Life Extension Magazine®
As you read this sentence, a destructive biochemical process known as glycation is occurring in your body. Glycation arises when simple sugars react with proteins to generate protein “cross-links.” These cross-linked proteins are called advanced glycation end products (AGEs). Cataract development in the lens of the eye, the drop in kidney function,damage to the delicate endothelial cell layer of our blood vessels, and the unsightly sagging and wrinkling of our skin are all examples of glycation. In addition to protein glycation, many health-conscious people do not realize that lipid glycation plays an important role in the development of age-associated complications. In fact, a high rate of lipid glycation occurs in the plasma of patients with type 2 diabetes. Since accelerated aging is a hallmark of type 2 diabetes, the cellular toxicity caused by lipid glycation must be minimized to reduce the risk of complications of aging as well as diabetes. Late last year, Life Extension was able to offer members access to pyridoxamine, a form of vitamin B6 well-validated to reduce glycation. Regrettably, a pharmaceutical company’s petition to have the regulatory status of pyridoxamine changed from a dietary supplement to an investigational new drug was very recently published by the FDA. However, the good news is that another form of vitamin B6, pyridoxal-5’-phosphate (a different B6 vitamer), offers anti-glycation benefits that scientific studies suggest may have similar properties as pyridoxamine plus better inhibitory effects against lipid glycation. Links and Cross-Links: The Processes of GlycationLife Extension members understand that glycation is a crucial aspect of the aging process. In patients with type 2 diabetes, aging occurs at an accelerated rate, and one of the most important factors responsible for this enhanced rate of aging is glycation. There is a striking similarity between the complications of diabetes and the negative changes that are commonly recognized as aging. These include the gradual loss of youthful elasticity of our blood vessels, the unsightly sagging and wrinkling of our skin, the degradation of the lens in the eye, the delay in wound healing and increased susceptibility to infection associated with aging, and the progressive, age-induced decline in kidney function. All of these problems include a common factor—glycation.
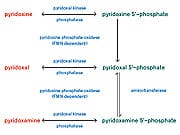
B6 Reduces GlycationOne of the best known anti-glycating agents is vitamin B6, which actually occurs in nature in several different chemical forms, or vitamers.22 The main dietary form of B6 is called pyridoxine, which is subsequently converted into pyridoxal-5’-phosphate. In fact, each of the major B6 vitamers are ultimately converted into pyridoxal-5’-phosphate in the body (see Figure 1 below). Although the different B6 vitamers possess slightly different biochemical properties, the most active forms in terms of anti-glycation capability are pyridoxamine and pyridoxal-5’-phosphate.23
Scientists first became interested in pyridoxamine because of its tremendous glycation-fighting activity. They began to research the benefits of both pyridoxamine and pyridoxal-5’-phosphate23 in reducing the age-accelerating aspects of glycation. As these data came to the attention of the pharmaceutical companies, they realized that there were important medical benefits to pyridoxamine that were unmatched by their current roster of pharmaceuticals in development. Unfortunately, a recent ruling by the FDA may remove pyridoxamine from consumers and place it in the hands of the pharmaceutical industry. In January 2009, responding to a petition by a pharmaceutical company, the FDA determined that pyridoxamine would not be considered a “dietary supplement,” paving the way for declaring pyridoxamine an “investigational new drug” for eventual sale as a prescription-only pharmaceutical.24 However, aging individuals cannot afford to wait as long as perhaps 10 years for the B6 vitamer pyridoxamine’s availability due to pharmaceutical greed and the inefficient quagmire that characterizes the FDA drug approval process. Pyridoxal-5’-phosphate offers an alternative. In addition to preventing formation of AGEs and ALEs, pyridoxal-5’-phosphate actually engages in “trapping” already formed AGEs and chaperoning them out of cells.25,26 Furthermore, there is striking scientific evidence that pyridoxal-5’-phosphate offers anti-glycation benefit that is the equal of several strong, natural inhibitors of glycation and comparable to some pharmaceutical agents. Kidney Disease and B6 VitamersThe loss of kidney function under the influence of chronic blood sugar elevation is one of the leading causes of disability among diabetic patients. Kidney damage leads to hypertension and exacerbates cardiovascular disease, both of which in turn produce additional kidney injury, so prevention of kidney disease is of utmost importance for diabetics.
