Life Extension Magazine®
Many people suffering from cardiovascular disease, stroke, migraines, and dementia could be suffering from the adverse effects of elevated levels of homocysteine in their blood. This condition has also been linked to other problems, including osteoporosis, birth defects, macular degeneration, and certain types of cancer.1 In most cases, doctors will not consider testing for homocysteine and could therefore be treating their patients without success. Most health-conscious people know their cholesterol level but few know their equally important homocysteine number. In this article, you’ll learn about a new non-prescription form of folic acid called bioactive folate (5-methyltetrahydrofolate, or 5-MTHF) and discover how it can prevent the homocysteine-related conditions that disable and kill many Americans. What you need to know
Why Doctors Don’t Check HomocysteineMany mainstream doctors still accept classic lipid-related risk factors for heart attack and stroke (e.g., high LDL and triglycerides and low HDL) as the gold standard upon which to base treatment. They continue to ignore the fact that in many cases, high blood levels of homocysteine also predict risk of vascular disease and stroke, especially in the elderly.2 Dr. Kilmer McCully first described the connection between homocysteine and atherosclerosis in 1969.3 His theory met with a rocky reception among members of the Harvard University Medical School faculty where he taught and conducted research. McCully’s revolutionary theory cast doubt on the widely accepted “lipid theory” of heart disease favored by his peers.
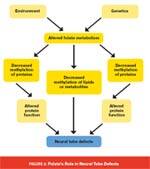
Mainstream doctors in the 1970s ignored McCully’s groundbreaking research just as many continue to do today. LDL levels (particularly elevated levels of oxidized LDL) remain an important risk factor for cardiovascular disease, yet more than half of all heart attack victims have blood cholesterol levels in the normal range. The pharmaceutical industry encourages doctors to prescribe highly profitable lipid-lowering drugs to prevent heart attack or stroke in their at-risk patients; there’s simply no monetary incentive for selling non-patentable dietary supplements that perform as well as prescription drugs in lowering homocysteine levels. There’s also little chance that drug sales reps will tell doctors that a bioactive form of folic acid called 5-MTHF (5-methyltetrahydrofolate) is now available as a non-prescription dietary supplement. Many physicians remain unaware that elevated homocysteine levels can lead to depression,4 osteo-porosis,5,6 headaches,7 and macular degeneration.8,9 Yet these and other health problems may be alleviated by lowering homocysteine blood levels. Homocysteine, Stroke, and FolateStroke is the third largest cause of death in the US, ranking behind cardiovascular disease and all forms of cancer.25 In 1998, the US and Canadian governments introduced a policy of folic acid fortification of enriched grain products.26 In 2006, researchers evaluated trends in stroke-related mortality before and after mandatory folic acid fortification in both countries and, as a comparison, during the same period in England and Wales, where fortification was not required.27 They found that average blood folate concentrations increased and homocysteine concentrations decreased in the United States after fortification. In addition, between 1998 and 2002, there was an accelerated decline in stroke mortality in the US with 12,900 fewer stroke deaths per year compared with the established trend seen between 1990 and 1997. Similar data were also seen in Canada, where there were 2,800 fewer stroke deaths per year between 1998 and 2002, while the decline in stroke mortality in England and Wales did not change significantly during this time period. Researchers concluded that folic acid supplementation could effectively reduce the risk of stroke in primary prevention. The findings add to the growing body of evidence that supports the theory that folic acid effectively reduces stroke death by reducing homocysteine levels in the blood. Homocysteine, Alzheimer’s Disease, and Active FolateUp to half of all Americans may carry a genetic variation that prevents the body, including the brain, from optimally using folic acid.23 Scientists believe that this genetic polymorphism may be linked with an increased risk of dementia.28 Fortunately, a non-prescription form of bioactive folate, 5-MTHF, can cross the blood-brain barrier and effectively reduce homocysteine. Elevated homocysteine has been linked with cognitive decline and Alzheimer’s disease.1,24,29 A 2002 study published in the New England Journal of Medicine reported that people with high blood levels of homocysteine had a greater risk of developing Alzheimer’s disease.30 Dementia developed in 111 study participants, of whom 83 were diagnosed with Alzheimer’s disease over an eight-year follow up. In those with a plasma homocysteine level greater than 14 µmol/L, the risk of Alzheimer’s disease nearly doubled. Investigators concluded, “An increased plasma homocysteine level is a strong, independent risk factor for the development of dementia and Alzheimer’s disease.” Active Folate: Relief For Migraine SufferersMigraine is a debilitating inflammatory blood vessel disease that may be triggered by damage inflicted by elevated blood levels of homocysteine to the endothelium of blood vessels in the brain.31 A recent study showed that treatment with B-complex vitamins, including 5-MTHF, might provide relief for migraine sufferers including those with the MTHFR C677T genotype, which typically limits the clinical effectiveness of supplemental folic acid.7 Headache frequency and pain severity were also reduced. The treatment effect proved successful in reducing homocysteine levels and migraine disability in study participants with the MTHFR C677T genotype.7 Researchers have long suspected that migraine headaches have a genetic component, because migraine sufferers often have family members who also have the condition. Studies suggest that MTHFR polymorphisms may account for the genetic predisposition to migraine in some individuals.32 Homocysteine’s Role in Macular DegenerationStudies of homocysteine’s role in age-related macular degeneration (both wet and dry types) reveal a strong link between the amino acid and the disease. In a group of 2,335 study participants who had evidence of age-related macular degeneration as detected from retinal photographs, researchers found that homocysteine blood levels greater than 15 µmol/L were associated with a more than three-fold increased likelihood of age-related macular degeneration in participants under age 75 years. They found a similar association for low vitamin B12 (under 125 pmol/L) among all study participants. Even in those with homocysteine under 15 µmol/L, low serum B12 was associated with nearly four-fold higher odds of age-related macular degeneration.8 In a larger and more recent study, Harvard researchers enrolled 5,442 women who were at high risk for cardiovascular disease. The women were given a placebo or 2,500 mcg folic acid, 50 mg vitamin B6, and 1,000 mcg vitamin B12. After seven years of treatment and follow-up, researchers recorded 55 cases of macular degeneration in the B vitamin treatment group and 82 in the placebo group. Investigators concluded that in women at high risk of cardiovascular disease, daily long-term supplementation with folic acid, B6, and B12 significantly reduced the risk of age-related macular degeneration.9 Folate, Vitamin B12, and Birth DisordersPublished data provide strong evidence that folate deficiency during the periconceptional period can lead to widespread alterations to DNA methylation in offspring, leading to disfigurement in childhood (Figure 2 bottom left).33 DNA methylation is the key to the maintenance of gene silencing, a process that relies on a dietary supply of methyl groups, as provided by B vitamins. Up to one-third of all pregnant and lactating women in the US may not be meeting their requirements for folate, despite mandatory folic acid-fortification of grain products.34 A long-term study demonstrated that women must begin supplementing with folate at least a year prior to conception, which practically speaking is only possible if women take supplements on a long-term daily basis. Women who took folic acid supplements for at least one year prior to conception experienced a 70% lower risk of premature delivery between the 20th and 28th week of gestation and a 50% decrease between the 28th and 32nd week compared with women who did not take folic acid supplements.35 Numerous other studies confirm that folic acid supplementation reduces the risk of neural tube defects and cleft lip and palate.36,37 Additional research reveals that supplementing with vitamin B12 may also reduce the risk—up to five times—of having a child with a neural tube defect, such as spina bifida.38
|
||||||||
Homocysteine is a Risk Factor For CancerEmerging research suggests that elevated levels of homocysteine may be a risk factor for cancer.39 A number of studies suggest that folate, through its effect on regulating DNA methylation and reducing homocysteine levels, may prevent cancers of the breast, bladder, lung, lymph, and colorectum.40-44 Conflicting evidence with respect to prostate cancer45 raises questions about the efficacy of folate for prostate cancer prevention. Some experts believe that when it comes to taking folate supplements, timing is everything. For example, some have proposed that folate may be beneficial when taken prior to old age because it decreases the likelihood of DNA mutations. Why Ordinary B Vitamin Supplements May Not Lower HomocysteineMany people who take B vitamin supplements are unable to sufficiently lower their homocysteine levels low enough to prevent disease.46 One possible reason is that folic acid, the form of the vitamin used in most supplements and enriched foods, is not bioavailable enough to sufficiently increase plasma folate levels in people with certain health problems or genotypes. Researchers recently found that giving bioactive 5-MTHF to patients with coronary artery disease resulted in a 700% higher plasma folate concentration compared with folic acid. This difference was irrespective of the patient’s genotype.23 Compared with bioactive 5-MTHF, folic acid supplements were relatively ineffective in increasing blood folate concentrations. Cooking and food processing destroy natural folates. Although red blood cells can retain folate for 40-50 days following discontinuation of supplementation, folic acid is poorly transported to the brain and is rapidly cleared from the central nervous system.1 Even though folic acid-fortified foods are ubiquitous in our food supply, and despite peoples’ best efforts to ensure adequate intake of the vitamin through supplementation, many individuals run the risk of not achieving sufficient plasma folate levels unless they supplement with bioactive folate. Bioactive Folate: A New Supplement Effective at Lowering HomocysteineBioactive folate, or 5-MTHF, is the predominant biologically active form of folate in cells, the blood, peripheral tissues, and particularly the brain.1 Until recently, 5-methyltetrahydrofolate was available only in expensive prescription medicines and medicinal food products. Now, this active form of folate, which provides increased bioavailability together with protection against homocysteine-related health problems, is available as a dietary supplement. Since 5-MTHF is the only form of folate used directly by the body, it doesn’t have to be converted and metabolized to be clinically useful, as does regular folic acid.1
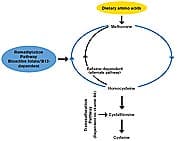
Folic acid, as used in ordinary dietary supplements and vitamin-fortified foods, must first be converted to bioactive 5-methyltetrahydrofolate in order to be clinically effective (Figure 3 to the Right). These steps require several enzymes, adequate liver and gastrointestinal function, and sufficient supplies of niacin (B3), pyridoxine (B6), riboflavin (B2), vitamin C, and zinc.1 Another advantage of 5-MTHF is that it is unlikely to mask a vitamin B12 deficiency, a shortcoming of folic acid. Folic acid, when administered as a single agent, may obscure the detection of vitamin B12 deficiency (specifically, folic acid may reverse the hematological manifestations of B12 deficiency, while not addressing the neurological manifestations). Research has shown that bioactive 5-MTHF (5-methyltetrahydrofolate) is less likely than folic acid to mask vitamin B12 deficiency.1 Who Would Benefit From Taking 5-MTHF Supplements?The low-dose requirements for 5-MTHF make it a relatively inexpensive supplement with superior clinical benefits over folic acid. People who would benefit from taking active folate include:
Individuals with the MTHFR C677T mutation are at higher risk of cardiovascular disease, stroke, pre-eclampsia (high blood pressure in pregnancy), and birth defects that occur during the development of the brain and spinal cord (neural tube defects).
How Much 5-MTHF Do You Need?A daily dose of 800-1,000 mcg (0.8 to 1.0 mg) bio-active folate is typically used in research studies to achieve clinically beneficial reduction in elevated plasma homocysteine concentrations. Even doses of 200-400 mcg (0.2 to 0.4 mg) have been shown to achieve this effect.47 A number of drugs and medical food products on the market contain very high amounts (5,000 mcg or 5 mg) of 5-MTHF. Unless specifically prescribed by a physician, food and drug products that contain such high doses are probably not required to normalize homocysteine levels in most people with mild-to-moderate elevations. Ancillary Supplements For Use With 5-MTHFIndividuals who wish to bring their elevated blood levels of homocysteine into the ideal range (under 8.0 µmol/L) may want to adopt a folate-boosting supplement protocol in addition to using bioactive folate 5-MTHF. This requires taking several supplements that work in concert to effectively reduce homocysteine and risk of disease. The following supplements can provide additional support:1
ConclusionElevated levels of homocysteine have been linked with a diverse spectrum of health disorders ranging from cardiovascular disease, stroke, and cancer to migraine, dementia, and macular degeneration. Most conventional physicians fail to detect and treat elevated levels of homocysteine in the blood. Folate is an essential nutrient for maintaining healthy homocysteine levels, yet up to half of Americans have a genetic inability to properly metabolize regular folic acid to its active form. Fortunately, the active form of folate is now available as a dietary supplement called 5-methyltetrahydrofolate, or 5-MTHF. By increasing blood folate levels 700% more effectively than ordinary folic acid, 5-MTHF offers advanced protection against the homocysteine-related disorders that threaten to compromise our longevity and well-being. If you have any questions on the scientific content of this article, please call a Life Extension Wellness Specialist at 1-800-226-2370. | ||||
| References | ||||
|

 ;
;