
High Blood Pressure (Hypertension)
High Blood Pressure (Hypertension)
Last Section Update: 08/2025
Contributor(s): Maureen Williams, ND; Debra Gordon, MS; Shayna Sandhaus, PhD; Stephen Tapanes, PhD
Table of Contents
- Overview
- Introduction
- Growing Body of Evidence for Benefits of Lower Blood Pressure Targets
- Life Extension’s Protocol for Optimal Blood Pressure Control
- Diligent Monitoring to Prevent Treatment-Related Adverse Events
- Blood Pressure Is Not the Only Contributor to Heart Disease
- Summary
- Update History
- References
1 Overview
Summary and Quick Facts for High Blood Pressure
- High blood pressure or hypertension, if left untreated, can lead to serious health conditions. Recent studies support a target blood pressure for most people of 120/80 mm Hg, with an optimal level of 115/75 mm Hg for many people.
- This protocol outlines a systematic approach to blood pressure control that combines natural interventions and lifestyle strategies with prescription antihypertensive drugs.
- When coupled with consistent at-home blood pressure monitoring and periodic blood testing to assess kidney function, this approach can help achieve a target blood pressure of 115/75 mm Hg while minimizing risk.
- Quercetin is found in many fruits and vegetables. Multiple cases showed that supplementation with quercetin lowered blood pressure and reduced risk of cardiovascular disease.
High Blood Pressure
Blood pressure is the measurement of systolic pressure (maximum pressure during one heartbeat) over diastolic pressure (minimum pressure between heartbeats). After the landmark SPRINT trial, published in 2015, found that lower blood pressure significantly reduced the risk of cardiovascular outcomes and death, the threshold for high blood pressure (hypertension) was lowered from 140/90 mm Hg to 130/80 mm Hg.
Natural interventions such as quercetin, coenzyme Q10, magnesium, and bioactive whey peptides, along with dietary and lifestyle changes, can help lower blood pressure.
What is Healthy Blood Pressure?
Healthy blood pressure is less than 120/80 mm Hg, and Life Extension recommends an optimal target of 115/75 mm Hg. Observational evidence suggests the risk of cardiovascular disease doubles for each increment of 20 mm Hg systolic and 10 mm Hg diastolic above 115/75 mm Hg.
Adding one or more blood pressure-lowering medications may be appropriate if dietary and lifestyle changes alone are insufficient to bring blood pressure into the healthy range.
People with diabetes, pre-existing cardiovascular or kidney disease, or who are over age 80 should be particularly careful with blood pressure-lowering medication. Always consult a doctor before implementing any changes to your daily regimen.
Note: High blood pressure is not the only risk factor for cardiovascular events. People interested in reducing their cardiovascular risks should follow these blood pressure-lowering strategies, and also read the Life Extension Magazine article titled “How to Circumvent 17 Independent Heart Attack Risk Factors.”
Dietary and Lifestyle Changes–The First Step in Controlling Blood Pressure
For otherwise healthy people with blood pressure above 120/80 mm Hg, dietary and lifestyle changes are often sufficient to bring blood pressure into a healthy range.
- Follow a healthy diet (eg, Dietary Approaches to Stop Hypertension [DASH] and Mediterranean diets)
- Reduce caloric intake
- Reduce sodium intake and consume sufficient dietary potassium
- Weight loss
- Regular exercise
- Limit alcohol consumption
- Limit use of non-steroidal anti-inflammatory drugs (NSAIDs)
- Get tested for sleep apnea, as it can increase the risk of high blood pressure
- Manage stress
Note: Using an at-home blood pressure monitor to regularly monitor blood pressure throughout the day is an effective way to track effectiveness of dietary and lifestyle changes and other interventions for blood pressure.
Medications to Help Lower Blood Pressure
If diet and lifestyle changes alone do not sufficiently lower blood pressure, blood pressure medication may be appropriate. Consult a qualified healthcare provider before beginning medical treatment for high blood pressure.
- First-line recommendation
- Angiotensin II receptor blockers (particularly telmisartan)
- Angiotensin-converting enzyme inhibitors
- Second-line recommendation
- Thiazide diuretics
- Third-line recommendation
- Calcium channel blockers
Simultaneous use of more than one blood pressure medication, called combination therapy, may be appropriate for people in whom a single drug fails to control blood pressure.
Note: Beta-blockers are generally not recommended as first-line therapy for blood pressure reduction except in those with an indication for beta-blocker therapy such as recent heart attack or heart failure.
What Natural Interventions May Help Lower Blood Pressure?
- Quercetin. Quercetin, a plant flavonoid shown to effectively lower blood pressure, is linked to lower cardiovascular risk. It is believed to act as an angiotensin receptor blocker.
- Myricetin. Myricetin is a flavonoid like quercetin, which also appears to act as an angiotensin receptor blocker. Myricetin may lower blood pressure in diabetics with hypertension.
- Stevioside. Stevioside, an extract from the Stevia plant commonly used as a sweetener in the United States, can also act as a calcium channel blocker to reduce blood pressure.
- Melatonin. Melatonin helps relax blood vessels and inhibit the sympathetic nervous system, both of which can reduce blood pressure. A meta-analysis indicated melatonin supplementation was effective at lowering nocturnal blood pressure.
- Fish oil. Fish oils and omega-3 fatty acids are associated with lower cardiovascular risk. Fish oil supplements have been shown to lower blood pressure in numerous clinical studies.
- Coenzyme Q10. Coenzyme Q10 has strong antihypertensive and cardioprotective effects, which may be due to its protection of various vasodilators.
- Magnesium. Magnesium supplementation can lower blood pressure in people with hypertension. It can improve endothelial function and act as a calcium channel blocker and vasodilator.
- Other natural interventions include whey protein peptides, grape polyphenols, pomegranate, olive leaf extract, celery seed extract, and hesperidin.
2 Introduction
Life Extension has long warned that blood pressure exceeding 115/75 mm Hg can be deadly.1-4 A large study published in 2015 helped corroborate our contention, but also pointed out some important caveats that aging individuals should be aware of before embarking on an aggressive blood-pressure-lowering regimen.
The SPRINT trial results were published November 9th, 2015 in the New England Journal of Medicine.5
This randomized controlled trial that enrolled over 9,300 subjects showed that non-diabetics at increased cardiovascular risk can substantially reduce their risk of cardiovascular events and death by lowering their blood pressure to levels below what were clinical standards at that time.
One group of SPRINT trial participants was intensively treated with blood-pressure-lowering medications to a target systolic blood pressure of less than 120 mm Hg. The other group received medications with a treatment goal of achieving systolic blood pressure of less than 140 mm Hg. The intensively treated subjects took, on average, one additional blood pressure medication compared with the standard treatment group.
The trial was scheduled to last for 5 years, but was stopped after a median of only 3.3 years of follow up because subjects who underwent more intensive blood pressure lowering had a dramatic 25% risk reduction for a composite of cardiovascular outcomes and a 27% lower risk of death from any cause compared with the standard-treatment group.
But while the intensive treatment led to better cardiovascular and overall outcomes, it did increase the risk of kidney problems. As readers of Life Extension publications are aware, aggressive blood-pressure-lowering treatment has the potential to cause side effects, such as impaired kidney function.6,7 We have long recommended regular blood testing to assess kidney function, as well as overall health; this strategy is particularly important for people who undergo aggressive blood-pressure-lowering treatment. By periodically testing one’s glomerular filtration rate, creatinine, and cystatin-C, declining kidney function can be identified in time to initiate preventive measures.
Subsequent to publication of the SPRINT trial results, major shifts have occurred in the conventional interpretation of blood pressure levels. In late 2017, a consortium of prominent medical organizations focused on heart and vascular health released updated guidelines overhauling the definition of high blood pressure.8 The most significant change was that the threshold for overt Stage 1 hypertension dropped from 140/90 mm Hg to 130/80 mm Hg. This change caused millions of additional Americans to suddenly be classified as having high blood pressure.
While we at Life Extension applaud the new guidelines for increasing awareness of the dangers of blood pressure levels previously described only as “prehypertension,” it is disconcerting that it took these major medical organizations this long to lower blood pressure thresholds. Life Extension has advocated a target blood pressure for most people of 120/80 mm Hg for many years, with an optimal level of 115/75 mm Hg for many people.
This protocol outlines a systematic approach to blood pressure control that combines natural interventions—such as quercetin, myricetin and myricitrin, stevioside, and melatonin—and lifestyle strategies with prescription antihypertensive drugs. When coupled with consistent at-home blood pressure monitoring and periodic blood testing to assess kidney function, this approach can help achieve a target blood pressure of 115/75 mm Hg while minimizing risk.
3 Growing Body of Evidence for Benefits of Lower Blood Pressure Targets
The table below shows the blood pressure categorization thresholds based on the 2017 consortium guidelines.8

We at Life Extension agree that “normal” blood pressure levels are below 120/80—with an optimal target being 115/75 for most adults up to age 80. Concerningly, up to half of adults worldwide have blood pressure levels ranging from 120/80 to 139/89 mm Hg.9
A recent meta-analysis of observational studies showed that blood pressure in this range correlates with a nearly 20% increased risk of declining kidney function; the association was especially strong in older individuals.10
In 2006, an analysis of 8,960 middle-aged adults in the Atherosclerosis Risk in Communities (ARIC) study demonstrated that individuals with blood pressure levels ranging from 120/80 to 139/89 mm Hg had about double the risk of cardiovascular disease compared with people whose blood pressure was below 120/80 mm Hg.1
A meta-analysis of 61 prospective trials evaluated the relationship between blood pressure and cardiovascular-related mortality rates for one million individuals with no known history of cardiovascular disease. Researchers noted that at least down to 115/75 mm Hg, there is no threshold where lower blood pressure is not directly associated with lower mortality rates due to cardiovascular events. In addition, among people 40 to 69 years old, each 20 mm Hg difference in systolic blood pressure was associated with at least a twofold difference in overall mortality due to stroke, ischemic heart disease, or other cardiovascular events.2
With the publication of the SPRINT trial results, the evidence base supporting more aggressive blood pressure treatment targets in select populations continues to broaden. In late 2016, Life Extension Magazine published a comprehensive analysis of the SPRINT trial and an overview of the shortcomings of conventional views regarding blood pressure in an article titled “Lower Blood Pressure Empowers Longer Life.”
The table below shows the blood pressure categorization thresholds based on the 2017 consortium guidelines.8

We at Life Extension agree that “normal” blood pressure levels are below 120/80—with an optimal target being 115/75 for most adults up to age 80. This becomes concerning when considering the staggering prevalence of higher-than-normal blood pressure: up to half of adults worldwide have blood pressure levels ranging from 120/80 to 139/89 mm Hg.9
A recent meta-analysis of observational studies showed that blood pressure in this range correlates with a nearly 20% increased risk of declining kidney function; the association was especially strong in older individuals.10
In 2006, an analysis of 8,960 middle-aged adults in the Atherosclerosis Risk in Communities (ARIC) study demonstrated that individuals with blood pressure levels ranging from 120/80 to 139/89 mm Hg had about double the risk of cardiovascular disease compared with people whose blood pressure was below 120/80 mm Hg.1
A meta-analysis of 61 prospective trials evaluated the relationship between blood pressure and cardiovascular-related mortality rates for one million individuals with no known history of cardiovascular disease. Researchers noted that at least down to 115/75 mm Hg, there is no threshold where lower blood pressure is not directly associated with lower mortality rates due to cardiovascular events. In addition, among people 40 to 69 years old, each 20 mm Hg difference in systolic blood pressure was associated with at least a twofold difference in overall mortality due to stroke, ischemic heart disease, or other cardiovascular events.2
With the publication of the SPRINT trial results, the evidence base supporting more aggressive blood pressure treatment targets in select populations continues to broaden. In late 2016, Life Extension Magazine published a comprehensive analysis of the SPRINT trial and an overview of the shortcomings of conventional views regarding blood pressure in an article titled “Lower Blood Pressure Empowers Longer Life.”
4 Life Extension’s Protocol for Optimal Blood Pressure Control
For decades, Life Extension has suggested that most otherwise healthy adults up to age 80 should target a blood pressure of 115/75 mm Hg. However, some adults with pre-existing cardiovascular disease, diabetes, or significant kidney disease may not tolerate aggressive blood pressure control. Caution must be used when lowering blood pressure in all older adults, paying careful attention to heart function202-205 as well as renal and cognitive function. Baseline blood pressure and health status must be taken into account during the implementation of this protocol:
- Those whose baseline blood pressure exceeds 120/80 mm Hg and are otherwise healthy should begin by implementing phase 1 of this protocol, which consists of dietary and lifestyle changes as well as targeted dietary supplementation, for a 3-month trial period. If this 3-month trial does not produce consistent blood pressure readings of approximately 115/75 mm Hg, the patient should discuss with his or her physician whether it is appropriate to add one or more blood-pressure-lowering medications to their regimen, as described in phase 2 of this protocol.
- Diabetics whose baseline blood pressure exceeds 120/80 mm Hg should implement phase 1 of this protocol (diet and lifestyle changes along with targeted natural interventions), but should carefully weigh the risks and benefits of blood-pressure-lowering drug treatment. A large clinical trial conducted by the ACCORD Study Group showed that, in diabetics, intensive antihypertensive drug treatment targeting a systolic blood pressure of less than 120 mm Hg did not reduce a composite measure of fatal and nonfatal cardiovascular events, nor of most specific cardiovascular outcomes, compared with less-intensive treatment targeting a systolic blood pressure of less than 140 mm Hg. There was, however, a reduced risk of stroke in the group receiving more intensive therapy.12 A 2016 analysis of data on more than 73,000 subjects from 49 clinical trials investigated the link between blood pressure levels and treatment outcomes in diabetics. The research team found that antihypertensive treatment in diabetics whose systolic blood pressure was under 140 mm Hg at baseline led to worse outcomes. Specifically, diabetics whose systolic blood pressure was under 140 at baseline experienced a 15% increased risk of cardiovascular death as a result of further blood-pressure-lowering treatment, with no observed benefit.14 The American Diabetes Association recommends treatment targets of <140/90 for most diabetic patients, and <130/80 for diabetics at high cardiovascular risk as long as more intensive treatment does not create an undue burden on the patient.224
- People who have significant pre-existing cardiovascular or kidney disease (including previous kidney transplant) and individuals over age 80 may not tolerate aggressive blood-pressure-lowering drug therapy, and may need a slow, steady, individualized approach.
- Blood pressure targets for those over age 80 may need to be individualized based on the overall health and functional status of the patient. There is a lack of consensus in recent guideline statements as to the ideal treatment target for older patients generally. Over-aggressive blood-pressure-lowering treatment in the elderly can increase risk of adverse events.225
- Anyone already on a physician-supervised plan to lower their blood pressure should consult their physician before adjusting their program or plan.
Phase 1: Diet and Lifestyle Changes and Targeted Natural Interventions
The initial phase of Life Extension’s systematic strategy for blood pressure control involves diet and lifestyle changes combined with targeted natural interventions. Blood pressure readings should be taken twice daily for at least three days prior to embarking on this regimen, and the average blood pressure should be recorded. After initiating the phase 1 regimen, blood pressure should be monitored and recorded twice daily. The recommendations described here should be continued indefinitely, even if medications are required to maintain blood pressure in the target range of 115/75 to 120/80 mm Hg.
For people whose blood pressure exceeds 120/80 mm Hg, these dietary and lifestyle changes and natural interventions may be tried for three months before medications are considered.
Accurate and Consistent Blood Pressure Monitoring is Important
Many people may not realize that blood pressure treatment targets depend on the method by which blood pressure is measured. It is thus important that blood pressure is measured regularly, using a consistent technique. Regular at-home self-monitoring of blood pressure is crucial for anyone concerned with cardiovascular health.19 At-home blood pressure monitoring helps confirm that any intervention you are undertaking is keeping your readings near your target goal.
In a randomized controlled trial, 136 participants with uncontrolled high blood pressure were assigned to at-home blood pressure monitoring or usual care; their medication regimen was not modified. Those who regularly monitored their blood pressure at home saw significant reductions in systolic and diastolic blood pressure compared with those who did not self-monitor. At the end of the two-month trial, 32% of the self-monitoring group had blood pressure of less than 130/80 mm Hg, while only half as many participants (16%) who did not self-monitor saw their blood pressure fall below this level.20 Several other studies have found similar benefits associated with at-home blood pressure monitoring.21-24
In routine clinical practice, the mere presence of a physician or healthcare professional can lead to a phenomenon known as “white coat syndrome,” an acute rise in blood pressure when it is measured in a healthcare setting.227 In fact, blood pressure measurements in routine clinical practice are slightly higher than measurements made by using the recommended strategy of the American Heart Association (AHA), also utilized in the SPRINT trial.228,229 The AHA recommendation is to measure blood pressure three to five minutes after quiet rest using an automated oscillometric device, have proper positioning (ie, seated with feet flat and back supported), utilize appropriate cuff size and cuff positioning, and take the mean of two or more blood pressure readings taken on two or more separate occasions.
