
Celiac Disease and Non-Celiac Gluten Sensitivity
Celiac Disease and Non-Celiac Gluten Sensitivity
Last Section Update: 04/2015
Contributor(s): Robert Iafelice, MS/RD/LDN
1 Overview
Summary and Quick Facts for Celiac Disease and Gluten Sensitivity
- Wheat allergy, celiac disease and non-celiac gluten sensitivity can be confused with each other but are actually three separate conditions.
- This protocol will give you a better understanding of celiac disease and non-celiac gluten sensitivity, and how they differ. You will also learn about lifestyle changes and targeted nutritional support with several natural compounds that can complement the gluten-free diet by correcting nutritional deficiencies, promoting intestinal healing, balancing the immune system and reducing chronic inflammation.
- Maintaining a gluten-free diet for life is essential for those with celiac disease. The gluten-free diet is also considered the primary treatment for non-celiac gluten sensitivity (though some researchers advocate avoidance of FODMAPs as opposed to gluten).
Celiac disease is an inflammatory immune disorder that occurs when genetically susceptible individuals eat gluten, a protein in wheat, barley, and rye. The immune system damages the lining of the small intestine, which can cause nutrient malabsorption and symptoms ranging from diarrhea and constipation to skin rashes and depression.
After adopting a gluten-free diet, there are natural interventions that can help replenish deficiencies caused by celiac disease, like supplementing with iron, magnesium, vitamin D, and digestive enzymes.
Differences between Wheat Allergy, Celiac Disease, and Non-Celiac Gluten Sensitivity
Wheat allergy, celiac disease, and non-celiac gluten sensitivity can be confused with each other but are actually three separate conditions:
- A wheat allergy involves the reaction of a certain type of antibody (immunoglobulin E) to wheat, resulting in the release of histamine and other inflammatory substances. This can cause symptoms such as skin eruptions, runny nose, itching, and in rare cases, anaphylaxis and even death.
- Celiac disease is an autoimmune disease, not an allergy. Autoimmune disease reactions are enduring and highly destructive, whereas allergic reactions can appear and disappear within minutes to hours after contact with an allergen.
- When wheat allergy and celiac disease have been ruled out (see Diagnosis of Celiac Disease), having a beneficial response to dietary elimination of gluten-containing foods is considered non-celiac gluten sensitivity. In some people, it may actually be poorly-absorbed carbohydrates called FODMAPs that contribute to sensitivity reactions.
Signs and Symptoms of Celiac Disease
Gastrointestinal:
- Pain, bloating, gas
- Constipation and/or diarrhea
- Loss of appetite
- Nausea, vomiting
Brain and nervous system:
- Recurring headaches
- Decreased sensation in peripheral nerves
- Anxiety, panic attacks
- Depression
Other systems of the body:
- Fatigue
- Itchy skin rash (dermatitis herpetiformis)
- Unexplained infertility and miscarriage
- Iron-deficiency anemia and other nutritional deficiencies
- Arthritis
Diagnosis of Celiac Disease
- Gold standard for diagnosis is a biopsy of the small intestine
- A more simple “four-out-of-five” guideline has been proposed:
-
- Typical symptoms of celiac disease (see Signs and Symptoms of Celiac Disease)
- Celiac disease immunoglobulin A antibodies are strongly positive in the serum
- HLA DQ2 and/or HLA DQ8 genes are present
- Characteristic changes are evident on small intestine biopsy
- Positive clinical response to a gluten-free diet
Dietary and Lifestyle Changes
- Maintaining a gluten-free diet for life is essential for those with celiac disease.
- The gluten-free diet is considered the primary treatment for non-celiac gluten sensitivity (though some researchers advocate avoidance of FODMAPs as opposed to gluten).
Integrative Interventions
- Minerals: Iron, calcium, magnesium, zinc, and selenium are some of the minerals that can become deficient in people with celiac disease, generally due to malabsorption. Screening for deficiency and supplementing as necessary is important.
- Vitamins: B vitamins and the fat-soluble vitamins A, D, E, and K can also become deficient due to malabsorption. Supplementation with these vitamins has been shown to improve bone status and neurological health in celiac patients with deficiencies in these vitamins.
- Probiotics: In a three-week randomized controlled trial, supplementation with a particular Bifidobacterium infantis strain in untreated celiac disease patients resulted in improvements in indigestion, constipation, and acid reflux.
- Digestive Enzymes: Pancreatic exocrine insufficiency may inhibit the ability of the pancreas to secrete digestive enzymes, a condition relatively common in celiac patients. Ninety percent of patients treated with digestive enzymes experienced a reduction in symptoms like diarrhea.
- L-carnitine: In a randomized controlled trial, supplementation with L-carnitine for six months resulted in significant improvement in reported fatigue in adults with celiac disease.
2 Introduction
Celiac disease is an inflammatory immune disorder that occurs in genetically susceptible individuals. In people with celiac disease, ingestion of gluten—the protein fraction of wheat, barley, and rye—provokes an immune attack that inflames and damages the lining of the small intestine. This typically results in nutrient malabsorption along with a wide variety of symptoms, ranging from diarrhea and constipation to skin rashes and depression (Brown 2012; Korponay-Szabo 2012; Lerner 2014; A.D.A.M. 2014). The reaction to gluten triggered by celiac disease is not an allergy, but rather an insidious inflammatory immune condition (FARE 2015a).
Untreated celiac disease can lead to the development of a host of other conditions including osteoporosis, infertility, neurological disorders, other autoimmune diseases, and in some cases, cancer (Ventura 1999; Fasano, Catassi 2012; Rashtak 2012; Kagnoff 2007; Sapone 2012; Bai 2013; UCMC 2014).
The prevalence of celiac disease has been rising sharply in recent decades. The reasons for the increase are not fully understood, but are thought to be due in part to better detection and diagnosis rates, as well as an actual increase in incidence, which may be a result of increasing dietary gluten content or changes in infant feeding patterns, among other possibilities (Sapone 2012). At least three million Americans, or approximately 1% of the US population, have celiac disease, making it more than four times as common today than just 30 years ago (Lundin 2012; Rubio-Tapia 2009; Lohi 2007).
Celiac disease is largely a hidden epidemic, since it is estimated that an alarming 85–90% of individuals in the United States who have celiac disease remain undiagnosed (Ferri 2015). In the United States, it takes an average of four years for a symptomatic person with celiac disease to be diagnosed, a delay that markedly increases the risk of developing autoimmune disorders, neurological problems, osteoporosis, and cancer (UCMC 2014).
In addition, emerging evidence suggests the existence of a clinically distinct sensitivity reaction to gluten-containing grains. This more recently characterized disorder has been referred to as both non-celiac wheat sensitivity and non-celiac gluten sensitivity (Carroccio 2012; Marchioni Beery 2015). This condition was described in the 1980s, and evidence for the existence of non-celiac gluten sensitivity has grown in recent years. Estimates suggest non-celiac gluten sensitivity is six times as common as celiac disease (Jackson 2012; Catassi 2013; Lundin 2012; Czaja-Bulsa 2014; Sapone 2012). Some recent research indicates that it may not be gluten or other proteins that cause people without celiac disease to react to these foods, but rather difficult-to-digest carbohydrates called FODMAPs (Biesiekierski 2013).
People with non-celiac gluten sensitivity can experience a spectrum of symptoms similar to those caused by celiac disease, such as abdominal pain, diarrhea, joint pain, and depression. Non-celiac gluten sensitivity does not appear to be an autoimmune condition, making it distinct from celiac disease; neither is non-celiac gluten sensitivity an allergic condition, which distinguishes it from classical wheat allergy (Sapone 2012). In fact, there are currently no universally recognized laboratory tests or biomarkers for non-celiac gluten sensitivity; the condition can be diagnosed by a double-blind challenge with gluten-containing food (Jackson 2012; Catassi 2013; Lundin 2012).
Currently, the only recognized treatment for celiac disease is strict, lifelong adherence to a gluten-free diet. This means avoiding all foods containing wheat, rye, barley, and their derivatives, as well as gluten-containing non-foods such as medicines and supplements. People with celiac disease must also be careful when selecting foods such as oats that may be contaminated with gluten-containing grains (Bai 2013; Sapone 2012; Fasano, Catassi 2012).
However, the gluten-free diet is nutritionally inadequate in some cases, whether as a result of poor food choices or inherent deficiencies in the diet due to lack of nutritional fortification of gluten-free foods (Shepherd 2013). While the gluten-free diet is also used to treat non-celiac gluten sensitivity, the need for strict adherence or permanency is not clear (Sapone 2012).
This protocol will give you a better understanding of celiac disease and non-celiac gluten sensitivity, and how they differ. The gluten-free diet will be reviewed and new treatment frontiers will be discussed. You will also learn about lifestyle changes and targeted nutritional support with several natural compounds that can complement the gluten-free diet by correcting nutritional deficiencies, promoting intestinal healing, balancing the immune system, and reducing chronic inflammation.
3 What Is Gluten?
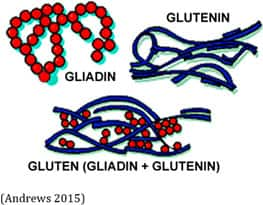
Gluten is not a single protein but rather a complex mixture of proteins, primarily gliadin and glutenin. Gluten forms when wheat flour is mixed with water to make dough. The combination of gliadin, a viscous (thick) protein, with glutenin, a long, elastic protein, yields gluten with its unique “visco-elastic” properties that are important in baking and food processing (Veraverbeke 2002; Allred 2010; Shewry 2002; Tatham 2000).
Thus, despite the common usage of the term “gluten-containing grains,” gluten itself is not actually present in the wheat seed; it is created during the formation of wheat flour dough (Hoseney 1990; Shewry 2002; Tilley 2001).
Rye and barley, the other “gluten” grains, do not actually form gluten (Tatham 2000). Instead, secalin proteins in rye and hordein proteins in barley contain toxic fragments that share properties with the wheat protein gliadin, most notably with regard to triggering celiac disease (Kagnoff 2007; Denham 2013). For convenience and simplicity, all three grains—wheat, rye, and barley—are considered “gluten-containing grains.”
Gliadin (wheat), secalin (rye), and hordein (barley) proteins are especially rich in two amino acid “building-blocks,” proline and glutamine, and thus are called “prolamins” (Stenman 2010; Hausch 2002; Denham 2013; Stern 2000). The bonds linking proline and glutamine on the gluten protein are resistant to digestion. This prevents complete breakdown of gluten into small, harmless molecules. Instead, large, undigested gluten fragments remain in the digestive tract (van den Broeck 2009; Kagnoff 2007; Denham 2013; Gass 2007).
In healthy people, these large, undigested fragments of gluten protein are eventually harmlessly excreted and do not provoke an immune response (Fasano 2009). For people with celiac disease, however, these glutamine- and proline-rich fragments, or peptides, are toxic (Sapone 2011; Holmes 2013; Volta 2013; Shan 2002).
4 Understanding Celiac Disease
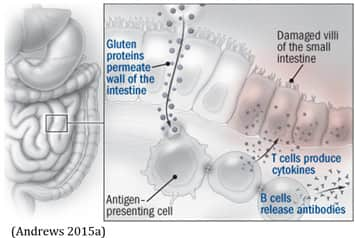
Much has been learned about how environmental and genetic factors interact with the immune system to inflame and damage the lining of the small intestine in celiac disease. The most important and best-characterized genetic components involved in the development of celiac disease are known as human leukocyte antigens (HLAs). More than 98% of people with celiac disease have at least one of two variants of a gene called HLA DQ. The HLA DQ2 variant of this gene is found in approximately 95% of celiac patients, while the HLA DQ8 variant is carried by the remainder. These genes play an integral role in priming the autoimmune response to gluten that occurs in celiac disease (Denham 2013; Sapone 2012; Green 2003).
Celiac disease is characterized by increased intestinal permeability (“leaky gut”), which allows gluten to seep through the compromised gut barrier and provoke a dysfunctional immune response (Fasano 2009; Fasano 2008; Lionetti 2011).
During this inflammatory immune assault against gluten, collateral damage to intestinal epithelial cells causes leakage of their contents into the fluid surrounding the cells. Included in this cellular discharge is a critical enzyme called tissue transglutaminase (Dieterich 1997; Fasano 2009). Tissue transglutaminase modifies the composition of the gliadin peptide, enabling gluten to bond strongly to the HLA DQ2 and DQ8 proteins. This then provokes an immune response from the T and B cells of the immune system and the production of antibodies against tissue transglutaminase (Lionetti 2011; Denham 2013; Fasano 2009).
The helper T cells of the immune system trigger the release of cytokines (signaling molecules), particularly interferon-gamma, which promote inflammation that specifically targets the lining of the small intestine, resulting in the autoimmune damage and flattening of the intestinal villi that are characteristic of celiac disease (Lionetti 2011; Kagnoff 2007; Denham 2013).
