Life Extension Magazine®
In an advance that reads like science fiction, researchers have created cells, which once transplanted into a living being, transform into an entire organ that is able to function as well as a natural organ!1
In this remarkable achievement, scientists were able to create a functioning thymus gland that helps oversee the regulation of our immune system. With aging, the thymus gland shrivels, virtually disappearing in normal adults as we approach age 40. This plays a role in the immune senescence encountered by maturing humans.2
The ability to develop a functioning thymus gland is therefore a major advance in anti-aging medicine.
Understanding Regenerative Medicine
The field of regenerative medicine3,4 is founded on the idea that aging tissues and organs can be repaired or replaced—not by transplantation from another human, but by regeneration from one’s own cells. A central goal of this specialty is the generation of intact, transplantable organs from one basic cell line derived from the same individual who will receive the transplanted organ.1
This approach removes any risks associated with current transplant medicine—which is the rejection of the new organ by the host’s immune system. In this case, the new organ is grown from the host’s own cells and therefore won’t be rejected.5 Readily available transplantable organs would also overcome the lengthy waiting lists that patients must currently endure because of the existing shortage of organs.6,7
In theory, a regenerative medicine specialist ought to be able to harvest a small number of cells from the intended host (by swabbing the inside of the cheek), expose those cells to various signals that would cause them to regress to a more primitive kind of cell (a stem cell), and then induce those stem-like cells to redevelop into the desired tissue types. The specialist would then simply transplant the “new” organ into the host.7-9
There has been surprising progress on the so-called “plug-and-play” organ production in laboratories around the world. Surprisingly, the difficulty isn’t getting functional cells for organs like kidneys, lungs, hearts, and livers—all of which have proved to be readily produced in culture.5,10,11 Rather, the major challenge has been getting the cells to organize themselves properly in order to achieve the correct structure that has been shown to be a vital driver of organ function.12,13
So far, functioning cells have been induced to grow on natural scaffolds derived from animal (mostly pig) organs, which have had the original cells removed to reduce rejection risk, and then repopulated with the recipient’s own functioning cells. In some cases, such organs have even been transplanted into rodent models with modest success, but to date the transplanted organs have failed to perform a full range of functions.6,10
That all changed in September of 2014, when an international research group reported their development of a fully intact and fully functional thymus gland from cells that were successfully transplanted into an animal host.1 This report is the first-ever publication about successful cellular reprogramming techniques that could generate an entire organ.
A transplantable thymus would be a tremendous breakthrough in human medicine—not only because of its implications for the field of regenerative medicine, but also because of the important role the thymus gland plays in maintaining a healthy immune system.
What You Need To Know
 |
The Thymus Gland And Immunity
- The thymus gland begins to decline in late adolescence, and has almost disappeared by the age of 40.
- The ability to restore normal thymic function would help reverse or arrest immune dysfunction.
- A groundbreaking study has now demonstrated the complete generation of a functioning, transplantable thymus gland in laboratory mice.
- The transplanted thymus produced normal numbers of functioning immune cells (T cells).
- When this technique is successfully applied to human beings, we can confidently anticipate the addition of years, even decades, to the current human life span.
Details From The Groundbreaking Study
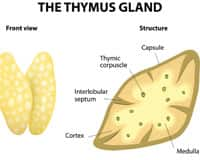
In September 2014, the journal Nature Cell Biology published a study by an international research group reporting their development of an intact, fully functioning thymus gland that was successfully transplanted into an animal host.1
For this study, researchers treated fibroblasts, which are cells that synthesize collagen and other extracellular substances, as the source material since they can be removed from healthy mice.1 The researchers used recently developed techniques to “force” expression of a gene that is required to trigger the specialization of cells into functioning thymus tissue.1
That gene technique involves a “transcription factor” with the unusual name of “forkhead box N1,” or Foxn1. Declining natural levels of Foxn1 have been implicated as a major cause of loss of thymus tissue with aging.14 Cells that have been “turned on” by the Foxn1 gene are capable of triggering the production of new naïve T cells, as well as “educating” and selecting them to perform their many functions.15-17
Once the basic, undifferentiated fibroblasts were genetically modified to express Foxn1 and allowed to grow, they were reprogrammed into functioning thymic epithelial cells, or TECs. These induced cells, dubbed “iTECs,” fully supported efficient development of the major T cell types while growing in culture.1
Remarkably, when these reprogrammed cells were transplanted from the culture dish into living mice, the iTECs proceeded to establish an intact, functional thymus with a natural architecture that contained all of the cell populations necessary to support formation of new T cells.1 Indeed, the recipients’ immune systems were rapidly populated by functioning T cells!
