
Skin Cancer
Skin Cancer
Last Section Update: 10/2025
1 Overview
Summary and Quick Facts for Skin Cancer
- Skin cancer is the most common form of cancer. Fortunately, it is also one of the most treatable, if caught early. That’s why it’s so important to pay attention to the skin and seek medical attention, if you notice any unusual skin changes.
- This protocol describes the different kinds of skin cancer, how they develop and how they are detected and treated. You will learn about promising, emerging treatment techniques with potential to further improve skin cancer treatment outcomes and several lifestyle choices and supplements that can protect skin against cancer-causing factors.
- Taking simple steps to avoid skin-damaging factors like excessive sun exposure can reduce the risk of skin cancer.
- Polypodium leucotomos is a tropical fern that contains compounds with skin-protective properties. Supplementation with P. leucotoms extract has been shown to protect the skin from sun damage.
Targeted interventions including avoiding excessive sun exposure, liberally applying sunscreen before sun exposure, and supporting skin health with scientifically studied nutrients like B-vitamins, Polypodium leucotomos, and red orange complex can protect skin from UV damage.
Causes and Risk Factors
- Exposure to sunlight/UV light is a primary risk factor for all skin cancers.
- All skin cancers are more common in men and with advancing age.
- Numerous freckles and/or moles can increase risk of skin cancer.
Signs and Symptoms
- Basal cell carcinoma, most of which occur on the head or neck
- Nodular: pearly pink or white, dome-shaped lesion
- Superficial: scaly plaques with raised, pearly white borders
- Micronodular and infiltrative: like nodular subtype but more malignant
- Morpheaform: firm, yellowish, ill-defined mass
- Squamous cell carcinoma, found on backs of hands and forearms and the head and neck
- Firm, smooth or scaly raised lesion, often ulcerated
- The “ABCDE guide” is often used to describe melanoma characteristics
- Asymmetrical shape
- Borders that are irregular
- Color that is not the same all over
- Diameter larger than a pencil eraser
- Evolving in shape, size or color
Diagnosis
- Skin cancers are diagnosed based on physical exam, patient history, and a variety of diagnostic techniques, including biopsy of skin and lymph nodes, dermoscopy, and imaging tests if the cancer has spread.
Conventional Treatment
- Treatments for non-melanoma skin cancers include Mohs microsurgery (which presents the best cure rates, lowest recurrence rates, and leaves the smallest surgical wounds), electrodessication and curettage, cryosurgery, and radiotherapy.
Novel and Emerging Therapies
- Vismodegib, an FDA-approved drug for advanced basal cell carcinoma, significantly decreased the appearance of new tumors and reduced the size of existing tumors in two phase II clinical trials.
- Daily low-dose aspirin usage reduced melanoma risk by 46% among women in a large Dutch population-based study.
- Tumor treating fields represent a noninvasive technique that has demonstrated efficacy in preventing proliferation of human melanoma cells in preclinical studies.
- Metformin, a first-line drug for type 2 diabetes, has been shown to reduce tumor growth rate in mice injected with human squamous carcinoma cells.
Lifestyle Considerations
- Perform skin self-exams for early detection
- Reduce UV exposure, including minimizing time spent outside during peak sun hours; heed the National Weather Service’s daily UV index; and wear long sleeves, sunglasses, and wide-brimmed hats
Integrative Interventions
- B-vitamins: Evidence suggests that nicotinamide (vitamin B3 derivative) offers the strongest support among B vitamins for reducing non-melanoma skin cancer risk. In a randomized trial of 386 high-risk patients, 500 mg twice daily for 12 months lowered new non-melanoma skin cancers by 23%, including a 30% reduction in squamous cell carcinoma. Meta-analyses and observational studies reinforce this benefit.
- Polypodium leucotomos: Pretreatment with Polypodium extract before exposure to UV radiation increased the minimal amount of radiation to which skin could be exposed before sunburn in 65% of high-risk melanoma patients in the study.
- Red Orange Complex: Supplementation with red orange complex in human volunteers led to a roughly 35% reduction in intensity of skin redness following UVB exposure, suggesting consumption of red orange complex may mitigate the likelihood of developing sunburn.
- Grape Seed Extract: Participants who consumed grape seed extract had a 74% decreased risk of cutaneous squamous cell carcinoma compared to those that did not.
- Melatonin: Evidence suggests higher urinary levels of a major melatonin metabolite may be associated with reduced skin cancer risk.
2 Introduction
Skin cancer is the most common form of cancer, affecting over 3.5 million people in the United States alone. The incidence of skin cancer is steadily increasing in frequency each year; although enhanced leisure time exposure to sunlight has been proposed as a cause, another factor may be an increase in skin cancer awareness through increased screening (Gloster 1996; Leiter 2008; ACS 2013a; Wiley 2012; Welch 2005).
The most prevalent skin cancers – melanoma, squamous cell carcinoma, and basal cell carcinoma – can affect individuals at any age, although diagnoses are significantly more likely with advancing age; incidence rates of non-melanoma skin cancers increase considerably in the 50s and peak in the 70s and 80s (Kim 2012).
Fortunately, skin cancer is a disease in which the patient is significantly empowered to influence the outcome of their encounter with malignancy. Simply by being aware of changes in the surface of the skin, especially areas often exposed to sunlight, and reporting any abnormalities to a physician in a timely manner, patients can help facilitate early detection and treatment of skin cancer. The impact of skin self-examination can be extraordinary because favorable skin cancer outcomes are highly dependent upon early diagnosis and treatment of malignant changes in the skin (ACS 2013b).
Technological advances in treatment options have improved outcomes even for patients with aggressive skin cancer variants. Significant survival improvements have been achieved in trials of targeted therapies for aggressive melanomas, while surgical techniques for treatment of early-stage skin cancers are achieving unprecedented precision (Kudchadkar 2013; Johnson 2013; Ma 2014; Liu 2013; Kim 2012; Paoli 2011; Hui 2012).
By taking advantage of the latest innovations in skin cancer prevention, including avoiding excessive sun exposure and supporting skin health with scientifically studied nutrients like Polypodium leucotomos and tea polyphenols, skin cancer risk can be dramatically reduced (Katiyar 2007; Katiyar 2011; Payette 2010; Yang 2011; Caccialanza 2011; Ramirez Boscá 2012; Zattra 2009; Aguilera 2013).
3 Background
Although several types of skin cancer have been identified, three types make up the vast majority of diagnoses: basal cell carcinoma, squamous cell carcinoma, and melanoma. Melanoma is the most dangerous of the skin cancers. It accounts for only 4% of all skin cancer diagnoses, but is responsible for more than 80% of skin cancer-related deaths (Miller 2006). The lifetime chance of a Caucasian developing an invasive melanoma of the skin is 1 in 35 among men and 1 in 54 among women (Siegel 2013). Melanoma readily invades local tissues and can metastasize (spread) to distant organs and establish new tumors. Advanced, metastatic melanoma is difficult to effectively treat (Miller 2006; Dunki-Jacobs 2013). While the incidence and mortality for most types of cancer in the United States have been steadily decreasing since the mid-1990s, the overall incidence of melanoma has increased in recent decades. Cutaneous (skin) melanoma currently ranks fifth for men and seventh for women in incidence of all new cancers diagnosed, with an estimated 76 690 diagnoses and 9480 deaths in 2013 (ACS 2013a; Siegel 2013). In the United States, statistical modeling indicates the incidence of new skin melanoma cases increased 2.6% per year between 2004 and 2014, while death rates remained stable (NCI 2014a). The cause of the increasing incidence of melanoma is controversial. A recent study that examined nine geographical areas in the United States suggests the increased incidence can be explained, to some degree, by an increase in screening and biopsies resulting in increased melanoma detection (Welch 2005). However, this conclusion is controversial, and disagreement exists as to identifying the possibility of fluctuation in melanoma database registrations (Lee 2009).
Non-melanoma skin cancers represent the majority of skin cancer diagnoses; about 80% of these are basal cell carcinomas and 20% are squamous cell carcinomas, with all other diagnoses representing only 1%. Non-melanoma skin cancers are usually not reported to cancer registries, so estimates of incidence are somewhat uncertain (Eisemann 2013). Although the incidence and prevalence of non-melanoma skin cancer is not being tracked by the National Cancer Institute (NCI), there are several studies that show that these cancers have become more frequent over the past 20 years (Firnhaber 2012). Surveys from the NCI in the early 1970s estimated 300 000 new cases per year (about 50% of all new cancers); other analyses from national Medicare data estimate the number of individuals in the United States with non-melanoma skin cancers in 2006 to be over 3.5 million (Rogers 2010). The average age of non-melanoma skin cancer diagnosis ranges from 59 to 65 years for men and 60 to 66 years for women (Kim 2012).
Squamous cell carcinomas have far greater capacity to metastasize in comparison with basal cell carcinoma of the skin (which may be locally invasive) and account for most deaths from non-melanoma skin cancers; they are also more likely to recur following removal compared to basal cell carcinomas. Most occur on the head, neck, and upper extremities, indicating their incidence is strongly correlated with sun-exposed skin. Basal cell carcinomas very rarely metastasize, but the risk of metastasis does appear to increase with tumor size (Kim 2012).
While genetics can influence the rate at which skin cancers can occur (Carless 2008; Dunki-Jacobs 2013), the predominant catalyst for skin carcinogenesis is exposure to ultraviolet (UV) radiation. Sun exposure leads to a measurable increase in risk of non-melanoma cancers (Leiter 2008; Fartasch 2012; Sánchez 2013) and has been implicated in melanoma progression (Erb 2008; Whiteman 2006). Not surprisingly, skin cancer incidence is more prevalent in individuals with light skin; the incidence of non-melanoma skin cancers in African Americans is 3.4 per 100 000 persons compared to 232.6 per 100 000 persons for white populations. White populations tend to have skin cancer rates 6-14 times higher than Hispanic populations living in the same areas. Lower skin cancer rates are attributed to the increased production of melanin, the UV-protective skin pigment, in darker-skinned individuals. In these individuals, squamous cell carcinoma is the more common cancer, often appearing in non-sun-exposed areas (eg, the lower extremities) and associated with trauma or chronic inflammation rather than exposure to UV radiation (Kim 2012).
Although skin cancer-related mortality figures at first appear sobering, skin cancers are quite treatable if identified early. Cure rates approach 99% for non-melanoma skin cancers (although recurrence rates can be high) and approximately 87% for melanoma (Kim 2012; Dunki-Jacobs 2013). Survival rates drop precipitously as skin cancers reach advanced stages; the percentages of patients who survive >5 years after being diagnosed with metastatic squamous cell carcinoma or melanoma are 26.8% and 15%, respectively (Kim 2012; ACS 2013a).
4 Biology and Development of Skin Cancer
Anatomy
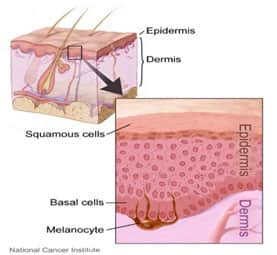
The skin is one of the largest organs in the body, tasked with protecting the body from moisture loss and bacterial invasion, helping maintain body temperature, detoxifying or excreting waste products and toxins, producing vitamin D, providing sensory function, and acting as a reservoir for blood. Structurally, the skin consists of two distinct layers: the outer epidermis is a self-renewing protective layer that is primarily cellular, while the inner dermis is mostly fibrous connective tissue and contains the skin’s vasculature, nerves, hair follicles, and glands. The epidermis consists predominantly of keratinocytes, a cell type formed deep within the epidermis (in a region called the basal layer). In this basal layer, precursor cells (stem cells) actively divide to produce new keratinocytes, which are pushed up to the surface of the skin as new cells are created (Marieb 2009).
As the keratinocytes migrate toward the skin surface, they flatten out (become squamous), fill with the protein keratin, and eventually die. These dead cells, which form the tough outer barrier of the epidermis, are constantly shed and replaced by new cells migrating up from the basal layer. Also within the epidermis are pigmented cells (melanocytes) that give skin its color, immune cells (dendrocytes), and sensory (Merkel) cells (Marieb 2009).
Development of Skin Cancer
Because of their constant exposure to the environment, skin cells are particularly susceptible to the types of damage that can lead to malignant (cancerous) transformation and abnormal cell growth. While the cells at the skin’s surface are shed and replenished quickly, cells in the deeper basal layer (such as the keratinocyte stem cells, melanocytes, or Merkel cells) reside in the epidermis for extended periods of time and can more readily accumulate the necessary genetic damage to become malignant (Thieu 2013).
Sunlight (particularly ultraviolet B [UVB] radiation) is the predominant causative factor catalyst in the malignant transformation of healthy skin cells. UVB radiation can initiate carcinogenesis in several ways. It directly induces breakages in cellular DNA and increases the activity of regulatory T-cells – cells of the immune system that can suppress the skin’s immune response and enable the proliferation of cancer cells (Erb 2008).
UVB radiation also plays a role in preventing the repair and elimination of heavily UV-damaged cells by suppressing the activity of cellular defenses. In several skin cancers, sunlight causes mutations in the p53 gene, which is critical for the protection of genome (DNA) integrity; p53 mutations increase skin cancer risk and have been identified in about 56% of basal cell carcinomas and in >90% of squamous cell carcinomas (Benjamin 2008; Erb 2008). Loss of the protective activities of p53 also enables tumor angiogenesis (the formation of blood vessels to help nourish growing tumors) and metastasis (the spread and establishment of cancer cells in distant organs) (Benjamin 2008). Finally, UV light induces the production of the inflammatory mediator prostaglandin E2 (PGE2) by increasing the activity of the inflammatory enzyme cyclooxygenase-2 (COX-2). Overactivation of the COX-2 enzyme may lead to chronic inflammation which is one of the driving forces behind the development of skin cancer (Rundhaug 2007).
5 Skin Cancer Types
The diversity of cell types in the epidermal and dermal skin layers lends the possibility of several different types of skin cancers upon malignant transformation of these cells. In practice, the vast majority of skin cancers in humans are one of three types: melanoma (a malignancy of melanocytes); or one of two non-melanoma skin cancers (basal cell carcinoma and squamous cell carcinoma, both malignancies of keratinocytes) (Thieu 2013; Lin 2011). Each has unique features and metastatic potential.
Melanoma
Melanoma results from the malignancy of pigmented melanocytes that reside deep in the epidermis; this occurs by mutation in one of several regulatory genes (eg, genes involved in regulating the cell cycle or that control cell division), ultimately allowing uncontrolled melanocyte growth and invasion of surrounding tissues. These defects may be hereditary or acquired (Erb 2008; Dunki-Jacobs 2013). UV exposure also has a role in this process (Rass 2008). Additionally, local immunosuppression caused by malignant melanocytes is thought to enable melanoma to escape host immune defenses (Erb 2008). Melanoma rapidly metastasizes, making it the most dangerous of the skin cancers (Uzarska 2013; Dunki-Jacobs 2013). While the median age at diagnosis is 61, melanoma occurs over a wide distribution of ages and is one of the most frequent cancers found in adolescents and young adults (Dunki-Jacobs 2013).
Non-Melanoma Skin Cancers
Basal cell carcinoma. Basal cell carcinomas, which represent 80% of non-melanoma skin cancers, are the most commonly diagnosed cancer in the United States (Thieu 2013). They arise from pluripotent keratinocytes (stem cells) located in the basal layer of the epidermis and around hair follicles (Erb 2008; Firnhaber 2012; Ponten 1994). UV radiation appears to be the principal catalyst in the malignant transformation of basal cells (Thieu 2013), and exposure to sunlight in childhood and adolescence may be an important determinant of malignancy (Firnhaber 2012). Up to 20% of these cancers occur in people under age 50. They are locally aggressive, but rarely metastasize to distant organs (Robinson 2003; Kurian 2013). In addition to the disruption in cellular defenses (p53 gene mutation), a basal cell malignancy may also be caused by mutations in genes that regulate the growth and maintenance of keratinocyte stem cells (Erb 2008).