Villous Atrophy
Nutrient absorption occurs in the small intestine, a tube roughly 20–22 feet long with an inner surface that, when magnified, appears wrinkled into hundreds of folds. Each fold is covered with thousands of fingerlike projections called villi (singular: villus). Each villus, in turn, is covered with thousands of tiny, tightly packed projections called microvilli. Together, the villi and microvilli increase the intestinal absorptive surface area 60–120 times (Helander 2014; CARTA 2015; CHP 2014). The villi also contain enzymes that help break down food particles to their smallest components: proteins into amino acids, and carbohydrates into simple sugars (Skovbjerg 1981; Nordström 1967).
In celiac disease, intestinal damage induced by the autoimmune reaction to gluten ingestion manifests as a flattening of the villi (a hallmark of celiac disease), which reduces the surface area available for nutrient absorption. The resulting malabsorption leads to both intestinal consequences such as indigestion and diarrhea, and non-intestinal consequences such as anemia and osteoporosis (Kagnoff 2007; Guandalini 2014; Lionetti 2011).
Wheat Allergy vs. Celiac Disease vs. Non-celiac Gluten SensitivityWheat allergy, celiac disease, and gluten sensitivity are three separate conditions arising from distinct processes, though they may share certain signs and symptoms (UCLA Health 2015; ACDA 2015a). The conventional medical definition of wheat or gluten allergy refers to a less common condition that affects about 0.4–0.5% of the general population and which classically involves Immunoglobulin E (IgE) antibodies to wheat proteins, including gluten (Sapone 2012; Catassi 2013; Lundin 2012; Volta 2013). IgE antibodies play a central role in immediate food allergy reactions, the kind that usually occur within minutes to several hours after ingesting the allergenic food, though the reaction can occur up to two days later (Katta 2014). An allergy involves the release of histamine and other inflammatory substances and can cause serious symptoms such as skin eruptions, runny nose, itching, tearing, coughing, and in rare cases, anaphylaxis and even death (Matsumura 1994; Sapone 2012). IgE reactions to foods can be measured by blood or dermal testing (FARE 2015b; FARE 2015c). Celiac disease is an autoimmune disease, not an allergy. Whereas allergic reactions tend to be transitory, appearing and disappearing within minutes to hours after contact with an allergen such as wheat, the autoimmune reactions of celiac disease are enduring and highly destructive. Recovery of the normal structure of the intestinal lining takes 6–24 months of adherence to a strict gluten-free diet (Ferri 2015; Mayo Clinic 2014; UCLA Health 2015; ACDA 2015a). Non-celiac gluten sensitivity refers to a more recently-recognized condition characterized primarily by a beneficial response to dietary elimination of wheat-containing foods for people in whom celiac disease and classical IgE-mediated wheat allergy have been ruled out. Whereas wheat or gluten allergy and celiac disease are well-defined conditions, science still has much to learn about non-celiac gluten sensitivity, including the mechanisms underlying the condition and how best to diagnose it. Non-celiac gluten sensitivity is neither an autoimmune condition nor a classical allergic condition characterized by rapid IgE-mediated reactions (UCLA Health 2015; Sapone 2012). One study found that over half of non-celiac gluten sensitivity patients tested positive for antigliadin immunoglobulin G (IgG) antibodies, and some researchers consider IgG antigliadin antibody testing useful in identifying cases of non-celiac gluten sensitivity (Volta, Tovoli 2012; Mansueto 2014). A recent clinical study suggests gluten or other proteins in wheat and related grains may not be the sole cause of non-celiac reactivity to wheat. In some people, fermentable, oligo-, di-, monosaccharides, and polyols (FODMAPs), which are poorly absorbed carbohydrates, may contribute to sensitivity reactions to wheat among people without overt celiac disease or wheat allergy (Biesiekierski 2013). Table 1: Comparison of Wheat Allergy, Celiac Disease, and Non-celiac Gluten Sensitivity
(UCLA Health 2015; Ferri 2015; Sapone 2012; FARE 2015a) More general information about food allergies and food sensitivities is available in the Life Extension Magazine article titled What’s Really Making You Sick? |
5 Understanding Non-Celiac Gluten Sensitivity
Some people who do not have celiac disease or a wheat or gluten allergy may still experience symptoms in response to gluten ingestion. These symptoms can resemble those associated with celiac disease and may include gastrointestinal symptoms and non-intestinal symptoms. This newly-recognized gluten-related syndrome has been named non-celiac wheat sensitivity or non-celiac gluten sensitivity. Current knowledge about non-celiac gluten sensitivity is limited, and many unresolved questions remain to be clarified (Volta 2013; Sapone 2012).
Non-celiac gluten sensitivity is not an autoimmune disease and does not result in the same damage to the lining of the small intestine that is characteristic of celiac disease. Overall, non-celiac gluten sensitivity is less severe than celiac disease. The mechanism underlying the development of non-celiac gluten sensitivity is not clear, though it is known to be different from that of celiac disease and wheat allergy. There are conflicting reports, for example, as to whether people with non-celiac gluten sensitivity have altered gut barrier function (Biesiekierski 2011; Sapone 2011; Vazquez-Roque 2013).
As in celiac disease, the fast-acting or “innate” immune system is activated in non-celiac gluten sensitivity, causing moderate inflammation of the intestinal lining. However, unlike celiac disease, most evidence indicates that the adaptive immune system (also called acquired or specific immunity) is not activated in non-celiac gluten sensitivity (Lundin 2012; Catassi 2013; Volta 2013; Sapone 2011; Goronzy 2012; Volta, Di Giorgio 2012).
Gluten proteins may not be the sole or main triggers of non-celiac gluten sensitivity; other non-gluten components in wheat may play a role as well (Bucci 2013; Biesiekierski 2013). For instance, amylase-trypsin inhibitors are non-gluten proteins in wheat that are resistant to digestion. They are the primary allergens in Baker’s asthma, an occupational asthma that afflicts bakers and others exposed to grain and wheat flour dust (Salcedo 2011; James 1997). They can provoke inflammation and immune reactions in the cells of celiac and non-celiac patients, though their potential role in non-celiac gluten sensitivity has yet to be explored (Junker 2012).
Other non-protein constituents of wheat could elicit gastrointestinal symptoms in people with non-celiac gluten sensitivity. FODMAPs—an acronym for fermentable oligo-, di-, and monosaccharides and polyols—are poorly digested and incompletely absorbed carbohydrates found in a wide range of foods including gluten-containing grains, fruits, foods made with high fructose corn syrup or sugar alcohols, and dairy (SHC 2014).
FODMAPs are easily fermented by gut bacteria, which can result in gas, bloating, and diarrhea, symptoms shared by both irritable bowel syndrome and non-celiac gluten sensitivity (Barrett 2012; Magge 2012).
A 2013 randomized controlled trial in 37 patients with non-celiac gluten sensitivity and irritable bowel syndrome found that gluten did not produce negative effects when FODMAPs were restricted in the diet. The study authors concluded that non-celiac gluten sensitivity may not be distinct from reactions to FODMAPs (Biesiekierski 2013).
6 Risk Factors
Several risk factors for celiac disease are well established:
- Race. Caucasians are generally at higher risk (Murray 2013; Lidums 2015; A.D.A.M. 2014).
- Gender. Among women, the prevalence is 1.5–2.8 times higher than among men (Fasano, Catassi 2012; Gujral 2012).
- Family history. First- and second-degree relatives of celiac patients have a 10- to 15-fold and a 2.5-fold increased risk, respectively, compared to the general population (Bai 2013; Silvester 2013; Fasano, Catassi 2012).
- Genetics. Positive HLA DQ2 and DQ8 genes are almost always present in celiac disease patients. Approximately 95% of celiac patients carry the HLA DQ2 gene, while the remainder express the HLA DQ8 gene. Without one of these gene variants, celiac disease is extremely rare and unlikely (Sapone 2012; Denham 2013; Fasano 2009).
- Early gluten exposure. Early introduction of gluten into an infant’s diet increases risk. There is a 5-fold increased risk in children fed gluten during their first three months of life; introduction of gluten to the diet of genetically susceptible infants should be delayed until the 4th to 6th month of life, and the mother should continue to breastfeed (Ferri 2015).
- Breastfeeding status. Not breastfeeding increases the risk of celiac disease in childhood, and breastfeeding is protective. Gradually introducing gluten-containing foods while breastfeeding decreases the risk up to 48%. It is not known whether breastfeeding prevents adult onset of celiac disease (Lidums 2015).
The risk factors for non-celiac gluten sensitivity are not currently known, though the condition is believed to occur more commonly in females and in young and middle age adults (Catassi 2013).
7 Signs, Symptoms, and Associated Conditions
Celiac Disease
Signs, symptoms, and possible manifestations. Symptoms of celiac disease are generally categorized as gastrointestinal (typical) or extraintestinal (atypical) (see Table 2). Other forms of celiac disease include silent, or symptomless celiac disease, as well as potential celiac disease in which autoantibodies are detected but the intestinal lining is undamaged (Sperandeo 2011; Fasano 2001; Di Sabatino 2009; Lionetti 2011). The absence of symptoms in silent celiac disease can be attributed to the ability of the small intestine to compensate for lost function in the damaged area, but only when the damage is minimal. As intestinal damage progresses, symptoms appear (Presutti 2007).
In recent years, more cases of atypical celiac disease and silent celiac disease have been recognized (Telega 2008; Roma 2009; Guandalini 2014; Fasano, Catassi 2012). Many of the signs and symptoms of typical and atypical celiac, as well as the diseases associated with celiac, can be at least partially attributed to the nutrient malabsorption that results from damage to the absorptive surface of the small intestine (Lidums 2015).
Table 2: Possible Presentations of Celiac Disease
| Gastrointestinal Presentation (Typical) | Extraintestinal Presentation (Atypical) |
|
|
(Guandalini 2014; Ferri 2015; Kelly 2014; Nikpour 2012; Lawson 2005; Caruso 2013; Krzywicka 2014; Snyder 2015)
Associated conditions. Celiac disease has been associated with a number of other conditions, especially autoimmune-related conditions such as type 1 diabetes and autoimmune thyroid disease (Barker 2008; Kelly 2014). There is also some evidence that celiac disease may increase risk of certain cancers. A large population study in over 30 000 adult celiac patients corroborated earlier findings of increased risk of certain cancers in celiac patients. In this population, non-Hodgkin lymphoma occurred at a 1.94-fold increased incidence rate, and incidence of small intestinal cancer was increased 4.29-fold, compared with rates expected in the general population. The incidence of colon cancer and basal cell carcinoma were also modestly elevated, by 1.35-fold and 1.13-fold, respectively (Ilus 2014; Green 2003). Table 3 summarizes conditions that may be associated with celiac disease.
Table 3: Diseases and Conditions Associated with Celiac Disease
| Strong Association | Weak Association |
|
|
(Guandalini 2014; Ferri 2015; Kelly 2014; Nikpour 2012; Lawson 2005; Caruso 2013; Krzywicka 2014; Snyder 2015; Elfström 2007)
Non-celiac Gluten Sensitivity
Many symptoms of non-celiac gluten sensitivity are similar to those of celiac disease. As in celiac disease, they include symptoms similar to irritable bowel syndrome such as abdominal pain, bloating, diarrhea, gas, and constipation; and atypical symptoms such as headache, fatigue, joint and muscle pain, dermatitis, depression, and anemia (Eswaran 2013; Sapone 2012; Volta 2013; Lundin 2012). The time interval between gluten exposure and onset of symptoms in celiac disease can be quite long—up to weeks and years—whereas it may be only a matter of a few hours to a few days in non-celiac gluten sensitivity (Catassi 2013; Sapone 2012; Volta, De Giorgio 2012).
There is no evidence that serious, long-term complications occur in non-celiac gluten sensitivity (Catassi 2013).
The Effects of Gluten on the Brain and Nervous SystemAmong atypical or extraintestinal signs of celiac disease, brain and nervous system problems are prominent (Hu 2006; Bushara 2005; Cascella 2011; Jackson 2012; Briani 2008; Rodrigo 2011; Nikpour 2012). One mechanism for gluten’s effects on the brain may involve autoimmune antibodies against a subtype of tissue transglutaminase called transglutaminase 6. This enzyme is structurally similar to transglutaminase types involved in intestinal and skin damage in celiac disease, but transglutaminase 6 is found primarily in the brain and nervous system. Antibodies against this enzyme have been identified in patients with gluten ataxia and schizophrenia. Importantly, they do not appear to be present in healthy individuals (Hadjivassiliou 2008; Cascella 2013). Whether antibodies to transglutaminase 6 are involved in other neurological and psychiatric conditions associated with celiac disease is not known, and will require further research. However, there is evidence that transglutaminase 6, and the transglutaminase enzyme family in general, may be important in the development of degenerative diseases of the brain (Thomas 2013; Jeitner 2009; De Vivo 2009; Martin 2011). Multiple sclerosis is an inflammatory autoimmune disorder that damages the insulating covers of nerve cells in the brain and spinal cord (Compston 2002). Recently, the prevalence of celiac disease was reported to be 5–10 times higher in a study in patients with multiple sclerosis compared to the general population. All the patients in this study had an excellent response to the gluten-free diet in an average of three years, with regard to both digestive and neurological symptoms (Rodrigo 2011). A 2014 case report showed celiac disease can mimic multiple sclerosis. A 43-year-old male patient with a history of diarrhea and colic since youth and neurological symptoms since age 18 was diagnosed with multiple sclerosis at age 34. Seven years later, though his blood tests did not show any of the typical celiac-related antibodies, he was diagnosed with celiac disease based on neurologic signs and symptoms, positive tests for HLA DQ2 and DQ8, and improvement in both digestive and neurological symptoms on a gluten-free diet (Finsterer 2014). |
8 Diagnosis
Celiac Disease
A blood test for immunoglobulin A (IgA) type autoimmune tissue transglutaminase antibodies is usually the first diagnostic step for celiac disease in most patients. Another test, IgA endomysial antibodies, may be used to confirm positive results. Deamidated gliadin peptide antibodies are tested for when serum IgA is low, a common occurrence in celiac disease (Ferri 2015; Rubio-Tapia 2013). Elevated antigliadin immunoglobulin G (IgG) antibody levels may also present in people with celiac disease who have recently consumed gluten. However, elevated IgG levels are not specific for celiac disease, and healthy people may sometimes have elevated IgG levels. Combined screening with IgG and IgA tests can be helpful in some cases, especially in people with IgA deficiency, which is thought to occur in about 2‒3% of individuals with celiac disease (ACDA 2015b).
