treatment since the late 1990s.
Skeptics point to studies in advanced melanoma and renal cell carcinoma patients where interleukin-2 provides only modest survival improvements. These narrow-focused cynics neglect evidence that interleukin-2 is most effective when administered before immune-suppressing surgery, radiation, and chemotherapy begins.33-37,47,48
Interleukin-2 Improves Survival 3-Fold!
Interleukin-2 (IL-2) enhances overall immune function, most notably by enhancing natural killer cell activity.13-15 Natural killer cells are among the body’s most important immune defenses against malignant and viral-infected cells.16-20 (Cells infected with certain viruses are more prone to convert to malignant cells.)21
IL-2 was long ago approved to treat kidney cancer22-26 and metastatic melanoma.27-29 Its efficacy was likely limited by the advanced disease stage patients are at by the time IL-2 is administered.30 There is toxicity associated with high-dose IL-2.31,32
Intriguing research suggests that administering moderate-dose IL-2 to patients before surgery and chemotherapy may improve survival and other outcomes.33-37 It does this by boosting immune function prior to it being impaired by conventional treatments.
Surgery results in significant immune impairment, something we warned against long before the mainstream considered it a factor in the poor survival rates seen in many types of cancer.38-43 Immune suppression that occurs during chemotherapy is a well-established treatment complication.44,45
In a study conducted on pancreatic cancer patients, half the group was administered moderate dose IL-2 for three consecutive days prior to a Whipple Procedure. Two years after the operation, 33% of patients pre-administered IL-2 were alive compared to only 10% of control surgical patients. Three-year survival was 22% in the IL-2 group compared to 0% of the controls.46
Surgical complications occurred in 80% of the control surgical patients compared with only 33% in the IL-2 pretreatment group. While the control group spent 19.5 days confined to the hospital after their Whipple Procedure, the IL-2 group escaped the hospital in 12 days.46
Life Extension has been recommending moderate-dose IL-2 since the 1990s, yet the mainstream oncologists behave as if these drugs are limited to advanced cancers for which they originally gained FDA-approval. The reality is that IL-2 and other immune-boosting drugs may have far greater efficacy when administered early in the disease process against of a wide range of solid tumors and some types of leukemia.
Why Cancer Patients Need To Boost Natural Killer Cell Activity
Natural killer cells are the part of the immune system that is capable of recognizing and killing virus-infected and malignant cells, while sparing normal cells.49,50
The importance of killing virus-infected cells is that cells infected with human papilloma virus (HPV) and other viruses have greater propensity to mutate into cancer cells. Chronic infection with some of these viruses also exhausts vital immune functions.51
In mice deficient in natural killer cells, tumors grow more aggressively and are more metastatic.52-54
Natural killer cells play an important role in the control of tumor growth.55
Infusion of immune enhancers like interleukin-2 boosts natural killer cell activity which can lead to the death of tumor cells.56
Leukemia patients have benefited using natural killer cells obtained from hematopoietic stem cell donors, which is an exciting area of cancer research.57-59
Non-drug ways of boosting natural killer cell activity include garlic,60-64 melatonin,65-67 Reishi extract,68-71 and other supplements used by Life Extension members. When treating cancer, however, interleukin-2 should be considered to provide an exponential improvement in natural killer cell activity prior to initiation of conventional treatments.
Contrast Mediocre Cancer Treatment To HIV
Cancer is not relegated to modern times. It has killed human beings forever, but has become prominent as people live longer and cancer incidence markedly increases. Pancreatic cancer, for instance, increases sharply in individuals over age 50, and most patients are 60 to 80 years old when diagnosed.72
HIV rose to prominence in the early 1980s, though the virus existed in the human population before then. The problem was that no one paid attention until thousands started dying.
Within 15 years of HIV infection becoming pandemic, effective anti-viral “cocktails” were discovered that turned AIDS from a death sentence into a manageable chronic disease.73-75
In 1981, AIDS was a disease of unknown origin.76 It is controllable today because of rapid scientific innovation. It is controllable today because of rapid scientific innovation. Pancreatic cancer, on the other hand, still kills virtually all its victims with the best hope for long-term survival being the Whipple Procedure first refined in 1935.8
So why were AIDS treatments discovered so quickly while effective cancer therapies languish?
The difference was the aggressive way that experimental multi-modal therapies were implemented in HIV/AIDS patients compared to the suffocating bureaucracy that stymies cancer research.
In the early days of AIDS treatment, any therapy that might work was tried immediately on dying patients and the results evaluated and documented. These treatments were often administered by those infected with HIV who faced pending death if a cure were not discovered quickly. The FDA was cast by the wayside as AIDS activists made certain that potentially effective treatments were not obstructed by bureaucratic red tape.77
We at Life Extension are proud of the part we played in saving the lives of AIDS patients by defying FDA attempts to shut us down. An editorial published late last year in the New England Journal of Medicine revealed how HIV revolutionized the way global health is pursued, and how it resulted in accelerated delivery of innovative life saving treatments.78
New England Journal Of Medicine Praises Work Of Early AIDS Activists
 |
Allan Brandt, PhD, is a professor of medical history at Harvard Medical School. Dr. Brandt’s perspective titled “How AIDS Invented Global Health” was published in the June 6, 2013, edition of the New England Journal of Medicine.79 Here are some quotes from his perspective:
- “AIDS has reshaped conventional wisdoms in public health, research practice, cultural attitudes, and social behaviors.”
- “The rapid development of effective antiretroviral treatments, in turn, could not have occurred without new forms of disease advocacy and activism.”
- “But AIDS activists explicitly crossed a vast chasm of expertise. They went to FDA meetings and events steeped in often arcane science of HIV, prepared to offer concrete proposals to speed research, reformulate trials, and accelerate regulatory processes.”
- “This approach went well beyond the traditional bioethical formulations of autonomy and consent. As many clinicians and scientists acknowledged, AIDS activists, including many people with AIDS, served as collaborators and colleagues rather than constituents and subjects, changing the trajectory of research and treatment.”
Omitted from Dr. Brandt’s complimentary statements were the harassment, persecution, and incarceration of AIDS activists by government agencies that sought to suppress burgeoning development of AIDS therapies.80,81
We Were Jailed!
The FDA did not like our aggressive stance when it came to accelerating medical research, particularly as it related to helping AIDS victims. The FDA did everything in its power to shut Life Extension down and imprison us for life.82 According to the FDA, we were ripping off dying AIDS patients by recommending unproven therapies.
The Journal of the American Medical Association (Nov 27, 2013) featured an article describing a 54% reduction in the risk of progressing from HIV to full-blown AIDS using selenium and multi-vitamins.83 Life Extension first recommended these nutrients in the October 1985 edition of this publication (called at that time Anti-Aging News).
While the study published in the Journal of the American Medical Association was conducted in a region of Africa where malnutrition is rampant, and the study had other flaws (like a 25% dropout rate in both groups), the delay in HIV-induced immune suppression in patients taking these nutrients was remarkable.
A number of previous studies support the benefits of certain nutrients in delaying HIV progression79,84-86 Even FDA Consumer Magazine eventually acknowledged the value of AIDS patients using nutrient supplements.
We also recommended a drug called isoprinosine to AIDS patients in the October 1985 issue of Anti-Aging News. This contributed to our being arrested by the FDA because isoprinosine was not an approved drug. In the June 21, 1990, edition of the New England Journal of Medicine, a study found that HIV-infected humans who took isoprinosine were eight times less likely to progress to AIDS compared to placebo.87 This was not enough, however, to keep us from being indicted in 1991.
What helped save us was the continuing publication of research findings corroborating that isoprinosine and certain nutrients significantly delayed disease progression in HIV-infected patients, thus negating the FDA’s argument that we were “ripping off AIDS patients” by recommending “unproven” therapies.
The FDA was on the wrong side when it sought to destroy us in the 1980-1990s. Regrettably, millions of Americans continue to perish from needless bureaucratic red tape from virtually all diseases except AIDS. The reason AIDS is the exception is that AIDS activists made it clear to the FDA that there would be no bureaucratic delays in delivering experimental therapies to HIV-infected patients. The FDA capitulated and this enabled rapid medical innovation to occur in a free market environment.
Cancer patients, on the other hand, sit by like timid sheep, as the FDA decides which experimental therapy they are “allowed” to try and how far their disease must progress before the experimental therapy is made available on a so-called “compassionate-use” basis. FDA’s granting of “compassionate-use” sometimes occurs weeks after the patient dies, or is so close to death that it has no chance of working.
“In conclusion, our data suggest the relevance of NK (natural killer) cells as primary effectors not only against high-risk leukemias, but also solid tumors.” 44
Quote from study published in the April 2013 edition of the journal Oncoimmunology
Not Fast Enough!
In 2010, the Life Extension Foundation® pledged a substantial amount of money to a prestigious cancer research institute to evaluate many of the components contained in our published Pancreatic Cancer Treatment Protocol. The institution eagerly pushed this project forward, generating reams of paperwork in order to obtain Institutional Review Board approval.
Here we are in 2014, and the total number of pancreatic patients enrolled in this study is zero.
Bureaucratic delays like this are beyond rational understanding. These are human lives we are talking about!
When we devised unique treatments for AIDS in the 1980s, they were provided to dying AIDS patients almost overnight. Not all of them worked, but the ones that did built on a foundation that has resulted in HIV patients living for decades, as opposed to pancreatic cancer patients who often die in a matter of months.
Contrast the rapid development of AIDS therapies to most pancreatic cancer patients who die even after enduring the Whipple Procedure that was first described in 1935. It is clear that methods employed by AIDS activists are far superior to today’s regulatory quagmire that stymies cancer research.
Citizens Should Revolt
Cancer will likely kill over 570,000 Americans this year.88
Already-approved treatments could be saving lives, such as administering moderate dose interleukin-2 early in the disease process. Yet even these simple treatment enhancements are ignored by the oncology mainstream that prefers to practice assembly line medicine.
These kinds of delays would have never been tolerated by AIDS activists, who experimented with any potentially effective drug on large numbers of dying patients to quickly discover what worked and what didn’t.
The New England Journal of Medicine credits the work of AIDS pioneers as revolutionizing the way medical research is conducted today. We at Life Extension disagree with this Pollyanna assessment, as cancer therapies we uncovered decades ago remain bogged down in FDA red tape. Many are not being pursued at all despite a continuous stream of favorable data flowing out of research facilities.
The slogan in the black box below was chanted by AIDS activists who surrounded FDA headquarters in 1988 and shut down the agency for one day:89,90
“Act Up, Speak Out… Silence = Death!”
Protest Now Rather Than Wait For Funerals
I do not know why every cancer patient and their family does not march on Washington to demand the same exemption from bureaucratic suffocation that enabled HIV to become a manageable disease in a relatively brief window of time.
Perhaps cancer patients should write their family and friends and state something to the effect:
“In lieu of attending my funeral, would you mind marching on the Capitol in Washington D.C. and insist that cancer patients have unfettered access to any therapy that might work.”
For longer life,
William Faloon
References
- Specht G, Stinshoff K. Walther Kausch (1867-1928) and his significance in pancreatic surgery. Zentralbl Chir. 2001 Jun;126(6):479-81.
- Available at: http://www.grandroundsjournal.com/articles/gr07l0001/gr07l0001.pdf. Accessed March 6 , 2014.
- Birkmeyer JD, Siewers AE, Finlayson EV, et al. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002 Apr 11;346(15):1128-37.
- Ho CK , Kleeff J,Friess H, Büchler MW. Complications of pancreatic surgery. HPB (Oxford). 2005;7(2):99-108.
- Shrikhande SV, D’Souza MA. Pancreatic fistula after pancreatectomy: evolving definitions, preventive strategies and modern management. World J Gastroenterol . 2008 Oct 14;14(38):5789-96.
- Wente MN, Bassi C, Dervenis C, et al. Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery . 2007 Nov;142(5):761-8.
- Ferrara MJ, Lohse C, Kudva YC, et al. Immediate post-resection diabetes mellitus after pancreaticoduodenectomy: incidence and risk factors. HPB (Oxford) . 2013 Mar;15(3):170-4.
- Available at: http://www.webmd.com/cancer/pancreatic-cancer/whipple-procedure. Accessed March 6, 2014.
- Available at: http://money.cnn.com/2008/03/02/news/companies/elkind_jobs.fortune/index.htm?postversion=2008030510. Accessed March 6, 2014.
- Available at: http://www.forbes.com/sites/erikkain/2011/10/05/steve-jobs-has-died-at-age-56. Accessed March 6, 2014.
- Available at: http://abcnews.go.com/health/cancerpreventionandtreatment/steve-jobs-pancreatic-cancer-timeline/story?id=14681812. Accessed March 6, 2014
- Available at: http://usatoday30.usatoday.com/news/health/medical/health/medical/cancer/story/2011-08-24/apple-ceo-steve-jobs-resigns-after-battling-pancreatic-cancer/50127460/1. Accessed March 6, 2014.
- Weigent DA, Stanton GJ, Johnson HM. Interleukin 2 enhances natural killer cell activity through induction of gamma interferon. Infect Immun. 1983 Sep;41(3):992-7.
- Kehrl JH, Dukovich M, Whalen G, Katz P, Fauci AS, Greene WC. Novel interleukin 2 (IL-2) receptor appears to mediate IL-2-induced activation of natural killer cells. J Clin Invest. 1988 Jan;81(1):200-5.
- Yao HC, Liu SQ, Yu K, Zhou M, Wang LX. Interleukin-2 enhances the cytotoxic activity of circulating natural killer cells in patients with chronic heart failure. Heart Vessels. 2009 Jul;24(4):283-6.
- Yokoyama WM, Altfeld M, Hsu KC. Natural killer cells: tolerance to self and innate immunity to viral infection and malignancy. Biol Blood Marrow Transplant. 2010 Jan;16(1 Suppl):S97-S105.
- Hwang I, Scott JM, Kakarla T, et al. Activation mechanisms of natural killer cells during influenza virus infection. PLoS One. 2012 7(12):e51858.
- Brandstadter JD, Yang Y. Natural killer cell responses to viral infection. J Innate Immun. 2011;3(3):274-9.
- Chisholm SE, Reyburn HT. Recognition of vaccinia virus-infected cells by human natural killer cells depends on natural cytotoxicity receptors. J Virol . 2006 Mar;80(5):2225-33.
- Viel S, Charrier E, Marçais A, et al. Monitoring NK cell activity in patients with hematological malignancies. Oncoimmunology. 2013 Sep 1;2(9):e26011.
- zur Hausen H. Immortalization of human cells and their malignant conversion by high risk human papillomavirus genotypes. Semin Cancer Biol. 1999 Dec;9(6):405-11.
- Rosenberg SA, Lotze MT, Muul LM, et al. Observations on the systemic administration of autologous lymphokine-activated killer cells and recombinant interleukin-2 to patients with metastatic cancer. N Engl J Med. 1985 Dec 5;313(23):1485-92.
- Salup RR, Wiltrout RH. Adjuvant immunotherapy of established murine renal cancer by interleukin 2-stimulated cytotoxic lymphocytes. Cancer Res. 1986 Jul;46(7):3358-63.
- Marumo K, Ueno M, Muraki J, Baba S, Tazaki H. Augmentation of cell-mediated cytotoxicity against renal carcinoma cells by recombinant interleukin 2. Urology. 1987 Oct;30(4):327-32.
- Wang J, Walle A, Gordon B, et al. Adoptive immunotherapy for stage IV renal cell carcinoma: a novel protocol utilizing periodate and interleukin-2-activated autologous leukocytes and continuous infusions of low-dose interleukin-2. Am J Med. 1987 Dec;83(6):1016-23.
- Fisher RI, Coltman CA Jr, Doroshow JH, et al. Metastatic renal cancer treated with interleukin-2 and lymphokine-activated killer cells. A phase II clinical trial. Ann Intern Med. 1988 Apr;108(4):518-23.
- Chu MB, Fesler MJ, Armbrecht ES, et al. High-dose interleukin-2 (HD IL-2) therapy should be considered for the treatment of patients with melanoma brain metastases. Chemother Res Pract. 2013 2013:726925.
- Atkins MB, Kunkel L, Sznol M, Rosenberg SA. High-dose recombinant interleukin-2 therapy in patients with metastatic melanoma: long-term survival update. Cancer J Sci Am. 2000 Feb;6 Suppl 1:S11-4.
- Keilholz U, Conradt C, Legha SS, et al. Results of interleukin-2-based treatment in advanced melanoma: a case record-based analysis of 631 patients. J Clin Oncol. 1998 Sep;16(9):2921-9.
- Petrella T, Quirt I, Verma S, et al. Single-agent interleukin-2 in the treatment of metastatic melanoma. Curr Oncol. 2007 Feb;14(1):21-6.
- Acquavella N, Kluger H, Rhee J, et al. Toxicity and activity of a twice daily high-dose bolus interleukin 2 regimen in patients with metastatic melanoma and metastatic renal cell cancer. J Immunother. 2008 Jul-Aug;31(6):569-76.
- Schwartz RN, Stover L, Dutcher J. Managing toxicities of high-dose interleukin-2. Oncology. 2002 Nov;16(11 Suppl 13):11-20.
- Brivio F, Lissoni P, Rovelli F, et al. Effects of IL-2 preoperative immunotherapy on surgery-induced changes in angiogenic regulation and its prevention of VEGF increase and IL-12 decline. Hepatogastroenterology. 2002 Mar-Apr;49(44):385-7.
- Böhm M, Ittenson A, Klatte T, et al. Pretreatment with interleukin-2 modulates perioperative immunodysfunction in patients with renal cell carcinoma. Folia Biol (Praha). 2003 49(2):63-8.
- Nichols PH, Ramsden CW, Ward U, Sedman PC, Primrose JN. Perioperative immunotherapy with recombinant interleukin 2 in patients undergoing surgery for colorectal cancer. Cancer Res. 1992 Oct 15;52(20):5765-9.
- Lissoni P, Brivio F, Fumagalli L, Di Fede G, Brera G. Enhancement of the efficacy of chemotherapy with oxaliplatin plus 5-fluorouracil by pretreatment with IL-2 subcutaneous immunotherapy in metastatic colorectal cancer patients with lymphocytopenia prior to therapy. In Vivo. 2005 Nov-Dec;19(6):1077-80.
- Ades EW, McKemie CR 3rd, Wright S, Peacocke N, Pantazis C, Lockhart WL 3rd.Chemotherapy subsequent to recombinant interleukin-2 immunotherapy: protocol for enhanced tumoricidal activity. Nat Immun Cell Growth Regul. 1987 6(5):260-8.
- Da Costa ML, Redmond P, Bouchier-Hayes DJ. The effect of laparotomy and laparoscopy on the establishment of spontaneous tumor metastases. Surgery. 1998 Sep;124(3):516-25.
- Shakhar G, Blumenfeld B. Glucocorticoid involvement in suppression of NK activity following surgery in rats. J Neuroimmunol. 2003 May;138(1-2):83-91.
- Rosenne E, Shakhar G, Melamed R, Schwartz Y, Erdreich-Epstein A, Ben-Eliyahu S. Inducing a mode of NK-resistance to suppression by stress and surgery: a potential approach based on low dose of poly I-C to reduce postoperative cancer metastasis. Brain Behav Immun. 2007 May;21(4):395-408.
- Marik PE, Flemmer M. The immune response to surgery and trauma: Implications for treatment. J Trauma Acute Care Surg. 2012 Oct;73(4):801-8.
- Yokoyama Y, Sakamoto K, Arai M, Akagi M. Radiation and surgical stress induce a significant impairment in cellular immunity in patients with esophageal cancer. Jpn J Surg. 1989 Sep;19(5):535-43.
- Sano T, Morita S, Tominaga R, et al. Adaptive immunity is severely impaired by open-heart surgery. Jpn J Thorac Cardiovasc Surg. 2002 May;50(5):201-5.
- Rasmussen L, Arvin A. Chemotherapy-induced immunosuppression. Environ Health Perspect. 1982 Feb;43:21-5.
- Zandvoort A, Lodewijk ME, Klok PA, et al. After chemotherapy, functional humoral response capacity is restored before complete restoration of lymphoid compartments. Clin Exp Immunol . 2003 Jan;131(1):8-16.
- Angelini C, Bovo G, Muselli P, et al. Preoperative interleukin-2 immunotherapy in pancreatic cancer: preliminary results. Hepatogastroenterology. 2006 Jan-Feb;53(67):141-4.
- Hietanen T, Kellokumpu-Lehtinen P, Pitkänen M. Action of recombinant and interleukin 2 in modulating radiation effects on viability and cytotoxicity of large granular lymphocytes. Int J Radiat Biol. 1995 Feb;67(2):119-26.
- Boise LH, Minn AJ, June CH, Lindsten T, Thompson CB. Growth factors can enhance lymphocyte survival without committing the cell to undergo cell division. Proc Natl Acad Sci U S A. 1995 Jun 6;92(12):5491-5.
- Oberoi P, Wels WS. Arming NK cells with enhanced antitumor activity: CARs and beyond. Oncoimmunology. 2013 Aug 1;2(8):e25220.
- Sanchez-Correa B, Morgado S, Gayoso I, Bergua JM, Casado JG, Arcos MJ, Bengochea ML, Duran E, Solana R, Tarazona R. Human NK cells in acute myeloid leukaemia patients: analysis of NK cell-activating receptors and their ligands. Cancer Immunol Immunother . 2011 Aug;60(8):1195-205.
- Brunner S, Herndler-Brandstetter D, Weinberger B, Grubeck-Loebenstein B. Persistent viral infections and immune aging. Ageing Res Rev. 2011 Jul;10(3):362-9.
- Kozlowski JM, Fidler IJ, Campbell D, Xu ZL, Kaighn ME, Hart IR. Metastatic behavior of human tumor cell lines grown in the nude mouse. Cancer Res. 1984 Aug;44(8):3522-9.
- Kim S, Iizuka K, Aguila HL, Weissman IL, Yokoyama WM. In vivo natural killer cell activities revealed by natural killer cell-deficient mice. Proc Natl Acad Sci USA. 2000 Mar 14;97(6):2731-6.
- Smyth MJ, Swann J, Cretney E, Zerafa N, Yokoyama WM, Hayakawa Y. NKG2D function protects the host from tumor initiation. J Exp Med. 2005 Sep 5;202(5):583-8.
- Vacca P, Martini S, Mingari MC, Moretta L. NK cells from malignant pleural effusions are potent antitumor effectors: A clue for adoptive immunotherapy? Oncoimmunology . 2013 Apr 1;2(4):e23638.
- Bhat R, Watzl C. Serial killing of tumor cells by human natural killer cells—enhancement by therapeutic antibodies. PLoS One. 2007 Mar 28;2(3):e326.
- Bradstock KF. The use of hematopoietic growth factors in the treatment of acute leukemia. Curr Pharm Des. 2002 8(5):343-55.
- Ruggeri L, Mancusi A, Burchielli E, Aversa F, Martelli MF, Velardi A. Natural killer cell alloreactivity in allogeneic hematopoietic transplantation. Curr Opin Oncol. 2007 Mar;19(2):142-7.
- Locatelli F, Pende D, Mingari MC, et al. Cellular and molecular basis of haploidentical hematopoietic stem cell transplantation in the successful treatment of high-risk leukemias: role of alloreactive NK cells. Front Immunol. 2013 Feb 1;4:15.
- Ishikawa H, Saeki T, Otani T, et al. Aged garlic extract prevents a decline of NK cell number and activity in patients with advanced cancer. J Nutr. 2006 Mar;136(3 Suppl):816S-20S.
- Nantz MP, Rowe CA, Muller CE, Creasy RA, Stanilka JM, Percival SS. Supplementation with aged garlic extract improves both NK and gd-T cell function and reduces the severity of cold and flu symptoms: a randomized, double-blind, placebo-controlled nutrition intervention. Clin Nutr. 2012 Jun;31(3):337-44.
- Tang Z, Sheng Z, Liu S, Jian X, Sun K, Yan M. [The preventing function of garlic on experimental oral precancer and its effect on natural killer cells, T-lymphocytes and interleukin-2]. Hunan Yi Ke Da Xue Xue Bao. 1997 22(3):246-8.
- Kyo E, Uda N, Suzuki A, et al. Immunomodulation and antitumor activities of Aged Garlic Extract. Phytomedicine. 1998 Aug;5(4):259-67.
- Butt MS, Sultan MT, Butt MS, Iqbal J. Garlic: nature’s protection against physiological threats. Crit Rev Food Sci Nutr. 2009 Jun;49(6):538-51.
- Miller SC, Pandi-Perumal SR, Esquifino AI, Cardinali DP, Maestroni GJ. The role of melatonin in immuno-enhancement: potential application in cancer. Int J Exp Pathol. 2006 Apr;87(2):81-7.
- Currier NL, Miller SC. Echinacea purpurea and melatonin augment natural-killer cells in leukemic mice and prolong life span. J Altern Complement Med. 2001 Jun;7(3):241-51.
- Srinivasan V, Spence DW, Pandi-Perumal SR, Trakht I, Cardinali DP. Therapeutic actions of melatonin in cancer: possible mechanisms. Integr Cancer Ther. 2008 Sep;7(3):189-203.
- Lin ZB. Cellular and molecular mechanisms of immuno-modulation by Ganoderma lucidum. J Pharmacol Sci. 2005 Oct;99(2):144-53.
- Gao Y, Zhou S, Jiang W, Huang M, Dai X. Effects of ganopoly (a Ganoderma lucidum polysaccharide extract) on the immune functions in advanced-stage cancer patients. Immunol Invest. 2003 Aug;32(3):201-15.
- Zheng S, Jia Y, Zhao J, Wei Q, Liu Y. Ganoderma lucidum polysaccharides eradicates the blocking effect of fibrinogen on NK cytotoxicity against melanoma cells. Oncol Lett. 2012 Mar;3(3):613-16.
- Zhu XL, Lin ZB. Effects of Ganoderma lucidum polysaccharides on proliferation and cytotoxicity of cytokine-induced killer cells. Acta Pharmacol Sin. 2005 Sep;26(9):1130-7.
- Bast RC Jr, Kufe DW, Pollock RE, et al., editors. Holland-Frei Cancer Medicine. 5th edition. Hamilton (ON): BC Decker; 2000. Available at: http://www.ncbi.nlm.nih.gov/books/nbk20889.
- Arts EJ, Hazuda DJ. HIV-1 antiretroviral drug therapy. Cold Spring Harb Perspect Med. 2012 Apr;2(4):a007161.
- De Clercq E. Anti-HIV drugs: 25 compounds approved within 25 years after the discovery of HIV. Int J Antimicrob Agents. 2009 Apr;33(4):307-20.
- Sahay S, Reddy KS, Dhayarkar S. Optimizing adherence to antiretroviral therapy. Indian J Med Res. 2011 Dec;134(6):835-49.
- Adler MW. ABC of Aids: Development of the epidemic. BMJ. 2001 May 19;322(7296):1226-9.
- Available at: http://www.fda.gov/forconsumers/byaudience/forpatientadvocates/hivandaidsactivities/ucm258087.htm. Accessed March 6, 2014.
- Available at: http://www.nejm.org/doi/full/10.1056/nejmp1305297. Accessed March 14, 2014.
- Fawzi WW, Msamanga GI, Spiegelman D, et al. A randomized trial of multivitamin supplements and HIV disease progression and mortality. N Engl J Med . 2004 Jul 1;351(1):23-32.
- [No authors listed] AIDS advocates returning to their activism roots. Protesters welcome arrests and publicity. AIDS Alert. 2004 Aug;19(8):90-3.
- Available at: http://www.tcnj.edu/~borland/2006-aids/cassy2.htm Accessed March 14, 2014.
- Available at: https://www.lifeextension.com/magazine. Accessed March 14, 2014.
- Baum MK, Campa A, Lai S, et al. Effect of micronutrient supplementation on disease progression in asymptomatic, antiretroviral-naive, HIV-infected adults in Botswana: a randomized clinical trial. JAMA. 2013 Nov 27;310(20):2154-63.
- Fawzi WW, Msamanga GI, Kupka R, et al. Multivitamin supplementation improves hematologic status in HIV-infected women and their children in Tanzania. Am J Clin Nutr. 2007 May;85(5):1335-43.
- Botros D, Somarriba G, Neri D, Miller TL. Interventions to address chronic disease and HIV: strategies to promote exercise and nutrition among HIV-infected individuals. Curr HIV/AIDS Rep. 2012 Dec;9(4):351-63.
- Mehta S, Fawzi W. Effects of vitamins, including vitamin A, on HIV/AIDS patients. Vitam Horm. 2007 75:355-83.
- Pedersen C, Sandström E, Petersen CS, et al. The efficacy of inosine pranobex in preventing the acquired immunodeficiency syndrome in patients with human immunodeficiency virus infection. The Scandinavian Isoprinosine Study Group. N Engl J Med. 1990 Jun 1;322(25):1757-63.
- Available at: http://www.cdc.gov/nchs/fastats/deaths.htm. Accessed March 17, 2014.
- Available at: http://theconversation.com/how-the-dallas-buyers-club-changed-hiv-treatment-in-the-us-22664. Accessed March 17, 2014.
- Available at: http://www.edgeboston.com/health_fitness/hiv_aids/news//154077/act-up_co-f. Accessed March 17, 2014.