Squamous cell carcinoma. Squamous cell carcinomas represent about 20% of non-melanoma skin cancer cases (Alam 2001). They are thought to originate from keratinocytes (like basal cell carcinomas), but from a more superficial layer of the epidermis, not the basal layer (Erb 2008). Also in contrast to basal cell carcinomas, there is a higher risk of metastasis (Alam 2001; Jennings 2010). Squamous cell carcinomas are strongly associated with UV radiation exposure as well as other phenotypic markers (fair skin, light eyes, light-colored hair; see section titled “Causes and Risk Factors”). Their association with cumulative sunlight exposure makes them an uncommon skin cancer in people younger than 50 years old (Firnhaber 2012). Squamous cell carcinomas may also be induced through exposure to chemical carcinogens (Thieu 2013).
Other non-melanoma skin cancers are rare, making up <1% of all skin cancers (ACS 2014a). These include (Thieu 2013; Rehal 2013; Lazareth 2013; Sebastien 1993; Lober 1994):
- Merkel cell carcinoma – a rare, very aggressive malignancy of the sensory Merkel cells with high rates of local recurrence and distant metastasis;
- Sebaceous carcinoma – malignancy of sebaceous gland cells;
- Eccrine porocarcinoma – malignancy of sweat gland cells;
- Atypical fibroxanthoma – a dermal tumor with low-moderate metastatic potential; and
- Microcystic adnexal carcinoma – a locally invasive tumor derived from sweat gland cells.
6 Causes and Risk Factors
Sunlight/Ultraviolet Light Exposure
Exposure to sunlight/UV light is thought to be a primary risk factor for melanoma and non-melanoma skin cancers (Erb 2008; Etzkorn 2013). The depletion of the ozone layer is predicted to increase this risk; the World Health Organization has estimated that a 10% decrease of ozone level will result in 300 000 more non-melanoma and 4500 melanoma skin cancers every year (Erb 2008). A lifetime of working outdoors was found to nearly triple the risk of squamous cell carcinoma in one study (Sánchez 2013). Intentional exposure to UV radiation carries additional cancer risk; the use of tanning beds is associated with a 1.5-fold increase in the risk of basal cell carcinoma and a 2.5-fold increase in the risk of squamous cell carcinoma, and extended UV therapy for psoriasis (ie, >250 treatments of psoralen [a phototoxic compound] combined with UVA exposure) may increase skin cancer risk more than 10-fold (Firnhaber 2012). Although cumulative exposure to UV irradiation is associated with skin cancer risk, the nature of exposure may also be a factor (Miller 2006). Intensive sun exposure to the point of severe or blistering sunburn in childhood and teen years is a risk factor for both melanoma and basal cell carcinoma, whereas excessive chronic cumulative exposure is a risk factor for squamous cell carcinoma (Jou 2012).
Age and Gender
Melanoma and non-melanoma skin cancers are more common in men than women (Etzkorn 2013; Siegel 2013) and significantly increase in frequency with age (Dunki-Jacobs 2013; Etzkorn 2013).
Hair Color
Lighter hair color is associated with an increased risk of basal cell carcinoma; in a comprehensive review of 29 studies, dark-haired participants had a decreased risk of basal cell carcinoma compared to those with red hair (2-fold increased risk), blond hair (1.4-fold increased risk), or light brown hair (1.3-fold increased risk) (Khalesi 2013).
Skin Phototype
Skin types can be described by a continuous scale from I (white skin, always burns, never tans) to VI (dark brown/black skin, never burns, tans deeply) (Roberts 2009). Risk of non-melanoma skin cancers increases with lower skin phototype grade (Etzkorn 2013). Fair-skinned individuals and individuals with skin that burns but does not tan have a 2-fold increased risk of basal cell carcinoma compared to darker-skinned individuals (Khalesi 2013).
Genetics and Family History of Skin Cancer
A personal history of skin cancer increases risk of a non-melanoma cancer (Etzkorn 2013). A family history of skin cancer increases risk of squamous cell carcinoma by 6.5-fold (Sánchez 2013), and a history of two or more first-degree relatives with melanoma increases skin cancer risk more than 10-fold (Firnhaber 2012). Inherited mutations/polymorphisms (altered forms) of certain genes involved in cell cycle signaling (MC1R, CDKN2A) have been identified as risk factors for melanoma or non-melanoma skin cancers; mutations of the CDKN2A gene have been thought to account for approximately 20-40% of familial melanoma cases (Hughes-Davies 1998; Pacifico 2007; Carless 2008; Dunki-Jacobs 2013; Cust 2013; Valverde 1996; Jones 1999).
Concurrent Skin Conditions
Squamous cell carcinoma risk is increased in individuals with other dermatological conditions, including actinic conjunctivitis (2.7-fold increased risk), poikiloderma of Civatte (3.3-fold increased risk), facial actinic keratosis (9.3-fold increased risk), or numerous freckles (3.7-fold increased risk) (Sánchez 2013). Freckling is not associated with increased risk of basal cell carcinoma (Khalesi 2013). Individuals with more than 100 nevi (benign, pigmented growths of the skin, commonly called “moles” or “birthmarks”), or at least 5 atypical nevi, are at more than 10 times the risk of skin cancer than the general population (Firnhaber 2012).
Smoking
Smoking more than 10 cigarettes a day almost tripled squamous cell carcinoma risk in one study (Sánchez 2013).
Immunosuppression
Suppression of immune function (such as that caused by immunosuppressant drugs) increases skin cancer risk, especially for squamous cell carcinoma (Erb 2008). In one study, researchers found that organ transplant recipients, who are frequently on immunosuppressant drugs, had a 65-fold increased risk of squamous cell carcinoma compared with the general population (Firnhaber 2012).
7 Signs and Symptoms
Basal Cell Carcinoma
There are five major subtypes of basal cell carcinoma, each with a different appearance. Most basal cell carcinomas (85%) occur on the head and neck, with at least a third in areas that receive little or no sun exposure (Firnhaber 2012).
Nodular. Nodular basal cell carcinomas have a variable appearance, but commonly present as a pearly pink or white, dome-shaped raised lesion with prominent surface reddening due to dilated blood vessels.
Superficial. Superficial basal cell carcinomas are scaly plaques that resemble eczema or psoriasis, with characteristic raised, pearly white borders. Superficial basal cell carcinoma is the least invasive of the subtypes and most commonly occurs on the trunk and extremities.
Micronodular and infiltrative. These are more malignant subtypes of basal cell carcinoma that are difficult to distinguish visually from the nodular subtype.
Morpheaform. Morpheaform basal cell carcinoma lacks the pearly appearance of the nodular subtype, appearing as a firm, yellowish, ill-defined mass.
Squamous Cell Carcinoma
Squamous cell carcinoma commonly appears as a firm, smooth or scaly raised lesion, often ulcerated; patients commonly describe it as bleeding after minimal trauma. Squamous cell carcinomas are often localized to the backs of the hands and forearms as well as areas of the head and neck that receive maximal sun exposure (Firnhaber 2012; Ferri 2014).
Melanoma
Melanoma usually appears as an irregularly pigmented skin lesion that changes over time. They most commonly occur spontaneously, but occasionally arise from a pre-existing nevus (birthmark or mole). The ABCDE rule is a useful guide to identifying signs of possible melanoma. The characteristics of a melanoma are (ACS 2013b):
- Asymmetrical shape;
- Borders that are irregular, ragged, or blurred;
- Color that is not the same all over and may include shades of black, brown, pink, red, white, or blue;
- Diameter of more than 6 mm (the size of a pencil eraser);
- Evolving or changing in shape, size, or color.
It is important to remember that these characteristics are a guideline, and not every melanoma follows these rules (Dunki-Jacobs 2013). Other signs associated with melanoma are a sore that does not heal; spread of pigment from a spot to the surrounding skin; redness or swelling outside the border of a mole; and itchiness, pain, or changes in the surface of a mole (ACS 2013b).
8 Diagnosis and Staging
Diagnosis
Skin cancers are diagnosed based on physical exam, patient history, and a variety of diagnostic techniques. Proper diagnosis is important to not only confirm the presence of malignancy, but also to stage the cancer progression to determine a suitable treatment and prognosis (outcome) (Fecher 2012; Usatine 2011; Carillo 2012).
Biopsy. Suspected skin cancer lesions are initially biopsied (sampling of the affected tissue) using a shaving technique for raised tumors or a punch biopsy (in which a circular plug of tissue is removed) for abnormal skin. For very small tumors, complete excision of the tumor may be part of the initial diagnosis (Firnhaber 2012). Biopsied tissue is examined for malignancy by histological staining and microscopy (Al-Arashi 2007; Patil 2011).
Dermoscopy. Dermoscopy (also called dermatoscopy or skin surface microscopy) employs a handheld magnifying device (dermoscope) to improve the examination of suspect skin lesions. It allows visualization of structures below the skin surface that are not otherwise visible to the naked eye. Dermoscopy improves the accuracy in diagnosing melanoma and pigmented lesions by as much as 30% (Rao 2012).
Lymph node mapping and biopsy. Lymph nodes are small, oval-shaped organs that are distributed throughout the body (Marieb 2009; ACS 2014b). They are important organs of the immune system, and they house the lymphocytes involved in immune response. Cancers often spread to lymph nodes before invading other areas of the body. To map lymph nodes, a small amount of radioactive tracer may be injected near the skin tumor to determine the nearby lymph node(s) to which the cancer is likely to spread (sentinel lymph nodes) (Harwood 2005; Stoeckli 2001). These nodes can be biopsied to check for the presence of cancer cells.
Imaging tests. Imaging tests include the following (ACS 2013c):
- Computed tomography. Computed tomography (CT) is a three-dimensional X-ray imaging technique for visualizing internal structures; it is especially useful for determining the presence of metastasis to distant organs.
- Positron emission tomography. Positron emission tomography (PET) can locate areas of malignancy by tracing the deposition of a radioactive glucose analog (given by injection). Since cancer cells have a high metabolic rate, they take up the radioactive glucose more readily than normal cells and can be identified by a PET scan.
- Chest radiographs. Chest radiographs (“X-rays”) may be used to detect metastases to the lungs.
- Magnetic resonance imaging. Magnetic resonance imaging (MRI), which images soft tissues using high magnetic fields, may be used to detect metastasis to the brain or spinal cord.
Novel imaging. Advances in imaging technology are improving diagnostic precision.
- Multispectral imaging. Multispectral imagers construct images of suspected skin tumors using specific wavelengths of light to provide additional information on a lesion such as its borders, vasculature, or melanin content. This additional information can be used to strengthen a clinician’s diagnosis or determine whether a biopsy may be needed. Melafind is an imager/software system that takes images of a suspected melanoma at 10 different wavelengths of light, determining its border and collecting information on color variation, asymmetry, and changes in texture or perimeter. It compares these data to an internal database of over 9000 images of biopsied melanoma tumors and provides the clinician with a recommendation on whether the lesion should be biopsied (Ferris 2012). Melafind has been shown to increase the sensitivity of detection of melanomas when used by trained dermatologists (Wells 2012), although it has a low specificity and may over-recommend biopsy of lesions that are benign (Cukras 2013).
Blood tests. Blood chemistry analysis may be helpful in melanoma diagnosis; the enzyme lactate dehydrogenase (LDH) may be elevated in people with melanoma. A complete blood count (CBC) and liver function tests may also be part of a skin cancer workup (ACS 2013c).
Staging
Once melanoma or non-melanoma skin cancers have been identified, they are next staged. Staging is a standardized method for describing the size or extent of the original tumor and whether it has spread to other regions in the body. By understanding the severity or extent of a cancer, physicians are able to select an appropriate treatment and provide a realistic prognosis for the disease. Additionally, understanding the stage of a cancer may help identify clinical trials of emerging cancer treatments that may be available (NCI 2013).
The standard staging system for most cancers is the American Joint Committee on Cancer (AJCC) TNM system. In the TNM system, physical examination and other diagnostic techniques are used to assess 3 categories of information about the cancer (Warner 2011; NCI 2013):
- T classification: tumor size, location, and penetration into other tissues
- N classification: spread of the cancer into nearby lymph nodes
- M classification: spread of the cancer to other organs (metastasis)
Depending on each of the T, N, and M classifications, the cancer is given a “stage” from 0 (pre-cancerous; best prognosis) to IV (most progressive, most difficult to treat). Each type of cancer has its own TNM system, which considers unique features of the cancer, while some cancers (brain and spinal cord, for example) use different protocols for staging (NCI 2013).
For melanoma, the clinical staging (based on physical exam and biopsy of the main melanoma) is shown in Table I.
Table I. Cutaneous Melanoma Clinical Staging System
(Balch 2009; NCI 2014b)
| Stage | Description (T, N, and M values are indicated in parentheses) |
0 |
There is a presence of abnormal or pre-cancerous cells only in the topmost layer of skin (also called melanoma in situ). There is no evidence of its spread to local lymph nodes or distant organs. (Tis, N0, M0) |
Ia |
The melanoma is 1.0 mm or less in thickness and is not ulcerated (there is no breakdown of the skin over the melanoma). There is no evidence of its spread to local lymph nodes or distant organs. (T1a, N0, M0) |
Ib |
The melanoma is 1.0 mm or less in thickness and ulcerated OR 1.01–2.0 mm in thickness and not ulcerated. There is no evidence of its spread to local lymph nodes or distant organs. (T1b or T2a, N0, M0) |
IIa |
The melanoma is 1.01–2.0 mm in thickness and ulcerated OR 2.01–4.0 mm in thickness and not ulcerated. There is no evidence of its spread to local lymph nodes or distant organs. (T2b or T3a, N0, M0) |
IIb |
The melanoma is 2.01–4.0 mm in thickness and ulcerated OR more than 4.0 mm in thickness and not ulcerated. There is no evidence of its spread to local lymph nodes or distant organs. (T3b or T4a, N0, M0) |
IIc |
The melanoma is more than 4.0 mm in thickness and ulcerated. There is no evidence of its spread to local lymph nodes or distant organs. (T4b, N0, M0) |
III |
The melanoma can be of any thickness and may/may not be ulcerated. There is evidence of its spread to one or more lymph nodes near the affected skin area. There is no evidence of its spread to distant organs. (any T, N1-3, M0) |
IV |
The melanoma can be of any thickness and may/may not be ulcerated. It may/may not have spread to local lymph nodes. It has spread to other organs, such as distant areas of skin or lymph nodes, the lungs, or other visceral sites. (any T, any N, M1) |
Squamous cell and basal cell carcinomas share a different set of criteria for staging (Table II). For these cancers, certain “high risk features” (characteristics of tumors associated with a poor prognosis) are also considered in the staging protocol. High risk features include (ACS 2014c):
- Tumor is thicker than 2 mm;
- Tumor has invaded deeper skin layers (lower dermis or subcutis);
- Tumor has grown into tiny nerves in the skin (perineural invasion);
- Tumor started on an ear or on the lips;
- Tumor cells look very abnormal when seen under a microscope (poorly differentiated or undifferentiated).
Table II: Non-melanoma Staging System
| Stage | Description (T, N, and M values are indicated in parentheses) |
0 |
There is a presence of abnormal or pre-cancerous cells only in the topmost layer of skin (also called carcinoma in situ). There is no evidence of its spread to local lymph nodes or distant organs. (Tis, N0, M0) |
I |
The tumor is 2 cm or less across and has ≤1 high-risk feature. There is no evidence of its spread to local lymph nodes or distant organs. (T1, N0, M0) |
II |
The tumor is more than 2 cm across and has 2 or more high-risk features. There is no evidence of its spread to local lymph nodes or distant organs. (T2, N0, M0) |
III |
The tumor has grown into facial bones (such as the jaw or around the eye). There is no evidence of its spread to local lymph nodes or distant organs; OR The tumor is of any size or has grown into facial bones. It has spread to 1 nearby lymph node on the same side of the body as the main tumor and the node is 3 cm or less in size. There is no evidence of its spread to distant organs. (T3, N0, M0) or (T1-3, N1, M0) |
IV |
The tumor is of any size or has grown into facial bones. It has spread to lymph nodes on the same or on the other side of the body, none of which are larger than 6 cm in diameter. There is no evidence of its spread to distant organs; OR The tumor is of any size. It has spread to any lymph node that is nearby and is larger than 6 cm in diameter. There is no evidence of its spread to distant organs; OR The tumor has grown into other bones of the body or into the skull. It may or may not have spread into the lymph nodes, but has not spread to distant organs; OR Any tumor that has spread to distant organs. (T1-3, N2, M0) or (Any T, N3, M0) or (T4, any N, M0) or (any T, any N, M1) |
9 Conventional Treatment
Treatments for melanoma and non-melanoma skin cancers include surgery, radiation therapy, topical and systemic drugs. The treatment choice depends on the cancer staging, risk of recurrence, and risk of metastasis. Tumors with high-risk behavior (based on location and size, well-defined versus ill-defined borders, primary versus recurrent disease, immunosuppression status, sites of prior radiotherapy, invasion into local nerves, evidence of chronic inflammation or rapid growth) are placed on high-risk treatment pathways (Kim 2012). Cure rates of non-melanoma skin cancers can reach 96-99% but are associated with high rates of recurrence. Five-year recurrence rates as high as 50% have been reported by some authors (Nguyen 2002; Firnhaber 2012), although lower rates have also been reported and vary by treatment type (Kim 2012).
Electrodessication and Curettage
Electrodessication and curettage (ED&C) is one of the most widely used treatments for squamous and basal cell carcinomas. It uses high voltage current to destroy the tumor and surrounding healthy tissue, which is removed by scraping with a special instrument (curette). The process is repeated until the tumor is removed. Low-risk areas (the trunk and extremities) are ideal locations for treatment by ED&C (Galiczynski 2011). Five-year tumor recurrence rates following ED&C are 7.7% (Kim 2012).
Cryosurgery
Cryosurgery has been used for both benign and malignant dermatological lesions. A cryogen (usually liquid nitrogen) is applied to the lesion with an applicator device, lowering the tissue temperature to -50°C to -60°C. This causes necrosis (death) of tumor cells and local healthy tissue. Cryosurgery is not suitable for aggressive tumors, those with ill-defined borders, or those that extend deeper than 10 mm (Galiczynski 2011). Five-year tumor recurrence rates following cryosurgery are 7.5% for non-melanoma skin cancers (Kim 2012).
Surgical Excision
Surgical excision is the standard option for surgical management of most non-melanoma skin cancers. The objective of surgical excision is to remove the entire tumor in addition to several millimeters of healthy tissue around the lesion as a safety margin. The size of excision margins varies by the size and aggressiveness of the tumor, with squamous cell carcinoma tumors over 2 cm in size requiring at least 10 mm of healthy tissue to be excised around the tumor. Taking sufficient margins during excision increases the probability that all of the tumor has been removed; this can be verified by microscopic evaluation of the excision margins. Surgical excision of basal cell carcinoma on the head, neck, trunk, and extremities has a five-year cure rate >95%, although the resultant surgical wounds can have cosmetic or functional impacts for the patient (Lazareth 2013).
Mohs Microsurgery
Mohs microsurgery is the preferred method of treatment for all non-melanoma high-risk tumors based on reported efficacy, with five-year tumor recurrence rates for basal cell carcinoma of 1-3.3% (Lazareth 2013; Kim 2012; Paoli 2011; Rowe 1989). It also offers significant advantages for surgical excision of melanoma (Hui 2012). In the Mohs technique, the tumor is excised with a very thin margin, and the excised tumor is immediately analyzed for the presence of any malignant cells within the margins of the excision. If malignant cells are found, these are mapped back onto the skin incision so the surgeon may precisely remove the additional cancerous tissue. The process is repeated until all surgical margins are clear of cancer cells. Mohs microsurgery presents the best cure rates, lowest recurrence rates, and leaves the smallest surgical wounds. The major disadvantages are expense and limited access to surgeons trained in the technique (Lazareth 2013; Larson 2013). Patients can locate physicians trained in Mohs surgery through the Surgeon Finder resource on the website of the American College of Mohs Surgery (http://acms.execinc.com/edibo/surgeonfinder).
Radiotherapy
Radiation can be an effective therapy for non-melanoma skin cancers up to 15 mm in diameter located in high-risk areas (Kim 2012). It offers reasonable cosmetic as well as functional outcomes and is a consideration for some types of cancerous lesions arising on or near the eyelids, nose, ears, and lips (Lazareth 2013). Superficial radiation therapy delivers low voltage (orthovoltage) X-ray radiation that rapidly loses its strength as it penetrates tissues to treat superficial cancers. Cancerous lesions up to about 6 cm deep require treatment by higher energy (megavoltage) electron beam radiation generated by linear accelerators to adequately penetrate tissues. A third type of radiotherapy, brachytherapy or interstitial therapy, delivers small beads of radioactive material directly into the cancerous lesion. Because the intensity of brachytherapy radiation decreases rapidly with distance, it allows a very high radiation dose to be delivered right at the source of the malignancy, while sparing noncancerous surrounding tissue (Hulyalkar 2011). Radiotherapy is a useful adjunct therapy for incompletely excised tumors, or those that have spread to local or distant lymph nodes (Lazareth 2013). Five-year tumor recurrence rates for basal cell carcinoma following radiotherapy are 8.7% (Kim 2012). Because of the potential to cause DNA damage and induce additional tumor formation in the treated area, radiotherapy is not a preferred option in young or immunocompromised individuals (Lazareth 2013).
Superficial Therapies
Topical therapies have been used either alone (monotherapy) or as an adjunct to other therapies for the treatment of non-melanoma skin cancers (Amini 2010). They are convenient, can generally preserve skin appearance and integrity, and can be used to treat large areas. The immunomodulating agent imiquimod (Aldara®) and the DNA synthesis inhibitor 5-fluorouracil (5-FU) are among the two most commonly used topical agents (Desai 2012; MacFarlane 2010). Imiquimod is approved by the Food and Drug Administration (FDA) for treatment of superficial basal cell carcinoma <2 cm in diameter. 5-FU has been used as a treatment for other solid tumors (breast and colorectal) when given intravenously and is indicated for the topical treatment of pre-cancerous skin lesions (actinic keratosis) as well as squamous cell carcinoma in situ (Galiczynski 2011). Few randomized trials compared the efficacy of topical treatment of squamous or basal cell carcinomas to that of surgery or radiation (Kim 2012), and the evidence of their use as monotherapy is limited. Use of topical imiquimod or 5-FU may be more appropriate for patients with small tumors in low-risk areas who are unwilling to undergo better-established therapies (Firnhaber 2012).
10 Novel and Emerging Therapies
Vismodegib
Vismodegib (Erivedge®) is the first antitumor drug to target the hedgehog signaling pathway, which is a set of proteins that interact to regulate the growth of keratinocyte stem cells (Amin 2013; Erb 2008). Dysregulation of the hedgehog signaling pathway is associated with aggressive subtypes of cancers that are prone to metastasis (Carpenter 2012). Vismodegib was FDA-approved for treatment of advanced and metastatic basal cell carcinoma in 2012. In two phase II trials of patients with metastatic basal cell carcinoma or basal cell nevus syndrome (a genetic disorder that predisposes patients to basal cell carcinoma), vismodegib (150 mg/day) significantly decreased the appearance of new tumors and reduced the size of existing tumors (Amin 2013).
Tumor Treating Fields
Tumor treating fields (TTFields) represent a noninvasive antitumor technique that uses low-intensity alternating electrical fields to disrupt tumor cell division and induce cell death. Different cancer types can be specifically targeted by “tuning” the electrical field frequency delivered by the TTField generator. It has been approved for the treatment of recurrent glioblastoma in the United States. Ongoing preclinical studies have demonstrated efficacy in preventing proliferation of isolated human melanoma cells, intradermal melanoma in rats, and the spread of injected malignant melanoma cells in mice (Davies 2013).
Laser Therapy
Several case reports have detailed success in treating basal cell carcinoma and squamous cell carcinoma in situ using pulsed CO2 or pulsed dye laser light (Brightman 2011). Lasers ablate (vaporize) tumor tissue from the skin, offering better visualization for the surgeon and less tissue injury than some other current treatments, such as ED&C (Shimizu 2011). Tumor clearance may be a concern for deeper tumors treated by laser. In a large case report of 44 patients with a form of squamous cell carcinoma, CO2 laser treatment was associated with a tumor clearance rate of 97.6% overall, with a tumor recurrence of 6.8% over 8-52 months (Covadonga Martínez González 2008).
Targeted Therapies
Vemurafenib and Trametinib. BRAF is a gene involved in the regulation of cell growth and proliferation; mutations of this gene leave it in a permanently (constitutively) active state and can result in uncontrolled cellular proliferation. Approximately 50% of melanomas contain BRAF mutations (Kudchadkar 2013). Therapies that inhibit the activity of BRAF and related regulatory factors represent a novel systemic approach to increase survival in patients with metastatic melanoma (melanoma that has spread to distant sites). Before either of these drugs is prescribed, presence of BRAF mutations must first be confirmed.
- Vemurafenib. Vemurafenib (Zelboraf®) is a BRAF inhibitor that has demonstrated significant antitumor activity in clinical trials and was shown to improve survival in patients with metastatic melanoma that carries the BRAF mutation (a median survival of 13.6 months in patients taking vemurafenib vs. 9.7 months in patients on standard chemotherapy). Side effects include fatigue, joint pain, edema, nausea, and heart rhythm changes (ie, a longer QT interval on electrocardiogram) (Johnson 2013). By altering cell-signaling pathways, vemurafenib has also been shown to paradoxically increase the risk of cutaneous squamous cell carcinoma, with lesions appearing in up to 20% of patients (Johnson 2013; Ma 2014).
- Trametinib. Trametinib (Mekinist®) was approved as a single therapy in 2013 and as a combination therapy in 2014 against malignant melanoma (NCI 2014c; Wright 2013). It targets cancer cells by indirectly inhibiting the activity of mutant BRAF genes (it actually inhibits cell cycle signaling proteins called MEK1 and MEK2) (Johnson 2013). As a single therapy, the most common side effects of trametinib include rash, diarrhea, hair loss, dermatitis, and edema (Liu 2013).
Immunotherapies
Interferon alpha. Interferon alpha (IFN-α) is an inflammatory signaling molecule produced by cells of the immune system in response to pathogens (primarily viruses) (Bekisz 2004); its antitumor effect comes from its ability to increase the activity of natural killer cells of the innate immune system (Quigley 2013). IFN-α has been used for the local treatment of cutaneous melanoma and as an adjuvant (additional) therapy to surgical resection (Quigley 2013; Tarhini 2012). As an adjuvant therapy, IFN-α has been shown to improve survival and reduce tumor recurrence in patients with high-risk melanoma (Tarhini 2012). Case reports also suggest IFN-α may be used to treat metastatic melanoma of the skin (Quigley 2013). IFN-α is given by injection. Side effects include depression, anxiety, and infection (Ma 2014).
Interleukin-2. Interleukin-2 is a cell-signaling molecule that helps orchestrate the development of specialized immune cells involved in anticancer immunity (Lotze 1985; van der Vliet 2007). Intravenous high-dose interleukin-2 therapy was approved by the US FDA in 1998 for the treatment of metastatic melanoma. As of 2013, it remained the only FDA-approved treatment regimen associated with continuous remission lasting longer than 5 years. Unfortunately, the overall response rate is only 16% and high-dose interleukin-2 therapy is associated with significant toxicity. Thus, its use has been restricted to patients who have advanced melanoma but are otherwise in good health (Guan 2012; Chu 2013; Weide 2010).
One of the major toxicities associated with high-dose interleukin-2 therapy is injury of the delicate endothelial cells that line the inside of blood vessels, leading to a potentially life-threatening condition called vascular leak syndrome (Guan 2012; Chu 2013). Interestingly, adding oral supplementation with resveratrol, a phytochemical found in Japanese knotweed and grapes (Chen 2013; Latruffe 2013), to high-dose interleukin-2 therapy significantly mitigated the development of vascular leak syndrome in a murine model of metastatic melanoma. Moreover, the combination of resveratrol and high-dose interleukin-2 was more effective in inhibiting tumor growth and metastasis than high-dose interleukin-2 alone. The dose of resveratrol given to the mice was equivalent to about 648 mg for an 80 kg adult human. The scientists who conducted the study remarked “Our results suggested the potential use of resveratrol in [high-dose interleukin-2] treatment against melanoma” (Guan 2012).
Other avenues are being pursued in hopes of optimizing the efficacy and safety profile of interleukin-2 therapy for melanoma. One promising approach involves administering interleukin-2 directly into tumors as opposed to systemic administration. In one study on 48 subjects with metastatic melanoma, intratumoral injection of interleukin-2 three times weekly led to a complete local response that lasted for at least 6 months in 70% of patients. Only mild to moderate toxicity was observed. The initial dose of interleukin-2 used in this study was 3 million IU. The dose was escalated as the study progressed depending on how well each subject tolerated the treatment (Weide 2010). Other researchers have investigated the possibility of administering interleukin-2 complexed with nanoparticles. In mice bearing engrafted melanoma, injection of interleukin-2-containing nanoparticles into the tissue surrounding the tumors suppressed tumor growth and improved the animals’ survival (Yao 2011).
Ipilimumab. Ipilimumab (Yervoy®) is a targeted immunotherapy that was approved in 2011 for the treatment of metastatic melanoma that is refractory to other treatments. Ipilimumab is a monoclonal antibody that binds to CTLA-4, a protein involved in regulating the activity of immune cells (T-lymphocytes or T-cells). By binding and inactivating CTLA-4, ipilimumab causes the continuous activation of T-cells, which is thought to stimulate the immune response against melanoma (Kudchadkar 2013). In a phase III clinical trial, ipilimumab improved median survival in patients with an unresectable stage III or IV melanoma compared to patients given only a cancer vaccine (10 months vs. 6.5 months, respectively). Side effects are autoimmune in nature and include pruritus (itchy skin), diarrhea, rash, colitis, and vitiligo (skin depigmentation) (Liu 2013). Although only a fairly low number of patients (10%) respond to ipilimumab, it produces significant results in those that do respond and may be useful in difficult-to-treat melanomas or as an adjuvant to other treatments (Davar 2013).
Ingenol Mebutate
Ingenol mebutate (Picato®) is a topical antitumor therapy extracted from the sap of Euphorbia peplus, a plant traditionally used as a treatment for skin diseases (Bahner 2013). It is an FDA-approved treatment for actinic keratosis, a type of UV-induced precancerous lesion that has the potential of progressing to squamous cell carcinoma (Firnhaber 2012). Ingenol is being investigated as a potential topical treatment for non-melanoma skin cancers. It is thought to work by rapidly disrupting mitochondrial membrane integrity in tumor cells, leading to cell death, and by inducing antibody-mediated eradication of residual diseased cells (Ogbourne 2004; Siller 2010). A pilot trial of high-dose (0.05%) ingenol gel in patients with superficial basal cell carcinoma demonstrated tumor clearance in 63% of patients (Siller 2010). Application of Euphorbia sap containing ingenol to squamous or basal cell carcinoma lesions showed a complete clinical response (no evidence of tumor by visual inspection) in 75-82% of patients at one month following treatment in an uncontrolled trial, with 50-57% of patients with no visual evidence of tumor at an average of 15 months post-treatment (Ramsay 2011).
Metformin
Metformin, an oral hypoglycemic agent and a first-line drug treatment for type 2 diabetes, possesses a tumor-suppressing effect that may be useful in treating skin cancer (Badr 2013). In mice injected with human squamous cell carcinoma cells, metformin reduced tumor growth rate and biomarkers of cancer cell proliferation (Chaudhary 2012). A similar study demonstrated metformin’s ability to inhibit the formation of chemically-induced skin tumors and reduce the size of existing tumors; this effect was more pronounced in obese mice than overweight control mice (Checkley 2013). Part of the antitumor activity in these studies was attributed to metformin’s ability to reduce activity of pro-inflammatory metabolic pathways. Metformin also inhibited growth of human melanoma cells in cell culture experiments (Badr 2013).
Aspirin and Non-Steroidal Anti-Inflammatory Drugs
Non-steroidal anti-inflammatory drugs (NSAIDs) and aspirin inhibit the cyclooxygenase (COX) enzymes, which are involved in several aspects of cancer biology (Zhan 2007). Use of NSAIDs or aspirin has been associated with reduced risk of several cancers, including skin cancers (Johannesdottir 2012). In a study on nearly 200 000 individuals in Denmark, ever having taken aspirin or NSAIDs was associated with a 15% reduced risk of squamous cell carcinoma and a 13% reduced risk of malignant melanoma. The risk reduction became more pronounced with higher intensity and longer duration of aspirin and NSAID use (Johannesdottir 2012).
In cell culture experiments, aspirin appears to be selectively toxic toward melanoma cancer cells (Vad 2008). Two large observational studies have demonstrated a significant reduction in the risk of cutaneous melanoma with frequent aspirin usage. In a large Dutch population-based study of 8104 subjects, daily low-dose aspirin usage (30-100 mg/day) for at least 3 years reduced melanoma risk by 46% among women (Joosse 2009). In the Women’s Health Initiative observational study of over 59 000 post-menopausal women, aspirin usage reduced cutaneous melanoma risk by 21% (Gamba 2013).
A topical preparation of the NSAID diclofenac sodium at 3% concentration (Solaraze®) is FDA-approved to treat actinic keratosis, a premalignant skin condition, which may lead to skin cancer. Topical diclofenac sodium is thought to treat actinic keratosis by inhibiting the COX-2 enzyme, inducing apoptosis (programmed cell death), and inhibiting the formation of new blood vessels (angiogenesis). It has demonstrated good efficacy, with one study reporting 58% of subjects treated with Solaraze achieved complete clearance at 30-days post-treatment (Martin 2012).
More Information about repurposing drugs for use in the context of cancer in general is available at Life Extension’s Drug Repurposing in Cancer protocol.
11 Lifestyle Considerations
Skin Self-Examination
Although there are several effective treatments for melanoma and non-melanoma skin cancers, the probability of successful outcomes increase considerably with early detection. Survival rates for melanoma drop significantly with growth of the primary tumor or spread to local lymph nodes (Balch 2009). Regular skin self-examinations can aid in detecting skin cancers early when they are easier to treat. It is important to inspect the entire skin surface for suspicious lesions. For non-melanoma skin cancers, look for new growths, spots, bumps, or non-healing sores, especially in areas exposed to sunlight. For melanoma, the ABCDE rule (refer to “Signs and Symptoms” section) (ACS 2013b) is a useful guide to identifying signs of possible melanoma. It is important to remember that these characteristics are a guideline, and not every melanoma follows these rules (Dunki-Jacobs 2013). Any suspicious skin lesion should be referred to a qualified health care professional for further evaluation.
Reduce UV Exposure
UV radiation is a risk factor for melanoma and non-melanoma skin cancers, and reducing sun exposure can mitigate this risk. This includes minimizing time spent outside during peak sun hours; heeding the National Weather Service’s daily UV index; and wearing long sleeves, sunglasses, and wide-brimmed hats (Jou 2012). The use of sunscreen has been generally recommended, although few studies have examined its efficacy in the reduction of skin cancer risk. It is important to remember that potential benefits of sunscreen are negated if it causes the user to increase time spent in the sun (Etzkorn 2013).
12 Nutrients
Conventional approaches are highly effective in the treatment of skin cancer, and the earlier the cancer is detected, the better the likelihood of a successful outcome. While the integrative interventions listed below may complement conventional skin cancer treatment, anyone who identifies a suspicious lesion on their skin should seek medical evaluation from a dermatologist right away.
B Vitamins
B complex vitamins are a group of water-soluble nutrients that support numerous cellular processes through their role as coenzymes. Adequate levels of B vitamins are necessary for skin health. In fact, deficiencies of some B vitamins, including B2 (riboflavin), B3 (niacin), B6 (pyridoxine), B7 (biotin), B8 (inositol), and B12 (cobalamin), cause characteristic skin disorders (Schellack 2019; Srihari 2024).
A meta-analysis of data from 18 randomized controlled trials involving 74,498 participants found supplementing with folic acid (mostly in dosages ranging from 680–8,500 mcg DFE per day), alone or with B6 (mostly 10–250 mg per day) and/or B12 (mostly 400–2,000 mcg per day), reduced melanoma risk (Zhang 2016).
One observational study searched for possible causal relationships between nutrients and skin cancer using genetic information from 19,077 non-melanoma skin cancer patients and 314,193 people without skin cancer. The study found higher vitamin B6 levels were correlated with lower non-melanoma skin cancer risk in a way that may be causal (rather than coincidental) (Chen 2025).
The above studies notwithstanding, the specific evidence on the various B vitamins and skin cancer-related outcomes is relatively sparse and mixed. However, nicotinamide is an exception in that there has been more robust, rigorous research into its role in skin cancer risk reduction (Camillo 2025).
Nicotinamide. Nicotinamide is a derivative of vitamin B3 (niacin). It is used in the body to make nicotinamide adenine dinucleotide (NAD+) and nicotinamide adenine dinucleotide phosphate (NADP+), coenzymes that play a critical role in numerous biochemical reactions inside cells that involve oxidation and reduction—the transfer of electrons. Nicotinamide has antioxidant and anti-inflammatory activities but does not cause side effects commonly triggered by other forms of niacin, such as flushing, itching, low blood pressure, and headaches, even at doses up to 3 grams (Camillo 2025). Nicotinamide is used to treat various skin conditions, including acne, rosacea, atopic dermatitis, and melasma (a type of skin discoloration). It may also help prevent skin cancer by promoting DNA repair and preventing ultraviolet light-induced immunosuppression in the skin (Camillo 2025; Nazarali 2017).
Some observational evidence indicates a possible role for nicotinamide in preventing skin cancer. An observational study evaluated skin cancer diagnoses in 12,287 subjects, some of whom were transplant recipients, who had received 500 mg of nicotinamide twice daily for 30 days or longer and in 21,479 matched individuals who had not received nicotinamide. The study found nicotinamide use was associated with a 14% lower likelihood of a skin cancer diagnosis. In those who started nicotinamide after a first skin cancer diagnosis, the likelihood of a second skin cancer was reduced by 54%. The association was strongest for squamous cell carcinoma (Breglio 2025).
Nicotinamide may help prevent non-melanoma skin cancer in patients at high risk. A randomized placebo-controlled trial that included 386 subjects with a history of at least two non-melanoma skin cancers in the previous five years found 500 mg of nicotinamide twice daily for 12 months reduced new non-melanoma skin cancers overall by 23%, basal cell carcinomas by 20%, and squamous cell carcinomas by 30% (Chen 2015). In a randomized placebo-controlled trial in 76 subjects with four or more actinic keratoses (precancerous sun-induced lesions), 500 mg of nicotinamide twice daily for four months decreased the number of actinic keratoses and reduced the risk of developing non-melanoma skin cancer (Surjana 2012; Jiminez 2023).
One meta-analysis pooled findings from 29 randomized controlled trials that reported on skin cancer outcomes in 3,039 participants who received nicotinamide therapy or served as controls. Most trials included participants who were not at high risk of skin cancer. The analysis found nicotinamide therapy (ranging from 500–1,000 mg daily or a 1% topical gel) was associated with 50% lower overall skin cancer risk. As for tolerability, this meta-analysis found that digestive side effects such as diarrhea were about 5.5% more frequent in the nicotinamide group compared with the control group (Mainville 2022).
Topical solutions containing 5% nicotinamide have been found to prevent UV light-induced immunosuppression in skin when used just before or just after light exposure, suggesting they may protect against skin cancer (Sivapirabu 2009; Damian 2008). However, in a crossover trial in 30 volunteers, oral nicotinamide was significantly more effective for protecting against UV light-induced immunosuppression in the skin: light-induced immunosuppression was reduced 59% after three days of treatment with a 5% topical nicotinamide lotion once daily and 66% after three days of oral nicotinamide at 500 mg twice daily (Thanos 2012). Clinical trials are needed to determine whether topical nicotinamide can reduce skin cancer risk.
Oral nicotinamide has been shown to be safe at doses of up to 3 grams daily; however, doses of 3.5 grams or higher may cause headache, dizziness, vomiting, increased liver enzyme levels, and liver toxicity (Camillo 2025).
Probiotics
In addition to their potential functions in gastrointestinal health, animal models suggest certain strains of probiotic bacteria may minimize UV-induced skin photodamage, potentially by modulating inflammatory response. Administration of the Bifidobacterium breve strain Yakult (BBY) to hairless mice for 9 and 14 days reduced skin elastase activity (a marker for photodamage) and inflammatory cytokine interleukin-1 beta (IL-1β) production arising from 4 consecutive days of UV-irradiation exposure (Sugimoto 2012). Lipoteichoic acid, a component of the cell wall of the probiotic strain Lactobacillus rhamnosus GG, had immunostimulatory effects and was photoprotective in hairless mice exposed to UV radiation and delayed the appearance of skin tumors (Weill 2012). A similar protection from UV immune suppression was observed in hairless mice after 10 days of supplementation with Lactobacillus johnsonii La1 (Guéniche 2006).
Tea Polyphenols
The anti-inflammatory effects of green tea polyphenols have been substantiated by dozens of laboratory and animal studies (Singh 2010). The polyphenol epigallocatechin gallate (EGCG) exerts its anti-inflammatory effects by inhibition of the NF-κβ signaling pathway, which results in decreased expression of several inflammatory mediators (such as IL-1β) in cell culture experiments (de Mejia 2009). Laboratory experiments have shown that EGCG suppresses melanoma proliferation through the inhibition of NF-κβ, which in turn led to decreased cell growth (Ellis 2011). Several studies of topical and oral green tea in mice have demonstrated the inhibition or delay of UVB-induced skin tumor development by reducing inflammation, inhibiting angiogenesis (recruitment of blood vessels by the tumor), reducing DNA damage, promoting rapid DNA repair, and reducing tissue adiposity (Katiyar 2007; Katiyar 2011; Payette 2010; Yang 2011). In a study conducted on human skin samples, topical green or white tea conferred protection against UV-induced detriments to cutaneous immunity (Camouse 2009).
Polypodium
Polypodium leucotomos, a species of tropical fern with anti-inflammatory and antioxidant properties, has long been used by Native Americans as a treatment for inflammatory disorders (Jansen 2013; Aguilera 2013). Polypodium extracts have been studied as an adjunctive therapy for several skin conditions, atopic dermatitis (Ramirez Boscá 2012), and photodermatoses (sunlight-induced skin rashes) (Caccialanza 2011). In mice fed Polypodium extract (300 mg/kg) for 10 days prior to exposure to UV irradiation, Polypodium pre-treatment lowered the activity of COX-2 (an inflammatory enzyme implicated in cancer progression) compared with control animals. Evidence of DNA damage was also reduced in the extract-fed mice (Zattra 2009). In 61 patients with melanoma, pre-treatment with Polypodium extract (1080 mg) before exposure to UV-radiation increased the minimal erythematous dose (the amount of UV radiation to which skin can be exposed before sunburn) compared to baseline (Aguilera 2013). This reduction in UV sensitivity was seen in 65% of the patients in the study.
Red Orange Complex
Red oranges, also known as blood oranges, have rinds resembling traditional oranges, but contain deeply-pigmented flesh rich in bioactive compounds. Three red orange varieties from the Mediterranean region – Citrus sinensis var. Moro, Tarocco, and Sanguinello – have gained scientific interest as an abundant source of powerful phytoceuticals including anthocyanins, flavones, hydroxycinnamic acids, and ascorbic acid (Bonina 2002; Saija 1998). These red orange constituents have been shown to exert potent antioxidant action and bolster intrinsic free radical defenses within the body when administered as a supplement at 50 mg per day to diabetics (Bonina 2002). Red orange constituents also appear to benefit cardiovascular health; in a study on 19 subjects between ages 27 and 56 with increased cardiovascular risk, supplementation with 500 mL of red orange juice daily for 7 days significantly improved a measure of endothelial function and reduced markers of systemic inflammation in blood samples compared to placebo (Buscemi 2012). Evidence for photoprotective properties of red orange extract make it appealing as a skin-protection agent. One study showed that topical application of red orange extract reduced UVB-induced erythema in healthy volunteers (Saija 1998). In a study on 18 volunteers aged 26-47, subjects took 100 mg of red orange complex twice daily for 15 days, then researchers tested the sensitivity of participants’ skin to UVB radiation. Supplementation with red orange complex led to a roughly 35% reduction in the intensity of skin erythema following UVB exposure, suggesting consumption of 200 mg daily of red orange complex may mitigate the likelihood of developing sunburn (Bonina 2008).
Grape Seed Extract
Grape seed extract contains proanthocyanidins, phytochemicals that exhibit antioxidant and anti-inflammatory properties (Jensen 2010). In mouse models and cell culture experiments of UV-induced skin damage, grape seed proanthocyanidins have enhanced DNA repair (Vaid, Sharma 2010), reduced immune suppression (Vaid 2011) and oxidative stress (Sharma 2007), and prevented tumor growth (Jensen 2010). In a case-control observational study, supplement usage was compared between 415 patients diagnosed with squamous cell carcinoma and 415 age-, sex-, and race-matched control subjects with no history of skin cancer. The comparison revealed that participants who consumed grape seed extract had a 74% decreased risk of cutaneous squamous cell carcinoma compared to those that did not (Asgari 2011). The same study demonstrated a borderline significant risk reduction among multivitamin users. No information on dose or duration of supplement use was obtained in this study. When applied topically to 10 healthy volunteers, grape seed extract decreased the incidence of sunburned keratinocytes and cells with UV-induced mutations in the DNA-protective protein p53 after experimental exposure to two doses of UV irradiation (Yuan 2012).
Silymarin
Silymarin, a mixture of several related flavonolignan compounds from milk thistle (Abenavoli 2010; Vaid, Katiyar 2010), may aid in the prevention of photocarcinogenesis by several mechanisms. In mouse models, topical application of silymarin reduced UV-induced hydrogen peroxide production (a major source of oxidative stress), reduced infiltration of inflammatory white blood cells into UV-irradiated skin, and reduced UV-induced production of inflammatory mediators (prostaglandins and COX-2) (Vaid, Katiyar 2010). Oxidative damage and inflammation have both been implicated in skin carcinogenesis. Additional mouse studies have demonstrated that silymarin may reduce UV-induced immune suppression (Katiyar 2002), markers of DNA damage (thymine dimers) (Dhanalakshmi 2004), and tumorigenesis (Katiyar 1997).
Fish Oil
Fish oil is a source of omega-3 fatty acids (eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA]), which cannot be efficiently synthesized by humans but are nonetheless essential for several metabolic processes. Omega-3 fatty acids may help reduce UV-induced immune suppression (an important component of skin carcinogenesis). In a study of 79 women with cutaneous nickel allergies, participants took 5 g/day of supplemental omega-3 (70% EPA plus 10% DHA) for 3 months. After supplementation, skin was exposed to 3 daily immunosuppressing doses of solar UV irradiation and then exposed to nickel sulfate ointment. UV-induced immune suppression appeared to be mitigated in the participants taking omega-3 supplements compared to the control group, although the results were not statistically significant (Pilkington 2013).
Pine Bark Extract
Extracts of the bark of the French maritime pine, Pinus pinaster, contain phenolic and polyphenolic flavonoids that are reported to possess multiple biological effects, including antioxidant, anti-inflammatory, and anti-carcinogenic properties (Sime 2004). Studies in mouse models have demonstrated that pine bark extracts may reduce UV-induced skin carcinogenesis by several mechanisms. Oral administration of pine bark extract (60, 200, or 600 mg/kg) twice daily to hairless mice significantly inhibited UV-induced photo-aging and increased the diameter and length of skin blood vessels. It also prevented increases in cellular biomarkers of UV damage (8-hydroxy-2’-deoxyguanosine-positive, vascular endothelial growth factor) (Kimura 2010). Long-term supplementation with pine bark extract protected chronically UV-exposed mice (10 min/day for 10 months) from squamous cell carcinoma formation (Pavlou 2009). In a third study, topical application of pine bark extract reduced immunosuppression and delayed tumor growth in chronically UV-exposed (5 UV doses/week for 10 weeks) hairless mice (Sime 2004).
Melatonin
Melatonin is the main secretory product of the pineal gland in the brain and regulator of circadian rhythm; its recognized anti-oxidation and membrane stabilization actions may make it a promising candidate as a sun-protective agent. In a systematic review of 4 randomized controlled human trials (58 total participants), topical melatonin (0.5-2.5%) demonstrated an overall significant suppression of UV-induced erythema (sunburn) when applied prior to UV exposure (Scheuer 2013). Melatonin has also demonstrated reductions of chemical-induced skin carcinogenesis in a mouse model (Man'cheva 2011). Evidence suggests that higher urinary levels of a major melatonin metabolite may be associated with reduced skin cancer risk. In a study on 140 subjects – 70 with skin cancer and 70 healthy controls – healthy individuals without skin cancer had significantly higher urinary levels of 6-sulfatoxymelatonin over a 24-hour period compared to skin cancer patients. The researchers who conducted the study concluded “It seems that a low level of 24-hour urinary 6-sulfatoxymelatonin renders human beings prone to skin cancer” (Ghaderi 2014).
Vitamin D
Vitamin D is important for healthy skin. It is involved in the growth regulation of keratinocytes and helps maintain barrier and immune function in the skin (Rosen 2012; Umar 2018). Vitamin D and its metabolites protect healthy skin cells by inhibiting DNA damage from UV exposure and inducing DNA repair mechanisms (Bocheva 2021). Vitamin D also suppresses proliferation of skin cancer cells, suggesting vitamin D and its metabolites may be protective against skin cancers (Gandini 2009; Bocheva 2021; Sutedja 2022).
Several studies have examined the link between vitamin D intake, status, and melanoma. In a cross-sectional study of 498 adults at risk of skin cancer, regular use of vitamin D supplements (doses not specified) was associated with a 55% reduced risk of melanoma occurrence compared with non-users (Kanasuo 2022). A 2022 meta-analysis including over 200,000 cases of melanoma concluded that vitamin D intake and serum levels were not closely related with risk of melanoma occurrence; however, serum vitamin D levels were inversely associated with melanoma thickness, which is correlated with melanoma mortality (Song 2022). And in a prospective study of over 1,000 participants, lower vitamin D serum levels (<16 ng/mL vs. ≥16 ng/mL) were associated with poorer melanoma outcomes, including increased likelihood of disease recurrence, increased tumor thickness, and nearly twice the odds of dying from melanoma (Fang 2016). Other studies have also found higher relapse-free survival in those with higher serum vitamin D levels (≥25 ng/mL) and worse overall survival in those with lower levels (<10 ng/mL) in people with melanoma (Berwick 2013; Newton-Bishop 2009; Garcia-Darder 2022).
While the illustration of a direct preventative effect of vitamin D on skin cancer awaits further study, supplemental vitamin D may be beneficial in maintaining adequate vitamin D levels without sun exposure. Approximately 90% of vitamin D is produced in the skin by exposure to sunlight (specifically UVB radiation [ie, the same radiation that increases cancer risk]) (Stroud 2008), and supplemental vitamin D may be a better option for maintaining vitamin D levels, especially in individuals at elevated risk for skin cancer (Barysch 2010; Martin-Gorgojo 2021).
Resveratrol and Pterostilbene
Much of the recent research on the phytochemical resveratrol has focused on its potential in cancer chemoprevention and treatment, inflammation, and oxidative stress. In cell culture, it has been shown to induce death in human epidermoid carcinoma and melanoma cells and reduce pro-oxidant production by cancer cells (Ndiaye 2011). It was also shown experimentally to increase the anti-cancer toxicity of the chemotherapy drugs temozolomide (Temodar®), cisplatin (Platinol®), and etoposide (Etopophos®), and the efficacy of radiation therapy against melanoma cell lines, while having no effect on normal human fibroblasts (Osmond 2012; Heiduschka 2014). In mouse models, oral and topical resveratrol significantly reduced UVB-mediated inflammation, free-radical generation, infiltration of white blood cells, tumorigenesis, and malignant conversion of benign skin papillomas to squamous cell carcinomas (Ndiaye 2011). Peritumoral injections of resveratrol in a mouse model of uveal (ocular) melanoma demonstrated its ability to induce cancer cell death and tumor regression (van Ginkel 2008). An analog of resveratrol, pterostilbene, may also inhibit skin carcinogenesis by reducing inflammatory gene expression; in a mouse model, it reduced expression of the pro-inflammatory enzyme COX-2 and the multiplicity of skin tumors (papilloma) induced by chemical carcinogens (Tsai 2012).
Curcumin
Curcumin has been investigated in a variety of human cancers including pancreatic, prostate, breast, and head and neck cancer (Sonavane 2012). In cell culture, curcumin inhibits growth of squamous cell carcinoma and melanoma cells (Sonavane 2012; Caltagirone 2000). In mouse models, topical application of low dose curcumin along with the carcinogen 12-O-tetradecanoylphorbol-13-acetate (TPA) reduced the ability of the carcinogen to induce skin tumor formation and DNA damage (Huang 1997). Both topical and dietary curcumin equally reduced the growth of human squamous cell carcinoma cells implanted in the skin of mice (Sonavane 2012).
Quercetin
Quercetin intake has been associated with decreases in colorectal and lung cancer risk; its potential anti-carcinogenic effects stem from its anti-inflammatory and antioxidant activity (Jung 2013). In cell culture experiments, quercetin was toxic against melanoma cancer cells (Sak 2014; Caltagirone 2000) and reduced the incidence of skin carcinogenesis caused by the carcinogens 12-O-tetradecanoylphorbol-13-acetate (TPA) and 7,12-dimethylbenz(a)anthracene (DMBA) by 35% when supplied at 0.02% in the diet of mice (Jung 2013). In a mouse model of melanoma metastasis, quercetin reduced the number of melanomas in mouse lungs by up to 71% when fed to mice at a concentration of 50 mg/kg (or about 324 mg for an 80 kg human) (Caltagirone 2000).
Coenzyme Q10
Coenzyme Q10 (CoQ10) is essential for normal cellular respiration and energy production, and its deficiency has been suggested to contribute to abnormal cell division patterns, which may favor cancer cell growth. Levels of CoQ10 are reduced in several cancers, including melanoma, and patients with melanoma who presented with low CoQ10 levels were found to have significantly higher risks of metastatic dissemination in one study (Rusciani 2006). In patients with stage I and II melanoma that had been treated by surgical excision, adjuvant therapy with CoQ10 (200 mg, twice daily) plus IFN-α for 3 years resulted in only about one-tenth the occurrence of tumor metastasis over an 8-year period compared to patients treated with interferon alone (Rusciani 2007).
Carnosol
Carnosol is an antioxidant principle from rosemary (Rosmarinus officinalis), which along with the related compound carnosic acid, is responsible for about 90% of rosemary’s antioxidant activity. In cell culture, it sensitizes melanoma cells to the damaging effects of X-ray radiation, increasing cellular death by 34% compared to radiation alone (Alcaraz 2013). It also inhibited the migration and invasion of melanoma cells in a cell culture assay (Huang 2005).
Mistletoe
Injectable preparations from the European mistletoe (Viscum album L.) are prescribed for cancer patients in several European countries (Horneber 2008). Several small pilot studies investigating mistletoe extract in cancer therapy have shown variable efficacy (Ernst 2003; Horneber 2008). One epidemiological study looked at patients who received surgical treatment for stage II or III melanoma at one of 35 German or Swiss hospitals and medical practices between 1985 and 2001. A group of 329 patients that received fermented mistletoe extract injections as part of their post-operative therapy were compared to 357 patients who did not. Over a 6.8 year period, patients that received mistletoe therapy demonstrated a significant increase in overall survival compared to the control group, although the rate of metastasis was not different between the groups except for lung/mediastinal metastases, which were significantly more rare in the group receiving mistletoe (Augustin 2005).
Honokiol
Honokiol is a derivative of Magnolia officinalis, a traditional Chinese therapy with antiangiogenic, anxiolytic, anti-inflammatory, and antitumor properties (Mannal 2011). In cell culture experiments, Honokiol extracts were shown to be toxic to human melanoma cells, by stopping their proliferation and growth, and increasing the production of apoptotic enzymes and mitochondrial depolarization (signs of apoptosis – programmed cell death) (Mannal 2011; Kaushik 2012). In a mouse model, topical application of high dose (60 mcg) Honokiol to the mice prior to UV exposure reduced tumor incidence by 40% compared to control animals (Guillermo 2012).
Ukrain
Ukrain is a semisynthetic derivative of alkaloids from Chelidonium majus that has been reported to have a selective toxic effect on tumor cells (Skivka 2011). It is licensed for use as a cancer therapy in several former Soviet states. Ukrain is given by intravenous injection and has been studied as a primary or adjuvant therapy for treating several human cancers (colorectal, bladder, pancreatic, and breast) (Ernst 2005). Published case reports suggest its efficacy in maintaining remission in two patients with melanoma (Hamler 1996; Stabuc 1996). In mouse models, Ukrain injections (7 times per day every third day) reduced tumor growth from both low- and high-metastatic melanoma variants (Skivka 2011).
Modified Citrus Pectin
Modified citrus pectin (MCP) is a water-soluble, indigestible polysaccharide isolated from the peel and pulp of citrus fruits, and it is modified using high pH and temperature treatment (Glinsky 2009). MCP, which has been studied as a metal chelating agent, has been reported to reduce heavy metal burden in several human studies (Crinnion 2008). Its potential activity against metastatic cancer is related to its ability to inhibit the function of the pro-carcinogenic factor galectin-3. Galectin-3 is a galactose-binding protein produced in excess in cancer cells, which protects them from self-destruction and facilitates their adhesion to the walls of blood vessels during metastasis (Glinsky 2009). Pectin is rich in galactoside residues and may bind and inhibit the activity of galectin-3; in mouse models, pre-incubation of MCP with B16-F1 melanoma cells reduced their ability to cause lung metastases by 90% when both were injected into test mice (Platt 1992). MCP also reduced the ability of melanoma cells to anchor and grow on a semi-solid growth surface in a cell culture experiment (Inohara 1994).
Fermented Wheat Germ Extract
Fermented wheat germ extract (Avemar®, AvéULTRA®), a patented natural preparation obtained via fermentation of wheat germ with the yeast Saccharomyces cerevisiae, has shown promise as an anticancer agent in several preclinical and clinical studies (Hidvegi 2002; Mueller 2011). It is approved in Europe as a “dietary food for special medical purposes for cancer patients” (Demidov 2008).
Studies indicate fermented wheat germ extract combats cancer via several mechanisms including inhibition of glucose uptake in cancer cells, suppression of metastasis, mitigation of cancer cell proliferation, and modulation of the immune system. It also appears not to reduce efficacy or enhance toxicity of conventional chemotherapy. Fermented wheat germ extract contains a multitude of molecular compounds, but evidence suggests that two glucosides – 2-methoxy benzoquinone and 2,6-dimethoxy benzoquinone – are partially responsible for its biological properties (Mueller 2011).
In a randomized clinical trial of 52 patients with high-risk stage III skin melanoma, adjuvant chemotherapy with dacarbazine was compared to dacarbazine plus fermented wheat germ extract. Following surgery, 26 patients were allocated to receive dacarbazine alone and 26 were allocated to receive dacarbazine plus 8.5 g daily of fermented wheat germ extract. Dacarbazine was given in 5-day infusion cycles once monthly for up to 4 months or until the disease progressed and treatment with fermented wheat germ extract alone continued for up to 12 months. At the 7-year follow-up period, progression-free survival was greater in the fermented wheat germ extract plus dacarbazine group (mean 55.8 months) compared to the dacarbazine-only group (mean 29.9 months). Overall survival was also greater in the subjects that received fermented wheat germ extract (mean 66.2 months) compared to those that received dacarbazine alone (mean 44.7 months). The researchers concluded “The inclusion of Avemar into the adjuvant protocols of high-risk skin melanoma patients is highly recommended” (Demidov 2008).
Active Hexose Correlated Compound
Active hexose correlated compound (AHCC) is derived from several species of Basidiomycete mushrooms. It is comprised of polysaccharides, amino acids, lipids, and minerals. Evidence has revealed AHCC to be a powerful immunomodulator, bolstering resistance to infections. It also possesses anticancer properties. In a preclinical study, AHCC delayed tumor development in mice inoculated with melanoma cells. It also enhanced growth and activity of several antitumor immune cells in this study. These findings led the researchers to conclude “…our results demonstrate that AHCC can enhance tumor immune surveillance through regulating both innate and adaptive immune responses” (Gao 2006).
AHCC may represent a valuable adjuvant therapy for individuals receiving systemic cancer treatment, as it has been shown to reduce the adverse effects of chemotherapy in preclinical and clinical studies (Ito 2014; Shigama 2009; Hangai 2013; Hirose 2007). A safety study found that AHCC is generally well tolerated in healthy volunteers and does not cause laboratory abnormalities (Spierings 2007).
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
Abenavoli, L., Capasso, R., Milic, N., and Capasso, F. Milk thistle in liver diseases: past, present, future. Phytother Res. 2010;24(10):1423–32.
ACS. American Cancer Society. 2013a. Cancer Facts and Figure 2013. Atlanta: American Cancer Society. Available at: http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-036845.pdf. Accessed 12/17/2013.
ACS. American Cancer Society. How is melanoma skin cancer diagnosed? Available at: http://www.cancer.org/cancer/skincancer-melanoma/detailedguide/melanoma-skin-cancer-diagnosed. Last updated 10/29/2013c. [Accessed December 4, 2013].
ACS. American Cancer Society. Lymph Nodes and Cancer. What is the lymph system? Available at: http://www.cancer.org/cancer/cancerbasics/lymph-nodes-and-cancer. Last updated 3/6/2014b. Accessed 3/31/2014.
ACS. American Cancer Society. Skin Cancer: Basal and Squamous Cell. What are basal and squamous cell skin cancers? Available at: http://www.cancer.org/cancer/skincancer-basalandsquamouscell/detailedguide/skin-cancer-basal-and-squamous-cell-what-is-basal-and-squamous-cell. Last updated 2/20/2014a. Accessed 3/31/2014.
ACS. American Cancer Society. Skin Cancer: Basal and Squamous Cell. How are basal and squamous cell skin cancers staged? Available at: http://www.cancer.org/cancer/skincancer-basalandsquamouscell/detailedguide/skin-cancer-basal-and-squamous-cell-staging. Last updated 2/2/2014c. Accessed 3/28/2014.
ACS. American Cancer Society. What should I look for? Available at: http://www.cancer.org/cancer/skincancer-melanoma/moreinformation/skincancerpreventionandearlydetection/skin-cancer-prevention-and-early-detection-what-to-look-for. Last updated 1/25/2013b. [Accessed December 4, 2013].
Aguilera, P., Carrera, C., Puig-Butille, J. A., Benefits of oral Polypodium Leucotomos extract in MM high-risk patients. J Eur Acad Dermatol Venereol. 2013;27(9):1095–100.
Alam, M., and Ratner, D. Cutaneous squamous-cell carcinoma. N. Engl. J. Med. 2001;344(13):975–83.
Al-Arashi MY, Salomatina E, Yaroslavsky AN. Multimodal confocal microscopy for diagnosing nonmelanoma skin cancers. Lasers in surgery and medicine. Oct 2007;39(9):696-705.
Alcaraz, M., Achel, D. G., Olivares, A., Olmos, E., Alcaraz-Saura, M., and Castillo, J. Carnosol, radiation and melanoma: a translational possibility. Clin Transl Oncol. 2013;15(9):712–9.
Allen NC, Martin AJ, Snaidr VA, et al. Nicotinamide for Skin-Cancer Chemoprevention in Transplant Recipients. The New England journal of medicine. Mar 2 2023;388(9):804-812. doi:10.1056/NEJMoa2203086. https://pubmed.ncbi.nlm.nih.gov/36856616/ https://www.nejm.org/doi/pdf/10.1056/NEJMoa2203086?download=true
Amin, S. H., Motamedi, K. K., Ochsner, M. C., Song, T. E., and Hybarger, C. P. Mechanisms and efficacy of vismodegib in the treatment of basal cell carcinoma. Discov Med. 2013;16(89):229–32.
Amini S, Viera MH, Valins W, Berman B. Nonsurgical innovations in the treatment of nonmelanoma skin cancer. The Journal of clinical and aesthetic dermatology. Jun 2010;3(6):20-34.
Asgari, M. M., Chren, M.-M., Warton, E. M., Friedman, G. D., and White, E. Supplement use and risk of cutaneous squamous cell carcinoma. J. Am. Acad. Dermatol. 2011;65(6):1145–51.
Augustin, M., Bock, P. R., Hanisch, J., Karasmann, M., and Schneider, B. Safety and efficacy of the long-term adjuvant treatment of primary intermediate- to high-risk malignant melanoma (UICC/AJCC stage II and III) with a standardized fermented European mistletoe (Viscum album L.) extract. Results from a multicenter, comparative, epidemiological cohort study in Germany and Switzerland. Arzneimittelforschung. 2005;55(1):38–49.
Badr, D., Kurban, M., and Abbas, O. Metformin in dermatology: an overview. J Eur Acad Dermatol Venereol. 2013;27(11):1329–35.
Bahner, J. D., and Bordeaux, J. S. Non-melanoma skin cancers: photodynamic therapy, cryotherapy, 5-fluorouracil, imiquimod, diclofenac, or what? Facts and controversies. Clin. Dermatol. 2013;31(6):792–8.
Balch, C. M., Gershenwald, J. E., Soong, S. J., Final Version of 2009 AJCC Melanoma Staging and Classification. Journal of Clinical Oncology. 2009;27(36):6199–206.
Barysch, M. J., Hofbauer, G. F., and Dummer, R. Vitamin D, ultraviolet exposure, and skin cancer in the elderly. Gerontology. 2010;56(4):410–3.
Bekisz J, Schmeisser H, Hernandez J, Goldman ND, Zoon KC. Human interferons alpha, beta and omega. Growth factors (Chur, Switzerland). Dec 2004;22(4):243-251.
Benjamin, C. L., Melnikova, V. O., and Ananthaswamy, H. N. P53 protein and pathogenesis of melanoma and nonmelanoma skin cancer. Adv Exp Med Biol. 2008;624:265–82.
Berwick, M., and Erdei, E. O. Vitamin D and melanoma incidence and mortality. Pigment Cell Melanoma Res. 2013;26(1):9–15.
Bocheva G, Slominski RM, Slominski AT. The Impact of Vitamin D on Skin Aging. International journal of molecular sciences. Aug 23 2021;22(16)doi:10.3390/ijms22169097. https://pubmed.ncbi.nlm.nih.gov/34445803/
Bonina F. Effect of the supplementation with Red Orange Complex® on ultraviolet-induced skin damage in human volunteers. Bionap report. Santa Venerina, CT, Italy. 2008.
Bonina FP, Leotta C, Scalia G, Puglia C, Trombetta D, Tringali G, . . . Saija A. Evaluation of oxidative stress in diabetic patients after supplementation with a standardised red orange extract. Diabetes, nutrition & metabolism. Feb 2002;15(1):14-19.
Breglio KF, Knox KM, Hwang J, et al. Nicotinamide for Skin Cancer Chemoprevention. JAMA Dermatology. 2025;doi:10.1001/jamadermatol.2025.3238. https://doi.org/10.1001/jamadermatol.2025.3238
Brightman, L., Warycha, M., Anolik, R., and Geronemus, R. Do lasers or topicals really work for nonmelanoma skin cancers? Semin Cutan Med Surg. 2011;30(1):14–25.
Buscemi S, Rosafio G, Arcoleo G, Mattina A, Canino B, Montana M, . . . Rini G. Effects of red orange juice intake on endothelial function and inflammatory markers in adult subjects with increased cardiovascular risk. The American journal of clinical nutrition. May 2012;95(5):1089-1095.
Caccialanza, M., Recalcati, S., and Piccinno, R. Oral polypodium leucotomos extract photoprotective activity in 57 patients with idiopathic photodermatoses. G Ital Dermatol Venereol. 2011;146(2):85–7.
Caltagirone, S., Rossi, C., Poggi, A., Flavonoids apigenin and quercetin inhibit melanoma growth and metastatic potential. Int J Cancer. 2000;87(4):595–600.
Camillo L, Zavattaro E, Savoia P. Nicotinamide: A Multifaceted Molecule in Skin Health and Beyond. Medicina (Kaunas, Lithuania). Feb 1 2025;61(2)doi:10.3390/medicina61020254. https://pubmed.ncbi.nlm.nih.gov/40005371/
Camouse MM, Domingo DS, Swain FR, Conrad EP, Matsui MS, Maes D, . . . Baron ED. Topical application of green and white tea extracts provides protection from solar-simulated ultraviolet light in human skin. Experimental dermatology. Jun 2009;18(6):522-526.
Carillo J. Basal cell carcinoma. First Consult. Available at: www.clinicalkey.com. Last updated 9/14/2012. Accessed 3/31/2014.
Carless, M. A., and Griffiths, L. R. Cytogenetics of melanoma and nonmelanoma skin cancer. Adv Exp Med Biol. 2008;624:227–40.
Carpenter RL, Lo HW. Hedgehog pathway and GLI1 isoforms in human cancer. Discovery medicine. Feb 2012;13(69):105-113.
Chaudhary, S. C., Kurundkar, D., Elmets, C. A., Kopelovich, L., and Athar, M. Metformin, an antidiabetic agent reduces growth of cutaneous squamous cell carcinoma by targeting mTOR signaling pathway. Photochem. Photobiol. 2012;88(5):1149–56.
Checkley, L. A., Rho, O., Angel, J. M. Metformin Inhibits Skin Tumor Promotion in Overweight and Obese Mice. Cancer Prevention Research (Phil). 2013. [Epub ahead of print]
Chen AC, Martin AJ, Choy B, et al. A Phase 3 Randomized Trial of Nicotinamide for Skin-Cancer Chemoprevention. The New England journal of medicine. Oct 22 2015;373(17):1618-26. doi:10.1056/NEJMoa1506197. https://www.ncbi.nlm.nih.gov/pubmed/26488693
Chen AC, Martin AJ, Dalziell RA, et al. A phase II randomized controlled trial of nicotinamide for skin cancer chemoprevention in renal transplant recipients. Br J Dermatol. Nov 2016;175(5):1073-1075. doi:10.1111/bjd.14662. https://pubmed.ncbi.nlm.nih.gov/27061568/
Chen H, Tuck T, Ji X, Zhou X, Kelly G, Cuerrier A, Zhang J. Quality assessment of Japanese knotweed (Fallopia japonica) grown on Prince Edward Island as a source of resveratrol. Journal of agricultural and food chemistry. Jul 3 2013;61(26):6383-6392.
Chen W, Pang L, Wei X, et al. Micronutrients and skin cancer risk: a Mendelian randomization study. World journal of surgical oncology. May 11 2025;23(1):180. doi:10.1186/s12957-025-03814-1. https://pubmed.ncbi.nlm.nih.gov/40350422/
Chu MB, Fesler MJ, Armbrecht ES, Fosko SW, Hsueh E, Richart JM. High-Dose Interleukin-2 (HD IL-2) Therapy Should Be Considered for Treatment of Patients with Melanoma Brain Metastases. Chemotherapy research and practice. 2013;2013:726925.
Covadonga Martínez González, M., Del Pozo, J., Paradela, S., Fernández Jorge, B., Fernández Torres, R., and Fonseca, E. Bowen's disease treated by carbon dioxide laser. A series of 44 patients. Journal of Dermatological Treatment. 2008;19(5):293–9.
Crinnion, W. Is modified citrus pectin an effective mobilizer of heavy metals in humans? Altern Med Rev. 2008;13(4):283–6.
Cukras, A. R. On the comparison of diagnosis and management of melanoma between dermatologists and MelaFind. JAMA Dermatol. 2013;149(5):622–3.
Cust AE, Goumas C, Vuong K, Davies JR, Barrett JH, Holland EA, . . . Jenkins MA. MC1R genotype as a predictor of early-onset melanoma, compared with self-reported and physician-measured traditional risk factors: an Australian case-control-family study. BMC cancer. Sep 4 2013;13(1):406.
Damian DL, Patterson CR, Stapelberg M, Park J, Barnetson RS, Halliday GM. UV radiation-induced immunosuppression is greater in men and prevented by topical nicotinamide. J Invest Dermatol. Feb 2008;128(2):447-54. doi:10.1038/sj.jid.5701058. https://pubmed.ncbi.nlm.nih.gov/17882270/
Davar, D., Tarhini, A. A., and Kirkwood, J. M. Adjuvant immunotherapy of melanoma and development of new approaches using the neoadjuvant approach. Clin. Dermatol. 2013;31(3):237–50.
Davies, A. M., Weinberg, U., and Palti, Y. Tumor treating fields: a new frontier in cancer therapy. Ann N Y Acad Sci. 2013;1291:86–95.
de Mejia, E. G., Ramirez-Mares, M. V., and Puangpraphant, S. Bioactive components of tea: cancer, inflammation and behavior. Brain Behav. Immun. 2009;23(6):721–31.
Demidov LV, Manziuk LV, Kharkevitch GY, Pirogova NA, Artamonova EV. Adjuvant fermented wheat germ extract (Avemar) nutraceutical improves survival of high-risk skin melanoma patients: a randomized, pilot, phase II clinical study with a 7-year follow-up. Cancer biotherapy & radiopharmaceuticals. Aug 2008;23(4):477-482.
Desai T, Chen CL, Desai A, Kirby W. Basic pharmacology of topical imiquimod, 5-fluorouracil, and diclofenac for the dermatologic surgeon. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. Jan 2012;38(1):97-103.
Dhanalakshmi, S., Mallikarjuna, G. U., Singh, R. P., and Agarwal, R. Silibinin prevents ultraviolet radiation-caused skin damages in SKH-1 hairless mice via a decrease in thymine dimer positive cells and an up-regulation of p53-p21/Cip1 in epidermis. Carcinogenesis. 2004;25(8):1459–65.
Drago F, Ciccarese G, Cogorno L, Calvi C, Marsano LA, Parodi A. Prevention of non-melanoma skin cancers with nicotinamide in transplant recipients: a case-control study. European journal of dermatology : EJD. Aug 1 2017;27(4):382-385. https://pubmed.ncbi.nlm.nih.gov/28468736/
Dunki-Jacobs, E. M., Callender, G. G., and McMasters, K. M. Current management of melanoma. Curr Probl Surg. 2013;50(8):351–82.
Eisemann, N., Waldmann, A., Geller, A. C. Non-Melanoma Skin Cancer Incidence and Impact of Skin Cancer Screening on Incidence. 2013:1–8.
Ellis, L. Z., Liu, W., Luo, Y. Green tea polyphenol epigallocatechin-3-gallate suppresses melanoma growth by inhibiting inflammasome and IL-1β secretion. Biochem Biophys Res Commun. 2011;414(3):551–6.
Erb, P., Ji, J., Kump, E., Mielgo, A., and Wernli, M. Apoptosis and pathogenesis of melanoma and nonmelanoma skin cancer. Adv Exp Med Biol. 2008;624:283–95.
Ernst, E., and Schmidt, K. Ukrain - a new cancer cure? A systematic review of randomised clinical trials. BMC Cancer. 2005;5:69.
Ernst, E., Schmidt, K., and Steuer-Vogt, M. K. Mistletoe for cancer? Int J Cancer. 2003;107(2):262–7.
Etzkorn, J. R., Parikh, R. P., Marzban, S. S. Identifying risk factors using a skin cancer screening program. Cancer Control. 2013;20(4):248–54.
Fang S, Sui D, Wang Y, et al. Association of Vitamin D Levels With Outcome in Patients With Melanoma After Adjustment For C-Reactive Protein. J Clin Oncol. May 20 2016;34(15):1741-7. doi:10.1200/jco.2015.64.1357. https://pubmed.ncbi.nlm.nih.gov/27001565/
Fartasch M, Diepgen TL, Schmitt J, Drexler H. The relationship between occupational sun exposure and non-melanoma skin cancer: clinical basics, epidemiology, occupational disease evaluation, and prevention. Deutsches Arzteblatt international. Oct 2012;109(43):715-720.
Fecher LA. Melanoma. First Consult. Available at: www.clinicalkey.com. Last updated 8/22/2012. Accessed 3/31/2014.
Ferri FF. Squamous Cell Carcinoma. Ferri’s Clinical Advisor. Available at: www.clinicalkey.com. 2014. Accessed 3/31/2014.
Ferris, L. K., and Harris, R. J. New diagnostic aids for melanoma. Dermatol Clin. 2012;30(3):535–45.
Firnhaber, J. M. Diagnosis and treatment of Basal cell and squamous cell carcinoma. Am Fam Physician. 2012;86(2):161–8.
Galiczynski, E. M., and Vidimos, A. T. Nonsurgical treatment of nonmelanoma skin cancer. Dermatol Clin. 2011;29(2):297–309–x.
Gamba, C. A., Swetter, S. M., Stefanick, M. L. Aspirin is associated with lower melanoma risk among postmenopausal Caucasian women: the Women's Health Initiative. Cancer. 2013;119(8):1562–9.
Gandini, S., Raimondi, S., Gnagnarella, P., Doré, J.-F., Maisonneuve, P., and Testori, A. Vitamin D and skin cancer: a meta-analysis. Eur J Cancer. 2009;45(4):634–41.
Gao Y, Zhang D, Sun B, Fujii H, Kosuna K, Yin Z. Active hexose correlated compound enhances tumor surveillance through regulating both innate and adaptive immune responses. Cancer immunology, immunotherapy : CII. Oct 2006;55(10):1258-1266.
Ghaderi R, Sehatbakhsh S, Bakhshaee M, Sharifzadeh GR. Urinary melatonin levels and skin malignancy. Iranian journal of medical sciences. Jan 2014;39(1):64-67.
Glinsky, V. V., and Raz, A. Modified citrus pectin anti-metastatic properties: one bullet, multiple targets. Carbohydr. Res. 2009;344(14):1788–91.
Gloster HM, Jr., Brodland DG. The epidemiology of skin cancer. Dermatologic surgery : official publication for American Society for Dermatologic Surgery [et al.]. Mar 1996;22(3):217-226.
Gracia-Darder I, Carrera C, Alamon-Reig F, Puig S, Malvehy J, Podlipnik S. Vitamin D deficiency in melanoma patients is associated with worse overall survival: a retrospective cohort study. Melanoma research. Oct 1 2022;32(5):384-387. doi:10.1097/cmr.0000000000000842. https://pubmed.ncbi.nlm.nih.gov/35979670/
Guan H, Singh NP, Singh UP, Nagarkatti PS, Nagarkatti M. Resveratrol prevents endothelial cells injury in high-dose interleukin-2 therapy against melanoma. PloS one. 2012;7(4):e35650.
Guéniche, A., Benyacoub, J., Buetler, T. M., Smola, H., and Blum, S. Supplementation with oral probiotic bacteria maintains cutaneous immune homeostasis after UV exposure. Eur J Dermatol. 2006;16(5):511–7.
Guillermo, R. F., Chilampalli, C., Zhang, X., Zeman, D., Fahmy, H., and Dwivedi, C. Time and dose-response effects of honokiol on UVB-induced skin cancer development. Drug Discov Ther. 2012;6(3):140–6.
Hamler, F., Hiesmayr, W., Korsh, O., and Melnyk, A. Ukrain monotherapy in malignant melanoma (case report). Drugs Exp Clin Res. 1996;22(3-5):235–7.
Hangai S, Iwase S, Kawaguchi T, Kogure Y, Miyaji T, Matsunaga T, . . . Yamaguchi T. Effect of active hexose-correlated compound in women receiving adjuvant chemotherapy for breast cancer: a retrospective study. Journal of alternative and complementary medicine (New York, N.Y.). Nov 2013;19(11):905-910.
Harwood, M., Wu, H., Tanabe, K., and Bercovitch, L. Metastatic basal cell carcinoma diagnosed by sentinel lymph node biopsy. J. Am. Acad. Dermatol. 2005;53(3):474–7.
Heiduschka, G., Lill, C., Seemann, R. The effect of resveratrol in combination with irradiation and chemotherapy: Study using Merkel cell carcinoma cell lines. Strahlenther Onkol. 2014;190(1):75-80.
Hidvegi M, Tomoskozine RF, Lapis K, Raso E, Szende B. Immunostimulatory and metastasis inhibiting fermented vegetal material. Google Patents; 2002.
Hirose A, Sato E, Fujii H, Sun B, Nishioka H, Aruoma OI. The influence of active hexose correlated compound (AHCC) on cisplatin-evoked chemotherapeutic and side effects in tumor-bearing mice. Toxicology and applied pharmacology. Jul 15 2007;222(2):152-158.
Horneber, M. A., Bueschel, G., Huber, R., Linde, K., and Rostock, M. Mistletoe therapy in oncology. Cochrane Database Syst Rev. 2008;(2):CD003297.
Huang, M. T., Ma, W., Yen, P. Inhibitory effects of topical application of low doses of curcumin on 12-O-tetradecanoylphorbol-13-acetate-induced tumor promotion and oxidized DNA bases in mouse epidermis. Carcinogenesis. 1997;18(1):83–8.
Huang, S.-C., Ho, C.-T., Lin-Shiau, S.-Y., and Lin, J.-K. Carnosol inhibits the invasion of B16/F10 mouse melanoma cells by suppressing metalloproteinase-9 through down-regulating nuclear factor-kappa B and c-Jun. Biochem. Pharmacol. 2005;69(2):221–32.
Hughes-Davies TH. CDKN2A mutations in multiple primary melanomas. The New England journal of medicine. Jul 30 1998;339(5):347-348.
Hui, A. M., Jacobson, M., Markowitz, O., Brooks, N. A., and Siegel, D. M. Mohs micrographic surgery for the treatment of melanoma. Dermatol Clin. 2012;30(3):503–15.
Hulyalkar, R., Rakkhit, T., and Garcia-Zuazaga, J. The role of radiation therapy in the management of skin cancers. Dermatol Clin. 2011;29(2):287–96.
Inohara, H., and Raz, A. Effects of natural complex carbohydrate (citrus pectin) on murine melanoma cell properties related to galectin-3 functions. Glycoconj. J. 1994;11(6):527–32.
Ito T, Urushima H, Sakaue M, Yukawa S, Honda H, Hirai K, . . . Kondo K. Reduction of Adverse Effects by a Mushroom Product, Active Hexose Correlated Compound (AHCC) in Patients With Advanced Cancer During Chemotherapy-The Significance of the Levels of HHV-6 DNA in Saliva as a Surrogate Biomarker During Chemotherapy. Nutrition and cancer. Apr 2014;66(3):377-382.
Jansen, R., Wang, S. Q., Burnett, M., Osterwalder, U., and Lim, H. W. Photoprotection: Part I. Photoprotection by naturally occurring, physical, and systemic agents. J. Am. Acad. Dermatol. 2013;69(6):853.e1–853.e12.
Jennings L, Schmults CD. Management of high-risk cutaneous squamous cell carcinoma. The Journal of clinical and aesthetic dermatology. Apr 2010;3(4):39-48.
Jensen, J. D., Wing, G. J., and Dellavalle, R. P. Nutrition and melanoma prevention. Clin. Dermatol. 2010;28(6):644–9.
Jiminez V, Yusuf N. An update on clinical trials for chemoprevention of human skin cancer. J Cancer Metastasis Treat. 2023;9(1)doi:10.20517/2394-4722.2022.99. https://pubmed.ncbi.nlm.nih.gov/37786882/
Johannesdottir SA, Chang ET, Mehnert F, Schmidt M, Olesen AB, Sorensen HT. Nonsteroidal anti-inflammatory drugs and the risk of skin cancer: a population-based case-control study. Cancer. Oct 1 2012;118(19):4768-4776.
Johnson, D. B., and Sosman, J. A. Update on the targeted therapy of melanoma. Curr Treat Options Oncol. 2013;14(2):280–92.
Jones FI, Ramachandran S, Lear J, Smith A, Bowers B, Ollier WE, . . . Strange RC. The melanocyte stimulating hormone receptor polymorphism: association of the V92M and A294H alleles with basal cell carcinoma. Clinica chimica acta; international journal of clinical chemistry. Apr 1999;282(1-2):125-134.
Joosse, A., Koomen, E. R., Casparie, M. K., Herings, R. M. C., Guchelaar, H.-J., and Nijsten, T. Non-steroidal anti-inflammatory drugs and melanoma risk: large Dutch population-based case-control study. J. Invest. Dermatol. 2009;129(11):2620–7.
Jou, P. C., Feldman, R. J., and Tomecki, K. J. UV protection and sunscreens: what to tell patients. Cleve Clin J Med. 2012;79(6):427–36.
Jung, M., Bu, S. Y., Tak, K.-H., Park, J.-E., and Kim, E. Anticarcinogenic effect of quercetin by inhibition of insulin-like growth factor (IGF)-1 signaling in mouse skin cancer. Nutr Res Pract. 2013;7(6):439–45.
Kanasuo E, Siiskonen H, Haimakainen S, Komulainen J, Harvima IT. Regular use of vitamin D supplement is associated with fewer melanoma cases compared to non-use: a cross-sectional study in 498 adult subjects at risk of skin cancers. Melanoma research. Dec 28 2022;doi:10.1097/cmr.0000000000000870. https://pubmed.ncbi.nlm.nih.gov/36580363/
Katiyar, S. K. Green tea prevents non-melanoma skin cancer by enhancing DNA repair. Arch. Biochem. Biophys. 2011;508(2):152–8.
Katiyar, S. K. Treatment of silymarin, a plant flavonoid, prevents ultraviolet light-induced immune suppression and oxidative stress in mouse skin. Int. J. Oncol. 2002;21(6):1213–22.
Katiyar, S. K., Korman, N. J., Mukhtar, H., and Agarwal, R. Protective effects of silymarin against photocarcinogenesis in a mouse skin model. J Natl Cancer Inst. 1997;89(8):556–66.
Katiyar, S., Elmets, C. A., and Katiyar, S. K. Green tea and skin cancer: photoimmunology, angiogenesis and DNA repair. J Nutr Biochem. 2007;18(5):287–96.
Kaushik, G., Ramalingam, S., Subramaniam, D. Honokiol induces cytotoxic and cytostatic effects in malignant melanoma cancer cells. Am. J. Surg. 2012;204(6):868–73.
Khalesi, M., Whiteman, D. C., Tran, B., Kimlin, M. G., Olsen, C. M., and Neale, R. E. A meta-analysis of pigmentary characteristics, sun sensitivity, freckling and melanocytic nevi and risk of basal cell carcinoma of the skin. Cancer Epidemiol. 2013;37(5):534–43.
Kim, R. H., and Armstrong, A. W. Nonmelanoma skin cancer. Dermatol Clin. 2012;30(1):125–39–ix.
Kimura, Y., and Sumiyoshi, M. French maritime pine bark (Pinus maritima Lam.) extract (Flavangenol) prevents chronic UVB radiation-induced skin damage and carcinogenesis in melanin-possessing hairless mice. Photochem. Photobiol. 2010;86(4):955–63.
Kudchadkar, R. R., Gonzalez, R., and Lewis, K. New targeted therapies in melanoma. Cancer Control. 2013;20(4):282–8.
Kurian, R. R., Di Palma, S., and Barrett, A. W. Basal Cell Carcinoma Metastatic to Parotid Gland. Head Neck Pathol. 2013. [Epub ahead of print]
Larson, B., Abeytunge, S., Seltzer, E., Rajadhyaksha, M., and Nehal, K. Detection of skin cancer margins in Mohs excisions with high-speed strip mosaicing confocal microscopy: a feasibility study. Br. J. Dermatol. 2013;169(4):922–6.
Latruffe N, Rifler JP. Bioactive polyphenols from grapes and wine emphasized with resveratrol. Current pharmaceutical design. 2013;19(34):6053-6063.
Lazareth, V. Management of non-melanoma skin cancer. Semin Oncol Nurs. 2013;29(3):182–94.
Lee KC, Weinstock MA. Melanoma is up: are we up to the challenge? J Invest Dermatol. 2009;129(7):1604-6.
Leiter U, Garbe C. Epidemiology of melanoma and nonmelanoma skin cancer--the role of sunlight. Advances in experimental medicine and biology. 2008;624:89-103.
Lin JS, Eder M, Weinmann S, et al. Behavioral Counseling to Prevent Skin Cancer: Systematic Evidence Review to Update the 2003 U.S. Preventive Services Task Force Recommendation [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2011 Feb. (Evidence Syntheses, No. 82.) 1, Introduction. Available at: http://www.ncbi.nlm.nih.gov/books/NKB53511/.
Liu, L. S., and Colegio, O. R. Molecularly targeted therapies for melanoma. Int. J. Dermatol. 2013;52(5):523–30.
Lober CW, Larbig GG. Microcystic adnexal carcinoma (sclerosing sweat duct carcinoma). Southern medical journal. Feb 1994;87(2):259-262.
Lotze MT, Matory YL, Ettinghausen SE, Rayner AA, Sharrow SO, Seipp CA, . . . Rosenberg SA. In vivo administration of purified human interleukin 2. II. Half life, immunologic effects, and expansion of peripheral lymphoid cells in vivo with recombinant IL 2. Journal of immunology (Baltimore, Md. : 1950). Oct 1985;135(4):2865-2875.
Ma, C., and Armstrong, A. W. Severe adverse events from the treatment of advanced melanoma: a systematic review of severe side effects associated with ipilimumab, vemurafenib, interferon alfa-2b, dacarbazine and interleukin-2. J Dermatolog Treat. 2014;25(5):401–8.
MacFarlane D. Skin Cancer Management: A Practical Approach. SpringerLink: Bücher. Springer, 2010.
Mainville L, Smilga AS, Fortin PR. Effect of Nicotinamide in Skin Cancer and Actinic Keratoses Chemoprophylaxis, and Adverse Effects Related to Nicotinamide: A Systematic Review and Meta-Analysis. Journal of cutaneous medicine and surgery. May-Jun 2022;26(3):297-308. doi:10.1177/12034754221078201. https://pubmed.ncbi.nlm.nih.gov/35134311/
Man'cheva, T. A., Demidov, D. V., Plotnikova, N. A., Kharitonova, T. V., Pashkevich, I. V., and Anisimov, V. N. Melatonin and metformin inhibit skin carcinogenesis and lipid peroxidation induced by benz(a)pyrene in female mice. Bull. Exp. Biol. Med. 2011;151(3):363–5.
Mannal, P. W., Schneider, J., Tangada, A., McDonald, D., and McFadden, D. W. Honokiol produces anti-neoplastic effects on melanoma cells in vitro. J Surg Oncol. 2011;104(3):260–4.
Marieb, E., and Hoehn, K. Human anatomy & physiology. 8 ed. 2009. San Francisco: Pearson Benjamin Cummings.
Martin GM, Stockfleth E. Diclofenac sodium 3% gel for the management of actinic keratosis: 10+ years of cumulative evidence of efficacy and safety. Journal of drugs in dermatology : JDD. May 2012;11(5):600-608.
Martin-Gorgojo A, Gilaberte Y, Nagore E. Vitamin D and Skin Cancer: An Epidemiological, Patient-Centered Update and Review. Nutrients. Nov 28 2021;13(12)doi:10.3390/nu13124292. https://pubmed.ncbi.nlm.nih.gov/34959844/
Miller, A. J., and Mihm, M. C., Jr. Melanoma. N. Engl. J. Med. 2006;355(1):51–65.
Mueller T, Voigt W. Fermented wheat germ extract--nutritional supplement or anticancer drug? Nutrition journal. 2011;10:89.
Nazarali S, Kuzel P. Vitamin B Derivative (Nicotinamide)Appears to Reduce Skin Cancer Risk. Skin Therapy Lett. Sep 2017;22(5):1-4. https://pubmed.ncbi.nlm.nih.gov/28888216/
NCI. National Cancer Institute. Cancer Staging Fact Sheet - National Cancer Institute. cancer.gov. 2013; Available at: http://www.cancer.gov/cancertopics/factsheet/detection/staging [Accessed November 1, 2013].
NCI. National Cancer Institute. Cancer Topical page. Cancer Drug Information. FDA Approval for Trametinib. Available at: http://www.cancer.gov/cancertopics/druginfo/fda-trametinib. 1/16/2014c. Accessed 3/31/2014.
NCI. National Cancer Institute. Melanoma Treatment (PDQ®). Stages of Melanoma. Available at: http://www.cancer.gov/cancertopics/pdq/treatment/melanoma/Patient/page2#Keypoint12. 2/7/2014b. Accessed 3/31/2014.
NCI. National Cancer Institute. SEER – Surveillance, Epidemiology, and End Results Program. SEER Stat Facts Sheet: Melanoma of the Skin. Available at: http://seer.cancer.gov/statfacts/html/melan.html. Accessed 3/28/2014a.
Ndiaye, M., Philippe, C., Mukhtar, H., and Ahmad, N. The grape antioxidant resveratrol for skin disorders: promise, prospects, and challenges. Arch. Biochem. Biophys. 2011;508(2):164–70.
Newton-Bishop JA, Beswick S, Randerson-Moor J, et al. Serum 25-hydroxyvitamin D3 levels are associated with breslow thickness at presentation and survival from melanoma. J Clin Oncol. Nov 10 2009;27(32):5439-44. doi:10.1200/jco.2009.22.1135. https://pubmed.ncbi.nlm.nih.gov/19770375/
Nguyen, T. H., and Ho, D. Q.-D. Nonmelanoma skin cancer. Curr Treat Options Oncol. 2002;3(3):193–203.
Ogbourne SM, Suhrbier A, Jones B, Cozzi SJ, Boyle GM, Morris M, . . . Parsons PG. Antitumor activity of 3-ingenyl angelate: plasma membrane and mitochondrial disruption and necrotic cell death. Cancer research. Apr 15 2004;64(8):2833-2839.
Osmond, G. W., Augustine, C. K., Zipfel, P. A., Padussis, J., and Tyler, D. S. Enhancing melanoma treatment with resveratrol. J. Surg. Res. 2012;172(1):109–15.
Pacifico A, Leone G. Role of p53 and CDKN2A inactivation in human squamous cell carcinomas. Journal of biomedicine & biotechnology. 2007;2007(3):43418.
Paoli, J., Daryoni, S., Wennberg, A.-M. 5-year recurrence rates of Mohs micrographic surgery for aggressive and recurrent facial basal cell carcinoma. Acta Derm. Venereol. 2011;91(6):689–93.
Patil CA, Kirshnamoorthi H, Ellis DL, van Leeuwen TG, Mahadevan-Jansen A. A clinical instrument for combined raman spectroscopy-optical coherence tomography of skin cancers. Lasers in surgery and medicine. Feb 2011;43(2):143-151.
Pavlou, P., Rallis, M., Deliconstantinos, G., Papaioannou, G., and Grando, S. A. In-vivo data on the influence of tobacco smoke and UV light on murine skin. Toxicol Ind Health. 2009;25(4-5):231–9.
Payette, M. J., Whalen, J., and Grant-Kels, J. M. Nutrition and nonmelanoma skin cancers. Clin. Dermatol. 2010;28(6):650–62.
Pilkington, S. M., Massey, K. A., Bennett, S. P. Randomized controlled trial of oral omega-3 PUFA in solar-simulated radiation-induced suppression of human cutaneous immune responses. American Journal of Clinical Nutrition. 2013;97(3):646–52.
Platt, D., and Raz, A. Modulation of the lung colonization of B16-F1 melanoma cells by citrus pectin. J Natl Cancer Inst. 1992;84(6):438–42.
Ponten F, Ren Z, Nister M, Westermark B, Ponten J. Epithelial-stromal interactions in basal cell cancer: the PDGF system. The Journal of investigative dermatology. Mar 1994;102(3):304-309.
Quigley, E. A., and Halpern, A. C. Microinvasive melanoma: cutaneous pharmacotherapeutic approaches. Am J Clin Dermatol. 2013;14(2):125–37.
Ramirez Boscá, A., Zapater, P., Betlloch, I. Polypodium leucotomos extract in atopic dermatitis: a randomized, double-blind, placebo-controlled, multicenter trial. Actas Dermosifiliogr. 2012;103(7):599–607.
Ramsay, J. R., Suhrbier, A., Aylward, J. H. The sap from Euphorbia peplus is effective against human nonmelanoma skin cancers. Br. J. Dermatol. 2011;164(3):633–6.
Rao, B. K., and Ahn, C. S. Dermatoscopy for melanoma and pigmented lesions. Dermatol Clin. 2012;30(3):413–34.
Rass, K., and Reichrath, J. UV damage and DNA repair in malignant melanoma and nonmelanoma skin cancer. Adv Exp Med Biol. 2008;624:162–78.
Rehal, B., Merin, M. R., and Barr, K. Metastatic eccrine porocarcinoma. Cutis. 2013;92(2):67–70.
Roberts, W. E. Skin type classification systems old and new. Dermatol Clin. 2009;27(4):529–33–viii.
Robinson, J. K., and Dahiya, M. Basal cell carcinoma with pulmonary and lymph node metastasis causing death. Arch Dermatol. 2003;139(5):643.
Rogers, H. W., Weinstock, M. A., Harris, A. R. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch Dermatol. 2010;146(3):283–7.
Rosen, C. J., Adams, J. S., Bikle, D. D. The nonskeletal effects of vitamin D: an Endocrine Society scientific statement. Endocr. Rev. 2012;33(3):456–92.
Rowe, D. E., Carroll, R. J., and Day, C. L. Long-term recurrence rates in previously untreated (primary) basal cell carcinoma: implications for patient follow-up. J Dermatol Surg Oncol. 1989;15(3):315–28.
Rundhaug, J. E., Mikulec, C., Pavone, A., and Fischer, S. M. A role for cyclooxygenase-2 in ultraviolet light-induced skin carcinogenesis. Mol Carcinog. 2007;46(8):692–8.
Rusciani, L., Proietti, I., Paradisi, A. Recombinant interferon alpha-2b and coenzyme Q10 as a postsurgical adjuvant therapy for melanoma: a 3-year trial with recombinant interferon-alpha and 5-year follow-up. Melanoma Res. 2007;17(3):177–83.
Rusciani, L., Proietti, I., Rusciani, A. Low plasma coenzyme Q10 levels as an independent prognostic factor for melanoma progression. J. Am. Acad. Dermatol. 2006;54(2):234–41.
Saija A, Tomaino A, Lo Cascio R, Rapisarda P, Dederen JC. In vitro antioxidant activity and in vivo photoprotective effect of a red orange extract. International journal of cosmetic science. Dec 1998;20(6):331-342.
Sak, K. Site-Specific Anticancer Effects of Dietary Flavonoid Quercetin. Nutr Cancer. 2014;66(2):177-93.
Sánchez, G., and Nova, J. Risk factors for squamous cell carcinoma, a study by the National Dermatology Centre of Colombia. Actas Dermosifiliogr. 2013;104(8):672–8.
Schellack G, Harirari P, Schellack N. Vitamin B-complex deficiency, supplementation and management. SA Pharmaceutical Journal. 2019;86(3):23-29. doi:doi:10.10520/EJC-16fb37f64d. https://journals.co.za/doi/abs/10.10520/EJC-16fb37f64d
Scheuer, C., Pommergaard, H.-C., Rosenberg, J., and Gögenur, I. Melatonin's protective effect against UV radiation: a systematic review of clinical and experimental studies. Photodermatol Photoimmunol Photomed. 2013. [Epub ahead of print]
Sebastien TS, Nelson BR, Lowe L, Baker S, Johnson TM. Microcystic adnexal carcinoma. Journal of the American Academy of Dermatology. Nov 1993;29(5 Pt 2):840-845.
Sharma, S. D., Meeran, S. M., and Katiyar, S. K. Dietary grape seed proanthocyanidins inhibit UVB-induced oxidative stress and activation of mitogen-activated protein kinases and nuclear factor-kappaB signaling in in vivo SKH-1 hairless mice. Mol. Cancer Ther. 2007;6(3):995–1005.
Shigama K, Nakaya A, Wakame K, Nishioka H, Fujii H. Alleviating effect of active hexose correlated compound (AHCC) for anticancer drug-induced side effects in non-tumor-bearing mice. Journal of experimental therapeutics & oncology. 2009;8(1):43-51.
Shimizu, I., Cruz, A., Chang, K. H., and Dufresne, R. G. Treatment of squamous cell carcinoma in situ: a review. Dermatol Surg. 2011;37(10):1394–411.
Siegel, R., Naishadham, D., and Jemal, A. Cancer statistics, 2013. CA: A Cancer Journal for Clinicians. 2013;63(1):11–30.
Siller, G., Rosen, R., Freeman, M., Welburn, P., Katsamas, J., and Ogbourne, S. M. PEP005 (ingenol mebutate) gel for the topical treatment of superficial basal cell carcinoma: results of a randomized phase IIa trial. Australas. J. Dermatol. 2010;51(2):99–105.
Sime, S., and Reeve, V. E. Protection from inflammation, immunosuppression and carcinogenesis induced by UV radiation in mice by topical Pycnogenol. Photochem. Photobiol. 2004;79(2):193–8.
Singh, R., Akhtar, N., and Haqqi, T. M. Green tea polyphenol epigallocatechin-3-gallate: inflammation and arthritis. [corrected]. Life Sci. 2010;86(25-26):907–18.
Sivapirabu G, Yiasemides E, Halliday GM, Park J, Damian DL. Topical nicotinamide modulates cellular energy metabolism and provides broad-spectrum protection against ultraviolet radiation-induced immunosuppression in humans. Br J Dermatol. Dec 2009;161(6):1357-64. doi:10.1111/j.1365-2133.2009.09244.x. https://pubmed.ncbi.nlm.nih.gov/19804594/
Skivka, L., Susak, Y., Trompak, O. The effect of monotherapy and combined therapy with NSC-631570 (Ukrain) on growth of low- and high-metastasizing B16 melanoma in mice. Journal of Oncology Pharmacy Practice. 2011;17(4):339–49.
Sonavane, K., Phillips, J., Ekshyyan, O. Topical curcumin-based cream is equivalent to dietary curcumin in a skin cancer model. J Skin Cancer. 2012;2012:147863.
Song Y, Lu H, Cheng Y. To identify the association between dietary vitamin D intake and serum levels and risk or prognostic factors for melanoma-systematic review and meta-analysis. BMJ open. Aug 26 2022;12(8):e052442. doi:10.1136/bmjopen-2021-052442. https://pubmed.ncbi.nlm.nih.gov/36028262/
Spierings EL, Fujii H, Sun B, Walshe T. A Phase I study of the safety of the nutritional supplement, active hexose correlated compound, AHCC, in healthy volunteers. Journal of nutritional science and vitaminology. Dec 2007;53(6):536-539.
Srihari S, Garg N, Iyer S. Cutaneous Manifestations of Nutritional Deficiencies. Indian Journal of Postgraduate Dermatology. 3doi:10.25259/IJPGD_188_2024. https://doi.org/10.25259/IJPGD_188_2024
Stabuc, B., and Benedicic, D. Ukrain with chemotherapy in malignant melanoma (case report). Drugs Exp Clin Res. 1996;22(3-5):231–3.
Stoeckli, S. Sentinel lymph node evaluation in squamous cell carcinoma of the head and neck. Otolaryngology - Head and Neck Surgery. 2001;125(3):221–6.
Stroud, M. L., Stilgoe, S., Stott, V. E., Alhabian, O., and Salman, K. Vitamin D-a review. Australian family physician. 2008;37(12):1002–5.
Sugimoto, S., Ishii, Y., Izawa, N. Photoprotective effects of Bifidobacterium breve supplementation against skin damage induced by ultraviolet irradiation in hairless mice. Photodermatol Photoimmunol Photomed. 2012;28(6):312–9.
Surjana D, Halliday GM, Martin AJ, Moloney FJ, Damian DL. Oral nicotinamide reduces actinic keratoses in phase II double-blinded randomized controlled trials. J Invest Dermatol. May 2012;132(5):1497-500. doi:10.1038/jid.2011.459. https://pubmed.ncbi.nlm.nih.gov/22297641/
Sutedja EK, Arianto TR, Lesmana R, Suwarsa O, Setiabudiawan B. The Chemoprotective Role of Vitamin D in Skin Cancer: A Systematic Review. Cancer management and research. 2022;14:3551-3565. doi:10.2147/cmar.S389591. https://pubmed.ncbi.nlm.nih.gov/36583029/
Tarhini, A. A., Gogas, H., and Kirkwood, J. M. IFN-α in the treatment of melanoma. J Immunol. 2012;189(8):3789–93.
Thanos SM, Halliday GM, Damian DL. Nicotinamide reduces photodynamic therapy-induced immunosuppression in humans. Br J Dermatol. Sep 2012;167(3):631-6. doi:10.1111/j.1365-2133.2012.11109.x. https://pubmed.ncbi.nlm.nih.gov/22709272/
Thieu, K., Ruiz, M. E., and Owens, D. M. Cells of origin and tumor-initiating cells for nonmelanoma skin cancers. Cancer Lett. 2013;338(1):82–8.
Tsai, M.-L., Lai, C.-S., Chang, Y.-H., Chen, W.-J., Ho, C.-T., and Pan, M.-H. Pterostilbene, a natural analogue of resveratrol, potently inhibits 7,12-dimethylbenz[a]anthracene (DMBA)/12-O-tetradecanoylphorbol-13-acetate (TPA)-induced mouse skin carcinogenesis. Food Funct. 2012;3(11):1185–94.
Umar M, Sastry KS, Al Ali F, Al-Khulaifi M, Wang E, Chouchane AI. Vitamin D and the Pathophysiology of Inflammatory Skin Diseases. Skin Pharmacol Physiol. 2018;31(2):74-86. doi:10.1159/000485132. https://pubmed.ncbi.nlm.nih.gov/29306952/
Usatine R, Scherer JE. Cutaneous squamous cell carcinoma. First Consult. Available at: www.clinicalkey.com. Last updated 8/15/2011. Accessed 3/31/2014.
Uzarska M, Czajkowski R, Schwartz RA, Bajek A, Zegarska B, Drewa T. Chemoprevention of skin melanoma: facts and myths. Melanoma research. Dec 2013;23(6):426-433.
Vad, N. M., Yount, G., and Moridani, M. Y. Biochemical mechanism of acetylsalicylic acid (Aspirin) selective toxicity toward melanoma cell lines. Melanoma Res. 2008;18(6):386–99.
Vaid, M., and Katiyar, S. K. Molecular mechanisms of inhibition of photocarcinogenesis by silymarin, a phytochemical from milk thistle (Silybum marianum L. Gaertn.) (Review). Int. J. Oncol. 2010;36(5):1053–60.
Vaid, M., Sharma, S. D., and Katiyar, S. K. Proanthocyanidins inhibit photocarcinogenesis through enhancement of DNA repair and xeroderma pigmentosum group A-dependent mechanism. Cancer Prevention Research. 2010;3(12):1621–9.
Vaid, M., Singh, T., Li, A. Proanthocyanidins inhibit UV-induced immunosuppression through IL-12-dependent stimulation of CD8+ effector T cells and inactivation of CD4+ T cells. Cancer Prevention Research. 2011;4(2):238–47.
Valverde P, Healy E, Sikkink S, Haldane F, Thody AJ, Carothers A, . . . Rees JL. The Asp84Glu variant of the melanocortin 1 receptor (MC1R) is associated with melanoma. Human molecular genetics. Oct 1996;5(10):1663-1666.
van der Vliet HJ, Koon HB, Yue SC, Uzunparmak B, Seery V, Gavin MA, . . . Exley MA. Effects of the administration of high-dose interleukin-2 on immunoregulatory cell subsets in patients with advanced melanoma and renal cell cancer. Clinical cancer research : an official journal of the American Association for Cancer Research. Apr 1 2007;13(7):2100-2108.
van Ginkel, P. R., Darjatmoko, S. R., Sareen, D., Resveratrol inhibits uveal melanoma tumor growth via early mitochondrial dysfunction. Invest Ophthalmol Vis Sci. 2008;49(4):1299–306.
Warner, C. L., and Cockerell, C. J. The new seventh edition American Joint Committee on Cancer staging of cutaneous non-melanoma skin cancer: a critical review. Am J Clin Dermatol. 2011;12(3):147–54.
Weide B, Derhovanessian E, Pflugfelder A, Eigentler TK, Radny P, Zelba H, . . . Garbe C. High response rate after intratumoral treatment with interleukin-2: results from a phase 2 study in 51 patients with metastasized melanoma. Cancer. Sep 1 2010;116(17):4139-4146.
Weill, F. S., Cela, E. M., Paz, M. L., Ferrari, A., Leoni, J., and González Maglio, D. H. Lipoteichoic acid from Lactobacillus rhamnosus GG as an oral photoprotective agent against UV-induced carcinogenesis. Br J Nutr. 2012;:1–10.
Welch HG, Woloshin S, Schwartz LM. Skin biopsy rates and incidence of melanoma: population based ecological study. BMJ. 2005;331:481-4.
Wells, R., Gutkowicz-Krusin, D., Veledar, E., Toledano, A., and Chen, S. C. Comparison of diagnostic and management sensitivity to melanoma between dermatologists and MelaFind: a pilot study. Arch Dermatol. 2012;148(9):1083–4.
Whiteman DC, Stickley M, Watt P, Hughes MC, Davis MB, Green AC. Anatomic site, sun exposure, and risk of cutaneous melanoma. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. Jul 1 2006;24(19):3172-3177.
Wikimedia Commons. File: Layers of the skin. Available at: http://commons.wikimedia.org/wiki/File:Layers_of_the_skin.jpg. Last updated 2/10/2013. Accessed 2/3/2014.
Wiley J. Perspectives in Dermatopathology. The ‘epidemic’ of melanoma between under- and overdiagnosis. J Cutan Pathol. 2012;39:9-16.
Wright CJ, McCormack PL. Trametinib: first global approval. Drugs. Jul 2013;73(11):1245-1254.
Yang, C. S., Wang, H., Li, G. X., Yang, Z., Guan, F., and Jin, H. Cancer prevention by tea: Evidence from laboratory studies. Pharmacol. Res. 2011;64(2):113–22.
Yao H, Ng SS, Huo LF, Chow BK, Shen Z, Yang M, . . . Lin MC. Effective melanoma immunotherapy with interleukin-2 delivered by a novel polymeric nanoparticle. Molecular cancer therapeutics. Jun 2011;10(6):1082-1092.
Yuan, X.-Y., Liu, W., Hao, J.-C., Gu, W.-J., and Zhao, Y.-S. Topical grape seed proanthocyandin extract reduces sunburn cells and mutant p53 positive epidermal cell formation, and prevents depletion of Langerhans cells in an acute sunburn model. Photomed Laser Surg. 2012;30(1):20–5.
Zattra, E., Coleman, C., Arad, S. Polypodium leucotomos extract decreases UV-induced Cox-2 expression and inflammation, enhances DNA repair, and decreases mutagenesis in hairless mice. Am. J. Pathol. 2009;175(5):1952–61.
Zhan H, Zheng H. The role of topical cyclo-oxygenase-2 inhibitors in skin cancer: treatment and prevention. American journal of clinical dermatology. 2007;8(4):195-200.
Zhang SL, Chen TS, Ma CY, et al. Effect of vitamin B supplementation on cancer incidence, death due to cancer, and total mortality: A PRISMA-compliant cumulative meta-analysis of randomized controlled trials. Medicine. Aug 2016;95(31):e3485. doi:10.1097/md.0000000000003485. https://pmc.ncbi.nlm.nih.gov/articles/PMC4979769/