A convenient screening panel, the Celiac Disease Antibody Screen, tests for immunoglobulin A antibodies to tissue transglutaminase, the most important blood test for celiac disease; serum immunoglobulin A levels, which can be low in celiac disease; and deamidated gliadin immunoglobulin A (Rubio-Tapia 2013).
Blood tests alone are often not considered adequate to confirm a diagnosis of celiac disease. The gold standard for a celiac diagnosis is a biopsy of the small intestine demonstrating flattened villi (Guandalini 2014; Gujral 2012; Rubio-Tapia 2013).
Genetic testing for HLA DQ2 and HLA DQ8 may be performed for three reasons: negative tests rule out almost all cases of celiac disease, including those already on a gluten-free diet; negative tests may help clarify the picture when the result of other testing is unclear; and these tests are used to screen close relatives of patients for genetic risk of celiac disease (those who are positive go on to receive conventional testing as listed previously) (Ferri 2015; Kelly 2014; Lidums 2015).
A simple “four-out-of-five” guideline has been proposed for the diagnosis of celiac disease, which would require that a minimum of four out of the five criteria below be met. Current practice guidelines, however, rely on more complex diagnostic algorithms. Generally, a biopsy demonstrating villous atrophy is still considered the “gold standard” for celiac diagnosis (Husby 2012; Rubio-Tapia 2013; Guandalini 2014; Ludvigsson 2014; Sapone 2012).
Four-Out-Of-Five Diagnosis Guideline (Sapone 2012)
- Typical symptoms of celiac disease (See “Signs and Symptoms”).
- Serum celiac disease immunoglobulin A antibodies are strongly positive.
- HLA DQ2 and/or HLA DQ8 genes are present.
- Characteristic changes, known as celiac enteropathy, are evident on small intestine biopsy.
- Positive clinical response to a gluten-free diet.
Distinguishing Celiac Disease from Irritable Bowel SyndromeThe misdiagnosis of celiac disease as irritable bowel syndrome (IBS) deserves special mention. Individuals diagnosed with IBS are considered to be at four- to seven-fold increased risk of celiac disease compared with the general population (Cristofori 2014; Sanders 2001). However, proper diagnosis of IBS necessarily includes ruling out conditions such as celiac disease (Moleski 2013). Typical gastrointestinal presentation of celiac disease may closely mimic the symptoms of IBS, including recurrent abdominal discomfort, bloating, constipation, and diarrhea (Lidums 2015; Verdu 2009). Patients who have received a diagnosis of IBS should confirm that celiac disease was adequately tested for and ruled out (Rodrigo 2013; Ford 2009; Zwolińska-Wcisło 2009; Spiegel 2004; Sanders 2001). More information on IBS can be found in Life Extension’s Irritable Bowel Syndrome protocol. |
Non-celiac Gluten Sensitivity
While there are currently no universally recognized laboratory tests or biomarkers for non-celiac gluten sensitivity, wheat allergy and celiac disease must be ruled out in order to make a non-celiac gluten sensitivity diagnosis (Czaja-Bulsa 2014; Sapone 2012). The best available procedure to confirm non-celiac gluten sensitivity is a double-blind, placebo-controlled gluten challenge that demonstrates that gluten-related symptoms are relieved by a gluten-free diet (Volta 2013; Sapone 2012).
9 The Gluten-Free Diet: An Overview
The only established treatment for celiac disease is strict, lifelong adherence to a gluten-free diet (Ferri 2015; Kelly 2014; Lidums 2015; Fasano, Catassi 2012). Absolute compliance to the gluten-free diet is vital, as even a tiny amount of gluten can lead to intestinal damage in those with celiac disease. In most cases, with strict adherence, symptoms are halted, existing intestinal damage is healed, and further damage is prevented (UCMC 2013).
The gluten-free diet excludes all foods that contain wheat (including spelt, triticale, and kamut), rye, and barley (Dirks 2004; Bai 2013). This means avoiding these grains, as well as pasta, cereals, and processed foods—unless they are labeled “gluten-free.” The status of oats in the gluten-free diet is less clear; it is thought that most celiac patients can eat modest amounts of pure oats, though some celiac patients do not tolerate oats (Fasano, Catassi 2012; Real 2012). Unless labeled “gluten-free,” oats should be avoided as they can be contaminated with gluten-containing grains (UCMC 2013; Kagnoff 2007).
There are many gluten-free alternatives to wheat: amaranth, buckwheat, corn, millet, rice (all varieties), quinoa, sorghum, teff, yucca, potato, nuts, flax, and all beans and legumes are naturally gluten-free (CDF 2015). There are also alternatives to gluten-containing flour, including potato, rice, soy, tapioca, carob, and bean flour (Zingone 2010; Eberman 2005).
The gluten-free diet is typically considered the primary treatment for non-celiac gluten sensitivity (though some researchers advocate avoidance of FODMAPs as opposed to gluten). While the level of gluten tolerance varies among individuals with non-celiac gluten sensitivity, with rare exceptions, most of these individuals can eat trace amounts of gluten without negative health consequences (Molina-Infante 2014; Sapone 2012; Volta 2013).
Unfortunately, lifelong strict adherence to a gluten-free diet is often difficult. Gluten is common in many types of food, and gluten-free products are often expensive and not widely available. Many people have difficulty maintaining the diet (Vahedi 2003; Abdulkarim 2002).
Gluten-Free Foods: Some Examples
Table 4 contains examples of foods that are allowed and those that are not allowed when eating gluten-free. This is not a complete list, however. It is important to read all ingredient labels carefully to make sure that a food does not contain gluten.
Table 4: Allowed and Disallowed Foods in the Gluten-Free Diet
| Allowed Grains and Starches | ||
|
amaranth |
legumes/beans |
sorghum |
|
Foods To Avoid |
||
|
wheat
|
barley |
|
|
Other Wheat Products to Avoid |
||
|
bromated flour |
graham flour |
self-rising flour |
|
Processed Foods that May Contain Wheat, Barley, or Rye* |
||
|
bouillon cubes |
French fries |
seasoned tortilla chips |
(NIDDK 2012)
* Most of these foods can be found gluten-free. When in doubt, carefully read food labels and check with the
food manufacturer.
Avoiding FODMAPs: Potential Benefits for Those with Non-celiac Gluten Sensitivity?Although several studies have identified specific reactions to the protein components of wheat and related grains in some people with non-celiac gluten sensitivity, limited evidence suggests that poorly absorbed carbohydrates called fermentable, oligo-, di-, monosaccharides, and polyols (FODMAPs) may drive symptoms in some individuals (Biesiekierski 2013; El-Salhy 2015). In one study of people with self-reported non-celiac gluten sensitivity, reducing FODMAP intake significantly reduced gastrointestinal symptoms (Biesiekierski 2013). Thus, avoiding dietary FODMAPs may be beneficial for gluten-sensitive individuals who fail to achieve relief by specifically avoiding gluten. Limiting FODMAP intake has also been shown to improve symptoms among individuals with irritable bowel syndrome (IBS), which has several similarities with non-celiac gluten sensitivity (Lowe 2014; Staudacher 2014). The low-FODMAP diet emphasizes avoidance of fructose, lactose, fructans, galactans, and polyols. Some examples of foods eliminated in a low-FODMAP diet are wheat, barley, and rye; high-lactose dairy; foods made with high-fructose corn syrup; certain fruits; soy protein; mushrooms; among others (SUMC 2014). Monash University in Australia has developed a comprehensive low-FODMAP diet program to assist individuals who wish to try this dietary approach. More information is available on Monash University’s website, here: http://www.med.monash.edu/cecs/gastro/fodmap/. |
10 Novel and Emerging Therapies
In light of the limitations of the gluten-free diet, several new treatment strategies for celiac disease are currently under development. These novel and emerging therapies can be classified into four general categories: permeability inhibition, immunomodulation, grain modification, and gluten detoxification.
While ongoing research on novel therapies is targeting celiac disease, some of the dietary modification and gluten detoxification strategies described in this section may provide solutions for people with non-celiac gluten sensitivity.
Permeability Inhibition
Zonulin antagonism. Zonulin is a protein that regulates the tight junctions that “glue” intestinal cells together and control what passes through the gut wall. In celiac disease, overproduction of zonulin causes these tight junctions to come apart and remain open, increasing intestinal permeability (Tripathi 2009; Fasano 2012a). This “leaky gut” then allows large molecules, including gluten fragments, to enter the bloodstream, where they trigger an immune response and inflammation (Fasano 2012b; Fasano 2011; van Elburg 1993).
An investigational drug named larazotide acetate blocks zonulin from making the gut permeable, and appears to be effective in preventing gluten-related symptoms in celiac disease (Gopalakrishnan 2012). In a randomized controlled trial in patients with celiac disease, acute gluten exposure caused a 70% increase in intestinal permeability in the placebo group, while no change in permeability was seen in the larazotide group. The patients treated with larazotide tolerated the drug well and experienced fewer gastrointestinal symptoms. Moreover, larazotide-treated subjects exhibited a lessened inflammatory response to gluten in comparison with the placebo group, as determined by changes in levels of several inflammatory cytokines (Paterson 2007).
A more recent phase IIb trial evaluated larazotide in 342 celiac patients who did not respond to a gluten-free diet. Compared with controls, patients taking the zonulin-inhibitor at the lowest dose (0.5 mg daily) had significantly fewer gastrointestinal symptoms and headaches, as well as less fatigue. No changes in serum levels of anti-tissue transglutaminase or gliadin antibodies were seen (AGA 2014; Castillo 2014). The drug producer, Alba Therapeutics, is planning for phase III clinical trials of lazarotide’s efficacy and safety (AZO Network 2014). Larazotide acetate has been granted fast-track designation by the Food and Drug Administration (FDA), which is a process by which therapies that help address an unmet medical need may be approved and become available in the market more quickly (PR Newswire 2014).
Immunomodulation
Vaccination. A preventive vaccine may be one of the most attractive and exciting novel therapies for patients with celiac disease (Aziz 2011). The theory behind vaccination is that tolerance to gluten can be induced through repeated small exposures to strongly immunogenic fragments of gluten. The immune system becomes trained to not react to gluten, thus preventing an autoimmune attack in the small intestine (Bakshi 2012). Just this type of gluten vaccine (Nexvax2) is under development. Nexvax2 is a mixture of the three toxic peptides—gliadin (wheat), hordein (barley), and secalin (rye)—that provoke the immune system in individuals with HLA DQ2-positive genes. Nexvax2 was shown to be safe and well tolerated in a three-week phase I clinical trial in celiac patients (Castillo 2014; Rashtak 2012; Bakshi 2012). Phase II and III clinical trials will be necessary before FDA approval.
Inhibition of tissue transglutaminase 2. Tissue transglutaminase 2 (tTG2) is an enzyme that leaks out of damaged cells and modifies gluten. It is this modified gluten that is presented to immune cells, eventually leading to autoimmune and inflammatory responses in the intestine. As tTG2 is vital to the development of celiac disease, selective inhibition of tTG2 could be another potential therapeutic approach for celiac disease (Kupfer 2012; Esposito 2007; Siegel 2007).
However, tTG2 has many other diverse biological functions such as wound healing and assisting the ingestion of microbes or other foreign particles by immune cells (Siegel 2007). Therefore, inhibiting tTG2 may result in systemic side effects. Nevertheless, in animal studies a series of compounds called dihydroisoxazoles were shown to efficiently inhibit tTG2 with low systemic toxicity (Choi 2005; Watts 2006). Inhibition of tTG2 is still in the discovery phase with no clinical trials yet initiated.
Blocking HLA DQ2 and HLA DQ8. In celiac disease, the human leukocyte antigens HLA DQ2 and HLA DQ8 bind with gluten peptides and induce an immune reaction that leads to disease development. Blocking the binding sites of these HLA antigens may provide yet another treatment alternative. Various gluten analogues (structurally similar to gluten) have been developed that effectively bind and inhibit HLA DQ2, which is present in over 90% of cases of celiac disease (Juse 2010; Huan 2011; Kapoerchan 2008; Xia 2007). The primary concern with these HLA antagonists is the possibility of interfering with other similar HLAs that are involved in normal immune surveillance. This strategy is still in the discovery phase (Rashtak 2012).
Anti-interferon-gamma and anti-TNF-alpha therapies. Interferon-gamma and tumor necrosis factor-alpha (TNF-α) are cytokines (signaling molecules) that are activated by gluten’s interaction with T cells of the immune system. These cytokines contribute to the inflammatory cascade in celiac disease. Interventions that block these cytokines have been investigated in celiac disease. Infliximab (Remicade), an anti-TNF-α monoclonal antibody used in the treatment of inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis, was beneficial in two case reports of unresponsive celiac disease (Guo 2013; Nilsen 1998; Pena 1998; Gillett 2002; Costantino 2008; Woodward 2013).
Anti-interleukin-15 therapy. Interleukin-15 (IL-15) is another cytokine that plays a
significant role in the development of celiac disease. Genetically altered mice that produce excess IL-15 in
intestinal cells develop inflammation along with flattening of the villi in the small intestine, much like
the damage that occurs in celiac disease. One trial showed that in mice, blocking IL-15 with antibodies
reversed this autoimmune damage (Yokoyama 2009). AMG 714, an anti-IL-15 antibody, is being evaluated in
phase II clinical trials for the autoimmune diseases rheumatoid arthritis (Amgen 2007) and psoriasis (Amgen
2014). Targeting IL-15 is a plausible treatment strategy for celiac disease as well, though human trials are
still lacking (Sollid 2011).
CCR9 antagonists. The chemokine receptor CCR9 acts as a trafficking or homing molecule that
selectively causes T cells to move to the small intestine, where they promote persistent inflammation. This
homing receptor has been associated with celiac disease (Olaussen 2007). An antagonist to CCR9 called
CCX282-B induced a clinical response and maintenance of clinical remission in Crohn’s disease (Keshav
2013). A phase II trial of CCX282-B in celiac disease has been completed, but results are pending. Due to a
lack of specificity, this drug may increase the risk of gastrointestinal infections (Stoven 2013; Sollid
2011).
Grain Modification
Wheat variants. The development and production of variant wheat strains that contain reduced levels of toxic gluten proteins is one novel approach currently under exploration. Success in this endeavor might allow people to consume wheat while reducing their exposure to gluten. Several studies have reported that various species of ancient wheat (einkorn) and some strains of pasta wheat (durum) contain little to no immunogenic gluten peptides. It appears that ancient forms of wheat contained less total gluten and toxic gluten fragments than modern bread wheat (Molberg 2005; Spaenij-Dekking 2005).
Research efforts are focused on breeding wheat strains similar to ancient wheat that lack harmful gluten peptides. Identification and breeding of grain varieties with less immunotoxic profiles is in development (Molberg 2005).
Gluten Detoxification
Enzyme therapy. Enzyme therapy is a promising new intervention designed to detoxify gluten. Certain protein-digesting enzymes (proteases) derived from plants and microorganisms can completely break down gluten peptides into harmless fragments. One such enzyme preparation, AN-PEP, is a proline-specific protease extracted from the common fungus Aspergillus niger. This enzyme specifically cleaves the proline-rich gliadin peptides, making them less immunogenic. Experiments with cell cultures demonstrated that AN-PEP can withstand the strongly acidic environment of the stomach, and fully digest gluten and immunogenic peptides (Stepniak 2006). Human clinical trials are underway to confirm this.
Another protease, ALV003, is further along in development and appears to have greater therapeutic potential. Composed of a proline-specific protease and a glutamine-specific protease called EP-B2, this two-enzyme combination was shown to be superior to either enzyme alone (Siegel 2006; Siegel 2012). According to a phase II trial, ALV003 appeared to reduce gluten-induced small intestinal damage in patients with celiac disease who were challenged with up to 2 g gluten per day while following a gluten-free diet (Lähdeaho 2014). A 500-person multi-center trial is currently ongoing (Adelman 2014).
11 Nutrients
Iron
Iron-deficiency anemia is one of the most common extraintestinal signs of celiac disease (Horowitz 2011; Caruso 2013); iron deficiency without anemia may also occur. Although iron deficiency is usually corrected by a gluten-free diet, restoration of iron levels can require 6–12 months after the lining of the small intestine has healed (Caruso 2013). Blood tests for iron deficiency, including a complete blood count and ferritin level, are important in diagnosis (Ferri 2015) and iron supplementation is considered part of necessary care for celiac patients, in part due to the nutritional insufficiency possible with a gluten-free diet (Thompson 2000; Kupper 2005; Lidums 2015; Kelly 2014).
Calcium
In celiac disease, damage to the intestinal lining often leads to calcium malabsorption and decreased serum levels of calcium (Rastogi 2012; Pantaleoni 2014; Babar 2011; Szymczak 2012), which then lead to bone loss, osteopenia, osteoporosis, and increased fracture risk (AGA 2001; Pantaleoni 2014). Calcium levels and bone mineral density improve on a gluten-free diet, but only after one to two years (Caruso 2013; Molteni 1995). However, a gluten-free diet can be low in calcium, and can lead to calcium deficiency (Kupper 2005; UCMC 2013; Blazina 2010) and subsequent low bone mineral density (Blazina 2010). Thus, supplementation with calcium is important for patients with celiac disease (Rastogi 2012; Mancini 2011; Szymczak 2012; Blazina 2010; Lucendo 2013; Caruso 2013; Bianchi 2008).
Vitamin D
Vitamin D deficiency is a common problem in celiac disease and is associated with a range of musculoskeletal disorders including bone pain, muscle disorders (myopathy), loss of bone mineral density, osteopenia, and osteoporosis (Mulder 2011; Rastogi 2012; Rabelink 2011; Karaahmet 2014; Caruso 2013). One study found low vitamin D levels in 64% of men and 71% of women with celiac disease (Kemppainen 1999). Importantly, however, this study defined vitamin D “deficiency” as below 10 ng/mL 25-hydroxyvitamin D, which is far below levels considered optimal. Also, evidence suggests serum vitamin D levels decrease with age in individuals with celiac disease, and low levels occur even in sunny climates (Lerner 2012). A case report described a celiac patient with myopathy and a vitamin D level below 4 ng/dL. The patient was started on a gluten-free diet and initiated vitamin D supplementation at a dose of 300 000 IU once every two weeks, while under physician supervision. The patient’s symptoms resolved within a week (Karaahmet 2014).
Supplementation with vitamin D and calcium, along with a gluten-free diet, has been shown to markedly improve bone status in celiac patients (Duerksen 2012; Bianchi 2008; Rastogi 2012), and supplementation with vitamin D and calcium is widely recommended for celiac patients (Szymczak 2012; Lucendo 2013; Rastogi 2012). Vitamin D has many additional functions beyond its role in the musculoskeletal system, particularly modulation of the immune system and anti-inflammatory effects. Data from laboratory studies provide evidence for vitamin D’s potential to decrease systemic inflammation and prevent autoimmune disease in humans (Richards 2007; Kriegel 2011; Agmon-Levin 2013; Antico 2012). Specifically, vitamin D inhibits secretion of pro-inflammatory cytokines such as interferon-gamma and TNF-α, while promoting the production of anti-inflammatory cytokines including IL-4, IL-5, and IL-10 (Prietl 2013).
Blood tests for 25-hydroxyvitamin D can determine baseline levels and monitor changes in levels resulting from vitamin D supplementation and a gluten-free diet.
B Vitamins
Deficiencies of folate and vitamins B12 and B6 are common in celiac disease (Dickey 2002; Haapalahti 2005; Tikkakoski 2007; Dahele 2001; Reinken 1978; Caruso 2013), and supplementation with these B vitamins, and also niacin (vitamin B3) and riboflavin (vitamin B2), is considered an important part of conventional care for celiac patients (Hadithi 2009; Caruso 2013; Wierdsma 2013; Halfdanarson 2007; Lidums 2015). In one study, folate and pyridoxal 5’-phosphate, the active form of vitamin B6, were deficient in 37% and 20% of adult celiac patients, respectively, who had been on a gluten-free diet for 10 years and who exhibited biopsy-proven recovery. Among the participants in this study, mean daily folate and vitamin B12 intake were significantly lower in those with celiac than in controls (Hallert 2002).
Homocysteine is an amino acid derivative that can damage the lining of blood vessels and promote atherosclerotic disease (Eren 2014; Pushpakumar 2014). Elevated homocysteine is often attributable to a lack of vitamins B12, B6, and folate, which are necessary for the metabolism of homocysteine (Higdon 2014a; Higdon 2014b). Several studies have demonstrated elevated blood levels of homocysteine in celiac patients (Hallert 2002; Dickey 2008; Saibeni 2005). One study showed celiac patients who took vitamin supplements had higher blood levels of folate and vitamins B6 and B12, and lower levels of homocysteine, compared with those who did not use supplements (Hadithi 2009). Deficiency in folate and vitamin B12 can cause B-deficiency anemia, which is common in celiac disease, occurring in up to 34% of untreated patients (Wierdsma 2013; Halfdanarson 2007).
Magnesium
Magnesium is an essential mineral involved in hundreds of enzymatic reactions in the body (Higdon 2013a). Magnesium is also required for proper metabolism of vitamin D and calcium, and thus for bone health (Zofkova 1995; Hardwick 1991).
The importance of magnesium deficiency in celiac disease was first recognized over fifty years ago, when a detailed case report showed a dramatic improvement in magnesium absorption and status in a young adult celiac patient after she commenced a gluten-free diet (Goldman 1962). Another study investigated magnesium status in children and adolescents with celiac disease. While all of the patients with classical celiac disease (with malabsorption) were magnesium deficient, only 1 in 5 patients on a gluten-free diet (no malabsorption) and 1 in 5 patients with silent celiac disease (no malabsorption) were low in magnesium (Rujner 2001). A later study examined magnesium in 41 children and adolescents with celiac disease or normal intestinal villi who had been on a gluten-free diet for an average of 11 years; 28 patients with untreated atypical celiac disease and eight controls were also included in the study. The researchers found magnesium deficiency, as assessed by red blood cell magnesium levels, in 14.6% of those on the gluten-free diet and 25% of untreated patients (Rujner 2004).
An earlier study in 23 adult celiac patients found that all of them had depleted intracellular magnesium levels despite being asymptomatic (no malabsorption) and on a gluten-free diet. Magnesium therapy in these individuals resulted in increases in both red blood cell magnesium and bone mineral density, suggesting magnesium deficiency may play a role in the development of osteoporosis in people with celiac disease (Rude 1996). Screening for magnesium status and deficiency, and magnesium supplementation and dietary enrichment, are considered essential aspects of care for celiac patients (Ferri 2015; Lidums 2015; Caruso 2013).
Zinc
Zinc is an essential nutrient mineral that acts as a catalytic agent for over 300 separate enzymatic reactions in the body (Higdon 2013b). Zinc deficiency is common in untreated celiac disease, and this deficiency is not always corrected by a gluten-free diet; zinc supplementation is considered a necessary part of celiac treatment by some sources (Caruso 2013; Lidums 2015; Kupper 2005). Zinc deficiency may be a culprit in poor growth, immune function, and wound healing, and skin problems in celiac disease.
A review of studies of both untreated and treated celiac patients found a remarkable prevalence of zinc deficiency, as measured by either plasma or serum zinc. Eleven separate studies of untreated adults and children with celiac disease found zinc deficiency in up to 100% of patients. Three of nine studies of celiac patients on a gluten-free diet for as little as three months to as long as 10 years found zinc deficiency persisted in 20–40% of patients. On a gluten-free diet alone, zinc deficiency usually takes a full year to resolve, and monitoring of serum zinc levels in celiac patients has been suggested (Caruso 2013; Wierdsma 2013; Guevara Pacheco 2014).
Vitamin E
Vitamin E is an important fat-soluble vitamin and free radical scavenger that plays a major role in protecting cell membranes from oxidative damage (Higdon 2008). Several studies have reported low levels of vitamin E in individuals with celiac disease (Odetti 1998; Guevara Pacheco 2014; Henri-Bhargava 2008). Vitamin E deficiency has also been implicated in the development of neurological symptoms in celiac disease (Kleopa 2005; Mauro 1991; Battisti 1996; Henri-Bhargava 2008). Vitamin E supplementation, together with a gluten-free diet, has been shown to improve neurological impairment caused by vitamin E deficiency in celiac disease (Kleopa 2005; Henri-Bhargava 2008; Mauro 1991).
Vitamin A
Vitamin A is a fat-soluble nutrient that can become deficient in celiac disease patients as a result of malabsorption. Vitamin A is important for normal immune function, vision, and gene expression (Higdon 2007a). The treatment for vitamin A deficiency in celiac disease is both a gluten-free diet and supplementation (Lidums 2015). One study of newly diagnosed celiac patients found that 7.5% of them were deficient in vitamin A versus none in a healthy control group (Wierdsma 2013). A case study reaffirmed the importance of vitamin A, even in controlled celiac cases. A 64-year-old man, with biopsy-proven celiac disease controlled on a gluten-free diet, presented with recent onset of diarrhea as well as redness and blurring in one eye. During three weeks of treatment of his eye symptoms with medication, his condition deteriorated. Testing of vitamin A revealed a stark deficiency. The patient received an intramuscular injection containing 100 000 IU vitamin A, and dramatic improvement in vision and the condition of his eye was noted within a week. This patient’s follow-up included regular vitamin A treatment (Alwitry 2000).
Vitamin K
Vitamin K is another fat soluble vitamin that can become deficient in celiac disease as a result of malabsorption (Lidums 2015; Mager 2012; Kelly 2014; Lidums 2015). Vitamin K deficiency can contribute to easy bruising, an atypical sign of celiac disease; vitamin K deficiency has also been shown to increase risk of blood clots and bleeding events in individuals with celiac disease (Berthoux 2011; Chen 2007; Vaynshtein 2004; Lerner 2014; Lerner 2014; Chen 2007; Vaynshtein 2004). In a 2012 study in children and adolescents with celiac disease, dietary intake of vitamin K was assessed. At diagnosis, 41% of patients had intakes less than half of the recommended amount, while intake remained deficient in 31% of patients after one year on a gluten-free diet (Mager 2012).
Children with both symptomatic and asymptomatic celiac disease have been found to have reduced bone mass at diagnosis (Mager 2012; Turner 2009; Rajani 2010). Vitamin K enhances calcium metabolism and therefore plays an important role in bone health (Zittermann 2001; Booth 2009). Because dietary intake of calcium and vitamins K and D is often inadequate, including for patients on a gluten-free diet, routine supplementation with vitamin K at the time of diagnosis of celiac disease has been suggested (Mager 2012).
Selenium
Selenium is an integral part of the enzyme glutathione peroxidase, which plays a major role in cellular antioxidant defense (Higdon 2007b). In a study of 30 children with celiac disease, 80% had serum selenium levels below the normal range (Yuce 2004). Selenium is essential for thyroid hormone regulation, and selenium deficiency in celiac disease has been linked to an increased risk of autoimmune thyroid disease (Stazi 2008; Stazi 2010).
Probiotics
Probiotics are beneficial microorganisms that colonize the intestine and play an important role in intestinal health; the balance of these organisms appears to be deranged in patients with celiac disease (Pozo-Rubio 2012; Round 2009). In a three-week randomized controlled trial, supplementation with Bifidobacterium infantis NLS super strainin untreated celiac disease patients resulted in improvements in indigestion, constipation, and acid reflux (Smecuol 2013).
Various strains of probiotics are being developed to specifically target mechanisms of immune and intestinal damage in celiac disease. The probiotic strain Bifidobacterium lactis has been shown to protect cultured gut epithelial cells from damage induced by gluten, possibly by blocking gluten-induced increases in intestinal permeability (Lindfors 2008). Also, in a lab experiment, a blend of eight different strains of probiotic bacteria, including several Lactobacillus and Bifidobacterium species, was demonstrated to extensively pre-digest gluten proteins in wheat flour. When intestinal biopsy samples from celiac patients were exposed to these pre-digested gluten proteins, recruitment of immune cells was reduced. Researchers concluded that the probiotic formulation reduced or eliminated the toxicity of wheat gluten (De Angelis 2006).
L-Carnitine
L-carnitine is a compound made in the human body from the amino acids lysine and methionine and also obtained in our diet primarily from animal foods such as meat, fish, and dairy (Higdon 2012a).
In a randomized controlled trial, supplementation with 2 g L-carnitine daily for six months resulted in
significant improvement in reported fatigue in adults with celiac disease. The L-carnitine dosage was safe
and well tolerated (Ciacci 2007).
L-carnitine is required to shuttle long chain fatty acids into the mitochondria (power plants of the cell)
for muscle energy production. The intestinal damage in celiac disease results in a decrease in absorptive
surface area, increasing risk of L-carnitine deficiency. In one study, untreated celiac patients were found
to have low serum L-carnitine concentrations, which returned to normal with a gluten-free diet (Lerner
1993). Another study found low serum carnitine levels in children with celiac disease (Yuce 2004).
L-carnitine deficiency may produce fatigue, weakness, and difficulty gaining weight, all common symptoms in
celiac disease (Lerner 1993; Guandalini 2014).
Omega-3 Fatty Acids
Omega-3 fatty acids have important biological anti-inflammatory effects (Skulas-Ray 2015; Yan 2013; Calder 2015). When ingested through food or supplements, omega-3 fatty acids compete with the more pro-inflammatory omega-6 fatty acid arachidonic acid for incorporation into lipid-rich cell membranes (Westphal 2011; Calder 2007; Pischon 2003; Surette 2008).
A study of serum fatty acid composition in adults with newly diagnosed celiac disease found that concentrations of omega-3 fatty acids were significantly lower than those of a control group. After one year on a gluten-free diet and having achieved clinical remission, levels of omega-3 fatty acids increased but still remained well below control values (Solakivi 2009). A study in children with celiac disease and type 1 diabetes mellitus found their omega-3 fatty acid levels—including docosapentaenoic and docosahexaenoic acid (DHA)—were significantly lower than those in a control group (Tarnok 2015).
A study that used cultures of intestinal epithelial cells (enterocytes) supports the potential benefit of oral supplementation with DHA in reducing intestinal inflammation in celiac disease. In response to inflammation triggered by gluten, enterocytes release arachidonic acid. DHA was shown to block this release of arachidonic acid and the resulting inflammatory cascade that furthers disease development (Vincentini 2011; Ferretti 2012). Another mechanism by which omega-3 fatty acids reduce inflammation is through inhibition of nuclear factor-kappaB (NF-ĸB), a pro-inflammatory mediator (Lee 2009). DHA also stimulates a cellular receptor called PPAR-gamma, which activates genes that lessen the production of pro-inflammatory cytokines (Zapata-Gonzalez 2008).
Vitamin C
Vitamin C is a water-soluble free radical scavenger that is also essential to the structural integrity of tissues throughout the body (Higdon 2013c). In one experiment, cultured intestinal biopsies from celiac patients were challenged with the gliadin component of gluten with and without the addition of vitamin C. The addition of vitamin C to the cultured cells decreased the secretion of pro-inflammatory cytokines TNF-α, IL-6, and interferon-gamma. Vitamin C also completely inhibited the secretion of the pro-inflammatory cytokine IL-15, which is strongly implicated in the damage that occurs in celiac disease (Bernardo 2012; Gujral 2012).
Glutathione
Glutathione is one of the body’s most powerful defenses against destructive free radicals, and is essential to the liver’s detoxification functions (NLM 2015; Kidd 1997). Several studies have reported significant reductions in glutathione concentrations in the intestinal lining (Stojiljkovic 2009; Stojiljkovic 2012) and peripheral blood (Stojiljkovic 2007; Stojiljkovic 2012) of celiac patients compared with controls. As a result, concentrations of lipid peroxides, or rancid fat, in the intestinal lining increased. Lipid peroxides can promote a cascade of oxidative damage in tissues, contributing to the tissue damage in the intestine that is characteristic of celiac disease, and to the increased risk of cancer in celiac patients (Stojiljkovic 2007; Stojiljkovic 2009; Stojiljkovic 2012).
The body’s glutathione reserves can be increased by means of direct supplementation with glutathione (Richie 2014) or with the use of physiologic precursors such as cysteine (Lee 2013; Sekhar 2011) and N-acetyl cysteine (NLM 2015; Kidd 1997), as well as with alpha-lipoic acid, selenium, and whey protein (Chen 2011; Zavorsky 2007; Jiang 2012).
Pancreatic Enzymes
Exocrine pancreatic insufficiency, a condition in which not enough digestive enzymes are secreted into the small intestine (Toouli 2010), is relatively common in celiac disease patients who do not respond to a gluten-free diet (Ferri 2015). Damage to the small intestine may inhibit the ability of the pancreas to secrete digestive enzymes. One study found that in cases in which diarrhea persisted even after a gluten-free diet, there was a significantly higher prevalence of laboratory signs of pancreatic insufficiency. Patients treated with pancreatic enzymes experienced a 75% reduction in symptoms. Another study found that the severity of villous atrophy in the small intestine was significantly associated with laboratory signs of pancreatic exocrine insufficiency. Pancreatic enzyme insufficiency was found to occur in over half of celiac patients in one study, and to return to normal after one year on a gluten-free diet (Malterre 2009; Evans 2010; Weizman 1987; Regan 1980).
Additional Support
The following interventions have not been thoroughly studied in the context of celiac disease, but may nonetheless be of benefit by helping support intestinal health or combatting inflammatory responses or cellular metabolic derangements, which are thought to underlie several manifestations of celiac disease.
Curcumin. Curcumin is the principal polyphenolic compound in turmeric, a plant used in Indian Ayurvedic medicine for centuries (Higdon 2009). Curcumin has powerful anti-inflammatory and antioxidant properties and has demonstrated in laboratory, animal, and human models an ability to protect against a wide range of conditions including autoimmune disease and IBD (Gupta 2013; Aggarwal 2009; Brumatti 2014; Bright 2007).
Like Crohn’s disease and ulcerative colitis, celiac disease is an inflammatory autoimmune disorder (Ferri 2015). In celiac disease, the inflammatory mediator interferon-gamma promotes enhanced permeability and damage in the small intestine (DiRaimondo 2012; Nilsen 1998). Curcumin has been shown to suppress interferon-gamma, suggesting that curcumin may be beneficial in celiac disease (Fahey 2007).
Interestingly, curcumin may be able to ameliorate psychological manifestations of celiac disease and non-celiac gluten sensitivity as well. In a randomized controlled trial, a highly bioavailable form of curcumin was found to be as effective as the standard antidepressant medication fluoxetine (Prozac) in the treatment of major depressive disorder (Sanmukhani 2014; Antony 2008). Since depression and other psychological manifestations of celiac disease and non-celiac gluten sensitivity are being recognized with increasing frequency (Peters 2014; Smith 2012; Bushara 2005; Carta 2002; Corvaglia 1999; Addolorato 1996), this is another of curcumin’s benefits that may be valuable in these gluten-related disorders.
Alpha-lipoic acid. Alpha-lipoic acid is a naturally occurring compound centrally involved in energy production within the mitochondria of the cell. It is also a powerful free radical scavenger (Higdon 2012b). In addition, alpha-lipoic acid regenerates other free radical scavengers such as glutathione, vitamin C, vitamin E, and coenzyme Q10 (Chen 2011; Packer 2011; Jones 2002).
Alpha-lipoic acid’s ability to increase levels of glutathione is important in celiac disease, in which low glutathione levels are common (Stojiljkovic 2007; Stojiljkovic 2009; Stojiljkovic 2012). In an animal model, dietary supplementation with alpha-lipoic acid significantly increased both cellular glutathione concentrations and the activity of glutathione peroxidase, a key glutathione-dependent antioxidant enzyme. Compared with controls, levels of oxidized lipids (peroxidation) were significantly lower as well (Chen 2011). Other studies support alpha-lipoic acid’s potential to accelerate glutathione synthesis (Packer 2011; Kolgazi 2007; Suh 2004). In a rodent model of gut inflammation, a feature of celiac disease, treatment of rats with alpha-lipoic acid markedly reduced several measures of damage to the gut lining (Kolgazi 2007).
Boswellia serrata. In recent years, preparations from the gummy resin of Boswellia serrata and other boswellia species have become popular in parts of Europe for the treatment of various chronic inflammatory conditions including chronic bowel diseases. Clinical studies suggest boswellia is safe and effective in the treatment of autoimmune bowel diseases (Crohn’s disease and ulcerative colitis). Boswellic acids, which are active constituents of boswellia, exert anti-inflammatory effects through inhibition of proinflammatory pathways involving 5-lipoxygenase, leukotrienes, and TNF-α (Ammon 2006). In a similar manner, boswellia may help reduce the intestinal inflammation of celiac disease.
Green tea extract (EGCG). Animal studies have indicated that green tea extract and its chief constituent, epigallocatechin-3-gallate (EGCG), are capable of interrupting inflammatory tissue damage that contributes to the symptoms of celiac and other autoimmune disease. Helper T cells are key drivers in many autoimmune diseases including celiac disease (Monteleone 2001). One mechanism for the protective effect of EGCG is suppression of autoreactive T cells and reduced production of pro-inflammatory cytokines. EGCG was also shown to inhibit interferon-gamma, a dominant inflammatory mediator in celiac disease (Wu 2012).
In a human cell-line study, EGCG prevented interferon-gamma from increasing intestinal permeability (Watson 2004). In an animal model of colitis, green tea extract significantly diminished damage to the colon, as evidenced by reduced amounts of inflammatory markers, less diarrhea, and slowing of weight loss (Mazzon 2005).
Glutamine. Glutamine, an amino acid, is especially important for the lining of the gastrointestinal tract, particularly during stress and illness. Glutamine is the most abundant amino acid in the body, and a critical fuel for the cells lining the intestine. In fact, oral glutamine supplementation is capable of increasing the height of intestinal villi, helping the cells of the gut lining to proliferate, and maintaining integrity of the gut lining and preventing excessive permeability (de Vasconcelos 1998; Miller 1999). As a regulator of tight junction proteins that “glue” intestinal cells together (Li 2004), glutamine has been shown to exert a positive effect on gut barrier function (permeability) in a variety of clinical conditions (Beutheu 2013; Sevastiadou 2011; Choi 2007). In a 2012 randomized controlled trial in patients with Crohn’s disease in remission phase, oral glutamine administered at 0.23 g per pound body weight for two months significantly improved intestinal permeability and mucosal integrity (Benjamin 2012).
Glutamine supplementation has yet to be studied in celiac disease. However, since supplementation with glutamine has been demonstrated to safely enhance and maintain gut barrier function, its application in the increased permeability caused by celiac disease is an area of great scientific interest (Kuhn 2010; Beutheu 2013; Choi 2007; Benjamin 2012; Fasano 2011; Clemente 2003; Fasano 2005; Arrieta 2006).
Whey protein. Glutathione forms an integral part of the defense mechanisms that protect the intestinal mucosa (gut lining) from oxidative damage (Stojiljkovic 2009). Whey protein is a rich source of cysteine that is needed for the synthesis of glutathione (Alt Med Rev 2008). Several studies have shown that whey protein is highly effective for increasing glutathione levels in various types of cells (Zavorsky 2007; Vilela 2006; Kent 2003; Grey 2003; Middleton 2004). Additionally, in a 2012 study in patients with Crohn’s disease, whey protein significantly reduced intestinal permeability (Benjamin 2012). This benefit of whey on gut integrity may be useful in celiac disease, and whey protein can easily be incorporated into a gluten-free diet as a source of high-quality protein and glutathione precursors.
Zinc-carnosine. Zinc-carnosine, a free radical scavenger and anti-glycation agent, is composed of zinc and carnosine (Alhamdani 2007). In a randomized crossover trial, healthy participants were given either zinc-carnosine or placebo after five days of indomethacin (Indocin) treatment. The indomethacin caused a 3-fold increase in gut permeability in the placebo group, while zinc-carnosine prevented this rise in permeability in the treatment group. Although more studies are needed, zinc-carnosine appears to help maintain the integrity of the gut barrier (Mahmood 2007).
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
A.D.A.M. PubMed Health. Celiac disease - sprue. Available at: http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0001280/. 2/21/2014. Accessed 2/24/2015.
Abdulkarim AS, Burgart LJ, See J, et al. Etiology of nonresponsive celiac disease: results of a systematic approach. Am J Gastroenterol 2002;97(8):2016-2021.
ACDA. American Celiac Disease Alliance. Diagnosis: What are the appropriate screening tests for celiac disease? Available at: http://americanceliac.org/celiac-disease/diagnosis/ Accessed 4/24/2015b.
ACDA. American Celiac Disease Alliance. What is Celiac Disease? Available at: http://americanceliac.org/celiac-disease/. Last updated 2/18/2015a. Accessed 3/6/2015.
Addolorato G, Stefanini GF, Capristo E, et al. Anxiety and depression in adult untreated celiac subjects and in patients affected by inflammatory bowel disease: a personality “trait” or a reactive illness? Hepatogastroenterology. 1996;Nov-Dec;43(12):1513-7.
Adelman D. Alvine Pharmaceuticals, Inc. National Institutes of Health. A phase 2b, Randomized, Double-Blind, Placebo-Controlled Dose-Ranging Study of the Efficacy and Safety of ALV003 Treatment in Symptomatic Celiac Disease Patients Maintained on a Gluten-Free Diet. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2014- [cited 2014 Dec 2]. Available at: https://clinicaltrials.gov/ct2/show/NCT01917630?term=ALV003&rank=4. NLM Identifier: NCT01917630.
AGA. The American Gastroenterological Association. American Gastroenterological medical position statement: celiac sprue. Gastroenterology. 2001;120(6):1522-5.
AGA. The American Gastroenterological Association. The American Gastroenterological Association Institute. Digestive Disease Week 2014. The AGA Report. Available at: http://www.gastro.org/journals-publications/gi-hepatology-news/GI_-_Hepatology_News_2014_DDW_Report.pdf. Updated 2014. Accessed 12/10/14.
Aggarwal BB, Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41:40-59.
Agmon-Levin N, Theodor E, Segal RM, et al. Vitamin D in systemic and organ-specific autoimmune diseases. Clinic Rev Allerg Immunol. 2013;45:256-266.
Alhamdani MS, Al-Kassir AH, Abbas FK, Jaleel NA, Al-Taee MF. Antiglycation and antioxidant effect of carnosine against glucose degradation products in peritoneal mesothelial cells. Nephron. Clinical practice. 2007;107(1):c26-34.
Allred LK, Ritter BW. Recognition of gliadin and glutenin fractions in four commercial gluten assays. Journal of AOAC International. Jan-Feb 2010;93(1):190-196.
Alt Med Rev. Therapeutic applications of whey protein. Alternative Medicine Review. 2008;13(4):341-347.
Alwitry A. Vitamin A deficiency in coeliac disease. The British Journal of Ophthalmology. 2000;84(9):1075-1075.
Amgen, Inc. National Institutes of Health. AMG 714 20060349 Multiple Dose Study in Moderate to Severe Psoriasis Subjects. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2014- [cited 2014 Dec 9]. Available at: http://www.clinicaltrials.gov/ct2/show/NCT00443326?term=AMG+714&rank=1. NLM Identifier: NCT00443326
Amgen, Inc. National Institutes of Health. Phase 2 AMG 714 in Rheumatoid Arthritis. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2007- [cited 2014 Dec 8]. Available at: http://www.clinicaltrials.gov/ct2/show/NCT00433875?term=AMG714&rank=2. NLM Identifier: NCT00433875.
Ammon HP. Boswellic acids in chronic inflammatory diseases. Planta Med. 2006;72:1100-1116.
Andrews R. All about gluten. Precision Nutrition. Available at: http://www.precisionnutrition.com/all-about-gluten. 2015. Accessed 12/22/2014.
Andrews R. All about gluten. Precision Nutrition. Available at: http://www.precisionnutrition.com/all-about-gluten. 2015a. Accessed 12/28/2014.
Antico A, Tampoia M, Tozzoli R, et al. Can supplementation with vitamin D reduce the risk or modify the course of autoimmune diseases? a systematic review of the literature. Autoimmunity Reviews 2012;12:127-136.
Antony B, Merina B, Iyer VS, et al. Bioavailability of BCM-95 CG (Biocurcumax), a novel bioenhanced preparation of curcumin. Indian J Pharm Sci. 2008;Jul-Aug;70(4):445-9.
Arrieta MC, Bistritz L, Meddings JB. Alterations in intestinal permeability. Gut. 2006;55:1512:1520.
Aziz I, Evans KE, Papageorgiou V, et al. Are patients with celiac disease seeking alternative therapies to a gluten-free diet? J Gastrointest Liver Dis. 2011;Mar;20(1):27-31.
AZO Network. Alba Therapeutics reports positive results from Phase IIb trial of larazotide acetate for CeD treatment. Available at: http://www.news-medical.net/news/20140211/Alba-Therapeutics-reports-positive-results-from-Phase-IIb-trial-of-larazotide-acetate-for-CeD-treatment.aspx. 2/11/2014. Accessed 3/2/2015.
Babar MI, Ahmed I, Rao MS, et al. Celiac disease and celiac crisis in children. J Coll Physicians Surg Pak. 2011;Aug;21(8):487-90.
Bai JC, Fried M, Corazza GR, et al. World gastroenterology organization global guidelines on celiac disease. J Clin Gastroenterol 2013; Feb;47(2):121-126.
Bakshi A, Stephen S, Borum ML, et al. Emerging therapeutic options for celiac disease. Gastroenterol Hepatol. 2012;8(9):582-588.
Barker JM, Liu E. Celiac disease: pathophysiology, clinical manifestations, and associated autoimmune conditions. Advances in pediatrics. 2008;55:349-365.
Barrett JS, Gibson PR. Fermentable oligosaccharides, disaccharides, monosaccharides and polyols (FODMAPs) and nonallergic food intolerance: FODMAPs or food chemicals? Therapeutic advances in gastroenterology. Jul 2012;5(4):261-268.
Battisti C, Dotti MT, Formichi P, et al. Disappearance of skin lipofuscin storage and marked clinical improvement in adult onset coeliac disease and severe vitamin E deficiency after chronic vitamin E megatherapy. J Submicrosc Cytol Pathol. 1996;Jul;28(3):339-44.
Benjamin J, Makharia G, Ahuja V, et al. Glutamine and whey protein improve intestinal permeability and morphology in patients with Crohn’s disease: a randomized controlled trial. Dig Dis Sci. 2012;57:1000-1012.
Bernardo D, Martinez-Abad B, Vallejo-Diez S, et al. Ascorbate-dependent decrease of the mucosal immune inflammatory response to gliadin in celiac disease patients. Allergol Immunopathol (Madr). 2012;40(1):3-8.
Berthoux E, Fabien N, Chayvialle JA, Ninet J, Durieu I. [Adult celiac disease with thrombosis: a case series of seven patients. Role of thrombophilic factors]. La Revue de medecine interne / fondee ... par la Societe nationale francaise de medecine interne. Oct 2011;32(10):600-604.
Beutheu S, Ghouzali I, Galas L, et al. Glutamine and arginine improve permeability and tight junction protein expression in methotrexate-treated Caco-2 cells. Clinical Nutrition. 2013;32:863-869.
Bianchi ML, Bardella MT. Bone in celiac disease. Osteoporos Int. 2008;19:1705-1716.
Biesiekierski JR, Appl B, Newnham ED, et al. Gluten causes gastrointestinal symptoms in subjects without celiac disease: a double-blind randomized placebo-controlled trial. Am J Gastroenterol. 2011;106:508-514.
Biesiekierski JR, Peters SL, Newnham ED, et al. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology. 2013;Aug;145(2):320-8.
Blazina S, Bratanic N, Campa AS, et al. Bone mineral density and importance of strict gluten-free diet in children and adolescents with celiac disease. Bone. 2010;Sep;47(3):598-603.
Booth SL. Roles for vitamin K beyond coagulation. Annu Rev Nutr. 2009;29:89-110.
Briani C, Zara G, Alaedini A, et al. Neurological complications of celiac disease and autoimmune mechanisms: a prospective study. J Neuroimmunol. 2008;195:171-175.
Bright JJ. Curcumin and autoimmune disease. Adv Exp Med Biol. 2007;595:425-451.
Brown K, DeCoffe D, Molcan E, Gibson DL. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients. Aug 2012;4(8):1095-1119.
Brumatti LV, Marcuzzi A, Tricarico PM, et al. Curcumin and inflammatory bowel disease: potential and limits of innovative treatments. Molecules 2014;19:211270-21153.
Bucci C, Zingone F, Russo I, et al. Gliadin does not induce mucosal inflammation or basophil activation in patients with nonceliac gluten sensitivity. Clin Gastroenterol Hepatol. 2013;Oct;11(10):1294-1299.
Bushara KO. Neurologic presentation of celiac disease. Gastroenterology. 2005;128:S92-S97.
Calder PC. Immunomodulation by omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2007;77:327-335.
Calder PC. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochimica et biophysica acta. Apr 2015;1851(4):469-484.
Carroccio A, Mansueto P, Iacono G, et al. Non-celiac wheat sensitivity diagnosed by double-blind placebo-controlled challenge: exploring a new clinical entity. Am J Gastroenterol. 2012;107(12):1898-906.
Carta MG, Hardoy MC, Boi MF, et al. Association between panic disorder, major depressive disorder and celiac disease: a possible role of thyroid autoimmunity. J Psychosom Res. 2002;Sep;53(3):789-93.
CARTA. Center for Academic Research & Training in Anthropogeny. Small/Large Intestine Length Ratio. Available at: http://carta.anthropogeny.org/moca/topics/smalllarge-intestine-length-ratio. Accessed 2/25/2015.
Caruso R, Pallone F, Stasi E, et al. Appropriate nutrient supplementation in celiac disease. Ann Med. 2013;Dec;45(8):522-31.
Cascella NG, Kryszak D, Bhatti B, et al. Prevalence of celiac disease and gluten sensitivity in the United States clinical antipsychotic trials of intervention effectiveness study population. Schizophrenia Bulletin. 2011;37(1):94-100.
Cascella NG, Santora D, Gregory P, et al. Increased prevalence of transglutaminase 6 antibodies in sera from schizophrenia patients. Schizophrenia Bulletin. 2013;39(4):867-871.
Castillo NE, Theethira TG, Leffler DA. The present and the future in the diagnosis and management of celiac disease. Gastroenterol Rep (Oxf). 2014; Oct 17:1-9.
Catassi C, Bai JC, Bonaz B, et al. Non-celiac gluten sensitivity: the new frontier of gluten related disorders. Nutrients. 2013;Oct;5(10):3839-3853.
Catassi C, Fasano A. Celiac disease diagnosis: simple rules are better than complicated algorithms. Am J Med. 2010 Aug;123(8):691-3.
CDF. Celiac Disease Foundation. What Can I Eat? Available at: http://celiac.org/live-gluten-free/glutenfreediet/food-options/. Copyright 2015. Accessed 3/2/2015.
Chen CS, Cumbler EU, Triebling AT. Coagulopathy due to celiac disease presenting as intramuscular hemorrhage. Journal of general internal medicine. Nov 2007;22(11):1608-1612.
Chen P, Ma QG, Ji C, et al. Dietary lipoic acid influences antioxidant capability and oxidative status of broilers. Int J Mol Sci. 2011;12(12):8476-8488.
Choi K, Lee SS, Oh SJ, et al. The effect of oral glutamine on 5-fluorouracil/leucovorin-induced mucositis/stomatitis assessed by intestinal permeability test. Clin Nutr. 2007;26:57-62.
Choi K, Siegel M, Piper JL, et al. Chemistry and biology of dihydroisoxazole derivatives: selective inhibitors of human transglutaminase 2. Chem Biol. 2005;Apr;12:469-475.
CHP. Children's Hospital of Pittsburgh. Organs: Small and Large Intestines. Available at: http://www.chp.edu/CHP/organs+intestine. Last updated 12/5/2014. Accessed 2/25/2015.
Ciacci C, Peluso G, Iannoni E, et al. L-carnitine in the treatment of fatigue in adult celiac disease patients: a pilot study. Dig Liver Dis. 2007;39:922-928.
Clemente MG, De Virgiliis S, Kang JS, et al. Early effects of gliadin on enterocyte intracellular signaling involved in intestinal barrier function. Gut. 2003;52:218-223.
Compston A, Coles A. Multiple sclerosis. Lancet. 2002;Apr 6;359(9313):1221-31.
Corvaglia L, Catamo R, Pepe G, et al. Depression in adult untreated celiac subjects: diagnosis by the pediatrician. Am J Gastroenterol. 1999;Mar;94(3):839-43.
Costantino G, della Torre A, Lo Presti MA, et al. Treatment of life-threatening type 1 refractory celiac disease with long-term infliximab. Dig Liver Dis. 2008;40:74-7.
Cristofori F, Fontana C, Magista A, Capriati T, Indrio F, Castellaneta S, . . . Francavilla R. Increased prevalence of celiac disease among pediatric patients with irritable bowel syndrome: a 6-year prospective cohort study. JAMA pediatrics. Jun 2014;168(6):555-560.
Czaja-Bulsa G. Non coeliac gluten sensitivity – a new disease with gluten intolerance. Clin Nutr. 2014;Aug 29;1-6.
Dahele A, Ghosh S. Vitamin B12 deficiency in untreated celiac disease. Am J Gastroenterology. 2001;96(3):745-750.
De Angelis M, Rizzello CG, Fasano A, et al. VSL#3 probiotic preparation has the capacity to hydrolyze gliadin polypeptides responsible for Celiac Sprue. Biochim Biophys Acta. 2006;Jan;1762(1):80-93.
de Vasconcelos MI, Tirapegui J. [Nutritional importance of glutamine]. Arquivos de gastroenterologia. Jul-Sep 1998;35(3):207-215.
De Vivo G, Di Lorenzo R, Ricotta M, Gentile V. Role of the transglutaminase enzymes in the nervous system and their possible involvement in neurodegenerative diseases. Current medicinal chemistry. 2009;16(36):4767-4773.
Denham JM, Hill ID. Celiac disease and autoimmunity: review and controversies. Curr Allergy Asthma Rep. 2013;13:347-353.
Di Sabatino A, Corazza GR. Coeliac disease. Lancet. 2009;373:1480-93.
Dickey W, Ward M, Whittle CR, et al. Homocysteine and related B-vitamin status in coeliac disease: effects of gluten exclusion and histological recovery. Scand J Gastroenterol. 2008;43:682-688.
Dickey W. Low serum vitamin B12 is common in coeliac disease and is not due to autoimmune gastritis. Eur J Gastroenterol Hepatol. 2002;14(4):425-427.
Dieterich W, Ehnis T, Bauer M, et al. Identification of tissue transglutaminase as the autoantigen of celiac disease. Nat Med. 1997;3(7):797-801.
DiRaimondo TR, Klock C, Khosla C, et al. Interferon gamma activates transglutaminase 2 via a phosphatidylinositol-3-kinase-dependent pathway: implications for celiac sprue therapy. J Pharmacol Exp Ther. 2012;341(1):104-114.
Dirks MH. Practical considerations for the identification and follow-up of children with celiac disease. Paediatrics & child health. Dec 2004;9(10):714-718.
Duerksen DR. Dramatic effect of vitamin D supplementation and a gluten-free diet on bone mineral density in a patient with celiac disease. J Clin Densitom. 2012;Jan-Mar;15(1):120-123.
Eberman LE, Cleary MA. Celiac disease in an elite female collegiate volleyball athlete: a case report. Journal of athletic training. Oct-Dec 2005;40(4):360-364.
Elfström P, Montgomery SM, Kämpe O, et al. Risk of primary adrenal insufficiency in patients with celiac disease. J Clin Endocrinol Metab. 2007;92(9):3595-8.
El-Salhy M, Gundersen D. Diet in irritable bowel syndrome. Nutrition journal. Apr 14 2015;14(1):36.
Eren E, Ellidag HY, Aydin O, Yilmaz N. Homocysteine, Paraoxonase-1 and Vascular Endothelial Dysfunction: Omnibus viis Romam Pervenitur. Journal of clinical and diagnostic research: JCDR. Sep 2014;8(9):Ce01-04.
Esposito C, Caputo I, Troncone R. New therapeutic strategies for coeliac disease: tissue transglutaminase as a target. Curr Med Chem. 2007;14:2572-2580.
Eswaran S, Goel A, Chey WD. What role does wheat play in the symptoms of irritable bowel syndrome? Gastroenterology & hepatology. Feb 2013;9(2):85-91.
Evans KE, Leeds JS, Morley S, Sanders DS. Pancreatic insufficiency in adult celiac disease: do patients require long-term enzyme supplementation? Digestive diseases and sciences. Oct 2010;55(10):2999-3004.
Fahey AJ, Adrian Robins R, Constantinescu CS. Curcumin modulation of IFN-beta and IL-12 signalling and cytokine induction in human T cells. Journal of cellular and molecular medicine. Sep-Oct 2007;11(5):1129-1137.
FARE. Food Allergy Research & Education. About Food Allergies. Blood Tests. Available at: http://www.foodallergy.org/diagnosis-and-testing/blood-tests. Copyright 2015c. Accessed 3/6/2015.
FARE. Food Allergy Research & Education. About Food Allergies. Skin Prick Test. Available at: http://www.foodallergy.org/diagnosis-and-testing/skin-tests. Copyright 2015b. Accessed 3/6/2015.
FARE. Food Allergy Research & Education. About Food Allergies. Wheat Allergy. Available at: http://www.foodallergy.org/allergens/wheat-allergy. Copyright 2015a. Accessed 3/6/2015.
Fasano A, Catassi C. Celiac disease. N Engl J Med. 2012;Dec 20;367(25):2419-2426.
Fasano A, Catassi C. Current approaches to diagnosis and treatment of celiac disease: an evolving spectrum. Gastroenterology. 2001;Feb;120(3):636-651.
Fasano A, Shea-Donohue T. Mechanisms of disease: the role of intestinal barrier function in the pathogenesis of gastrointestinal autoimmune diseases. Nat Clin Pract Gastroenterol Hepatol. 2005;Sep;2(9):416-22.
Fasano A. Intestinal permeability and its regulation by zonulin: diagnostic and therapeutic implications. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association. Oct 2012a;10(10):1096-1100.
Fasano A. Leaky gut and autoimmune diseases. Clinic Rev Allerg Immunol. 2012b;42:71-78.
Fasano A. Physiological, pathological, and therapeutic implications of zonulin-mediated intestinal barrier modulation: living life on the edge of the wall. Am J Pathol. 2008;Nov;173:1243-52.
Fasano A. Surprises from celiac disease. Scientific American. 2009;301(2):54-61.
Fasano A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiological Reviews. 2011;Jan1;91(1):151-175.
Ferretti G, Bacchetti T, Masciangelo S, Saturni L. Celiac disease, inflammation and oxidative damage: a nutrigenetic approach. Nutrients. Apr 2012;4(4):243-257.
Ferri FF. Ferri's Clinical Advisor. Celiac Disease. Available at: www.clinicalkey.com. Copyright 2015. Accessed 2/3/2015.
Finsterer J, Leutmezer F. Celiac disease with cerebral and peripheral nerve involvement mimicking multiple sclerosis. J Med Life. 2014;Sep 15;7(3):440-4.
Ford AC, Chey WD, Talley NJ, et al. Yield of diagnostic tests for celiac disease in individuals with symptoms suggestive of Irritable bowel syndrome: systematic review and meta-analysis. Arch Intern Med. 2009;Apr 13;169(7):651-8.
Gass J, Bethune MT, Siegel M, et al. Combination enzyme therapy for gastric digestion of dietary gluten in patients with celiac sprue. Gastroenterology. 2007;133:472-480.
Gillett HR, Arnott IDR, McIntyre M, et al. Successful infliximab treatment for steroid-refractory celiac disease: a case report. Gastroenterology. 2002;122:800-805.
Goldman AS, Van Fossan DD, Baird EE. Magnesium deficiency in celiac disease. Pediatrics. Jun 1962;29:948-952.
Gopalakrishnan S, Tripathi A, Tamiz AP, et al. Larazotide acetate promotes tight junction assembly in epithelial cells. Peptides. 2012;35:95-101.
Goronzy JJ, Weyand CM. The Innate and Adaptive Immune Systems. In: Goldman's Cecil Medicine, 24th edition. Available at: www.clinicalkey.com. Copyright 2012 by Saunders, an imprint of Elsevier Inc.
Green PHR, Jabri B. Coeliac disease. Lancet. 2003;Aug 2;362:383-391.
Grey V, Mohammed SR, Smountas AA, et al. Improved glutathione status in young adult patients with cystic fibrosis supplemented with whey protein. J Cyst Fibros. 2003;2:195-198.
Guandalini S, Assiri A. Celiac disease: a review. JAMA Pediatr 2014;168(3):272-278.
Guevara Pacheco G, Chavez Cortes E, Castillo-Duran C. [Micronutrient deficiencies and celiac disease in Pediatrics]. Arch Argent Pediatr. 2014;Oct;112(5):457-63.
Gujral N, Freeman HJ, Thomson ABR. Celiac disease: Prevalence, diagnosis, pathogenesis and treatment. World J Gastroenterol. 2012;18(42):6036-6059.
Guo Y, Lu N, Bai A. Clinical use and mechanisms of infliximab treatment on inflammatory bowel disease: a recent update. BioMed research international. 2013;2013:581631.
Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;Jan;15(1):195-218.
Haapalahti M, Kulmala P, Karttunen TJ, et al. Nutritional status in adolescents and young adults with screen-detected celiac disease. J Pediatr Gastroenterol of Nutr. 2005;May;40:566-570.
Hadithi M, Mulder CJJ, Stam f, et al. Effect of B vitamin supplementation on plasma homocysteine levels in celiac disease. World J Gastroenterol. 2009;Feb 28;15(8):955-960.
Hadjivassiliou M, Aeschlimann P, Strigun A, et al. Autoantibodies in gluten ataxia recognize a novel neuronal transglutaminase. Ann Neurol. 2008;Sep;64(3):332-43.
Halfdanarson TR, Litzow MR, Murray JA. Hematologic manifestations of celiac disease. Blood. 2007;Jan 15;109(2):412-421.
Hallert C, Grant C, Grehn S, et al. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333-1339.
Hardwick LL, Jones MR, Brautbar N, Lee DB. Magnesium absorption: mechanisms and the influence of vitamin D, calcium and phosphate. The Journal of nutrition. Jan 1991;121(1):13-23.
Hausch F, Shan L, Santiago NA, et al. Intestinal digestive resistance of immunodominant gliadin peptides. Am J Physiol Gastrointest Liver Physiol. 2002;June5;283:G996-G1003.
Helander HF, Fandriks L. Surface area of the digestive tract – revisited. Scand J Gastroenterol. 2014;Jun;49(6):681-9.
Henri-Bhargava A, Melmed C, Glikstein R, et al. Neurologic impairment due to vitamin E and copper deficiencies in celiac disease. Neurology. 2008;Sep 9;71(11):860-1.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Magnesium. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin B12. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin B6. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Zinc. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin E. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin A. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Selenium. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. L-carnitine. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Curcumin. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Vitamin C. Data on file.