Scientists have evaluated various anti-glycation B6 vitamers in preventing diabetic complications.4,27 For example, scientists have shown that chemically combining pyridoxal, a B6 vitamer, with an AGE-fighting drug, aminoguanidine, enhanced the kidney protection conferred by the drug alone, and provided additional antioxidant effects as well.28 Basing their work on evidence that pyridoxamine could prevent development of kidney damage in experimental models of diabetes, scientists at the University of South Carolina hypothesized in 2003 that this B6 vitamer would provide similar benefits in experimental subjects that, while not diabetic, were obese.29 The researchers were also interested in identifying the impact of glycated lipids on cardiovascular damage and kidney disease. They divided the experimental cohort into three groups: a healthy lean group, an obese group without B6 vitamer treatment, and an obese group given pyridoxamine daily in their drinking water. The results were compelling: the obese untreated group had formation of glycated proteins and lipids at rates two to three times greater than those of the healthy lean group. Pyridoxamine supplementation, however, blocked those increases, while simultaneously decreasing plasma triglycerides, cholesterol, and the waste-product creatinine (a marker of kidney function). Physiologically, the supplemented obese group experienced reductions in both their previously elevated blood pressure and thickened blood vessel walls. Urinary protein excretion, a sign of kidney disease, was restored to near normal levels as well. The authors concluded that “Lipids are an important source of chemical modification of tissue proteins, even in the absence of hyperglycemia,” and that B6 vitamer treatment with pyridoxamine reduced protein and lipid glycation and “…protected against renal and vascular pathology in a nondiabetic model” (of obesity). Further evidence for a role of lipid glycation as a cause of human kidney disease came from Japanese scientists in 2005, in a study comparing glycated lipid levels in diabetic and non-diabetic patients with kidney disease.30 They found that levels in diabetic patients on chronic hemodialysis were nearly twice those in diabetic patients not on dialysis, whose levels in turn were slightly higher than those in non-diabetic dialysis patients. Levels of glycated lipids were lowest of all (by a substantial margin), in control patients with neither diabetes nor non-diabetic kidney disease. These findings are of great importance because, as we’ll see, research indicates that the B6 vitamer pyridoxal-5-phosphate compares very favorably with the B6 vitamer pyridoxamine at reducing lipid glycation. Harvard’s Joslin Diabetes group has published the results of a Phase II (safety and tolerability) study of the B6 vitamer pyridoxamine in diabetic patients with fully developed kidney disease.31 Their work involved two studies in which patients received pyridoxamine 50 or 250 mg twice daily, or placebo, for 24 weeks. B6 vitamer-treated patients experienced significantly slower deterioration of their serum creatinine (a standard biomarker of renal function) compared with placebo recipients. There was also a statistical trend towards lower levels of the inflammatory protein TGF-1 in urine, as well as urinary levels of AGEs, in the treated patients compared with placebo patients. Data on B6 vitamers’ benefits in renal disease continue to accumulate, with evidence for its effect on improving the health of experimental animals receiving peritoneal dialysis and on those having undergone kidney transplantation.32,33 These and similar studies are generating hopes that we will see similar protective effects in humans whose kidney function has deteriorated to the point of needing these advanced interventions. Eye Disease: The Impact of B6 Vitamers on Cataracts, Retinopathy, and IschemiaWith its high rate of metabolic activity and precision of function, the human eye is particularly vulnerable to the effects of glycated protein and lipids, as well as the structural and inflammatory effects they produce.34 The lens, which depends for its clarity on precise protein structure, becomes cloudy and eventually opacifies (cataract) when those proteins become cross-linked as the result of glycation.9,10,35 An important demonstration of this effect was provided in a 2002 study, in which scientific researchers studied the impact of high-glucose solutions on cultures of lens cells, with or without the presence of the B6 vitamers pyridoxine and pyridoxamine.36 Cells grown in high-glucose environments without B6 vitamer protection had substantially higher rates of glycation and protein cross-linking than did similar cells grown with the addition of pyridoxine or pyridoxamine. Ophthalmologists at Case Western Reserve University subsequently demonstrated an anti-cataract effect of a B6 vitamer in an experimental diabetes model with pyridoxamine.37 Treatment increased the activity of a chemical pathway that reacts with, and decreases, glycation before the process can damage the lens.
Japanese scientists showed in 2004 that the B6 vitamer pyridoxal was found to have anti-glycation benefits superior to the drug aminoguanidine in an experimental model of cataract formation. The scientists administered either pyridoxal or aminoguanidine to the study groups in drinking water for seven weeks. Pyridoxal, but not aminoguanidine, significantly improved motor nerve conduction velocity. The time to develop cataract was longer in the pyridoxal-treated group than in the aminoguanidine-treated group. The increase in lens opacification in culture medium containing high glucose levels was more efficiently reduced by pyridoxal than by aminoguanidine. Furthermore, the level of urinary 8-hydroxy-2’-deoxyguanosine, a marker of oxidative DNA damage, and the level of liver malondialdehyde plus 4-hydroxy-2-alkenals, a marker of tissue lipid peroxidation, both of which are elevated in diabetes, were significantly reduced by pyridoxal but not by aminoguanidine.38 Diabetic retinopathy is the term physicians use to describe the progressive destruction of retinal blood vessels under the effects of chronic blood sugar elevations. Ophthalmologists in Northern Ireland suspected this to be the case, and explored the possibility that B6 vitamers could protect against those changes.39 Treatment with the B6 vitamer pyridoxamine protected against the blood vessel changes, and decreased the amount of protein and lipid glycation in retinal tissue.
A 2002 study showed that the B6 vitamers pyridoxal-5’-phosphate and pyridoxal hydrochloride protect the retina of the eye from ischemia (lack of blood flow). Daily administration of the B6 vitamers pyridoxal hydrochloride and pyridoxal-5’-phosphate was performed for 10 consecutive days. On the sixth day, whole brain complete ischemia was produced by clipping the innominate and the left subclavian arteries for 20 minutes. The ischemia induced toxic cellular damage in the retina as shown by the loss of ganglion cells and the reduction of thickness of the ganglion cell, inner plexiform, and inner nuclear layers. Pyridoxal-5’-phosphate significantly prevented the ganglion cell loss and the reduction of thickness of the ganglion cell layer. Pyridoxal hydrochloride significantly prevented the ganglion cell loss as well as the reduction of thickness of ganglion cell, inner plexiform, and inner nuclear layers. These results suggest that the B6 vitamers pyridoxal-5’-phosphate and pyridoxal hydrochloride counteract the ischemic nerve cell death in the retina.40 | |||||||||
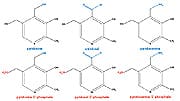
Alzheimer’s DiseaseBrain and nerve tissue are also highly vulnerable to the effects of glycation, and neurologists and gerontologists are beginning to suspect that glycation plays a significant, and perhaps central role in the development of age-related dementias such as Alzheimer’s disease. German neuroscientists reviewed the state of knowledge in 2001, pointing out that products of glycation accumulate within brain cells, causing them to clump and impairing their ability to perform vital transport processes, and ultimately leading to cell death.11 They also recognized that extracellular glycation exerts chronic oxidative stress on neurons, triggering toxic inflammatory cytokine release. The reviewers went on to speculate that, as a result, “Glycation inhibitors might also become interesting novel therapeutic drugs for treatment of Alzheimer’s disease.” A 2002 study from the University of California, Davis, for example, examined vitamin B status, including pyridoxal-5-phosphate, in 43 Alzheimer’s disease patients, comparing them with 37 control subjects.41 Alzheimer’s disease patients were more than 12 times more likely to have low pyridoxal-5’phosphate levels than were control patients. An even more comprehensive study by Norwegian geriatricians demonstrated lower levels of a number of nutrients in apparently well-nourished Alzheimer’s disease patients, compared with controls.42 In particular, pyridoxal-5’-phosphate levels were found to be 90.2 nmol/L in healthy control patients, and just 24.8 in Alzheimer’s disease subjects. Most remarkably, all of the analyses conducted by these researchers identified pyridoxal-5’-phosphate as the common factor capable of identifying Alzheimer’s disease patients with 100% reliability! Pyridoxal-5’-Phosphate: A B6 Vitamer with Anti-Glycation BenefitsThe scientific literature clearly shows that B6 vitamers can play an important role in reducing the damaging impact of glycation upon aging. The recent decision by the FDA charged with protecting and promoting the health of the public by maintaining safe access to our supply of food and drugs now paradoxically places access to the B6 vitamer pyridoxamine out of the reach of private citizens. The B6 vitamer pyridoxal-5’-phosphate has been shown in several studies to possess potent anti-glycation benefits.43-45 In addition, the fact that pyridoxal-5’-phosphate levels are especially low in Alzheimer’s patients raised scientific interest in this B6 vitamer’s role in supporting cognitive health beginning in 2004.42 B6 vitamers possess subtle biochemical differences that help account for their specific biochemical and metabolic properties. Pyridoxamine is converted to pyridoxamine-5’-phosphate and pyridoxal-5’-phosphate in the liver and intestine. Therefore, pyridoxamine exerts beneficial effects on inhibition of glycation, at least in part, by pyridoxal-5’-phosphate. Pyridoxal-5’-phosphate is a naturally occurring metabolite of vitamin B6, and inhibits AGE formation, but in a different way than pyridoxamine due to differences in chemical structure (see Figure 2 below). The difference in structure between pyridoxamine and pyridoxal-5’-phosphate helps explain the differences in their effect on AGE inhibition. The nucleophilic amino group of pyridoxamine binds to reactive carbonyl compounds to inhibit AGE formation. However, pyridoxal-5’-phosphate does not have any amino group, and instead has an aldehyde group and a phosphate group (see Figure 2 below).
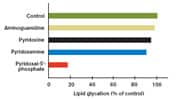
For example, in comparison to pyridoxamine, pyridoxal-5’-phosphate traps a major glucose degradation product called 3-deoxyglucosone that contributes to glycation-induced damage, preventing 3-deoxyglucosone from reacting with tissue proteins and lipids.26 In a fascinating study, Japanese researchers evaluated the process of peritoneal dialysis, a common procedure in humans with diabetic kidney failure.26 The researchers showed that pyridoxal-5’-phosphate inhibited AGE formation by trapping 3-deoxyglucosone, a major glucose degradation product in peritoneal dialysis fluid. The fact that pyridoxamine does not trap 3-deoxyglucosone, but pyridoxal-5’-phosphate does, is very likely due to differences in their chemical structure. Because 3-deoxyglucosone is a precursor of imidazolone, a significant glycation end product, trapping of 3-deoxyglucosone by pyridoxal-5’-phosphate directly reduces accumulation of imidazolone, a major glycation end product. Scientific researchers have directly compared the effect of oral supplementation with either pyridoxamine or pyridoxal-5’-phosphate on prevention of kidney disease in experimental models. In an interesting 2007 study, researchers evaluated the B6 vitamers pyridoxamine and pyridoxal-5’-phosphate in the prevention of kidney disease progression.25 In this experimental model, the scientists provided oral supplementation with either pyridoxal-5’-phosphate or pyridoxamine at a dose of 600 mg/kg body weight per day for 16 weeks, monitoring kidney function, AGE production, and biochemical markers of inflammation. Pyridoxal-5’-phosphate was found to significantly reduce protein loss in the urine as well as the microscopic evidence of damage to kidney structures, compared with the control diabetic group. There was a marked reduction in accumulation of AGEs in kidney tissue, with a concomitant reduction in inflammatory molecules and the aptly named receptor for advanced glycation end products (RAGE). Of interest, this experimental study of kidney disease showed that oral supplementation of the B6 vitamer pyridoxal-5’-phosphate was slightly better than pyridoxamine at preventing the development of diabetic nephropathy in the experimental test group. The scientists concluded, “Pyridoxal-5’-phosphate prevented progression of nephropathy in diabetic animals by inhibiting formation of AGEs. Pyridoxal-5’-phosphate is considered a promising active form of vitamin B6 for the treatment of AGE-linked disorders such as diabetic nephropathy.” Scientists have recently started to focus on the damage to DNA by glycation and free radical (oxidant) stress. New research suggests that glycation-induced damage to DNA can be a factor in the initiation of cancer as well as cellular aging.46 Using an elegant experimental model of DNA damage, Indian scientists tested various vitamins for their ability to prevent the damage. Of interest, pyridoxal-5’-phosphate showed maximal protection at preventing damage to DNA, and the scientists correlated this protective effect to inhibition of production of hydroxyl and superoxide free radicals. Recent research has identified lipid glycation, in addition to protein glycation, as an important factor in aging, kidney disease, diabetes, and pre-diabetic (insulin-resistant) states.30,47,48 Scientists have developed experimental models to test the inhibitory effects of different nutrients and drugs on lipid glycation. One such experimental model screened compounds that could prevent formation of so-called Amadori-PE, a product of lipid glycation with LDL cholesterol.49 Pyridoxal-5’-phosphate emerged as not only the best B6 vitamer at inhibiting lipid glycation, but significantly better than the drug aminoguanidine in this experimental model (see Figure 3 to the below).
SummaryB6 vitamers offer a variety of interesting and beneficial biochemical properties. Glycation of tissue proteins is a critical factor in the aging process as well as diseases of the cardiovascular system, kidneys, and eyes. New and emerging research strongly suggests that lipid glycation also plays a prominent role in aging as well as other diseases. B6 vitamers like pyridoxamine and pyridoxal-5’-phosphate provide protection against the ravages of glycation. Recent experimental studies show that oral supplementation with pyridoxal-5’-phosphate offers dramatic anti-glycation benefits in a model of diabetic kidney disease comparable to oral supplementation with pyridoxamine. Over the next decade, new and exciting scientific developments with B6 vitamers will help us in our quest to minimize accelerated aging induced by protein and lipid glycation.
If you have any questions on the scientific content of this article, please call a Life Extension Health Advisor at 1-800-226-2370. | |||||||||
| References | |||||||||
| 1. Am J Cardiovasc Drugs. 2003;3(5):315-20. 2. Ann NY Acad Sci. 2005 Jun;1043:529-32. 3. Cell Biochem Biophys. 2007;48(2-3):147-57. 4. Am J Kidney Dis. 2001 Oct;38(4 Suppl 1):S100-6. 5. Biochim Biophys Acta. 2001 Feb 14;1535(2):110-9. 6. Clin Chim Acta. 2002 Jul;321(1-2):69-76. 7. Pathol Biol (Paris). 2006 Sep;54(7):387-95. 8. Rejuvenation Res. 2006;9(2):264-73. 9. Invest Ophthalmol Vis Sci. 2008 Nov;49(11):4945-52. 10. Biochem J. 2007 Dec 1;408(2):251-8. 11. Biogerontology. 2001;2(1):19-34. 12. J Invest Dermatol. 2006 Feb;126(2):291-9. 13. Ann NY Acad Sci. 2005 Jun;1043:784-92. 14. Diabetes. 1992 Oct;41(Suppl 2):67-73. 15. Ann Clin Biochem. 1995 Sep;32( Pt 5):459-63. 16. Medicina (B Aires). 2000;60(5 Pt 1):645-56. 17. Angiology. 2005 Jul;56(4):431-8. 18. J Lipid Res. 2006 May;47(5):964-74. 19. Ann NY Acad Sci. 2005 Jun;1043:759-66. 20. Free Radic Biol Med. 2000 Jun 15;28(12):1708-16. 21. Drug Discov Today. 2006 Jul;11(13-14):646-54. 22. Mol Nutr Food Res. 2008 Mar;52(3):379-85. 23. Cell Mol Life Sci. 2005 Aug;62(15):1671-81. 24. www.fdalawblog.net/fda_law_blog_hyman_phelps/2009/01/fda-determines-that-pyridoxamine-is-excluded-from-the-definition-of-dietary-supplement-under-fdc-act.html. 25. J Am Soc Nephrol. 2005 Jan;16(1):144-50. 26. Nephrol Dial Transplant. 2007 Aug;22(8):2165-74. 27. Eur J Pharmacol. 1999 Aug 13;378(3):283-9. 28. Horm Metab Res. 2002 Jul;34(7):371-7. 29. Kidney Int. 2003 Jun;63(6):2123-33. 30. Ann NY Acad Sci. 2005 Jun;1043:280-3. 31. Am J Nephrol. 2007;27(6):605-14. 32. Nephrol Dial Transplant. 2008 Feb;23(2):518-24. 33. Kidney Int. 2005 Sep;68(3):1326-36. 34. Ophthalmologica. 2006;220(5):317-22. 35. Curr Eye Res. 2008 Aug;33(8):669-75. 36. Free Radic Biol Med. 2002 Dec 15;33(12):1615-21. 37. Ophthalmic Res. 2006;38(5):294-302. 38. Horm Metab Res. 2004 Mar;36(3):183-7. 39. Diabetes. 2002 Sep;51(9):2826-32. 40. Brain Res. 2002 Jun 14;940(1-2):36-43 41. Neurology. 2002 May 28;58(10):1471-5. 42. J Nutr Health Aging. 2004;8(5):407-13. 43. Biochim Biophys Acta. 1994 Jul 18;1226(3):286-90. 44. Ophthalmic Res. 1996;28(Suppl 1):65-8. 45. Nutr Sci Vitaminol (Tokyo). 1995 Feb;41(1):43-50. 46. J Pharm Biomed Anal. 2005 Apr 29;37(5):1173-7. 47. Amino Acids. 2007 Nov;33(4):615-21. 48. J Lipid Res. 2005 Nov;46(11):2514-24. 49. J Lipid Res. 2006 May;47(5):964-74. 50. Am J Clin Nutr. 1981 Jul;34(7):1328-37. 51. Shane B. Vitamin B6 and blood. In: Human Vitamin B6 Requirements: Proceedings of a Workshop. Washington, DC: National Academy Press; 1978:111-28. 52. J Nutr. 1980 Feb;110(2):275-85. |