A recent analysis of SPRINT data revealed that the mean systolic blood pressure was 7.3 mm Hg lower with the AHA recommended strategy compared with measurements taken in the traditional clinical setting. The finding that blood pressure readings taken by the method utilized in the SPRINT trial are lower than those taken in traditional clinical settings have important implications for the management of blood pressure. The SPRINT trial suggested that more aggressive blood pressure control may improve outcomes. However, the more aggressive treatment targets may not be appropriate if blood pressure measurements are being taken by traditional in-office methods. This is another example of why regular at-home blood pressure monitoring is so important. The restful environment at home may produce readings more similar to those obtained by the SPRINT investigators because they took steps to ensure participants were relaxed before taking their blood pressure by automated device.229
Dietary and Lifestyle Changes
Healthy Diet (DASH- or Mediterranean-Type Dietary Pattern). The Dietary Approaches to Stop Hypertension (DASH) diet is high in fruits, vegetables, nuts, and low-fat dairy products, and emphasizes fish and chicken over red meat. DASH is low in saturated fat, cholesterol, sugar, and refined carbohydrates,25-27 and is especially high in fiber (31 g/day) and potassium (4.7 g/day), factors that likely contribute to its efficacy for treating hypertension.28 A sodium-restricted DASH diet may be particularly effective.29 Research has shown that the DASH diet, when combined with a weekly cognitive-behavioral weight loss intervention and supervised exercise sessions 3 times per week, helped reduce systolic blood pressure by 16.1 mm Hg and reduce weight by 19.2 lbs in overweight men and women with above-normal blood pressure.30
The Mediterranean dietary pattern emphasizes similar foods, and may include moderate amounts of wine with dinner. This dietary pattern has a robust research record associating greater adherence with reduced risk of cardiovascular disease, diabetes, neurodegenerative disease, cancer, and death from all causes, as well as with lower blood pressure.31-35
A group of a bioactive plant compounds called flavan-3-ols, which are particularly high in wine, tea, chocolate, nuts, and certain fruits, have been shown in interventional studies to provide cardiovascular benefits.230,231 A diet that contains higher flavan-3-ol has been associated with blood pressure reductions similar to those achieved with the Mediterranean diet. In a cross-sectional study of over 25,000 European participants, there was a significant association between participants with higher biomarker estimated flavan-3-ol intake and significantly lower blood pressure in both men and women.232
Caloric Restriction. Caloric restriction is the long-term reduction of dietary calories while still consuming adequate amounts of essential nutrients. Caloric restriction studies have demonstrated benefit for blood pressure, diabetes, metabolic syndrome, heart disease, and possibly for the prevention of cancer. Clinical studies generally restrict calorie intake by 10–30% compared with normal energy consumption. Studies of long-term caloric restriction in many species have demonstrated that it extends life and retards age-related diseases.36,37
Caloric restriction’s benefits are mediated through multiple physiologic pathways, including AMPK activation, reduction of inflammation, improved insulin sensitivity and nitric oxide metabolism, angiotensin-blocking effects, and reversal of age- and obesity-related changes in the nervous system.38-43 Even modest weight loss induced by caloric restriction results in improvement in arterial stiffness superior to that achieved by resistance training exercise.44
During a two-year clinical caloric restriction trial in which participants consumed 1750–2100 calories per day, an impressive reduction in blood pressure ensued: systolic blood pressure dropped by 25% and diastolic blood pressure dropped by 22%.45 In a two-year randomized controlled trial of less than 25% caloric restriction, multiple cardiovascular risk parameters improved more in the caloric restriction group, including reductions in systolic and diastolic blood pressure.46 A study of non-drug interventions compared a low-salt diet, calorie restriction, and exercise for an average of 23 months in 825 subjects with high blood pressure. Caloric restriction lowered systolic blood pressure by 6.6 mm Hg, on average. Among the caloric restriction group, there was a 17% chance of a blood pressure reduction of 10 mm Hg systolic and 5 mm Hg diastolic.47
Reduce Sodium Intake and Maintain Sufficient Dietary Potassium. Higher salt (sodium chloride) intake is associated with higher blood pressure and greater risk of cardiovascular disease. Limiting salt intake lowers blood pressure29 and cardiovascular risk, and salt reduction is part of standard dietary advice for blood pressure treatment.48 Similarly, evidence from observational studies and clinical trials consistently indicates that high levels of dietary potassium are associated with lower blood pressure.49,50
Diets like the DASH diet and Mediterranean diet encourage intake of unprocessed/minimally processed vegetables, fruits, legumes, whole grains, and nuts in part to ensure a healthy balance of sodium and potassium. As part of these diets, it is also encouraged to prepare unpackaged food at home as much as possible. According to the FDA, approximately 70% of sodium in the typical American diet comes from eating packaged or preprepared foods; therefore, simply eating plant-based foods may not be sufficient to combat high blood pressure if they are highly processed.270
Sodium intake can be further minimized by replacing regular table salt with a potassium-enriched salt. Randomized-controlled trials have consistently shown that potassium-enriched salt is an effective option for improving potassium levels, while also reducing blood pressure, risk of cardiovascular disease, and mortality. A systematic review and meta-analyses of 21 randomized-controlled trials, totaling close to 32,000 people, evaluated the effects of potassium-enriched salts with a concentration of 25–65% potassium chloride for a duration of 1–60 months. Mean baseline systolic and diastolic blood pressure ranged from 113–177 mm Hg and 71–105 mm Hg, respectively. The results showed that the use of potassium-enriched salts reduced mean systolic blood pressure by 4.61 mm Hg and mean diastolic blood pressure by 1.61 mm Hg. Total mortality was reported in five of these trials; the results showed a statistically significant 11% reduction in total mortality risk.271
A salt substitute that is 25% potassium chloride (75% sodium chloride) is generally recommended for those with hypertension unless they have advanced kidney disease, are using a potassium supplement, are using a potassium-sparing diuretic, or have other contraindications.272
Weight Loss. A reduction of systolic blood pressure by 5‒20 mm Hg accompanying weight loss has been observed in several studies.51 Weight loss may also allow a decrease in the required dose of blood pressure medications.48 Weight loss has been found to be especially important in adults under the age of 65. For adults over 65, weight had less impact on systolic blood pressure.52 Refer to the Weight Management protocol for an overview of a comprehensive approach to attaining a healthy weight.
Exercise. Regular exercise has been associated with average reductions in blood pressure of 3.2‒3.8 mm Hg (systolic) and 2.6‒3.5 mm Hg (diastolic) in thousands of subjects across many studies.53-55 Even something as simple as breaking up prolonged sitting with light intensity walking was associated in one study with lower blood pressure (-3 mm Hg systolic and -3 mm Hg diastolic).56
Limit Alcohol Intake. In adults ages 20‒84 without high blood pressure, the National Health and Nutrition Examination Survey (NHANES) showed that alcohol consumption above two drinks per day in men and one drink per day in women was associated with higher systolic blood pressure in both men and women.57 The good news is that limiting alcohol consumption to less than two drinks per day for men and less than one drink per day for women can reduce systolic blood pressure by 2‒4 mm Hg.58
Limit Use of NSAIDs & Acetaminophen. Nonsteroidal anti-inflammatory drugs (NSAIDs) and acetaminophen have been shown to increase blood pressure in those with and without hypertension.233-235 Low-dose aspirin does not have this effect.235 NSAIDs induce acute kidney injury and are especially problematic for individuals with existing kidney disease.236,237 The combination of NSAIDs, diuretics, ACE inhibitors or ARBs, and advanced age is associated with an especially high risk of kidney injury.237,238 In one study, researchers found that those with high blood pressure who had been taking NSAIDs for at least three months were 32% more likely to have chronic kidney disease than those who did not take NSAIDs, and those who used NSAIDs more than once daily were 23% more likely to have chronic kidney disease compared with nonusers.60
The PATH-BP trial examined the effect of regular acetaminophen use in 103 patients with hypertension. The study showed that two weeks of daily acetaminophen use (1 gram four times daily) led to an increase of about 5 mm Hg in average systolic blood pressure compared with placebo.233 In elderly individuals, sodium-containing forms of acetaminophen have also been found to increase the risk of high blood pressure compared with non-sodium-containing forms.239
A wide range of other medications and drugs can raise blood pressure, including alcohol, nicotine, and stimulant drugs; migraine medications; estrogens, including in birth control pills; decongestants such as pseudoephedrine and other over-the-counter cold medications; some antidepressants; and corticosteroids.240,241
Get Tested for Sleep Apnea. Symptoms of obstructive sleep apnea include snoring and choking, gasping, or silent breathing pauses during sleep. Sleep apnea is a major underappreciated risk factor for high blood pressure. Rates of sleep apnea have greatly increased over the past two decades and are now estimated to occur in 26% of adults between the ages of 30 and 70 years old.61 Sleep apnea causes a sudden drop in blood oxygen, which can lead to an increase in blood pressure.62 A meta-analysis of randomized controlled trials found that continuous positive airway pressure (CPAP), a treatment for sleep apnea, helped to decrease blood pressure in individuals with sleep apnea and resistant hypertension by nearly 5 mm Hg systolic and 3 mm Hg diastolic.63
Manage Stress. Acute stress can cause short-lived blood pressure spikes, and effectively managing stress may improve blood pressure and overall health. The body’s stress response results in release of the hormones cortisol and epinephrine, which elevate heart rate and cause blood vessel constriction, raising blood pressure.64 A meta-analysis found that individuals who had stronger responses to acute psychological stress were 21% more likely to develop hypertension.65Many strategies for reducing stress are described in Life Extension’s Stress Management protocol.
Maintain Good Oral Hygiene. Poor oral hygiene has been associated with cardiovascular risks, and particularly with high blood pressure.261,262 In people with existing hypertension, the presence of multiple dental caries has been shown to increase the risk of adverse outcomes like stroke and myocardial infarction.263 A meta-analysis that included over 250,000 participants found that those who brushed their teeth infrequently (approx. one or fewer times per day) had an 84% increased risk of hypertension compared with those who brushed their teeth more frequently (approx. three or more times per day), and each increment of daily tooth brushing decreased the risk of hypertension by 22%.264
The relationship between oral hygiene and hypertension appears to be mediated in part by the oral microbiome. Certain bacteria in the mouth reduce ingested nitrate and nitrite to produce nitric oxide, a multifunctional signaling molecule that plays an essential role in the function of the vascular endothelium. Disruption of nitrate-reducing bacteria in the oral microbiome, either through overgrowth of pathogenic bacteria as in periodontitis, or through overuse of antibacterial mouth rinses, can decrease nitric oxide levels and increase blood pressure.261 Frequent use of antibacterial mouth rinses (≥2 times daily) has been shown to raise blood pressure and increase the risk of hypertension.265,266
This effect appears to be strongly tied to chlorhexidine mouth rinses and changes in the oral microbiome that affect nitrite availability.267 There is some evidence that probiotics may help improve blood pressure, although further studies are needed to elucidate specific beneficial strains and the role of the oral microbiome in hypertension.268,269
Targeted Natural Interventions
Quercetin. Quercetin is a type of plant pigment called a flavonoid. Many fruits and vegetables contain quercetin. Studies over the last few decades have found that quercetin intake is linked to reduced cardiovascular disease. More recently, intervention studies in animals and humans have shown that quercetin supplementation lowers blood pressure.66 Quercetin is thought to lower blood pressure through multiple mechanisms, including functioning as an angiotensin receptor blocker (ARB). In fact, a 2015 study showed quercetin achieved a similar estimated receptor docking score for the angiotensin receptor as some pharmaceutical ARBs, including irbesartan and losartan.67
The blood-pressure-lowering capacity of quercetin has been observed in several clinical trials. For example, a trial in 93 overweight or obese individuals showed 150 mg of supplemental quercetin daily for six weeks reduced systolic blood pressure by 2.6 mm Hg.68 In a separate randomized controlled trial, 49 men consumed 150 mg of quercetin or a placebo each day for eight weeks. Subjects who took quercetin experienced a 5.7 mm Hg reduction in 4-hour postprandial (after-meal) blood pressure, while placebo recipients’ blood pressure did not change significantly.69 A 2015 randomized controlled trial found that in hypertensive subjects six weeks of treatment with 162 mg of quercetin daily lowered 24-hour ambulatory blood pressure by 3.6 mm Hg compared with placebo.70 Other trials that used different quercetin doses and treatment durations have also shown that this flavonoid effectively lowers blood pressure.71,72
Myricetin and myricitrin. Like quercetin, myricetin is a flavonoid present in vegetables, fruits, nuts, berries, tea, and red wine.73,74 And also as with quercetin, it appears that myricetin functions as an angiotensin receptor blocker (ARB).67 Preclinical studies have shown that myricetin attenuates the rise in blood pressure in response to hypertensive stimuli in animals.75,76 Myricitrin, also a naturally occurring flavonoid, is converted to myricetin by intestinal flora.77 Both myricetin and myricitrin have anti-inflammatory properties,78,79 and myricitrin has shown anxiolytic properties in a preclinical study.80
In an uncontrolled 2014 pilot clinical study on diabetics with normal blood pressure, 600 mg of the myricitrin-containing herb Eugenia punicifolia daily for three months led to an 11 mm Hg reduction in systolic blood pressure, and a 6 mm Hg reduction in diastolic blood pressure.81
Stevioside. A glycoside derived from the leaves of Stevia rebaudiana, stevioside is commonly known in the United States as a sweetener. In South America and Asia, extracts of the Stevia plant have been used traditionally to control blood sugar levels.82,83 Preclinical evidence has shown that stevioside may help control blood pressure by functioning as a calcium channel blocker. Blocking calcium channels is how some medications like verapamil lower blood pressure.84-86
A 2015 meta-analysis of data from published studies found stevioside reduced blood pressure by nearly 12 mm Hg (for diastolic pressure) when used for more than one year. Overall, studies in this analysis that used predominantly stevioside as the intervention showed an average reduction of 4.5 mm Hg in systolic blood pressure. The dosage of stevioside used in the studies included in this analysis ranged from 750 to 1,500 mg daily.87
Melatonin. Melatonin is well-known and widely used as a natural sleep aid. It is a hormone that the pineal gland releases at night to promote restful sleep and help regulate circadian (day-night) body rhythms.88
Melatonin has some other important but underappreciated health benefits: it appears to help control blood pressure by acting within the central nervous system as well as peripheral parts of the body. Peripherally, melatonin helps relax blood vessels and promote vasodilation, which reduces blood pressure. Melatonin can also inhibit the sympathetic nervous system, overstimulation of which can contribute to high blood pressure.89,90 Uncontrolled nocturnal hypertension is a serious problem that many people may not take into consideration. Elevated nighttime blood pressure contributes to cardiovascular disease and mortality, and is especially prevalent in people with sleep apnea.91-93
In a 2011 meta-analysis of data from seven studies (221 subjects in total), 2‒3 mg of controlled release melatonin at bedtime reduced nocturnal systolic blood pressure by 6 mm Hg, and nocturnal diastolic pressure by 3.5 mm Hg.94
Fish Oil. Fish oil is a source of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), polyunsaturated omega-3 fatty acids best known for their anti-inflammatory effects. Omega-3 fatty acids from fish oil are important cardioprotective nutrients, due to their ability to reduce inflammation and oxidative stress, stabilize cell membranes, improve disordered lipid levels and glucose metabolism, normalize blood vessel function, and inhibit formation of blood clots and atherosclerosis.242 In addition, DHA is used in the body to make compounds called resolvins, protectins, and maresins, known collectively as specialized pro-resolving mediators (SPMs), that have a critical role in ending inflammatory processes.243
A meta-analysis published in 2022 that included data from 71 randomized controlled trials, with a total of 4,973 participants, found supplementing with a combination of EPA plus DHA reduced both systolic and diastolic blood pressures. The researchers identified 2–3 grams per day of combined EPA plus DHA as an optimal dosage. This dosage resulted in an overall 2.61 mm Hg drop in systolic blood pressure and 1.64–1.80 mm Hg drop in diastolic blood pressure. No additional blood pressure reductions were seen using dosages higher than 3 grams per day, especially in those with normal baseline blood pressures. The greatest effects were seen in older individuals, those with systolic blood pressure ≥ 140 mm Hg, and those with abnormal lipid levels, and findings suggested these populations may possibly benefit from doses slightly higher than 3 grams per day.244
Coenzyme Q10. An in-depth review examined randomized controlled trials that used coenzyme Q10 (CoQ10) supplementation, for a minimum of three weeks, to treat primary hypertension. Treatment with CoQ10 resulted in an average decrease in systolic blood pressure of 11 mm Hg and an average decrease in diastolic blood pressure of 7 mm Hg.101 CoQ10’s antihypertensive effects may be a result of nitric oxide-dependent vasodilation and protection of nitric oxide from free radical-mediated degradation; CoQ10 may also boost production and increase tissue sensitivity of prostacyclin, a potent vasodilator that prevents blood clotting.102 CoQ10 supplementation has demonstrated efficacy for decreasing risk of major cardiovascular events and death from cardiovascular and all causes; improving endothelial function, an important concern in high blood pressure, including in statin-treated diabetics; and increasing activity of the free radical modulating enzyme superoxide dismutase.103-106 In a five-year randomized trial of 443 elderly individuals, CoQ10 combined with selenium reduced cardiovascular mortality by half compared with placebo.107
Magnesium. Magnesium supplementation dose-dependently lowers blood pressure in people who have high blood pressure.108 Based on data from epidemiologic and observational studies, individuals with high blood pressure tend to have lower intakes of magnesium than those with normal blood pressure, and low magnesium intake is associated with greater risk of having and dying from heart disease. Magnesium intake of 500–1000 mg per day is associated with lower systolic and diastolic blood pressure. Magnesium also appears to increase the effectiveness of anti-hypertensive medications. Magnesium’s effectiveness is attributable to its ability to improve endothelial function, act as a natural calcium channel blocker and vasodilator, and possible anti-inflammatory activity.109-113
In a meta-analysis of seven studies that enrolled patients with systolic blood pressure over 155 mm Hg who had taken antihypertensive medication for at least six months, magnesium supplementation was shown to significantly lower systolic and diastolic blood pressure by an average of 18.7 mm Hg and 10.9 mm Hg, respectively.114 Low serum magnesium is associated with a decline in kidney function and an increased risk of chronic kidney disease.115 Magnesium status can be assessed using the red blood cell magnesium test.116
A randomized controlled trial investigated the effects of 400 mg of daily magnesium citrate supplementation for 12 weeks on metabolic and physiological parameters in 54 participants with metabolic syndrome. Magnesium was given to 27 subjects and the remaining 27 received placebo. Ionized magnesium levels increased significantly in the supplemented group, although serum magnesium levels remained unchanged. Participants receiving magnesium showed notable reductions in systolic (from 135 to 126 mm Hg) and diastolic (from 84 to 79 mm Hg) blood pressure and interleukin-6 levels (from 4.9 to 3.0 pg/mL), along with a significant improvement in vitamin D levels.280
Whey Protein. Whey is a liquid byproduct of cheesemaking. For centuries whey was treated as waste, but is now recognized as a valuable source of high-quality protein, peptides, and amino acids.245,246 Whey protein powder is an extract from the liquid whey.247 Numerous controlled clinical trials have demonstrated whey protein’s ability to lower blood pressure and improve vascular function. These studies have also shown that whey protein improves other metabolic risk factors, including insulin sensitivity and glucose and lipid levels.120,248 Two separate meta-analyses concluded that whey protein consumption results in lower systolic and diastolic blood pressure, as well as other metabolic improvements such as improved lipid levels, lower fasting glucose, and improved body composition.249,250
In a dietary challenge trial comparing the eight-hour response to 28 grams of whey protein, casein, or maltodextrin, whey resulted in a significantly greater drop in systolic blood pressure that lasted up to five hours. Arterial stiffness also improved.251 A similar rapid effect of whey protein on blood pressure was revealed in a study in normal-weight men with an average age of 75 years. A nutrient drink supplemented with 70 grams whey protein alone, or mixed with carbohydrate and fat, lowered systolic blood pressure by 22–24 mmHg in the two to three hours after the drink was consumed. The combination drink lowered diastolic blood pressure as well.252 An eight-week trial in 38 adults with pre-hypertension and mild hypertension showed that two daily doses of 28 grams of whey protein daily for eight weeks lowered systolic and diastolic blood pressure, and improved endothelial function as measured by flow-mediated dilation.253 Another randomized controlled trial in 54 adults found that in obese and overweight individuals who received 30 grams of whey protein for 12 weeks, systolic blood pressure was significantly lower, as were body fat, fat percentage, and waist circumference, compared with the control group. Endothelial function also improved. The authors of this paper concluded that body fat loss was the mechanism for the blood pressure benefit.254
Fat loss and improvements in arterial stiffness and endothelial function all likely contribute to the blood pressure benefits of whey protein, but they do not completely explain the rapid lowering effect that occurs within hours. It has been suggested, however, that whey protein’s effects may also be mediated by inhibition of the angiotensin converting enzyme (ACE) and by interaction with opioid neurotransmitter receptors.246
Trials showing these benefits generally have used roughly one to three ounces (28–84 grams) of whey protein powder per day.249,250
Grape Polyphenols. A meta-analysis of randomized controlled trials found that, compared with controls, grape polyphenol treatment lowered systolic blood pressure. The effective dosages used in these clinical trials ranged from 200 to 733 mg/day, over a period of 2–12 weeks. The blood pressure-lowering effect was most pronounced in study participants with metabolic syndrome.128 This effect is probably mediated in large part by grape polyphenols’ antioxidant properties and ability to substantially improve endothelial function in both healthy subjects and those at cardiovascular risk.127,255 The endothelial benefit of grape polyphenols may be explained by a modulation of nitric oxide metabolism.129
Evidence has continued to accumulate from preclinical studies,256,257 randomized controlled trials,255,258 and meta-analyses259,260 that grape polyphenols and grape seed extract are potent modifiers of vascular endothelial function, yielding improvements in blood pressure.
Pomegranate Extract. Pomegranate juice and its polyphenolic extract have established cardioprotective benefits that include reduction in systolic and diastolic blood pressure.131-133 Pomegranate juice naturally inhibits the angiotensin-converting enzyme, mimicking the mechanism of antihypertensive ACE inhibitor drugs.134,135 Animal studies indicate that pomegranate juice extract also interferes with the hypertensive effect of angiotensin II, a property similar to angiotensin II receptor blocker (ARB) medications, and has a beneficial influence on endothelial nitric oxide.136,137
Olive Leaf Extract. A 12-week randomized clinical study compared the blood-pressure-lowering effects of olive leaf extract to the ACE-inhibitor medication captopril in patients with stage I hypertension. Both the olive leaf and the captopril group experienced reductions in systolic and diastolic pressure, with no important difference in the effects of the two compounds. In the olive leaf extract group, systolic and diastolic blood pressure decreased on average by 11.5 mm Hg and 4.8 mm Hg, respectively.138 This study confirmed results of earlier trials that found olive leaf extract effective for hypertension.139,140 Olive leaf extract has been shown to increase insulin sensitivity in men at risk of developing metabolic syndrome, a condition often accompanied by high blood pressure.141 Olive leaf’s antihypertensive effect may be attributable to several of the same mechanisms as prescription blood pressure drugs, including calcium-channel blocking, ACE inhibition, and angiotensin II antagonism.142-145
Celery Seed Extract. A trial of standardized celery seed extract in 30 patients with mild-to-moderate hypertension found that six weeks of supplementation reduced systolic and diastolic blood pressure by 8.2 and 8.5 mm Hg, respectively.146 Long-term administration of celery seed extract to hypertensive rats resulted in a substantial reduction in blood pressure.147 Laboratory studies with celery extract have demonstrated vasodilatory and vasorelaxant activity, which may be mediated by calcium-channel-blocking activity.148,149
Hesperidin. The inner peels of sweet orange (Citrus sinensis) are a rich source of flavonoids, including hesperidin.206 Hesperidin and other citrus flavonoids enhance vascular health by improving endothelial function and reducing inflammation.207,208 In addition, hesperidin’s anticoagulant and lipid-regulating properties add to its potential utility in preventing and treating hypertension and other cardiovascular diseases.209
In a randomized controlled trial, 64 subjects with type 2 diabetes received either 500 mg of hesperidin per day or placebo for six weeks. Those taking hesperidin had greater reductions in systolic and diastolic blood pressure and inflammatory marker levels compared with placebo.210 A randomized controlled trial in subjects with metabolic syndrome found that 500 mg of hesperidin for three weeks improved endothelial function and levels of inflammatory markers.208
Citrus fruits and their juices have high concentrations of hesperidin.211 In a controlled clinical trial in healthy individuals, drinking orange juice and taking 500 mg of hesperidin daily for four weeks each resulted in changes in gene expression toward a less inflammatory and less atherosclerosis-promoting profile.212 In a similar four-week trial, orange juice and supplemental hesperidin were found to have similar positive effects, compared with placebo, on diastolic blood pressure and endothelial function of small blood vessels. This study, in moderately overweight men aged 50‒65, suggests hesperidin may be the active compound responsible for oranges’ cardioprotective effects.213 In animal research, hesperidin has been shown to reduce high blood pressure214 and reverse age-related arterial stiffness.215
Hesperidin and its breakdown product, hesperetin, have been found to increase the production of nitric oxide in cells from the inner lining of human blood vessels.216,217 Nitric oxide from these cells plays a key role in relaxing blood vessel walls and reducing high blood pressure.218 In the same study, after treatment with hesperidin, human blood vessel cells also produced less endothelin-1, a compound that constricts blood vessels and increases blood pressure, and fewer oxygen free radicals.216 Hesperidin’s ability to raise levels of vessel-dilating nitric oxide has also been demonstrated in animal studies.219,220 Animal and preclinical research indicates hesperidin may also relax blood vessel walls by altering the function of potassium channels that help regulate muscle contraction in blood vessel walls.221,222
Hesperidin, like other plant polyphenols, impacts the health and diversity of the microbial community of the gut. Because the gut microbiome plays a key role in regulating metabolism and inflammation, it is now thought that flavonoids like hesperidin may exert their positive effects on health and disease in part through interactions with gut microbes. The possible links between hesperidin, the gut microbiome, and improved metabolic and cardiovascular health is an exciting and emerging topic in nutrition research.211,223
Garlic. Garlic has been examined in many studies for its benefits primarily on blood lipid profiles, such as reducing high cholesterol and serum triglycerides. Evidence from multiple studies also suggest garlic can reduce systolic and diastolic blood pressure.273 A meta-analysis of 12 trials including 553 hypertensive participants found that garlic supplements lowered systolic and diastolic blood pressure by an average of 8.3 mm Hg and 5.5 mm Hg, respectively.274
A branded aged garlic extract, Kyolic, was found to effectively reduce blood pressure in multiple clinical studies. In one randomized controlled study conducted over 12 weeks, a total of 79 participants with uncontrolled systolic hypertension (≥ 140 mm Hg) were randomized to placebo or 240, 480, or 960 mg of aged garlic extract. In the 480 mg group, systolic blood pressure was significantly reduced by 11.8 mm Hg compared with placebo.275 In a separate controlled trial, participants taking 1.2 grams of Kyolic aged garlic extract daily for 12 weeks were found to have significant reductions in systolic (10 mm Hg) and diastolic (5.4 mm Hg) blood pressure.276
Black garlic is produced from the aging of raw garlic under high temperature and humidity conditions. The aging process changes the garlic to a dark color and increases the content of various compounds, including S-allyl cysteine (SAC). SAC is a major bioactive component in aged and black garlic that contributes to many of its health benefits.277 In one randomized controlled trial conducted over 12 weeks, 56 healthy individuals aged 25-65 years with borderline cardiovascular disease risk factors received placebo or 500 mg of aged black garlic, providing 2.5 mg SAC. After 12 weeks, those given aged black garlic had statistically significant and clinically meaningful reductions in systolic and diastolic blood pressure compared with placebo. Aged black garlic also showed significant improvements in total cholesterol, LDL cholesterol, serum triglycerides, and fasting blood glucose.278 A separate randomized trial including 67 participants with hypercholesterolemia found that 250 mg aged black garlic (providing 1.25 mg SAC) modestly reduced diastolic blood pressure compared with placebo.279
Taurine. Taurine promotes healthy blood pressure through several complementary actions, many of which have been demonstrated in preclinical models. A primary mechanism involves the modulation of intracellular sodium and calcium, ultimately helping to mitigate vascular stiffness and endothelial dysfunction. Additionally, taurine promotes blood vessel relaxation. Taurine further contributes to blood pressure regulation through its antioxidant and osmoregulatory (regulating cellular water content) properties.281 Taurine also appears to prevent harmful remodeling of endothelial cells by angiotensin II, a hormone that can cause blood vessels to constrict and stiffen.282
Multiple clinical trials and meta-analyses support the antihypertensive efficacy of taurine across diverse populations. A meta-analysis of seven randomized controlled trials found that taurine supplementation at doses of up to 6 grams/day over periods of up to 12 weeks reduced systolic and diastolic blood pressure by about 3 mm Hg each.283 A larger meta-analysis of 12 randomized controlled trials, generally in metabolically unhealthy populations, reported systolic blood pressure and diastolic blood pressure reductions of 4.67 mm Hg and 2.90 mm Hg, respectively, following supplementation with up to 6 grams taurine daily for 15 days to six months.284 A more extensive meta-analysis encompassing 20 randomized controlled trials confirmed a 4.00 mm Hg reduction in systolic blood pressure and 1.43 mm Hg reduction in diastolic blood pressure following taurine supplementation for 15 days to12 months. The dosage used in most of the studies in this analysis ranged from about 1.5 to 6 grams daily. Interestingly, greater effects were seen in hypertensive and prehypertensive individuals. There was some evidence that higher taurine dosages corresponded with greater blood pressure reductions, although this was more apparent for diastolic than systolic blood pressure.285
More recently, a 12-week trial published in 2025 included 144 patients with type 2 diabetes and found that 2.4 grams of taurine daily led to a 7 mm Hg reduction in systolic blood pressure compared with baseline (which was significantly better than placebo). Taurine supplementation also improved arterial stiffness, a key factor in cardiovascular health.286
Additional Support. The following natural compounds may be considered for additional cardiovascular and blood pressure support.
- Calcium and vitamin K. Calcium plays an important role in regulating the concentration of minerals that are essential to healthy blood pressure, including sodium, potassium, and magnesium. Calcium is also involved in smooth muscle cell contraction in blood vessels.150 In a review of 40 randomized controlled trials, an average daily calcium dose of 1,200 mg was associated with a modest reduction in systolic (almost 1.9 mm Hg) and diastolic (almost 1.0 mm Hg) blood pressure. In persons with habitually low calcium intake (≤ 800 mg/day), the hypotensive effect was greater.151
- Adequate vitamin K intake is crucial during calcium supplementation. Vitamin K prevents calcium deposition in non-bony tissues like the arteries, and protects against atherosclerotic heart disease. Vitamin K comes in several forms: vitamin K2, also known as menaquinone, has shown the greatest association with reduced cardiovascular risk. Vitamin K is also essential for proper blood coagulation and bone health, and is associated with a reduced risk of cancer and death from cancer.152-156
- L-arginine. An exhaustive analysis of 11 randomized controlled trials found that L-arginine supplementation, in dosages ranging from 4 to 24 g per day, lowered systolic blood pressure by 5.4 mm Hg and diastolic blood pressure by 2.7 mm Hg, measurably more so than placebo.157
- Chlorogenic acid. A randomized controlled trial in individuals with mild hypertension found that chlorogenic acid, which is abundant in green coffee beans and specially roasted brewed coffees, lowered systolic and diastolic blood pressure by as much as 5.6 mm Hg and 3.9 mm Hg, respectively, compared with placebo.158 Another clinical trial compared a coffee beverage that was specially treated to enhance chlorogenic acid activity with a control beverage. The chlorogenic acid-enriched coffee resulted in a reduction in blood pressure, while the control beverage had no effect.159
- Hawthorn extract. Many studies have shown that several species of hawthorn confer cardiovascular benefits. Hawthorn has long been used in Chinese herbal medicine, and it has been used in North America to treat heart problems since 1800. Clinical trials have shown that supplementation with hawthorn and hawthorn extracts improves mild heart failure, hypertension, and elevated blood lipids.160
- Resveratrol. Resveratrol is a polyphenol found in grapes, wine, Japanese knotweed, and several other plants. It has been extensively studied as a calorie-restriction mimetic because of its ability to influence metabolic pathways similarly to reducing caloric intake. A meta-analysis of six studies found that supplementation with at least 150 mg of resveratrol daily reduced systolic blood pressure by nearly 12 mm Hg while not significantly affecting diastolic blood pressure.161
Phase 2: Adding a Drug Regimen
Many people may not be able to lower their blood pressure to the range of 115/75 – 120/80 mm Hg with dietary and lifestyle changes alone (although those who practice caloric restriction may more easily attain this goal). For those whose blood pressure remains elevated, adding a single blood pressure medication, initially at a low dose, can help lower blood pressure. If a single medication is inadequate, then a second and possibly a third can be added, although side effect risk may increase in those who take high doses of multiple blood pressure medications. A trial period of up to one month should be allocated to assess the impact of each drug added to your regimen.
The regimen described may not be appropriate for aging adults with significant pre-existing cardiovascular disease, metabolic disease, renal disease, and/or major cognitive impairment.
This regimen represents Life Extension’s suggestions for most adults up to age 80 who have high blood pressure but are otherwise healthy. Please be aware that evidence suggests African American patients may respond better to initial therapy with a calcium channel blocker or a thiazide diuretic (whereas most other patients tend to show a greater response to angiotensin II receptor blockers or angiotensin-converting enzyme inhibitors).18
Maintaining optimal blood pressure is a lifelong endeavor and often requires a diligent, frequently individualized approach.
First-Line Recommendation: Angiotensin II Receptor Blocker (ARB) or Angiotensin-Converting Enzyme Inhibitor (ACEI). Angiotensin II is a peptide hormone naturally produced in the body. It exerts broad influence on blood pressure control. Blunting angiotensin II signaling is an established pharmacologic strategy for lowering blood pressure. This can be accomplished with angiotensin II receptor blockers (ARBs) or angiotensin-converting enzyme inhibitors (ACEIs). Both of these drug classes have been studied in large clinical trials.162,163 ARBs and ACEIs are similarly effective in reducing the risk of cardiovascular disease and stroke when used in the treatment of primary hypertension, but ARBs may be less likely to cause side effects.164
- Angiotensin II receptor blockers. Life Extension favors initiating therapy with the ARB telmisartan. A little-known side benefit of ARBs is they enhance insulin sensitivity, increase utilization of fat as energy, and improve mitochondrial function. Of all the drugs in this class, telmisartan, first approved to lower blood pressure in 1998,165 stands out as superior for potential longevity enhancement.166-168
- Angiotensin-converting enzyme inhibitors. Angiotensin-converting enzyme inhibitors (ACEIs) function differently than ARBs to lower blood pressure, but have similar indications and benefits.48,118,119 However, as many as 20% of patients prescribed ACEIs will develop a chronic cough, a side effect not associated with ARBs.187 ACEIs as a class tend to be associated with more adverse effects compared to ARBs. Also, evidence for potential longevity benefits associated with ARBs may not apply to ACEIs.
Studies indicate that telmisartan directly stimulates PPAR-gamma (peroxisome proliferator-activated receptor gamma), a key inducer of beneficial metabolic effects.169-171 PPAR-gamma activating properties have also been reported for other ARB drugs, but telmisartan is at least 10 times more powerful.169
Telmisartan has been shown in preclinical models to reduce weight gain, increase total energy expenditure, and increase expression of key mitochondrial enzymes in skeletal muscle better than a more popular drug in this class (Diovan).172-177 Also, telmisartan functions by multiple mechanisms to protect against arterial occlusion,178-180 including increasing beneficial endothelial nitric oxide.174,181,182 Lastly, several preclinical and lab studies suggest telmisartan may confer meaningful cancer chemopreventive effects.183-186
If you cannot achieve blood pressure readings of 115/75 – 120/80 mm Hg with diet and lifestyle changes and targeted dietary supplementation alone, consult with your personal physician concerning an initial dose of 20 or 40 mg of telmisartan daily; choosing between 20 and 40 mg should be left to the discretion of your physician because individual patient factors will determine which dose is more appropriate for you.
To read more about telmisartan and its many merits, refer to the March 2015 Life Extension Magazine article titled “Best Drug to Treat Hypertension.”
Second-Line Recommendation: Thiazide Diuretic (Chlorthalidone). Chlorthalidone, a type of thiazide diuretic, can help lower blood pressure by increasing urinary excretion of salt and water. This drug has been used in the treatment of hypertension and for cardiovascular event risk reduction for decades.188
If you cannot achieve your target blood pressure within one month of starting your first medication, add a thiazide diuretic to your daily regimen. Start at a low dose, and allow one month after starting the diuretic to see if your blood pressure falls to target range.
Although thiazides have a long record of safe use and a good risk-benefit ratio, these drugs may cause a drop in your blood electrolyte levels (eg, potassium, sodium, and chloride).189 You can monitor your electrolyte levels by having a routine blood chemistry panel performed periodically after you begin taking a thiazide diuretic.
Third-Line Recommendation: Calcium Channel Blocker. If you cannot achieve your target blood pressure by adhering to good eating habits, getting plenty of exercise, and taking blood-pressure-lowering supplements along with telmisartan and a thiazide diuretic, consider adding a calcium channel blocker.
Calcium channel blockers, such as amlodipine, influence several aspects of cardiovascular physiology, resulting in lowering of blood pressure. A recent systematic review concluded that calcium channel blockers in combination with thiazide diuretics may be a good option for elderly individuals who have isolated systolic hypertension.190
Beta-Blockers: Brief Review of Current Evidence
Sympathetic nervous system activity (our “fight or flight” system) tends to increase as we age, and it is thought that this increase may hasten the development of age-related mortality. Prescription drugs called beta-blockers inhibit the action of epinephrine, the main hormone of our sympathetic nervous system. Because of this mechanism, beta-blockers are used for hypertension and have been studied as a means to increase lifespan.
In a 2013 study, beta-blockers were shown to extend the lifespan of mice and fruit flies. In this study, metoprolol and nebivolol (both beta-blockers) increased the median lifespan of mice by 8.4% and fruit flies by 15‒23%.191
These changes were not due to calorie restriction or change in activity, as neither the treated mice nor the fruit flies showed changes in food intake or activity level. The authors suggest that the signaling pathway beta-blockers inhibit is a fundamental mechanism that itself regulates an animal’s lifespan.
Mice in the same study that were placed on a calorie-restricted diet had their lifespan extended by 23% (nearly 3 times the extension by beta-blockers). However, beta-blocker treatment doubled the number of liver tumors in the mice, suggesting the compounds were toxic to the liver, even when given at a relatively low dose.
In a more recent animal study, although atenolol (a beta-blocker) changed the mitochondrial membrane fatty acid profile to that of much longer-lived mammals and decreased oxidative stress, lifelong treatment of the mice did not result in any extension of lifespan.192 The authors concluded that side effects from the drug could have masked the decrease in aging that is usually caused by the change in membrane fatty acid profile.
Beta-blockers used to be considered a first-line treatment for hypertension, but there have recently been some worrisome findings about their use in otherwise healthy adults. The 2014 Evidence-Based Guideline for the Management of High Blood Pressure in Adults (JNC 8) no longer recommends beta-blockers as initial therapy since one study found that the use of beta-blockers resulted in a higher rate of cardiovascular death, myocardial infarction, and especially stroke as compared to use of an ARB.18
Also, there is some evidence that some beta-blockers are associated with increased risk of type 2 diabetes. Recent human studies do not support the use of beta-blockers in hypertensive adults who are otherwise healthy.
Table 1: Advantages and Caveats of Common Antihypertensive Drug Classes 48,187,193,194
|
Drug Class |
Advantages |
Caveats |
|
Angiotensin II Receptor Blockers (ARBs)
|
Once-daily dose; well tolerated; associated with improved quality of life; reduction in cardiovascular mortality (for telmisartan); indicated in patients who experience coughing from ACEIs; lower risk of swelling (angioedema) compared to ACEIs; considered first-line treatment for patients with hypertension and diabetes, congestive heart failure, chronic kidney disease |
Risk of high serum potassium; may cause small increase in serum creatinine in some people via decrease in glomerular filtration rate, but this is usually transient and reversible; should not be used by women who are pregnant or expecting to become pregnant |
|
Angiotensin-converting enzyme inhibitors (ACEIs)
|
Indicated in patients with high blood pressure and diabetes, heart failure, chronic kidney disease, or coronary artery disease; may reduce risk of heart attack, stroke, and cardiovascular death in high-risk patients over age 55 |
Frequent (5‒20%) side effect of cough; risk of excessively low blood pressure, excessively high serum potassium; rare risk of swelling (angioedema); should not be used by women who are pregnant or expecting to become pregnant |
|
Beta-Blockers
|
Indicated after myocardial infarction and in heart failure and atrial fibrillation patients |
Risk of depression, impotence, fatigue, bronchospasm, hypoglycemia, peripheral vascular disease, worsening of lipids, obscuring signs and symptoms of hypoglycemia in diabetics |
|
Calcium Channel Blockers
|
Indicated in patients with ischemic heart disease, asthma, kidney disease, peripheral vascular disease, salt sensitivity, and metabolic diseases; tend to be associated with improved quality of life; work independently of sodium consumption |
Risk of disturbance of normal heart rhythm; constipation; headache; dizziness; low blood pressure; overgrowth of gums |
|
Thiazide Diuretics
|
Once daily dosing; indicated in congestive heart failure and edema; improve cardiovascular outcomes |
Risk of potassium, magnesium, and sodium depletion, and excessive serum calcium levels; cardiac arrhythmias; sexual dysfunction; worsening of lipid and glucose levels |
|
Table 2: Initial Dosages of Common Antihypertensive Drugs |
|||
| Class/Drug | Initial Dose | Generic Form Available? | |
| Angiotensin II Receptor Blockers (ARBs) | |||
| Telmisartan | 20 mg per day is lowest, for volume-depleted patients (on diuretics); otherwise 40 mg per day | Yes | |
| Losartan | 25 mg per day | Yes | |
| Benicar | 20 mg per day | Olmesartan medoxomil, not available | |
| Edarbi | 40 mg per day | Azilsartan medoxomil, not available | |
| Irbesartan | 75 mg per day | Yes | |
| ACE-Inhibitor | |||
| Lisinopril | 5 mg per day | Yes | |
| Diuretic (Thiazide) | |||
| Chlorthalidone | 12.5 mg per day | Yes | |
| Beta-Blocker | |||
| Atenolol | 25 mg per day | Yes | |
| Calcium Channel Blocker | |||
| Amlodipine | 2.5 mg per day | Yes | |
Phase 3: Drug Dosage Titration
There are several approaches to drug and dosage adjustment in the management of high blood pressure. For example, one option is to start with a single medication and then slowly increase the dosage of that medication until the target blood pressure is achieved, side effects limit the dosage, or the maximum recommended dose is reached. In this approach, a second medication may not be added until the dosage limit of the first medication is reached, and the dosage of the second drug is maximized before adding a third.
Another approach, which Life Extension advocates, is to start with a low dose of a single medication, then add a second and third medication before beginning to slowly increase dosages of one drug at a time. This approach may reduce side effects, which may be more likely with higher dosage of a given drug than with lower dosages of multiple drugs.
Overall, decisions to add an additional medication or adjust the dosage of a current medication need to be discussed by the patient and his or her physician. Clinical factors may influence which approach the physician determines to be most appropriate. Many people ultimately require more than one medication to achieve target blood pressure.195
Figure 1: Suggested Blood Pressure Management Algorithm
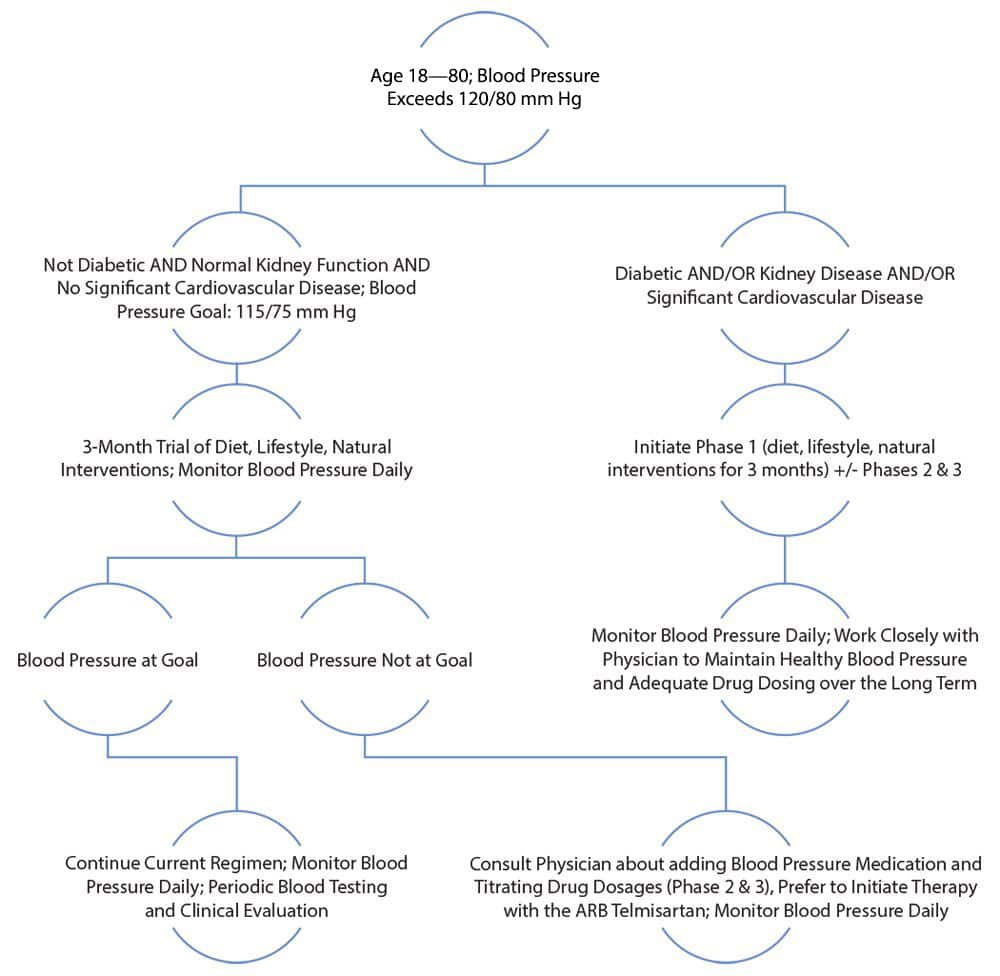
Blood Pressure Medications May Confer Greater Protection if Taken at Bedtime
A number of studies have shown that bedtime dosing of antihypertensive medication may improve treatment efficacy compared with traditional morning and evening dosage schedules.196-201
In one trial that enrolled 661 patients with chronic kidney disease, ambulatory blood pressure was measured at baseline, then tracked for over five years after adjusting the medication schedule according to one of two regimens. In the first group, all antihypertensive drugs were taken upon awakening, while a second group took at least one medication at bedtime. Not only did the second group have lower blood pressure during sleep, but a markedly greater percentage of them gained control over their daytime blood pressure compared with the morning dosing group.196
After analyzing the study data, researchers uncovered a dramatic reduction in the risk of cardiovascular events and associated mortality—those taking blood pressure medication at bedtime had only about one-third the risk of those who took all their blood pressure medication in the morning. Moreover, each 5 mm Hg reduction in blood pressure during sleep was tied to a 14% reduction in cardiovascular events during the follow-up period.
In a randomized clinical trial, 204 subjects with high blood pressure were assigned to take a combination of valsartan and hydrochlorothiazide either in the morning or before bed for 12 weeks. Subjects who took their blood pressure medication before bed had better blood pressure control at night than those who took their medication in the morning, and there was no difference in daytime seated blood pressure between the groups. Also, more subjects in the bedtime-dosing group had properly controlled ambulatory blood pressure. The researchers concluded that “…valsartan/[hydrochlorothiazide] combination should be preferably administered at bedtime for treatment of subjects with essential hypertension requiring combination therapy to achieve proper [blood pressure] control.”200
Several other trials have also shown that bedtime dosing of blood pressure medications offers superior blood pressure control compared with morning dosing.197-199,201
5 Diligent Monitoring to Prevent Treatment-Related Adverse Events
Those who take prescription antihypertensive medications must remain aware of the potential for these drugs to cause kidney dysfunction and electrolyte abnormalities in some people. Also, blood pressure that is too low can cause dizziness, especially upon standing, which may result in fainting and injurious falls. At-home blood pressure monitoring is critical, and should be performed twice daily, once in the morning and once in the evening.
In order to mitigate the chances of medication-related complications, particularly those related to kidney function, blood tests should be performed regularly to monitor treatment and health status. Changes in medications and medication dosage levels, and changes in overall health status (including urinary and fluid balance symptoms) are indications to repeat testing, bearing in mind that some alterations will manifest rapidly while others may appear gradually. It is important to remember that this applies to all medications, including over-the-counter drugs, not simply to blood pressure medications. For those in stable health, with no change in medication or dosage, testing should be performed every six months at minimum. All changes that could potentially impact kidney function should be closely followed using the indicated laboratory tests.
Important tests for people undertaking strategies to lower blood pressure include:
- Chemistry panel and complete blood count (CBC)
- Cystatin-C
- Orthostatic hypotension evaluation (clinical test)
6 Blood Pressure Is Not the Only Contributor to Heart Disease
Life Extension has identified at least 17 independent risk factors that increase your likelihood of succumbing to a deadly cardiovascular event. All of these risk factors must be addressed if one’s goal is optimal risk reduction.
Although high blood pressure is recognized worldwide as one of the strongest cardiovascular risk factors, focusing only on blood pressure and neglecting the other 16 independent risk factors could be a devastating oversight. These risk factors include:
- Excess LDL
- Excess total cholesterol
- Low HDL
- Excess blood glucose
- Excess homocysteine
- Excess C-reactive protein (CRP)
- Insufficient vitamin D
- Insufficient vitamin K
- Elevated triglycerides
- Low blood EPA and DHA
- Low testosterone (in men)
- Excess estrogen (in men)
- Excess insulin
- Nitric oxide deficit
- Excess fibrinogen
- Elevated levels of oxidized LDL
People interested in maximizing their cardiovascular risk reduction should follow the strategy outlined in this article to control their blood pressure, and also read the Life Extension Magazine article titled “How to Circumvent 17 Independent Heart Attack Risk Factors.”
7 Summary
Life Extension has long suggested that the lower blood pressure achieved by calorie restriction adherents is a critical component of the health and longevity benefits they enjoy. A growing body of evidence supports the benefit of blood pressure lower than 120/80 mm Hg for certain populations.1-3 The results from the SPRINT trial,5 published in 2015, lend additional support to a target systolic blood pressure of less than 120 mm Hg as opposed to a more conservative target of less than 140 mm Hg. For decades, Life Extension has advocated a blood pressure goal of 115/75 mm Hg.
The foundation for Life Extension’s approach is the evidence-based diet, lifestyle, and integrative intervention program outlined in this protocol; this aspect of the program should be continued indefinitely for those who want to maximize their health and longevity through optimal blood pressure control. For those who aren’t able to reach a target blood pressure of less than 120/80 mm Hg using these natural measures alone, Life Extension has presented a drug treatment protocol that represents an integration of the most current scientific knowledge, maximizing therapeutic benefit and minimizing the risk of adverse effects.
By working closely with your clinician, regularly monitoring your blood pressure and health status, and taking advantage of the cutting edge information presented here, you can marshal the longevity benefits of optimal blood pressure.
2025
- Aug: Added section on taurine to Life Extension’s Protocol for Optimal Blood Pressure
2024
- Nov: Updated section on magnesium in Life Extension’s Protocol for Optimal Blood Pressure
- Apr: Added section on garlic & updated section on dietary and lifestyle changes in Life Extension’s Protocol for Optimal Blood Pressure
2023
- Oct: Added section on maintaining good oral hygiene to Life Extension’s Protocol for Optimal Blood Pressure Control
- May: Updated sections on grape polyphenols and whey protein in Life Extension’s Protocol for Optimal Blood Pressure Control
2022
- Jun: Updated section on fish oil in Life Extension’s Protocol for Optimal Blood Pressure Control
- Mar: Added section on limiting use of NSAIDs and acetaminophen to the section on Optimal Blood Pressure Control
2011
- Aug: Comprehensive update & review
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
- Kshirsagar AV, Carpenter M, Bang H, Wyatt SB, Colindres RE. Blood pressure usually considered normal is associated with an elevated risk of cardiovascular disease. The American journal of medicine. Feb 2006;119(2):133-141.
- Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. Dec 14 2002;360(9349):1903-1913.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. May 15 2018;71(19):e127-e248. doi:10.1016/j.jacc.2017.11.006. https://www.ncbi.nlm.nih.gov/pubmed/29146535
- Weber MA, Schiffrin EL, White WB, et al. Clinical practice guidelines for the management of hypertension in the community: a statement by the American Society of Hypertension and the International Society of Hypertension. Journal of clinical hypertension (Greenwich, Conn.). Jan 2014;16(1):14-26.
- Sprint Research Group. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. The New England journal of medicine. Nov 9 2015;0(0):null.
- Bakris GL, Weir MR. Angiotensin-converting enzyme inhibitor-associated elevations in serum creatinine: is this a cause for concern? Archives of internal medicine. Mar 13 2000;160(5):685-693.
- Apperloo AJ, de Zeeuw D, de Jong PE. A short-term antihypertensive treatment-induced fall in glomerular filtration rate predicts long-term stability of renal function. Kidney international. Mar 1997;51(3):793-797.
- Whelton PK, Carey RM, Aronow WS, Casey DE, Collins KJ, Dennison Himmelfarb C, . . . Wright JT. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. 2017.
- Egan BM, Stevens-Fabry S. Prehypertension--prevalence, health risks, and management strategies. Nature reviews cardiology. May 2015;12(5):289-300.
- Garofalo C, Borrelli S, Pacilio M, et al. Hypertension and Prehypertension and Prediction of Development of Decreased Estimated GFR in the General Population: A Meta-analysis of Cohort Studies. American journal of kidney diseases: the official journal of the National Kidney Foundation. Oct 13 2015.
- Stocchetti N, Chieregato A, De Marchi M, Croci M, Benti R, Grimoldi N. High cerebral perfusion pressure improves low values of local brain tissue O2 tension (PtiO2) in focal lesions. Acta neurochirurgica. Supplement. 1998;71:162-165.
- The ACCORD Study Group. Effects of Intensive Blood-Pressure Control in Type 2 Diabetes Mellitus. New England Journal of Medicine. 2010;362(17):1575-1585.
- Barzilay JI, Howard AG, Evans GW, et al. Intensive Blood Pressure Treatment Does Not Improve Cardiovascular Outcomes in Centrally Obese Hypertensive Individuals With Diabetes: The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Blood Pressure Trial. Diabetes care. July 1, 2012 2012;35(7):1401-1405.
- Brunström M, Carlberg B. Effect of antihypertensive treatment at different blood pressure levels in patients with diabetes mellitus: systematic review and meta-analyses. BMJ (Clinical research ed.). 2016-02-25 00:06:06 2016;352.
- Odden MC, Shlipak MG, Whitson HE, et al. Risk factors for cardiovascular disease across the spectrum of older age: the Cardiovascular Health Study. Atherosclerosis. Nov 2014;237(1):336-342.
- Beckett NS, Peters R, Fletcher AE, et al. Treatment of hypertension in patients 80 years of age or older. The New England journal of medicine. May 1 2008;358(18):1887-1898.
- Oates DJ, Berlowitz DR, Glickman ME, Silliman RA, Borzecki AM. Blood pressure and survival in the oldest old. Journal of the American Geriatrics Society. Mar 2007;55(3):383-388.
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA : the journal of the American Medical Association. Feb 5 2014;311(5):507-520.
- Krakoff LR. Blood Pressure Out of the Office: Its Time Has Finally Come. American journal of hypertension. Nov 6 2015.
- Fuchs SC, Ferreira-da-Silva AL, Moreira LB, et al. Efficacy of isolated home blood pressure monitoring for blood pressure control: randomized controlled trial with ambulatory blood pressure monitoring - MONITOR study. Journal of hypertension. Jan 2012;30(1):75-80.
- Breaux-Shropshire TL, Judd E, Vucovich LA, Shropshire TS, Singh S. Does home blood pressure monitoring improve patient outcomes? A systematic review comparing home and ambulatory blood pressure monitoring on blood pressure control and patient outcomes. Integrated blood pressure control. 2015;8:43-49.
- Agena F, Prado Edos S, Souza PS, et al. Home blood pressure (BP) monitoring in kidney transplant recipients is more adequate to monitor BP than office BP. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association. Nov 2011;26(11):3745-3749.
- Imai Y, Obara T, Asamaya K, Ohkubo T. The reason why home blood pressure measurements are preferred over clinic or ambulatory blood pressure in Japan. Hypertension research : official journal of the Japanese Society of Hypertension. Aug 2013;36(8):661-672.
- Fuchs SC, Mello RG, Fuchs FC. Home blood pressure monitoring is better predictor of cardiovascular disease and target organ damage than office blood pressure: a systematic review and meta-analysis. Current cardiology reports. Nov 2013;15(11):413.
- Sacks FM, Appel LJ, Moore TJ, et al. A dietary approach to prevent hypertension: a review of the Dietary Approaches to Stop Hypertension (DASH) Study. Clinical cardiology. Jul 1999;22(7 Suppl):Iii6-10.
- Svetkey LP, Simons-Morton D, Vollmer WM, et al. Effects of dietary patterns on blood pressure: subgroup analysis of the Dietary Approaches to Stop Hypertension (DASH) randomized clinical trial. Archives of internal medicine. Feb 8 1999;159(3):285-293.
- Appel LJ, Moore TJ, Obarzanek E, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. The New England journal of medicine. Apr 17 1997;336(16):1117-1124.
- Streppel MT, Arends LR, van 't Veer P, Grobbee DE, Geleijnse JM. Dietary fiber and blood pressure: a meta-analysis of randomized placebo-controlled trials. Archives of internal medicine. Jan 24 2005;165(2):150-156.
- Sacks FM, Svetkey LP, Vollmer WM, et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. The New England journal of medicine. Jan 4 2001;344(1):3-10.
- Hinderliter AL, Sherwood A, Craighead LW, et al. The long-term effects of lifestyle change on blood pressure: One-year follow-up of the ENCORE study. American journal of hypertension. May 2014;27(5):734-741.
- Sikand G. Mediterranean, DASH & TLC Dietary Patterns: Mechanisms for CVD Reduction. Paper presented at: 2014 Clinical Lipid Update; March 16th 2014, 2014; Maui.
- Tognon G, Lissner L, Saebye D, Walker KZ, Heitmann BL. The Mediterranean diet in relation to mortality and CVD: a Danish cohort study. The British journal of nutrition. Jan 14 2014;111(1):151-159.
- Lourida I, Soni M, Thompson-Coon J, et al. Mediterranean diet, cognitive function, and dementia: a systematic review. Epidemiology (Cambridge, Mass.). Jul 2013;24(4):479-489.
- Perez-Lopez FR, Chedraui P, Haya J, Cuadros JL. Effects of the Mediterranean diet on longevity and age-related morbid conditions. Maturitas. Oct 20 2009;64(2):67-79.
- Martinez-Gonzalez MA, Salas-Salvado J, Estruch R, Corella D, Fito M, Ros E. Benefits of the Mediterranean Diet: Insights From the PREDIMED Study. Progress in cardiovascular diseases. Jul-Aug 2015;58(1):50-60.
- Palkowska E, Bartnikowska E, Owsiak D. [The use of low-caloric diet with modified fatty acids pool in the therapy of the metabolic syndrome]. Roczniki Panstwowego Zakladu Higieny. 2012;63(2):163-169.
- Trubitsyn AG. The Lag of the Proliferative Aging Clock Underlies the Lifespan-Extending Effect of Calorie Restriction. Current aging science. 2015;8(3):220-226.
- Stein PK, Soare A, Meyer TE, Cangemi R, Holloszy JO, Fontana L. Caloric restriction may reverse age-related autonomic decline in humans. Aging Cell. Aug 2012;11(4):644-650.
- McCarty MF. AMPK activation--protean potential for boosting healthspan. Age (Dordrecht, Netherlands). Apr 2014;36(2):641-663.
- Kishi T, Hirooka Y, Ogawa K, Konno S, Sunagawa K. Calorie restriction inhibits sympathetic nerve activity via anti-oxidant effect in the rostral ventrolateral medulla of obesity-induced hypertensive rats. Clinical and experimental hypertension (New York, N.Y. : 1993). 2011;33(4):240-245.
- Finckenberg P, Eriksson O, Baumann M, et al. Caloric restriction ameliorates angiotensin II-induced mitochondrial remodeling and cardiac hypertrophy. Hypertension. Jan 2012;59(1):76-84.
- de Cavanagh EM, Inserra F, Ferder L. Angiotensin II blockade: how its molecular targets may signal to mitochondria and slow aging. Coincidences with calorie restriction and mTOR inhibition. American journal of physiology. Heart and circulatory physiology. Jul 1 2015;309(1):H15-44.
- Liu Y, Chen H, Liu D. Mechanistic perspectives of calorie restriction on vascular homeostasis. Science China. Life sciences. Aug 2014;57(8):742-754.
- Jefferson ME, Nicklas BJ, Chmelo EA, et al. Effects of Resistance Training With and Without Caloric Restriction on Arterial Stiffness in Overweight and Obese Older Adults. American journal of hypertension. Aug 20 2015.
- Walford RL, Mock D, Verdery R, MacCallum T. Calorie restriction in biosphere 2: alterations in physiologic, hematologic, hormonal, and biochemical parameters in humans restricted for a 2-year period. The journals of gerontology. Series A, Biological sciences and medical sciences. Jun 2002;57(6):B211-224.
- Ravussin E, Redman LM, Rochon J, et al. A 2-Year Randomized Controlled Trial of Human Caloric Restriction: Feasibility and Effects on Predictors of Health Span and Longevity. The journals of gerontology. Series A, Biological sciences and medical sciences. Sep 2015;70(9):1097-1104.
- Riegel G, Moreira LB, Fuchs SC, et al. Long-term effectiveness of non-drug recommendations to treat hypertension in a clinical setting. American journal of hypertension. Nov 2012;25(11):1202-1208.
- Elsevier. First Consult: Hypertension.
- Perez V, Chang ET. Sodium-to-potassium ratio and blood pressure, hypertension, and related factors. Advances in nutrition (Bethesda, Md.). Nov 2014;5(6):712-741.
- Penton D, Czogalla J, Loffing J. Dietary potassium and the renal control of salt balance and blood pressure. Pflugers Arch. Mar 2015;467(3):513-530.
- Stevens VJ, Obarzanek E, Cook NR, et al. Long-term weight loss and changes in blood pressure: results of the Trials of Hypertension Prevention, phase II. Ann Intern Med. Jan 2 2001;134(1):1-11.
- Kan AW, Hussain T, Carson KA, et al. The Contribution of Age and Weight to Blood Pressure Levels Among Blacks and Whites Receiving Care in Community-Based Primary Care Practices. Preventing chronic disease. 2015;12:E161.
- Whelton SP, Chin A, Xin X, He J. Effect of aerobic exercise on blood pressure: a meta-analysis of randomized, controlled trials. Ann Intern Med. Apr 2 2002;136(7):493-503.
- Cornelissen VA, Fagard RH. Effect of resistance training on resting blood pressure: a meta-analysis of randomized controlled trials. Journal of hypertension. Feb 2005;23(2):251-259.
- Kelley GA, Kelley KS. Progressive resistance exercise and resting blood pressure : A meta-analysis of randomized controlled trials. Hypertension. Mar 2000;35(3):838-843.
- Larsen RN, Kingwell BA, Sethi P, Cerin E, Owen N, Dunstan DW. Breaking up prolonged sitting reduces resting blood pressure in overweight/obese adults. Nutrition, metabolism, and cardiovascular diseases : NMCD. Sep 2014;24(9):976-982.
- Bazzano LA, Green T, Harrison TN, Reynolds K. Dietary approaches to prevent hypertension. Current hypertension reports. Dec 2013;15(6):694-702.
- Xin X, He J, Frontini MG, Ogden LG, Motsamai OI, Whelton PK. Effects of alcohol reduction on blood pressure: a meta-analysis of randomized controlled trials. Hypertension. Nov 2001;38(5):1112-1117.
- Aljadhey H, Tu W, Hansen RA, Blalock SJ, Brater DC, Murray MD. Comparative effects of non-steroidal anti-inflammatory drugs (NSAIDs) on blood pressure in patients with hypertension. BMC cardiovascular disorders. 2012;12:93.
- Hsu CC, Wang H, Hsu YH, et al. Use of Nonsteroidal Anti-Inflammatory Drugs and Risk of Chronic Kidney Disease in Subjects With Hypertension: Nationwide Longitudinal Cohort Study. Hypertension. Sep 2015;66(3):524-533.
- Medicine AAoS. Rising prevalence of sleep apnea in U.S. threatens public health. 2014.
- Mayo Clinic. Diseases and Conditions. Sleep Apnea. http://www.mayoclinic.org/diseases-conditions/sleep-apnea/basics/complications/con-20020286. 8/25/2015. Accessed 11/19/2015.
- Liu L, Cao Q, Guo Z, Dai Q. Continuous Positive Airway Pressure in Patients With Obstructive Sleep Apnea and Resistant Hypertension: A Meta-Analysis of Randomized Controlled Trials. Journal of clinical hypertension (Greenwich, Conn.). Aug 17 2015.
- Hughes JW, Fresco DM, Myerscough R, van Dulmen MH, Carlson LE, Josephson R. Randomized controlled trial of mindfulness-based stress reduction for prehypertension. Psychosomatic medicine. Oct 2013;75(8):721-728.
- Gasperin D, Netuveli G, Dias-da-Costa JS, Pattussi MP. Effect of psychological stress on blood pressure increase: a meta-analysis of cohort studies. Cadernos de saude publica. Apr 2009;25(4):715-726.
- Larson AJ, Symons JD, Jalili T. Therapeutic potential of quercetin to decrease blood pressure: review of efficacy and mechanisms. Advances in nutrition (Bethesda, Md.). Jan 2012;3(1):39-46.
- Laskar MAC, M. D. In silico screening of some plant based natural products as angiotensin receptor blockers against cardiovascular diseases. World Journal of Pharmacy and Pharmaceutical Science. 2015;4(1):1248-1257.
- Egert S, Bosy-Westphal A, Seiberl J, et al. Quercetin reduces systolic blood pressure and plasma oxidised low-density lipoprotein concentrations in overweight subjects with a high-cardiovascular disease risk phenotype: a double-blinded, placebo-controlled cross-over study. The British journal of nutrition. Oct 2009;102(7):1065-1074.
- Pfeuffer M, Auinger A, Bley U, et al. Effect of quercetin on traits of the metabolic syndrome, endothelial function and inflammation in men with different APOE isoforms. Nutrition, metabolism, and cardiovascular diseases : NMCD. May 2013;23(5):403-409.
- Brull V, Burak C, Stoffel-Wagner B, et al. Effects of a quercetin-rich onion skin extract on 24 h ambulatory blood pressure and endothelial function in overweight-to-obese patients with (pre-)hypertension: a randomised double-blinded placebo-controlled cross-over trial. The British journal of nutrition. Oct 2015;114(8):1263-1277.
- Edwards RL, Lyon T, Litwin SE, Rabovsky A, Symons JD, Jalili T. Quercetin reduces blood pressure in hypertensive subjects. The Journal of nutrition. Nov 2007;137(11):2405-2411.
- Zahedi M, Ghiasvand R, Feizi A, Asgari G, Darvish L. Does Quercetin Improve Cardiovascular Risk factors and Inflammatory Biomarkers in Women with Type 2 Diabetes: A Double-blind Randomized Controlled Clinical Trial. International journal of preventive medicine. Jul 2013;4(7):777-785.
- Ross JA, Kasum CM. Dietary flavonoids: bioavailability, metabolic effects, and safety. Annual review of nutrition. 2002;22:19-34.
- Basli A, Soulet S, Chaher N, et al. Wine polyphenols: potential agents in neuroprotection. Oxidative medicine and cellular longevity. 2012;2012:805762.
- Godse S, Mohan M, Kasture V, Kasture S. Effect of myricetin on blood pressure and metabolic alterations in fructose hypertensive rats. Pharmaceutical biology. May 2010;48(5):494-498.
- Borde P, Mohan M, Kasture S. Effect of myricetin on deoxycorticosterone acetate (DOCA)-salt-hypertensive rats. Natural product research. Sep 2011;25(16):1549-1559.
- Hobbs CA, Swartz C, Maronpot R, et al. Genotoxicity evaluation of the flavonoid, myricitrin, and its aglycone, myricetin. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association. Sep 2015;83:283-292.
- Wang SJ, Tong Y, Lu S, et al. Anti-inflammatory activity of myricetin isolated from Myrica rubra Sieb. et Zucc. leaves. Planta medica. Oct 2010;76(14):1492-1496.
- Domitrovic R, Rashed K, Cvijanovic O, Vladimir-Knezevic S, Skoda M, Visnic A. Myricitrin exhibits antioxidant, anti-inflammatory and antifibrotic activity in carbon tetrachloride-intoxicated mice. Chemico-biological interactions. Mar 25 2015;230:21-29.
- Fernandez SP, Nguyen M, Yow TT, et al. The flavonoid glycosides, myricitrin, gossypin and naringin exert anxiolytic action in mice. Neurochemical research. Oct 2009;34(10):1867-1875.
- Sales DS, Carmona F, de Azevedo BC, et al. Eugenia punicifolia (Kunth) DC. as an adjuvant treatment for type-2 diabetes mellitus: a non-controlled, pilot study. Phytotherapy research : PTR. Dec 2014;28(12):1816-1821.
- Soejarto DD, Kinghorn AD, Farnsworth NR. Potential Sweetening Agents of Plant Origin. III. Organoleptic Evaluation of Stevia Leaf Herbarium Samples For Sweetness. Journal of natural products. 1982/09/01 1982;45(5):590-599.
- Gardana C, Simonetti P, Canzi E, Zanchi R, Pietta P. Metabolism of stevioside and rebaudioside A from Stevia rebaudiana extracts by human microflora. Journal of agricultural and food chemistry. Oct 22 2003;51(22):6618-6622.
- Liu JC, Kao PK, Chan P, et al. Mechanism of the antihypertensive effect of stevioside in anesthetized dogs. Pharmacology. Jan 2003;67(1):14-20.
- Melis MS. Influence of calcium on the blood pressure and renal effects of stevioside. Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas / Sociedade Brasileira de Biofisica ... [et al.]. 1992;25(9):943-949.
- Melis MS. Stevioside effect on renal function of normal and hypertensive rats. Journal of ethnopharmacology. Jun 1992;36(3):213-217.
- Onakpoya IJ, Heneghan CJ. Effect of the natural sweetener, steviol glycoside, on cardiovascular risk factors: A systematic review and meta-analysis of randomised clinical trials. European journal of preventive cardiology. Dec 2015;22(12):1575-1587.
- Altun A, Ugur-Altun B. Melatonin: therapeutic and clinical utilization. International journal of clinical practice. May 2007;61(5):835-845.
- Pechanova O, Paulis L, Simko F. Peripheral and Central Effects of Melatonin on Blood Pressure Regulation. International journal of molecular sciences. 10/08 07/03/received 09/17/revised 09/17/accepted 2014;15(10):17920-17937.
- Rodella LF, Favero G, Foglio E, et al. Vascular endothelial cells and dysfunctions: role of melatonin. Frontiers in bioscience (Elite edition). 2013;5:119-129.
- Li Y, Wang JG. Isolated nocturnal hypertension: a disease masked in the dark. Hypertension. Feb 2013;61(2):278-283.
- Kimura G. [Nocturnal hypertension and cardio-renal association]. Nihon rinsho. Japanese journal of clinical medicine. Aug 2014;72(8):1404-1409.
- Li F, Huang H, Song L, Hao H, Ying M. Effects of Obstructive Sleep Apnea Hypopnea Syndrome on Blood Pressure and C-Reactive Protein in Male Hypertension Patients. J Clin Med Res. Mar 2016;8(3):220-224.
- Grossman E, Laudon M, Zisapel N. Effect of melatonin on nocturnal blood pressure: meta-analysis of randomized controlled trials. Vasc Health Risk Manag. 2011;7:577-584.
- Ho MJ, Bellusci A, Wright JM. Blood pressure lowering efficacy of coenzyme Q10 for primary hypertension. The Cochrane database of systematic reviews. 2009(4):Cd007435.
- Wyman M, Leonard M, Morledge T. Coenzyme Q10: a therapy for hypertension and statin-induced myalgia? Cleveland Clinic journal of medicine. Jul 2010;77(7):435-442.
- Mortensen SA, Rosenfeldt F, Kumar A, et al. The Effect of Coenzyme Q on Morbidity and Mortality in Chronic Heart Failure: Results From Q-SYMBIO: A Randomized Double-Blind Trial. JACC. Heart failure. Sep 25 2014.
- Gao L, Mao Q, Cao J, Wang Y, Zhou X, Fan L. Effects of coenzyme Q10 on vascular endothelial function in humans: a meta-analysis of randomized controlled trials. Atherosclerosis. Apr 2012;221(2):311-316.
- Hamilton SJ, Chew GT, Watts GF. Coenzyme Q10 Improves Endothelial Dysfunction in Statin-Treated Type 2 Diabetic Patients. Diabetes care. May 1, 2009 2009;32(5):810-812.
- Kedziora-Kornatowska K, Czuczejko J, Motyl J, et al. Effects of coenzyme Q10 supplementation on activities of selected antioxidative enzymes and lipid peroxidation in hypertensive patients treated with indapamide. A pilot study. Archives of medical science : AMS. Aug 30 2010;6(4):513-518.
- Alehagen U, Johansson P, Björnstedt M, Rosén A, Dahlström U. Cardiovascular mortality and N-terminal-proBNP reduced after combined selenium and coenzyme Q10 supplementation: A 5-year prospective randomized double-blind placebo-controlled trial among elderly Swedish citizens. International journal of cardiology. 9/1/ 2013;167(5):1860-1866.
- Jee SH, Miller ER, 3rd, Guallar E, Singh VK, Appel LJ, Klag MJ. The effect of magnesium supplementation on blood pressure: a meta-analysis of randomized clinical trials. American journal of hypertension. Aug 2002;15(8):691-696.
- Shechter M, Shechter A. The Role of Magnesium in the Cardiovascular System. In: Watson RR, Preedy VR, Zibadi S, eds. Magnesium in Human Health and Disease: Humana Press; 2013:191-204.
- Houston M. Magnesium and Hypertension. In: Watson RR, Preedy VR, Zibadi S, eds. Magnesium in Human Health and Disease: Humana Press; 2013:183-189.
- Rosanoff A. Magnesium supplements may enhance the effect of antihypertensive medications in stage 1 hypertensive subjects. Magnesium research : official organ of the International Society for the Development of Research on Magnesium. Mar 2010;23(1):27-40.
- Qu X, Jin F, Hao Y, et al. Magnesium and the risk of cardiovascular events: a meta-analysis of prospective cohort studies. PloS one. 2013;8(3):e57720.
- Grober U, Schmidt J, Kisters K. Magnesium in Prevention and Therapy. Nutrients. 2015;7(9):8199-8226.
- Rosanoff A, Plesset MR. Oral magnesium supplements decrease high blood pressure (SBP>155 mmHg) in hypertensive subjects on anti-hypertensive medications: a targeted meta-analysis. Magnesium research : official organ of the International Society for the Development of Research on Magnesium. Jul-Sep 2013;26(3):93-99.
- Tin A, Grams ME, Maruthur NM, et al. Results from the Atherosclerosis Risk in Communities study suggest that low serum magnesium is associated with incident kidney disease. Kidney international. 04//print 2015;87(4):820-827.
- Witkowski M, Hubert J, Mazur A. Methods of assessment of magnesium status in humans: a systematic review. Magnesium research : official organ of the International Society for the Development of Research on Magnesium. Dec 2011;24(4):163-180.
- Gold Standard. Drug Monograph. Lisinopril. www.clinicalkey.com. Copyright 2015 by Elsevier, Inc. Last updated 8/5/2014. Accessed 11/19/2015.
- Gold Standard. Drug Monograph. Candesartan. www.clinicalkey.com. Copyright 2015 by Elsevier, Inc. Last updated 6/15/2012. Accessed 11/19/2015.
- Pal S, Ellis V. The chronic effects of whey proteins on blood pressure, vascular function, and inflammatory markers in overweight individuals. Obesity (Silver Spring, Md.). Jul 2010;18(7):1354-1359.
- Li SH, Tian HB, Zhao HJ, Chen LH, Cui LQ. The acute effects of grape polyphenols supplementation on endothelial function in adults: meta-analyses of controlled trials. PloS one. 2013;8(7):e69818.
- Li SH, Zhao P, Tian HB, Chen LH, Cui LQ. Effect of Grape Polyphenols on Blood Pressure: A Meta-Analysis of Randomized Controlled Trials. PloS one. 2015;10(9):e0137665.
- Edirisinghe I, Burton-Freeman B, Tissa Kappagoda C. Mechanism of the endothelium-dependent relaxation evoked by a grape seed extract. Clinical science (London, England : 1979). Feb 2008;114(4):331-337.
- Asgary S, Keshvari M, Sahebkar A, Hashemi M, Rafieian-Kopaei M. Clinical investigation of the acute effects of pomegranate juice on blood pressure and endothelial function in hypertensive individuals. ARYA atherosclerosis. Nov 2013;9(6):326-331.
- Asgary S, Sahebkar A, Afshani MR, Keshvari M, Haghjooyjavanmard S, Rafieian-Kopaei M. Clinical evaluation of blood pressure lowering, endothelial function improving, hypolipidemic and anti-inflammatory effects of pomegranate juice in hypertensive subjects. Phytotherapy research : PTR. Feb 2014;28(2):193-199.
- Tsang C, Smail NF, Almoosawi S, Davidson I, Al-Dujaili EA. Intake of polyphenol-rich pomegranate pure juice influences urinary glucocorticoids, blood pressure and homeostasis model assessment of insulin resistance in human volunteers. Journal of nutritional science. 2012;1:e9.
- Aviram M, Dornfeld L. Pomegranate juice consumption inhibits serum angiotensin converting enzyme activity and reduces systolic blood pressure. Atherosclerosis. Sep 2001;158(1):195-198.
- Aviram M, Rosenblat M, Gaitini D, et al. Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clinical nutrition (Edinburgh, Scotland). Jun 2004;23(3):423-433.
- de Nigris F, Balestrieri ML, Williams-Ignarro S, et al. The influence of pomegranate fruit extract in comparison to regular pomegranate juice and seed oil on nitric oxide and arterial function in obese Zucker rats. Nitric Oxide. Aug 2007;17(1):50-54.
- Mohan M, Waghulde H, Kasture S. Effect of pomegranate juice on Angiotensin II-induced hypertension in diabetic Wistar rats. Phytotherapy research : PTR. Jun 2010;24 Suppl 2:S196-203.
- Susalit E, Agus N, Effendi I, et al. Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: comparison with Captopril. Phytomedicine : international journal of phytotherapy and phytopharmacology. Feb 15 2011;18(4):251-258.
- Cherif S, Rahal N, Haouala M, et al. [A clinical trial of a titrated Olea extract in the treatment of essential arterial hypertension]. J Pharm Belg. Mar-Apr 1996;51(2):69-71.
- Perrinjaquet-Moccetti T, Busjahn A, Schmidlin C, Schmidt A, Bradl B, Aydogan C. Food supplementation with an olive (Olea europaea L.) leaf extract reduces blood pressure in borderline hypertensive monozygotic twins. Phytotherapy research : PTR. Sep 2008;22(9):1239-1242.
- de Bock M, Derraik JG, Brennan CM, et al. Olive (Olea europaea L.) leaf polyphenols improve insulin sensitivity in middle-aged overweight men: a randomized, placebo-controlled, crossover trial. PloS one. 2013;8(3):e57622.
- Scheffler A, Rauwald HW, Kampa B, Mann U, Mohr FW, Dhein S. Olea europaea leaf extract exerts L-type Ca(2+) channel antagonistic effects. Journal of ethnopharmacology. Nov 20 2008;120(2):233-240.
- Gilani AH, Khan AU, Shah AJ, Connor J, Jabeen Q. Blood pressure lowering effect of olive is mediated through calcium channel blockade. International journal of food sciences and nutrition. Dec 2005;56(8):613-620.
- Hansen K, Adsersen A, Christensen SB, Jensen SR, Nyman U, Smitt UW. Isolation of an angiotensin converting enzyme (ACE) inhibitor from Olea europaea and Olea lancea. Phytomedicine : international journal of phytotherapy and phytopharmacology. Mar 1996;2(4):319-325.
- Parzonko A, Czerwinska ME, Kiss AK, Naruszewicz M. Oleuropein and oleacein may restore biological functions of endothelial progenitor cells impaired by angiotensin II via activation of Nrf2/heme oxygenase-1 pathway. Phytomedicine : international journal of phytotherapy and phytopharmacology. Sep 15 2013;20(12):1088-1094.
- Madhavi D, Kagan D, Rao V. A Pilot Study to Evaluate the Antihypertensive Effect of a Celery Extract in Mild to Moderate Hypertensive Patients. Age. 2013;57:10.13.
- Moghadam MH, Imenshahidi M, Mohajeri SA. Antihypertensive effect of celery seed on rat blood pressure in chronic administration. Journal of medicinal food. Jun 2013;16(6):558-563.
- Ko FN, Huang TF, Teng CM. Vasodilatory action mechanisms of apigenin isolated from Apium graveolens in rat thoracic aorta. Biochimica et biophysica acta. Nov 14 1991;1115(1):69-74.
- Jorge VG, Angel JR, Adrian TS, et al. Vasorelaxant activity of extracts obtained from Apium graveolens: possible source for vasorelaxant molecules isolation with potential antihypertensive effect. Asian Pac J Trop Biomed. Oct 2013;3(10):776-779.
- Hamet P. The evaluation of the scientific evidence for a relationship between calcium and hypertension. The Journal of nutrition. Feb 1995;125(2 Suppl):311s-400s.
- van Mierlo LA, Arends LR, Streppel MT, et al. Blood pressure response to calcium supplementation: a meta-analysis of randomized controlled trials. Journal of human hypertension. Aug 2006;20(8):571-580.
- Geleijnse JM, Vermeer C, Grobbee DE, et al. Dietary intake of menaquinone is associated with a reduced risk of coronary heart disease: the Rotterdam Study. The Journal of nutrition. Nov 2004;134(11):3100-3105.
- Chow CK. Dietary intake of menaquinones and risk of cancer incidence and mortality. The American journal of clinical nutrition. December 1, 2010 2010;92(6):1533-1534.
- Shea MK, O'Donnell CJ, Hoffmann U, et al. Vitamin K supplementation and progression of coronary artery calcium in older men and women. The American journal of clinical nutrition. Jun 2009;89(6):1799-1807.
- DiNicolantonio JJ, Bhutani J, O'Keefe JH. The health benefits of vitamin K. Open Heart. 10/06 06/01/received 08/27/revised 09/18/accepted 2015;2(1):e000300.
- Shea MK, Holden RM. Vitamin K status and vascular calcification: evidence from observational and clinical studies. Advances in nutrition (Bethesda, Md.). Mar 2012;3(2):158-165.
- Dong JY, Qin LQ, Zhang Z, et al. Effect of oral L-arginine supplementation on blood pressure: a meta-analysis of randomized, double-blind, placebo-controlled trials. American heart journal. Dec 2011;162(6):959-965.
- Kozuma K, Tsuchiya S, Kohori J, Hase T, Tokimitsu I. Antihypertensive effect of green coffee bean extract on mildly hypertensive subjects. Hypertension research : official journal of the Japanese Society of Hypertension. Sep 2005;28(9):711-718.
- Yamaguchi T, Chikama A, Mori K, et al. Hydroxyhydroquinone-free coffee: a double-blind, randomized controlled dose-response study of blood pressure. Nutrition, metabolism, and cardiovascular diseases : NMCD. Jul 2008;18(6):408-414.
- Wang J, Xiong X, Feng B. Effect of crataegus usage in cardiovascular disease prevention: an evidence-based approach. Evidence-based complementary and alternative medicine : eCAM. 2013;2013:149363.
- Liu Y, Ma W, Zhang P, He S, Huang D. Effect of resveratrol on blood pressure: a meta-analysis of randomized controlled trials. Clinical nutrition (Edinburgh, Scotland). Feb 2015;34(1):27-34.
- Izzo JL, Jr., Weir MR. Angiotensin-converting enzyme inhibitors. Journal of clinical hypertension (Greenwich, Conn.). Sep 2011;13(9):667-675.
- Taylor AA, Siragy H, Nesbitt S. Angiotensin receptor blockers: pharmacology, efficacy, and safety. Journal of clinical hypertension (Greenwich, Conn.). Sep 2011;13(9):677-686.
- Li EC, Heran BS, Wright JM. Angiotensin converting enzyme (ACE) inhibitors versus angiotensin receptor blockers for primary hypertension. The Cochrane database of systematic reviews. 2014;8:Cd009096.
- Akhrass PR, McFarlane SI. Telmisartan and cardioprotection. Vascular Health and Risk Management. 11/15 2011;7:677-683.
- Lacourciere Y, Krzesinski JM, White WB, Davidai G, Schumacher H. Sustained antihypertensive activity of telmisartan compared with valsartan. Blood pressure monitoring. Aug 2004;9(4):203-210.
- Littlejohn T, Mroczek W, Marbury T, VanderMaelen CP, Dubiel RF. A prospective, randomized, open-label trial comparing telmisartan 80 mg with valsartan 80 mg in patients with mild to moderate hypertension using ambulatory blood pressure monitoring. The Canadian journal of cardiology. Sep 2000;16(9):1123-1132.
- Lacourciere Y, Lenis J, Orchard R, et al. A comparison of the efficacies and duration of action of the angiotensin II receptor blockers telmisartan and amlodipine. Blood pressure monitoring. Oct 1998;3(5):295-302.
- Benson SC, Pershadsingh HA, Ho CI, et al. Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARgamma-modulating activity. Hypertension. May 2004;43(5):993-1002.
- Schupp M, Janke J, Clasen R, Unger T, Kintscher U. Angiotensin type 1 receptor blockers induce peroxisome proliferator-activated receptor-gamma activity. Circulation. May 4 2004;109(17):2054-2057.
- Berger JP, Petro AE, Macnaul KL, et al. Distinct properties and advantages of a novel peroxisome proliferator-activated protein [gamma] selective modulator. Molecular endocrinology (Baltimore, Md.). Apr 2003;17(4):662-676.
- Takeuchi K, Yamamoto K, Ohishi M, et al. Telmisartan modulates mitochondrial function in vascular smooth muscle cells. Hypertension research : official journal of the Japanese Society of Hypertension. May 2013;36(5):433-439.
- He H, Yang D, Ma L, et al. Telmisartan prevents weight gain and obesity through activation of peroxisome proliferator-activated receptor-delta-dependent pathways. Hypertension. Apr 2010;55(4):869-879.
- Scalera F, Martens-Lobenhoffer J, Bukowska A, Lendeckel U, Tager M, Bode-Boger SM. Effect of telmisartan on nitric oxide--asymmetrical dimethylarginine system: role of angiotensin II type 1 receptor gamma and peroxisome proliferator activated receptor gamma signaling during endothelial aging. Hypertension. Mar 2008;51(3):696-703.
- Feng X, Luo Z, Ma L, et al. Angiotensin II receptor blocker telmisartan enhances running endurance of skeletal muscle through activation of the PPAR-delta/AMPK pathway. Journal of cellular and molecular medicine. Jul 2011;15(7):1572-1581.
- Sanchis-Gomar F, Lippi G. Telmisartan as metabolic modulator: a new perspective in sports doping? Journal of strength and conditioning research / National Strength & Conditioning Association. Mar 2012;26(3):608-610.
- Sugimoto K, Qi NR, Kazdova L, Pravenec M, Ogihara T, Kurtz TW. Telmisartan but not valsartan increases caloric expenditure and protects against weight gain and hepatic steatosis. Hypertension. May 2006;47(5):1003-1009.
- Goyal SN, Bharti S, Bhatia J, Nag TC, Ray R, Arya DS. Telmisartan, a dual ARB/partial PPAR-gamma agonist, protects myocardium from ischaemic reperfusion injury in experimental diabetes. Diabetes, obesity & metabolism. Jun 2011;13(6):533-541.
- Iwai M, Inaba S, Tomono Y, et al. Attenuation of focal brain ischemia by telmisartan, an angiotensin II type 1 receptor blocker, in atherosclerotic apolipoprotein E-deficient mice. Hypertension research : official journal of the Japanese Society of Hypertension. Jan 2008;31(1):161-168.
- Iwanami J, Mogi M, Tsukuda K, et al. Low dose of telmisartan prevents ischemic brain damage with peroxisome proliferator-activated receptor-gamma activation in diabetic mice. Journal of hypertension. Aug 2010;28(8):1730-1737.
- Yuen CY, Wong WT, Tian XY, et al. Telmisartan inhibits vasoconstriction via PPARgamma-dependent expression and activation of endothelial nitric oxide synthase. Cardiovascular research. Apr 1 2011;90(1):122-129.
- Myojo M, Nagata D, Fujita D, et al. Telmisartan activates endothelial nitric oxide synthase via Ser1177 phosphorylation in vascular endothelial cells. PloS one. 2014;9(5):e96948.
- de Araujo Junior RF, Leitao Oliveira AL, de Melo Silveira RF, de Oliveira Rocha HA, de Franca Cavalcanti P, de Araujo AA. Telmisartan induces apoptosis and regulates Bcl-2 in human renal cancer cells. Experimental biology and medicine (Maywood, N.J.). Jan 2015;240(1):34-44.
- Koyama N, Nishida Y, Ishii T, Yoshida T, Furukawa Y, Narahara H. Telmisartan induces growth inhibition, DNA double-strand breaks and apoptosis in human endometrial cancer cells. PloS one. 2014;9(3):e93050.
- Lee LD, Mafura B, Lauscher JC, Seeliger H, Kreis ME, Grone J. Antiproliferative and apoptotic effects of telmisartan in human colon cancer cells. Oncology letters. Dec 2014;8(6):2681-2686.
- Pu Z, Zhu M, Kong F. Telmisartan prevents proliferation and promotes apoptosis of human ovarian cancer cells through upregulating PPARgamma and downregulating MMP9 expression. Molecular medicine reports. Nov 6 2015.
- Ferri FF. Ferri's Clinical Advisor. Disease Overview: Hypertension. Copyright 2016 by Elsevier, Inc. www.clinicalkey.com. 11/19/2015.
- Moser M, Feig PU. FIfty years of thiazide diuretic therapy for hypertension. Archives of internal medicine. 2009;169(20):1851-1856.
- ePocrates. Drugs. hydrochlorothiazide. Adverse Reactions. https://online.epocrates.com/drugs/22205/hydrochlorothiazide/Adverse-Reactions. Accessed 11/19/2015.
- Rimoldi SF, Messerli FH, Chavez P, Stefanini GG, Scherrer U. Efficacy and safety of calcium channel blocker/diuretics combination therapy in hypertensive patients: a meta-analysis.
- Spindler SR, Mote PL, Li R, et al. beta1-Adrenergic receptor blockade extends the life span of Drosophila and long-lived mice. Age (Dordrecht, Netherlands). Dec 2013;35(6):2099-2109.
- Gomez A, Sanchez-Roman I, Gomez J, et al. Lifelong treatment with atenolol decreases membrane fatty acid unsaturation and oxidative stress in heart and skeletal muscle mitochondria and improves immunity and behavior, without changing mice longevity. Aging Cell. Jun 2014;13(3):551-560.
- NPS MEDICINEWISE. Medical tests. ACE and ARB - reductions in GFR. http://www.nps.org.au/medical-tests/tests-by-conditions/for-individuals/kidney-problems-and-disorders-tests/for-health-professionals/medicines-to-be-aware-of/ace-arb-reductions-in-gfr#References. 6/30/2012. Accessed 11/20/2015.
- Schoolwerth A. Cautions with Angiotensin Converting Enzyme Inhibitor (ACEI) and Angiotensin Receptor Blocker (ARB) Therapy. CKD Update: Attacking Chronic Kidney Disease. Vol. 1; Issue 3. Copyright 2004 by The National Kidney Foundation. http://www.imakenews.com/ckdupdate/e_article000327587.cfm?x=b11,0,w. Accessed 11/20/2015.
- Weber MA, Schiffrin EL, White WB, et al. Clinical practice guidelines for the management of hypertension in the community a statement by the American Society of Hypertension and the International Society of Hypertension. Journal of hypertension. Jan 2014;32(1):3-15.
- Hermida RC, Ayala DE, Mojon A, Fernandez JR. Bedtime dosing of antihypertensive medications reduces cardiovascular risk in CKD. Journal of the American Society of Nephrology : JASN. Dec 2011;22(12):2313-2321.
- Hermida RC, Ayala DE. Chronotherapy with the angiotensin-converting enzyme inhibitor ramipril in essential hypertension: improved blood pressure control with bedtime dosing. Hypertension. Jul 2009;54(1):40-46.
- Hermida RC, Ayala DE, Fontao MJ, Mojon A, Fernandez JR. Chronotherapy with valsartan/amlodipine fixed combination: improved blood pressure control of essential hypertension with bedtime dosing. Chronobiology international. Jul 2010;27(6):1287-1303.
- Hermida RC, Ayala DE, Mojon A, Fernandez JR. Chronotherapy with nifedipine GITS in hypertensive patients: improved efficacy and safety with bedtime dosing. American journal of hypertension. Aug 2008;21(8):948-954.
- Hermida RC, Ayala DE, Mojon A, Fontao MJ, Fernandez JR. Chronotherapy with valsartan/hydrochlorothiazide combination in essential hypertension: improved sleep-time blood pressure control with bedtime dosing. Chronobiology international. Aug 2011;28(7):601-610.
- Zeng J, Jia M, Ran H, et al. Fixed-combination of amlodipine and diuretic chronotherapy in the treatment of essential hypertension: improved blood pressure control with bedtime dosing-a multicenter, open-label randomized study. Hypertension research : official journal of the Japanese Society of Hypertension. Jun 2011;34(6):767-772.
- Cruickshank JM. The role of coronary perfusion pressure. European heart journal. Sep 1992;13 Suppl D:39-43.
- Owens P, O'Brien E. Hypotension in patients with coronary disease: can profound hypotensive events cause myocardial ischaemic events? Heart (British Cardiac Society). Oct 1999;82(4):477-481.
- Voudris V, Avramides D, Gatzov P, Malakos J, Skoularigis J, Manolis A, . . . Cokkinos DV. The effect of rapid decreases of blood pressure by different mechanisms on coronary flow and flow reserve in normal coronary arteries. American journal of hypertension. Dec 2003;16(12):1000-1005.
- Bangalore S, Messerli FH, Wun CC, Zuckerman AL, DeMicco D, Kostis JB, LaRosa JC. J-curve revisited: An analysis of blood pressure and cardiovascular events in the Treating to New Targets (TNT) Trial. European heart journal. Dec 2010;31(23):2897-2908.
- Lanza CM. CITRUS FRUITS | Processed and Derived Products of Oranges. In: Caballero B, ed. Encyclopedia of Food Sciences and Nutrition (Second Edition). Oxford: Academic Press; 2003:1346-1354.
- Benavente-Garcia O, Castillo J. Update on uses and properties of citrus flavonoids: new findings in anticancer, cardiovascular, and anti-inflammatory activity. J Agric Food Chem. Aug 13 2008;56(15):6185-6205.
- Rizza S, Muniyappa R, Iantorno M, Kim JA, Chen H, Pullikotil P, . . . Quon MJ. Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J Clin Endocrinol Metab. May 2011;96(5):E782-792.
- Roohbakhsh A, Parhiz H, Soltani F, Rezaee R, Iranshahi M. Molecular mechanisms behind the biological effects of hesperidin and hesperetin for the prevention of cancer and cardiovascular diseases. Life Sci. Mar 1 2015;124:64-74.
- Homayouni F, Haidari F, Hedayati M, Zakerkish M, Ahmadi K. Blood pressure lowering and anti-inflammatory effects of hesperidin in type 2 diabetes; a randomized double-blind controlled clinical trial. Phytotherapy research: PTR. Jun 2018;32(6):1073-1079.
- Williamson G. The role of polyphenols in modern nutrition. Nutrition Bulletin / Bnf. Sep 2017;42(3):226-235.
- Milenkovic D, Deval C, Dubray C, Mazur A, Morand C. Hesperidin displays relevant role in the nutrigenomic effect of orange juice on blood leukocytes in human volunteers: a randomized controlled cross-over study. PLoS One. 2011;6(11):e26669.
- Morand C, Dubray C, Milenkovic D, Lioger D, Martin JF, Scalbert A, Mazur A. Hesperidin contributes to the vascular protective effects of orange juice: a randomized crossover study in healthy volunteers. The American journal of clinical nutrition. Jan 2011;93(1):73-80.
- Yamamoto M, Jokura H, Suzuki A, Hase T, Shimotoyodome A. Effects of continuous ingestion of hesperidin and glucosyl hesperidin on vascular gene expression in spontaneously hypertensive rats. Journal of nutritional science and vitaminology. 2013;59(5):470-473.
- Ouyang A, Garner TB, Fleenor BS. Hesperidin reverses perivascular adipose-mediated aortic stiffness with aging. Experimental gerontology. Oct 15 2017;97:68-72.
- Chiou CS, Lin JW, Kao PF, Liu JC, Cheng TH, Chan P. Effects of hesperidin on cyclic strain-induced endothelin-1 release in human umbilical vein endothelial cells. Clinical and experimental pharmacology & physiology. Aug 2008;35(8):938-943.
- Liu L, Xu DM, Cheng YY. Distinct effects of naringenin and hesperetin on nitric oxide production from endothelial cells. Journal of agricultural and food chemistry. Feb 13 2008;56(3):824-829.
- Li Q, Youn JY, Cai H. Mechanisms and consequences of endothelial nitric oxide synthase dysfunction in hypertension. Journal of hypertension. Jun 2015;33(6):1128-1136.
- Ikemura M, Sasaki Y, Giddings JC, Yamamoto J. Preventive effects of hesperidin, glucosyl hesperidin and naringin on hypertension and cerebral thrombosis in stroke-prone spontaneously hypertensive rats. Phytotherapy research : PTR. Sep 2012;26(9):1272-1277.
- Mastantuono T, Battiloro L, Sabatino L, Chiurazzi M, Di Maro M, Muscariello E, . . . Lapi D. Effects of Citrus Flavonoids Against Microvascular Damage Induced by Hypoperfusion and Reperfusion in Rat Pial Circulation. Microcirculation (New York, N.Y. : 1994). Jul 2015;22(5):378-390.
- Dobias L, Petrova M, Vojtko R, Kristova V. Long-term Treatment with Hesperidin Improves Endothelium-dependent Vasodilation in Femoral Artery of Spontaneously Hypertensive Rats: The Involvement of NO-synthase and Kv Channels. Phytotherapy research : PTR. Oct 2016;30(10):1665-1671.
- Calderone V, Chericoni S, Martinelli C, Testai L, Nardi A, Morelli I, . . . Martinotti E. Vasorelaxing effects of flavonoids: investigation on the possible involvement of potassium channels. Naunyn-Schmiedeberg's archives of pharmacology. Oct 2004;370(4):290-298.
- Guirro M, Costa A, Gual-Grau A, Mayneris-Perxachs J, Torrell H, Herrero P, . . . Arola L. Multi-omics approach to elucidate the gut microbiota activity: Metaproteomics and metagenomics connection. Electrophoresis. Jul 2018;39(13):1692-1701.
- de Boer IH, Bangalore S, Benetos A, et al. Diabetes and Hypertension: A Position Statement by the American Diabetes Association. Diabetes Care. 2017;40(9):1273-1284.
- Benetos A, Petrovic M, Strandberg T. Hypertension Management in Older and Frail Older Patients. Circulation Research. 2019;124(7):1045-1060.
- AHA. Amerian Heart Association. Hypertensive Crisis: When You Should Call 911 for High Blood Pressure. https://www.heart.org/en/health-topics/high-blood-pressure/understanding-blood-pressure-readings/hypertensive-crisis-when-you-should-call-911-for-high-blood-pressure. Updated 11/30/2017. Accessed 10/29/2020.
- Pioli MR, Ritter AM, de Faria AP, Modolo R. White coat syndrome and its variations: differences and clinical impact. Integrated blood pressure control. 2018;11:73-79.
- Muntner P, Shimbo D, Carey RM, et al. Measurement of Blood Pressure in Humans: A Scientific Statement From the American Heart Association. Hypertension. 2019;73(5):e35-e66.
- Drawz PE, Agarwal A, Dwyer JP, et al. Concordance Between Blood Pressure in the Systolic Blood Pressure Intervention Trial and in Routine Clinical Practice. JAMA Intern Med. 2020.
- Ostertag LM, Kroon PA, Wood S, et al. Flavan-3-ol-enriched dark chocolate and white chocolate improve acute measures of platelet function in a gender-specific way--a randomized-controlled human intervention trial. Mol Nutr Food Res. 2013;57(2):191-202.
- Saarenhovi M, Salo P, Scheinin M, et al. The effect of an apple polyphenol extract rich in epicatechin and flavan-3-ol oligomers on brachial artery flow-mediated vasodilatory function in volunteers with elevated blood pressure. Nutr J. 2017;16(1):73.
- Ottaviani JI, Britten A, Lucarelli D, et al. Biomarker-estimated flavan-3-ol intake is associated with lower blood pressure in cross-sectional analysis in EPIC Norfolk. Sci Rep. 2020;10(1):17964.
- MacIntyre IM, Turtle EJ, Farrah TE, et al. Regular Acetaminophen Use and Blood Pressure in People With Hypertension: The PATH-BP Trial. Circulation. Feb 8 2022;145(6):416-423. doi:10.1161/CIRCULATIONAHA.121.056015
- Schjerning AM, McGettigan P, Gislason G. Cardiovascular effects and safety of (non-aspirin) NSAIDs. Nature reviews Cardiology. Sep 2020;17(9):574-584. doi:10.1038/s41569-020-0366-z
- Townsend RR. NSAIDs and acetaminophen: Effects on blood pressure and hypertension. UpToDate. Updated 3/14/2022. Accessed 3/17/2022, https://www.uptodate.com/contents/nsaids-and-acetaminophen-effects-on-blood-pressure-and-hypertension#H2
- Baker M, Perazella MA. NSAIDs in CKD: Are They Safe? American Journal of Kidney Diseases. 2020;76(4):546-557. doi:10.1053/j.ajkd.2020.03.023
- Luciano R, Perazella MA. NSAIDs: Acute kidney injury. UpToDate. Updated 11/18/2021. https://www.uptodate.com/contents/nsaids-acute-kidney-injury
- MEDSAFE. NSAIDs and Acute Kidney Injury. Updated 6/6/2013. Accessed 3/17/2022, https://www.medsafe.govt.nz/profs/PUArticles/June2013NSAIDS.htm#5
- Zeng C, Rosenberg L, Li X, et al. Sodium-containing acetaminophen and cardiovascular outcomes in individuals with and without hypertension. European Heart Journal. 2022:ehac059. doi:10.1093/eurheartj/ehac059
- Unger T, Borghi C, Charchar F, et al. 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension. Jun 2020;75(6):1334-1357. doi:10.1161/HYPERTENSIONAHA.120.15026
- MedlinePlus. High blood pressure - medicine-related. National Library of Medicine. Updated 2/18/2022. Accessed 3/17/2022, https://medlineplus.gov/ency/article/000155.htm
- Drenjancevic I, Pitha J. Omega-3 Polyunsaturated Fatty Acids-Vascular and Cardiac Effects on the Cellular and Molecular Level (Narrative Review). Int J Mol Sci. Feb 14 2022;23(4)doi:10.3390/ijms23042104. https://www.ncbi.nlm.nih.gov/pubmed/35216214
- Ferreira I, Falcato F, Bandarra N, Rauter AP. Resolvins, Protectins, and Maresins: DHA-Derived Specialized Pro-Resolving Mediators, Biosynthetic Pathways, Synthetic Approaches, and Their Role in Inflammation. Molecules. Mar 3 2022;27(5)doi:10.3390/molecules27051677. https://www.ncbi.nlm.nih.gov/pubmed/35268778
- Zhang X, Ritonja JA, Zhou N, Chen BE, Li X. Omega-3 Polyunsaturated Fatty Acids Intake and Blood Pressure: A Dose-Response Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. Jun 1 2022:e025071. doi:10.1161/JAHA.121.025071. https://www.ncbi.nlm.nih.gov/pubmed/35647665
- Smithers GW. Whey and whey proteins—From ‘gutter-to-gold’. International Dairy Journal. 2008/07/01/ 2008;18(7):695-704. doi:https://doi.org/10.1016/j.idairyj.2008.03.008
- Car H, Koprowicz T, Tokajuk A, Tokajuk A. [The effect of natural whey proteins on mechanisms of blood pressure regulation]. Postepy Hig Med Dosw (Online). Feb 6 2014;68:172-8. Wpływ naturalnych białek serwatki na mechanizmy regulacji ciśnienia tętniczego krwi. doi:10.5604/17322693.1088752
- Goulding DA, Fox PF, O’Mahony JA. Chapter 2 - Milk proteins: An overview. In: Boland M, Singh H, eds. Milk Proteins (Third Edition). Academic Press; 2020:21-98.
- Pal S, Radavelli-Bagatini S. The effects of whey protein on cardiometabolic risk factors. Obesity Reviews. 2013;14(4):324-343. doi:https://doi.org/10.1111/obr.12005
- Badely M, Sepandi M, Samadi M, Parastouei K, Taghdir M. The effect of whey protein on the components of metabolic syndrome in overweight and obese individuals; a systematic review and meta-analysis. Diabetes Metab Syndr. Nov-Dec 2019;13(6):3121-3131. doi:10.1016/j.dsx.2019.11.001
- Wirunsawanya K, Upala S, Jaruvongvanich V, Sanguankeo A. Whey Protein Supplementation Improves Body Composition and Cardiovascular Risk Factors in Overweight and Obese Patients: A Systematic Review and Meta-Analysis. J Am Coll Nutr. Jan 2018;37(1):60-70. doi:10.1080/07315724.2017.1344591
- Fekete AA, Giromini C, Chatzidiakou Y, Givens DI, Lovegrove JA. Whey protein lowers systolic blood pressure and Ca-caseinate reduces serum TAG after a high-fat meal in mildly hypertensive adults. Sci Rep. Mar 22 2018;8(1):5026. doi:10.1038/s41598-018-23333-2
- Oberoi A, Giezenaar C, Lange K, et al. Acute effects of whey protein, alone and mixed with other macronutrients, on blood pressure and heart rate in older men. BMC geriatrics. Jun 28 2022;22(1):535. doi:10.1186/s12877-022-03213-1
- Fekete AA, Giromini C, Chatzidiakou Y, Givens DI, Lovegrove JA. Whey protein lowers blood pressure and improves endothelial function and lipid biomarkers in adults with prehypertension and mild hypertension: results from the chronic Whey2Go randomized controlled trial. Am J Clin Nutr. Dec 2016;104(6):1534-1544. doi:10.3945/ajcn.116.137919
- Yang J, Wang HP, Tong X, et al. Effect of whey protein on blood pressure in pre- and mildly hypertensive adults: A randomized controlled study. Food science & nutrition. May 2019;7(5):1857-1864. doi:10.1002/fsn3.1040
- Odai T, Terauchi M, Kato K, Hirose A, Miyasaka N. Effects of Grape Seed Proanthocyanidin Extract on Vascular Endothelial Function in Participants with Prehypertension: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients. Nov 20 2019;11(12)doi:10.3390/nu11122844.
- Tropea T, Greenwood SL, Sibley CP, Cottrell EC. Grape Seed Extract Polyphenols Improve Resistance Artery Function in Pregnant eNOS(-/-) Mice. Front Physiol. 2020;11:588000. doi:10.3389/fphys.2020.588000.
- Mas-Capdevila A, Iglesias-Carres L, Arola-Arnal A, Suárez M, Bravo FI, Muguerza B. Changes in arterial blood pressure caused by long-term administration of grape seed proanthocyanidins in rats with established hypertension. Food Funct. Oct 21 2020;11(10):8735-8742. doi:10.1039/d0fo00981d.
- Schön C, Allegrini P, Engelhart-Jentzsch K, Riva A, Petrangolini G. Grape Seed Extract Positively Modulates Blood Pressure and Perceived Stress: A Randomized, Double-Blind, Placebo-Controlled Study in Healthy Volunteers. Nutrients. Feb 17 2021;13(2)doi:10.3390/nu13020654. https://mdpi-res.com/d_attachment/nutrients/nutrients-13-00654/article_deploy/nutrients-13-00654-v3.pdf?version=1613786803
- Foshati S, Nouripour F, Sadeghi E, Amani R. The effect of grape (Vitis vinifera) seed extract supplementation on flow-mediated dilation, blood pressure, and heart rate: A systematic review and meta-analysis of controlled trials with duration- and dose-response analysis. Pharmacological research : the official journal of the Italian Pharmacological Society. Jan 2022;175:105905. doi:10.1016/j.phrs.2021.105905.
- Weaver SR, Rendeiro C, McGettrick HM, Philp A, Lucas SJE. Fine wine or sour grapes? A systematic review and meta-analysis of the impact of red wine polyphenols on vascular health. European journal of nutrition. Feb 2021;60(1):1-28. doi:10.1007/s00394-020-02247-8.
- Pignatelli P, Fabietti G, Ricci A, Piattelli A, Curia MC. How Periodontal Disease and Presence of Nitric Oxide Reducing Oral Bacteria Can Affect Blood Pressure. International journal of molecular sciences. Oct 13 2020;21(20)doi:10.3390/ijms21207538.
- Hwang SY, Oh H, Rhee MY, Kang S, Kim HY. Association of periodontitis, missing teeth, and oral hygiene behaviors with the incidence of hypertension in middle-aged and older adults in Korea: A 10-year follow-up study. J Periodontol. Sep 2022;93(9):1283-1293. doi:10.1002/jper.21-0706.
- Kim J, Kim HJ, Jeon J, Song TJ. Association between oral health and cardiovascular outcomes in patients with hypertension: a nationwide cohort study. J Hypertens. Feb 1 2022;40(2):374-381. doi:10.1097/hjh.0000000000003022.
- Zou L, Zhang M, Fu W, Liu Y, Wen J, Lu Z. Meta-analysis on the association between the frequency of tooth brushing and hypertension risk. Journal of clinical hypertension (Greenwich, Conn). Jun 2022;24(6):689-697. doi:10.1111/jch.14498.
- Joshipura K, Muñoz-Torres F, Fernández-Santiago J, Patel RP, Lopez-Candales A. Over-the-counter mouthwash use, nitric oxide and hypertension risk. Blood pressure. Apr 2020;29(2):103-112. doi:10.1080/08037051.2019.1680270.
- Senkus KE, Crowe-White KM. Influence of mouth rinse use on the enterosalivary pathway and blood pressure regulation: A systematic review. Crit Rev Food Sci Nutr. 2020;60(17):2874-2886. doi:10.1080/10408398.2019.1665495.
- Bescos R, Ashworth A, Cutler C, et al. Effects of Chlorhexidine mouthwash on the oral microbiome. Sci Rep. Mar 24 2020;10(1):5254. doi:10.1038/s41598-020-61912-4.
- Ejtahed HS, Ardeshirlarijani E, Tabatabaei-Malazy O, et al. Effect of probiotic foods and supplements on blood pressure: a systematic review of meta-analyses studies of controlled trials. J Diabetes Metab Disord. Jun 2020;19(1):617-623. doi:10.1007/s40200-020-00525-0.
- Yuan L, Li Y, Chen M, et al. Effects of probiotics on hypertension. Appl Microbiol Biotechnol. Feb 2023;107(4):1107-1117. doi:10.1007/s00253-023-12369-8.
- FDA. U.S. Food & Drug Administration. Sodium in Your Diet. Updated 3/5/2024. Accessed 4/9/2024, https://www.fda.gov/food/nutrition-education-resources-materials/sodium-your-diet
- Yin X, Rodgers A, Perkovic A, et al. Effects of salt substitutes on clinical outcomes: a systematic review and meta-analysis. Heart. Sep 26 2022;108(20):1608-1615. doi:10.1136/heartjnl-2022-321332. https://heart.bmj.com/content/108/20/1608
- Xu X, Zeng L, Jha V, et al. Potassium-Enriched Salt Substitutes: A Review of Recommendations in Clinical Management Guidelines. Hypertension. Mar 2024;81(3):400-414. doi:10.1161/hypertensionaha.123.21343. https://pubmed.ncbi.nlm.nih.gov/38284271/
- Imaizumi VM, Laurindo LF, Manzan B, et al. Garlic: A systematic review of the effects on cardiovascular diseases. Crit Rev Food Sci Nutr. 2023;63(24):6797-6819. doi:10.1080/10408398.2022.2043821. https://www.ncbi.nlm.nih.gov/pubmed/35193446
- Ried K. Garlic lowers blood pressure in hypertensive subjects, improves arterial stiffness and gut microbiota: A review and meta-analysis. Exp Ther Med. Feb 2020;19(2):1472-1478. doi:10.3892/etm.2019.8374. https://www.ncbi.nlm.nih.gov/pubmed/32010325
- Ried K, Frank OR, Stocks NP. Aged garlic extract reduces blood pressure in hypertensives: a dose-response trial. Eur J Clin Nutr. Jan 2013;67(1):64-70. doi:10.1038/ejcn.2012.178. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3561616/pdf/ejcn2012178a.pdf
- Ried K, Travica N, Sali A. The Effect of Kyolic Aged Garlic Extract on Gut Microbiota, Inflammation, and Cardiovascular Markers in Hypertensives: The GarGIC Trial. Front Nutr. 2018;5:122. doi:10.3389/fnut.2018.00122. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6297383/pdf/fnut-05-00122.pdf
- Rais N, Ved A, Ahmad R, et al. S-Allyl-L-Cysteine — A garlic Bioactive: Physicochemical Nature, Mechanism, Pharmacokinetics, and health promoting activities. Journal of Functional Foods. 2023/08/01/ 2023;107:105657. doi:https://doi.org/10.1016/j.jff.2023.105657. https://www.sciencedirect.com/science/article/pii/S1756464623002578
- Nirvanashetty S, Panda S. High Potency Aged Garlic Extract reduces Cardiovascular Disease Risk Factors in Healthy Participants: A Randomized, Double-Blind, Placebo Controlled Study. Open Acc J Comp & Alt Med. 10/20/2023 2023;5(1):585-593. doi:10.32474/OAJCAM.2023.05.000203.
- Valls RM, Companys J, Calderon-Perez L, et al. Effects of an Optimized Aged Garlic Extract on Cardiovascular Disease Risk Factors in Moderate Hypercholesterolemic Subjects: A Randomized, Crossover, Double-Blind, Sustainedand Controlled Study. Nutrients. Jan 18 2022;14(3)doi:10.3390/nu14030405. https://pubmed.ncbi.nlm.nih.gov/35276764/
- Kisters S, Kisters K, Werner T, et al. Positive effects of magnesium supplementation in metabolic syndrome. Int J Clin Pharmacol Ther. 2024; 62: 569-578. https://pubmed.ncbi.nlm.nih.gov/39380547/
- Bkaily G, Jazzar A, Normand A, Simon Y, Al-Khoury J, Jacques D. Taurine and cardiac disease: state of the art and perspectives. Canadian journal of physiology and pharmacology. Feb 2020;98(2):67-73. doi:10.1139/cjpp-2019-0313. https://www.ncbi.nlm.nih.gov/pubmed/31560859
- Yildiz O, Ulusoy KG. Effects of taurine on vascular tone. Amino Acids. Dec 2022;54(12):1527-1540. doi:10.1007/s00726-022-03198-6. https://www.ncbi.nlm.nih.gov/pubmed/35986125
- Waldron M, Patterson SD, Tallent J, Jeffries O. The Effects of Oral Taurine on Resting Blood Pressure in Humans: a Meta-Analysis. Curr Hypertens Rep. Jul 13 2018;20(9):81. doi:10.1007/s11906-018-0881-z. https://www.ncbi.nlm.nih.gov/pubmed/30006901
- Guan L, Miao P. The effects of taurine supplementation on obesity, blood pressure and lipid profile: A meta-analysis of randomized controlled trials. European journal of pharmacology. Oct 15 2020;885:173533. doi:10.1016/j.ejphar.2020.173533. https://www.ncbi.nlm.nih.gov/pubmed/32871172
- Tzang CC, Lin WC, Lin LH, et al. Insights into the cardiovascular benefits of taurine: a systematic review and meta-analysis. Nutr J. Aug 15 2024;23(1):93. doi:10.1186/s12937-024-00995-5. https://www.ncbi.nlm.nih.gov/pubmed/39148075
- Li Y, Wang Q, Liu Y, et al. Taurine ameliorates blood pressure and vascular function in patients with type 2 diabetes: Randomized, double-blind, placebo-controlled trial. iScience. Jun 20 2025;28(6):112719. doi:10.1016/j.isci.2025.112719. https://www.ncbi.nlm.nih.gov/pubmed/40546935