Higdon J. Oregon State University. Linus Pauling Institute. Micronutrient Information Center. Lipoic Acid. Data on file.
Holmes G. Non coeliac gluten sensitivity. Gastroenterology and hepatology from bed to bench. Summer 2013;6(3):115-119.
Horowitz S. Celiac Disease: New Directions in Diagnosis, Treatment, and Prevention. Alternative and Complementary Therapies. 2011;17(2):92-98.
Hoseney RC, Rodgers DE. The formation and properties of wheat flour doughs. Crit Rev Food Sci Nutr. 1990;29(2):73-93.
Hu WT, Murray JA, Greenaway MC, et al. Cognitive impairment and celiac disease. Arch Neurol. 2006;63:1440:1446.
Huan J, Meza-Romero R, Mooney JL, et al. Single-chain recombinant HLA-DQ2.5/peptide molecules block alpha2-gliadin-specific pathogenic CD4+ T-cell proliferation and attenuates production of inflammatory cytokines: a potential therapy for celiac disease. Mucosal Immunol. 2011;Jan;4(1):112-120.
Husby S, Koletzko S, Korponay-Szabo I, Mearin M, Phillips A, Shamir R, . . . Catassi C. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. Journal of pediatric gastroenterology and nutrition. 2012;54(1):136-160.
Ilus T, Kaukinen K, Virta LJ, et al. Incidence of malignancies in diagnosed celiac patients: a population-based estimate. Am J Gastroenterol. 2014;Sep;109(9):1471-7.
Jackson JR, Eaton WW, Cascella NG, et al. Neurologic and psychiatric manifestations of celiac disease and gluten sensitivity. Psychiatr Q. 2012;83:91-102.
James JM, Sixbey JP, Helm RM, Bannon GA, Burks AW. Wheat alpha-amylase inhibitor: a second route of allergic sensitization. The Journal of allergy and clinical immunology. Feb 1997;99(2):239-244.
Jeitner TM, Pinto JT, Krasnikov BF, Horswill M, Cooper AJL. Transglutaminases and neurodegeneration. Journal of neurochemistry. 2009;109(Suppl 1):160-166.
Jiang X, Dong J, Wang B, Yin X, Qin L. [Effects of organic selenium supplement on glutathione peroxidase activities: a meta-analysis of randomized controlled trials]. Wei sheng yan jiu = Journal of hygiene research. Jan 2012;41(1):120-123.
Jones W, Li X, Qu ZC, et al. Uptake recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med. 2002;33(1):83-93.
Junker Y, Zeissig S, Kim SJ, et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J Exp Med. 2012;209(13):2395-2408.
Juse U, van de Wal Y, Koning F, et al. Design of new high-affinity peptide ligands for human leukocyte antigen-DQ2 using a positional scanning peptide library. Hum Immunol. 2010;71:475-481.
Kagnoff M. Celiac disease: pathogenesis of a model immunogenetic disease. J Clin Invest. 2007;117(1):41-49.
Kapoerchan VV, Wiesner M, Overhand M, et al. Design of azidoproline containing gluten peptides to suppress CD4+ T-cell responses associated with celiac disease. Bioorg Med Chem. 2008;16:2053-2062.
Karaahmet OZ, Unlu E, Karaahmet F, Gurcay E, Cakci A. Myopathy related to vitamin D deficiency in patient with celiac disease. Muscle & nerve. Jul 2014;50(1):147-148.
Katta R, Schlichte M. Diet and dermatitis: food triggers. J Clin Aesthet Dermatol. 2014;Mar;7(3):30-6.
Kelly CP, Leffler DA. ePocrates online. Celiac disease. Available at: https://online.epocrates.com/noFrame/showPage?method=diseases&MonographId=636. Last updated 4/11/2014. Accessed 2/3/2015.
Kemppainen T, Kroger H, Janatuinen E. Osteoporosis in adult patients with celiac disease. Bone. 1999;24(3):249-255.
Kent KD, Harper WJ, Bomser JA. Effect of whey protein isolate on intracellular glutathione and oxidant-induced cell death in human prostate epithelial cells. Toxicol In Vitro. 2003;17:27-33.
Keshav S, Vanasek T, Niv Y, et al. A randomized controlled trial of the efficacy and safety of CCX282-B, an orally-administered blocker of chemokine receptor CCR9, for patients with Crohn’s disease. PLoS One 2013;8(3):e60094.
Kidd PM. Glutathione: systemic protectant against oxidative and free radical damage. Alternative medicine review: a journal of clinical therapeutic. 1997;2(3):155-176.
Kleopa KA, Kyriacou K, Zamba-Papanicolaou E, et al. Reversible inflammatory and vacuolar myopathy with vitamin E deficiency in celiac disease. Muscle Nerve. 2005;31:260-265.
Kolgazi M, Jahovic N, Yuksel M, et al. Alpha-lipoic acid modulates gut inflammation induced by trinitrobenzene sulfonic acid in rats. J Gastroenterol Hepatol. 2007;22:1859-1865.
Korponay-Szabo IR, Simon-Vecsei Z, De Leo L, Not T. Gluten-dependent intestinal autoimmune response. Current pharmaceutical design. 2012;18(35):5753-5758.
Kriegel MA, Manson JE, Costenbader KH. Does vitamin D affect risk of developing autoimmune disease?: a systematic review. Semin Arthritis Rheum. 2011;40:512-531.
Krzywicka B, Herman K, Kowalczyk-Zajac M, Pytrus T. Celiac disease and its impact on the oral health status - review of the literature. Advances in clinical and experimental medicine: official organ Wroclaw Medical University. Sep-Oct 2014;23(5):675-681.
Kuhn KS, Muscaritoli M, Wischmeyer P, Stehle P. Glutamine as indispensable nutrient in oncology: experimental and clinical evidence. European journal of nutrition. Jun 2010;49(4):197-210.
Kupfer SS, Jabri B. Pathophysiology of celiac disease. Gastrointestinal endoscopy clinics of North America. Oct 2012;22(4):639-660.
Kupper C. Dietary guidelines and implementation for celiac disease. Gastroenterology. Apr 2005;128(4 Suppl 1):S121-127.
Lähdeaho ML, Kaukenin K, Laurila K, et al. Glutenase ALV003 attenuates gluten-induced mucosal injury in patients with celiac disease. Gastroenterology. 2014;146:1649-1658.
Lawson A, West J, Aithal GP, Logan RF. Autoimmune cholestatic liver disease in people with coeliac disease: a population-based study of their association. Alimentary pharmacology & therapeutics. Feb 15 2005;21(4):401-405.
Lee S, Han KH, Nakamura Y, Kawakami S, Shimada K, Hayakawa T, . . . Fukushima M. Dietary L-cysteine improves the antioxidative potential and lipid metabolism in rats fed a normal diet. Bioscience, biotechnology, and biochemistry. 2013;77(7):1430-1434.
Lee SA, Kim HJ, Chang KC, et al. DHA and EPA down-regulate COX-2 expression through suppression of NF-kappaB activity in LPS-treated human umbilical vein endothelial cells. Korean J Physiol Pharmacol. 2009;Aug;13:301-307.
Lerner A, Blank M. Hypercoagulability in celiac disease--an update. Autoimmunity reviews. Nov 2014;13(11):1138-1141.
Lerner A, Gruener N, Iancu TC. Serum carnitine concentrations in coeliac disease. Gut. 1993;34:933-935.
Lerner A, Shapira Y, Agmon-Levin N, et al. The clinical significance of 25OH-vitamin D status in celiac disease. Clinic Rev Allerg Immunol. 2012;42:322-330.
Li N, Lewis P, Samuelson D, et al. Glutamine regulates Caco-2 tight junction proteins. Am J Physiol Gastrointest Liver Physiol. 2004;287:G726-G733.
Lidums I, Mechaber AJ. First Consult. Celiac disease. Available at: www.clinicalkey.com. Copyright 2015 by Elsevier Inc.
Lindfors K, Blomqvist T, Juuti-Uusitalo K, et al. Live probiotic Bifidobacterium lactis bacteria inhibit the toxic effects induced by wheat gliadin in epithelial cell culture. Clin Exp Immunol. 2008;152:552-558.
Lionetti E, Catassi C. New clues in celiac disease epidemiology, pathogenesis, clinical manifestations, and treatment. Int Rev Immunol. 2011;30:219-231.
Lohi S, Mustalahti K, Kaukinen K, et al. Increasing prevalence of celiac disease over time. Aliment Pharmacol Ther. 2007 Nov 1;26(9):1217-25.
Lowe AW, Moseley RH. Role of dietary FODMAPs in IBS-related symptoms. Gastroenterology. Jan 2014;146(1):1.
Lucendo AJ, Garcia-Manzanares A. Bone mineral density in adult coeliac disease: an updated review. Rev Esp Enferm Dig. 2013;Mar;105(3):154-62.
Ludvigsson JF, Bai JC, Biagi F, et al. Diagnosis and management of adult coeliac disease: guidelines from the British Society of Gastroenterology. Gut. June 10, 2014.
Lundin KEA, Alaedini A. Non-celiac gluten sensitivity. Gastrointest Endoscopy Clin N Am. 2012;22:723-734.
Mager DR, Qiao J, Turner J. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur J Clin Nutr. 2012;66:488-495.
Magge S, Lembo A. Low-FODMAP Diet for Treatment of Irritable Bowel Syndrome. Gastroenterology & hepatology. Nov 2012;8(11):739-745.
Mahmood A, FitzGerald AJ, Marchbank T, et al. Zinc carnosine, a health food supplement that stabilizes small bowel integrity and stimulates gut repair processes. Gut. 2007;56:168-175.
Malterre T. Digestive and nutritional considerations in celiac disease: could supplementation help? Alternative medicine review: a journal of clinical therapeutic. Sep 2009;14(3):247-257.
Mancini LA, Trojian T, Mancini AC. Celiac disease and the athlete. Curr Sports Med Rep. 2011;Mar-Apr;10(2):105-8.
Mansueto P, Seidita A, D'Alcamo A, Carroccio A. Non-celiac gluten sensitivity: literature review. Journal of the American College of Nutrition. 2014;33(1):39-54.
Marchioni Beery RM, Birk JW. Wheat-related disorders reviewed: making a grain of sense. Expert Rev Gastroenterol Hepatol. 2015;3:1-14. [Epub ahead of print]
Martin A, De Vivo G, Gentile V. Possible role of the transglutaminases in the pathogenesis of Alzheimer's disease and other neurodegenerative diseases. International journal of Alzheimer's disease. 2011;2011:865432.
Matsumura Y, Niitsuma T, Ito H. A study of factors contributing to bakers' allergy symptoms. Arerugi = [Allergy]. May 1994;43(5):625-633.
Mauro A, Orsi L, Mortara P, et al. Cerebellar syndrome in adult celiac disease with vitamin E deficiency. Acta Neurol Scand. 1991;84:167-170.
Mayo Clinic. Diseases and Conditions page. What allergy. Definition. Available at: http://www.mayoclinic.org/diseases-conditions/wheat-allergy/basics/definition/con-20031834. 5/17/2014. Accessed 3/6/2015.
Mazzon E, Muia C, Paola RD, et al. Green tea polyphenol extract attenuates colon injury induced by experimental colitis. Free Radic Res. 2005;Sep;39(9):1017-25.
Middleton N, Jelen P, Bell G. Whole blood and mononuclear cell glutathione response to dietary whey protein supplementation in sedentary and trained male human subjects. Int J Food Sci Nutr. 2004;55(2):131-141.
Miller AL. Therapeutic considerations of L-glutamine: a review of the literature. Alternative medicine review: a journal of clinical therapeutic. Aug 1999;4(4):239-248.
Molberg O, Kjersti Uhlen A, Jensen T, et al. Mapping of gluten T-cell epitopes in the bread wheat ancestors: implications for celiac disease. Gastroenterology. 2005;128:393-401.
Moleski SM. The Merck Manual online. Irritable Bowel Syndrome (IBS). Available at: http://www.merckmanuals.com/professional/gastrointestinal_disorders/irritable_bowel_syndrome_ibs/irritable_bowel_syndrome_ibs.html. Last updated 7/2013. Accessed 2/9/2015.
Molina-Infante J, Santolaria S, Montoro M, Esteve M, Fernandez-Banares F. [Non-celiac gluten sensitivity: a critical review of current evidence]. Gastroenterologia y hepatologia. Jun-Jul 2014;37(6):362-371.
Molteni N, Bardella MT, Vezzoli G, et al. Intestinal calcium absorption as shown by stable strontium test in celiac disease before and after gluten-free diet. Am J Gastroenterol. 1995; Nov;90(11):2025-8.
Monteleone G, Pender SL, Alstead E, Hauer AC, Lionetti P, McKenzie C, MacDonald TT. Role of interferon alpha in promoting T helper cell type 1 responses in the small intestine in coeliac disease. Gut. Mar 2001;48(3):425-429.
Mulder CJ, Cardile AP, Dickert J. Celiac disease presenting as severe osteopenia. Hawaii medical journal. