Life Extension Magazine®

You may have noticed that a lot of famous people are succumbing to pancreatic cancer.
It is not a coincidence.
Pancreatic cancer incidence has steadily increased in the United States by about 1.2% annually over the last 10 years.1
The lethality of a pancreatic malignancy has generated volumes of scientific data on how to reduce your risk of contracting it.
The good news for readers of Life Extension® magazine is they already follow many of these healthy diets and lifestyles.
What troubles me, however, is that a leading pancreatic cancer risk factor (tobacco use) has plummeted since the 1960s-1970s.
Yet, pancreatic cancer cases are not declining.

Prominent Figures Who Have Succumbed to Pancreatic Cancer
- John Lewis. Congressman, civil rights legend. 2020.
- Alex Trebek. Host of the game show Jeopardy! 2020.
- Ruth Bader Ginsburg. Supreme Court justice. 2020.
- Aretha Franklin. Queen of Soul. 2018.
- Luciano Pavarotti. Opera star. 2007.
- Dizzy Gillespie. Jazz trumpet great. 1993.
- Sally Ride. Astronaut. 2012.
- Patrick Swayze. Actor. 2009.
- Michael Landon. TV star. 1991.
- Gene Upshaw. Football great. 2008.
- Karl Lagerfeld. Designer. 2019.
- Bob Gibson. Hall-of-Fame baseball great. 2020.
This editorial describes what’s behind rising numbers of Americans who perish from pancreatic cancer and what can be done to reverse this deadly trend.
In 1967, when I was 12 years old, my best friend’s mother perished from pancreatic cancer. Her relatively young age and six young children made this a particularly tragic event.
At her funeral no one would have conceived that 54 years later there would be no cure.
Instead of curative therapy, most metastatic pancreatic cancer victims are offered palliative choices that provide only a limited amount of survival time depending upon treatment regimen. The survival variance often depends on what degree of chemotherapy toxicity the patient is willing to tolerate.
Surging Global Incidences of Pancreatic Cancer
A comprehensive analysis, published in the prestigious journal The Lancet, revealed a sharp increase of pancreatic cancer worldwide.2
From 1990 to 2017 the number of pancreatic cancer cases more than doubled from 195,000 to 448,000 cases.
Better reporting of causes of death over these decades is partially responsible for this statistical surge, but the increase, as noted in The Lancet, is of international concern.
The Lancet article concluded:
“ Prevention strategies should focus on modifiable risk factors. Development of screening programs for early detection and more effective treatment strategies for pancreatic cancer are needed.”
Few pancreatic cancer patients survive more than 18 months when relying on chemo drugs that were approved decades ago.
The lack of progress in better treatment options is inexcusable when one considers the advances made in other areas of medical practice. This includes cardiac stenting that was unheard of in the 1960s and is now a routine treatment for coronary artery disease.
Surgery for Pancreatic Cancer
By the time most pancreatic cancers are diagnosed, the malignancy has spread into the liver and adjacent tissues. Chemo at these advanced states becomes a death-delaying option.
Some patients are diagnosed early enough for potential curative surgery.
In 1935, a doctor named Allen Whipple devised a more effective way to remove a section of the pancreas and adjacent body parts.4,5
Dr. Whipple’s technique removes the head of the pancreas, along with portions of the stomach, first part of the small intestine, gallbladder, and the bile duct.
The impact of this procedure on the body, even in 2021, is severe, with a higher death rate compared to many other types of surgeries.6
The rearranged internal organs do not always hold together, and infection can spread inside the patient. This can lead to more surgeries, and in some cases the remainder of the pancreas and the spleen must be removed to correct problems from the first operation.
Chemotherapy for Metastatic Pancreatic Cancer
In 2018, a study was published in the New England Journal of Medicine that compared chemotherapy using either gemcitabine or FOLFIRINOX (fluorouracil, leucovorin, irinotecan, and oxaliplatin), an arduous chemotherapy regimen.3
At a median follow-up of 33.6 months, the median disease-free survival was 21.6 months in the FOLFIRINOX group, with 39.7% of patients disease-free and alive at three years. The gemcitabine group had median disease-free survival of only 12.8 months with 21.4% alive and disease-free at three years.
Serious adverse events (grade 3 or 4) occurred in 75.9% of the patients in the modified-FOLFIRINOX group compared to 52.9% in the gemcitabine group.
This study showed that chemotherapy that inflicted harsher side effects (FOLFIRINOX) improved survival time.
These data are based on an unusual patient population that first underwent a brutal surgical (Whipple) procedure followed by two choices of chemo regimens (gemcitabine or FOLFORINOX). The chemo alone did not enable these higher rates of survival.
There is a clear need for more effective treatments for pancreatic cancer.
Some patients do not heal well and leak pancreatic fluid from where body parts are sewn together. This happens so frequently that the surgeon leaves in drainage catheters for fluids to exit so they do not accumulate inside the patient.7-9
Despite these horrific side effects, most patients who survive the painful surgical ordeal (called the “Whipple Procedure”) will die from metastatic pancreatic cancer.
Risk Factors You Control

Significant investigative resources have been deployed to identify modifiable risk factors that can enable people to reduce their odds of developing pancreatic cancer.
A modifiable risk factor is one that YOU have control over.
Few readers of Life Extension® magazine use tobacco, but they should know that exposure to secondhand smoke can increase pancreatic cancer risk by over 50%.13
About 25% of pancreatic cancers relate to cigarettes.14 Yet smoking rates have plummeted since the 1960s-1970s while pancreatic cancer incidence has steadily increased.
Steve Jobs, Pancreatic Cancer Victim

Steve Jobs was criticized for delaying a Whipple Procedure for nine months after being diagnosed with pancreatic cancer.10
The initial approaches Steve Jobs tried (acupuncture, vegan diet, herbs, spiritualists) had no chance of eradicating his pancreatic tumor.

It’s hard to blame the then 49-year-old co-founder of Apple for not wanting his body cut up via a Whipple Procedure.
Steve Jobs eventually died at age 56 after undergoing multiple aggressive treatments, including a liver transplant.11,12
If one is diagnosed with pancreatic cancer at an early stage today, a procedure made clinically relevant back in 1935 (the Whipple Procedure) is still the best treatment option.
Better treatments are urgently needed.
One villain is the surging numbers of overweight and obese Americans. Close to half of all adults in the United States today are pre-diabetic or type II diabetic.15
Compared to normal-weight people, obese individuals have about a 50% increased risk of developing pancreatic cancer, as well as lower overall survival rates.16-18 That’s about the same risk as cigarette smokers, which helps explain why pancreatic cancer incidence is not declining despite reductions in tobacco use.
Both excess body weight and smoking are pancreatic cancer risk factors.19 Those who would never touch a cigarette may not realize the similar dangers inflicted from excess body fat.
Heavier individuals, especially when fat accumulates in the belly, are often in a chronic inflammatory state and have glucose control issues.20
Type II diabetes and chronic inflammation increase the risk of multiple degenerative disorders, including pancreatic cancer.21,22
To put the danger of inflammation in context, those with chronic pancreatitis have up to an eight-fold higher risk of developing pancreatic cancer.23
There is a lot to be gained from normalizing one’s body weight. The benefits include reduced inflammation, improved glycemic control, and lower risk for many cancers.
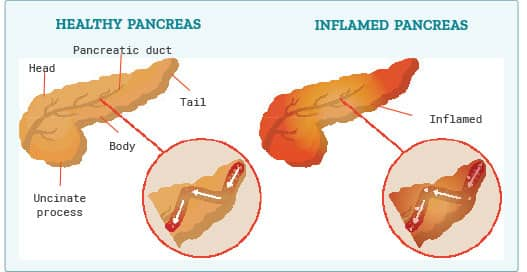
Chronic pancreatitis is long-term inflammation of the pancreas that typically causes severe pain in the center of your belly and/or extending through to your back. The cell damage inflicted by pancreatitis increases future pancreatic cancer incidence. Risk factors for pancreatitis include obesity and excess alcohol ingestion.24
Dietary Factors
What you eat and drink impacts your pancreatic cancer risk up to 30%-50%. Certain foods are associated with higher risk, while others confer protection.25-27
Consumption of red meats (especially when cooked at high temperature), fried foods, and foods containing nitrosamines (processed meats) may increase pancreatic cancer risk.28,29
One meta-analysis that included 11 case-control studies found that red meat intake increased pancreatic cancer risk by about 48%.30
Yet other studies of meat consumption and various cancer risks are inconsistent and less conclusive.31
On the protective side, high intake of vegetables and fruits decreased pancreatic cancer risk by 38% and 29%, respectively.30
Increased nut consumption was shown to significantly lower risk of pancreatic cancer.32,33
In a large UK study published in 2016, mortality for pancreatic cancer was lower for less-frequent meat eaters (about 45% lower mortality), as well as vegetarians and vegans (about 50% lower mortality) compared with regular meat eaters.34
For those who insist on eating some red meat, it’s good to know that reducing total red meat intake may confer risk reduction.
The article on page 50 of this month’s issue describes dietary factors most associated with pancreatic cancer risk reduction, including lycopene from tomatoes and carotenoids from other foods.
Role of Magnesium

In the December 2016 issue of Life Extension® magazine I wrote an article35 based on a landmark study showing a modest increase in magnesium intake from diet and supplements resulted in profound reductions in pancreatic cancer risk.36
What struck me about this study’s findings is that it did not require a large amount of additional magnesium to produce a meaningful reduction in pancreatic cancer risk.36
For example, compared to a person who ingests 300 mg a day of magnesium, an individual with a daily magnesium intake of 200 mg would be expected to have a 24% increased risk of pancreatic cancer.
Both intakes (200 mg and 300 mg a day of magnesium) are considered deficient even by government standards.
This 2015 published study involved over 66,000 men and women aged 50-76 years who were followed for an eight-year period. The subjects were divided into the following three groups based upon their magnesium intake:
- Optimal Intake - Defined as ingesting greater than or equal to 100% of the government recommended dietary allowance (RDA) for magnesium (420 mg a day for males and 320 mg a day for females)
- Sub-optimal Intake - Daily intake of only 75% to 99% of the government RDA for magnesium
- Deficient Intake - Less than 75% of the government RDA for magnesium (less than 315 mg a day for males and less than 240 mg a day for females)
Those who ingested 75%-99% of the government’s RDA for magnesium (sub-optimal intake) had a 42% greater risk of pancreatic cancer incidence compared with those ingesting greater than or equal to 100% of the magnesium RDA.
Those who ingested less than 75% of the government’s RDA for magnesium (deficient intake) had a striking 76% greater risk of pancreatic cancer incidence compared to those whose intake of magnesium was equal to or greater than the government’s (optimal intake) RDA.
When analyzing those who met or exceeded the government’s RDA for total magnesium intake, only those who took dietary supplements containing magnesium were able to consistently achieve the benefits.
What struck me about these findings is that the amount of added magnesium needed to meet the government’s RDA was exceedingly small.
For most people, taking one low-cost magnesium capsule a day, or obtaining it in a sufficient potency multi-nutrient formula, is all that may be needed to garner protective effects.
Intolerable Delays
The snail’s pace of progress against malignancies like pancreatic cancer should provoke societal outrage.
Like lambs standing in line awaiting slaughter, the public tolerates mediocre medicine that inflicts horrific suffering and needless deaths.
By contrast, we here at Life Extension® view bureaucratic roadblocks that impede delivery of better cancer treatments as intolerable delays that will be ridiculed by future historians.
I am not implying that there are an insufficient number of ongoing clinical trials. As of this writing there were about 500 human trials recruiting pancreatic cancer patients.
The urgency is for the 57,000 Americans who will be diagnosed with pancreatic cancer over the next 12 months. Where are the improved treatments for them?
Life Extension® has updated its Pancreatic Cancer Protocol (LifeExtension.com/pancreatic) to describe the use of repurposed drugs that may improve patient outcomes. Many of these treatments are not being incorporated into conventional practice.
Take Control!

About 10% to 20% of pancreatic tumors (adenocarcinomas) are thought to be due to a heritable cause that includes mutations in the BRCA1, BRCA2 and other cell regulatory genes.37,38
This implies that most (around 80%) of pancreatic cancers may be attributed to modifiable behavior patterns.
Hard data show the risk of developing pancreatic cancer can be reduced if people follow healthier diet and lifestyle practices.
Healthy choices not only help protect against most malignancies, but also reduce incidences of cardio-vascular disorders and dementia.
Most Americans engage in behaviors that spike disease risk. The tragic impact can be seen with increasing rates of common malignancies.
Beyond unhealthy lifestyle choices is a pharmaceutical industry that is failing to deliver meaningful improvements in treating pancreatic cancer and other deadly malignancies.
Mutated BRCA genes are associated with a high risk of breast, ovarian, and other cancers.39 Women who test BRCA positive sometimes undergo prophylactic removal of both breasts and their ovaries at young ages.
I cannot fathom why these barbaric procedures are tolerated in a world where rapid technological advances are taken for granted.
Young bodies are mutilated, toxic treatments deployed that do not cure, and oncology drugs prescribed that cost over $100,000. Yet pancreatic cancer deaths spiral upward each year.
Cancer Establishment's Position
A report published earlier this year showed cancer death rates went down 31% from 1991-2018 in response to reductions in smoking and to earlier diagnoses and some improved treatments.40 But this does not spare the more than 600,000 Americans who will perish from a malignancy over the next 12 months.
Greater priority given to discovering curative treatments is urgently needed. This will not happen until the public demands meaningful changes.
In the meantime, I urge readers to take control of their individual risk factors to reduce odds of contracting deadly digestive tract malignancies including colon, esophageal, and pancreatic cancers.
For longer life,
William Faloon
Life Extension® Buyers Club
References
- Available at: https://www.cancer.net/cancer-types/pancreatic-cancer/statistics. Accessed May 7, 2021.
- Collaborators GBDPC. The global, regional, and national burden of pancreatic cancer and its attributable risk factors in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2019 Dec;4(12):934-47.
- Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or Gemcitabine as Adjuvant Therapy for Pancreatic Cancer. N Engl J Med. 2018 Dec 20;379(25):2395-406.
- Are C, Dhir M, Ravipati L. History of pancreaticoduodenectomy: early misconceptions, initial milestones and the pioneers. HPB (Oxford). 2011 Jun;13(6):377-84.
- Whipple AO, Parsons WB, Mullins CR. Treatment of Carcinoma of the Ampulla of Vater. Ann Surg. 1935 Oct;102(4):763-79.
- Birkmeyer JD, Siewers AE, Finlayson EV, et al. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002 Apr 11;346(15):1128-37.
- Shrikhande SV, D’Souza MA. Pancreatic fistula after pancreatectomy: evolving definitions, preventive strategies and modern management. World J Gastroenterol. 2008 Oct 14;14(38):5789-96.
- Ho CK, Kleeff J, Friess H, et al. Complications of pancreatic surgery. HPB (Oxford). 2005;7(2):99-108.
- Nahm CB, Connor SJ, Samra JS, et al. Postoperative pancreatic fistula: a review of traditional and emerging concepts. Clin Exp Gastroenterol. 2018;11:105-18.
- Available at: https://money.cnn.com/2008/03/02/news/companies/elkind_jobs.fortune/index.htm. Accessed May 5, 2021.
- Available at: https://www.forbes.com/sites/erikkain/2011/10/05/steve-jobs-has-died-at-age-56/. Accessed May 5, 2021.
- Available at: https://abcnews.go.com/Health/CancerPreventionAndTreatment/steve-jobs-pancreatic-cancer-timeline/story?id=14681812. Accessed May 5, 2021.
- Vrieling A, Bueno-de-Mesquita HB, Boshuizen HC, et al. Cigarette smoking, environmental tobacco smoke exposure and pancreatic cancer risk in the European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2010 May 15;126(10):2394-403.
- Available at: https://www.cancer.org/cancer/pancreatic-cancer/causes-risks-prevention/risk-factors.html. Accessed May 7, 2021.
- Available at: https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf. Accessed May 5, 2021.
- Calle EE, Rodriguez C, Walker-Thurmond K, et al. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med. 2003 Apr 24;348(17):1625-38.
- Xu M, Jung X, Hines OJ, et al. Obesity and Pancreatic Cancer: Overview of Epidemiology and Potential Prevention by Weight Loss. Pancreas. 2018 Feb;47(2):158-62.
- Bracci PM. Obesity and pancreatic cancer: overview of epidemiologic evidence and biologic mechanisms. Mol Carcinog. 2012 Jan;51(1):53-63.
- Aune D, Greenwood DC, Chan DS, et al. Body mass index, abdominal fatness and pancreatic cancer risk: a systematic review and non-linear dose-response meta-analysis of prospective studies. Ann Oncol. 2012 Apr;23(4):843-52.
- Ellulu MS, Patimah I, Khaza’ai H, et al. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017 Jun;13(4):851-63.
- Furman D, Campisi J, Verdin E, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019 Dec;25(12):1822-32.
- Shadhu K, Xi C. Inflammation and pancreatic cancer: An updated review. Saudi J Gastroenterol. 2019 Jan-Feb;25(1):3-13.
- Kirkegard J, Mortensen FV, Cronin-Fenton D. Chronic Pancreatitis and Pancreatic Cancer Risk: A Systematic Review and Meta-analysis. Am J Gastroenterol. 2017 Sep;112(9):1366-72.
- Available at: https://pancreaticcanceraction.org/news/is-pancreatitis-a-risk-factor-for-pancreatic-cancer/. Accessed May 5, 2021.
- Midha S, Chawla S, Garg PK. Modifiable and non-modifiable risk factors for pancreatic cancer: A review. Cancer Lett. 2016 Oct 10;381(1):269-77.
- Michaud DS, Skinner HG, Wu K, et al. Dietary patterns and pancreatic cancer risk in men and women. J Natl Cancer Inst. 2005 Apr 6;97(7):518-24.
- Maisonneuve P, Lowenfels AB. Risk factors for pancreatic cancer: a summary review of meta-analytical studies. Int J Epidemiol. 2015 Feb;44(1):186-98.
- Lightsey D, National Council Against Health F, Quackwatch. Comment on ‘Red and processed meat consumption and risk of pancreatic cancer: meta-analysis of prospective studies’. Br J Cancer. 2012 Aug 7;107(4):754-5.
- Stolzenberg-Solomon RZ, Cross AJ, Silverman DT, et al. Meat and meat-mutagen intake and pancreatic cancer risk in the NIH-AARP cohort. Cancer Epidemiol Biomarkers Prev. 2007 Dec;16(12):2664-75.
- Paluszkiewicz P, Smolinska K, Debinska I, et al. Main dietary compounds and pancreatic cancer risk. The quantitative analysis of case-control and cohort studies. Cancer Epidemiol. 2012 Feb;36(1):60-7.
- McCullough ML, Jacobs EJ, Shah R, et al. Meat consumption and pancreatic cancer risk among men and women in the Cancer Prevention Study-II Nutrition Cohort. Cancer Causes Control. 2018 Jan;29(1):125-33.
- Bao Y, Hu FB, Giovannucci EL, et al. Nut consumption and risk of pancreatic cancer in women. Br J Cancer. 2013 Nov 26;109(11):2911-6.
- Wu L, Wang Z, Zhu J, et al. Nut consumption and risk of cancer and type 2 diabetes: a systematic review and meta-analysis. Nutr Rev. 2015 Jul;73(7):409-25.
- Appleby PN, Crowe FL, Bradbury KE, et al. Mortality in vegetarians and comparable nonvegetarians in the United Kingdom. Am J Clin Nutr. 2016 Jan;103(1):218-30.
- Available at: https://www.lifeextension.com/magazine/2016/12/will-magnesium-become-next-vitamin-d. Accessed May 5, 2021.
- Dibaba D, Xun P, Yokota K, et al. Magnesium intake and incidence of pancreatic cancer: the VITamins and Lifestyle study. Br J Cancer. 2015 Dec 1;113(11):1615-21.
- Bartsch DK, Gress TM, Langer P. Familial pancreatic cancer--current knowledge. Nat Rev Gastroenterol Hepatol. 2012 Aug;9(8):445-53.
- Bannon SA, Montiel MF, Goldstein JB, et al. High Prevalence of Hereditary Cancer Syndromes and Outcomes in Adults with Early-Onset Pancreatic Cancer.Cancer Prev Res (Phila). 2018 Nov;11(11):679-86.
- Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA. 2017 Jun 20;317(23):2402-16.
- Siegel RL, Miller KD, Fuchs HE, et al. Cancer Statistics, 2021. CA Cancer J Clin. 2021 Jan;71(1):7-33.
Photo credits
- John Lewis. © Getty Images / Chip Somodevilla / Staff
- Alex Trebek. © Getty Images / Amanda Edwards / Stringer
- Ruth Bader Ginsburg. © Getty Images / Mark Wilson / Staff
- Aretha Franklin. © Getty Images / Michael Ochs Archives / Stringer
- Luciano Pavarotti. © Getty Images / Michael Ochs Archives / Stringer
- Dizzy Gillespie. © Getty Images / Michael Ochs Archives / Stringer
- Sally Ride. © Getty Images / MPI / Stringer
- Patrick Swayze. © Getty Images / Ethan Miller / Staff
- Michael Landon. © Getty Images / NBC Television / Handout
- Gene Upshaw. © Getty Images / Rick Diamond / Staff
- Karl Lagerfeld. © Getty Images / Pascal Le Segretain / Staff
- Bob Gibson. © Getty Images / Ezra Shaw / Staff
- Steve Jobs. © Getty Images / Justin Sullivan / Staff