If replicated in humans, it is entirely conceivable that we might see widespread use of thymus transplantation to boost immune function in both young children with congenitally absent thymus tissue, and in the much larger population of aging adults whose thymus glands have deteriorated. Success in this area would help end many of the problems associated with immune senescence, and could add years to the overall human health span.
Why We Need More Transplantable Organs
 |
It is hard to overstate the crucial need for organs available for transplant, as the technology for implanting organs is rapidly outpacing their availability. In 2012, the latest year for which data are available, 16,487 kidney transplants were performed in the United States—but 95,022 transplant candidates remained on waiting lists by year’s end.12 A total of 2,600 additional kidneys were available for transplantation, but had to be discarded for technical reasons, even as more than 4,700 patients died awaiting a kidney.12
The promise of regenerative medicine is to make such waiting lists obsolete by allowing production of each needed organ exactly when it is needed, and made from precisely the same tissue type as the recipient because the original cells were derived from the recipient him- or herself. This would ensure a virtually inexhaustible source of organs for transplantation.12
Summary
Breakthrough research has now resulted in the production of a fully functioning, intact, and transplantable thymus in a mammal model. The techniques used to accomplish this feat take place entirely in a laboratory culture dish, and pose no discernible threat to the intended host, who is also the donor.
Self-donated tissue growing into functional, transplantable organs may turn out to be the wave of the future. In the case of the thymus gland…that future is closer than ever.
If you have any questions on the scientific content of this article, please call a Life Extension® Wellness Specialist at 1-866-864-3027.
References
- Bredenkamp N, Ulyanchenko S, O’Neill KE, Manley NR, Vaidya HJ, Blackburn CC. An organized and functional thymus generated from FOXN1-reprogrammed fibroblasts. Nat Cell Biol. 2014 Sep;16(9):902-8.
- Tosi P, Kraft R, Luzi P, et al. Involution patterns of the human thymus. I Size of the cortical area as a function of age. Clin Exp Immunol. 1982 Feb;47(2):497-504.
- Salgado AJ, Oliveira JM, Martins A, Teixeira FG, Silva NA, Neves NM, Sousa N, Reis RL. Tissue engineering and regenerative medicine: past, present, and future. Int Rev Neurobiol. 2013;108:1-33.
- Atala A. Regenerative medicine strategies. J Pediatr Surg. 2012 Jan;47(1):17-28.
- Bonvillain RW, Danchuk S, Sullivan DE, et al. A nonhuman primate model of lung regeneration: detergent-mediated decellularization and initial in vitro recellularization with mesenchymal stem cells. Tissue Eng Part A. 2012 Dec;18(23-24):2437-52.
- Soto-Gutierrez A, Wertheim JA, Ott HC, Gilbert TW. Perspectives on whole-organ assembly: moving toward transplantation on demand. J Clin Invest. 2012 Nov 1;122(11):3817-23.
- Peloso A, Katari R, Zambon JP, et al. Abdominal organ bioengineering: Current status and future perspectives. Minerva Chir. 2014 Oct 10.
- Park IH, Lerou PH, Zhao R, Huo H, Daley GQ. Generation of human-induced pluripotent stem cells. Nat Protoc. 2008;3(7):1180-6.
- Patel M, Yang S. Advances in reprogramming somatic cells to induced pluripotent stem cells. Stem Cell Rev. 2010 Sep;6(3):367-80.
- Arenas-Herrera JE, Ko IK, Atala A, Yoo JJ. Decellularization for whole organ bioengineering. Biomed Mater. 2013 Feb;8(1):014106.
- Xinaris C, Yokoo T. Reforming the kidney starting from a single-cell suspension. Nephron Exp Nephrol. 2014;126(2):107.
- Katari R, Peloso A, Zambon JP, et al. Renal bioengineering with scaffolds generated from human kidneys. Nephron Exp Nephrol. 2014;126(2):119.
- Salvatori M, Katari R, Patel T, et al. Extracellular matrix scaffold technology for bioartificial pancreas engineering: state of the art and future challenges. J Diabetes Sci Technol. 2014 Jan 1;8(1):159-69.
- Bredenkamp N, Nowell CS, Blackburn CC. Regeneration of the aged thymus by a single transcription factor. Development. 2014 Apr;141(8):1627-37.
- Romano R, Palamaro L, Fusco A, et al. FOXN1: A master regulator gene of thymic epithelial development program. Front Immunol. 2013;4:187.
- Ma D, Wang L, Wang S, Gao Y, Wei Y, Liu F. Foxn1 maintains thymic epithelial cells to support T-cell development via mcm2 in zebrafish. Proc Natl Acad Sci USA. 2012 Dec 18;109(51):21040-5.
- Zook EC, Krishack PA, Zhang S, et al. Overexpression of Foxn1 attenuates age-associated thymic involution and prevents the expansion of peripheral CD4 memory T cells. Blood. 2011 Nov 24;118(22):5723-31.

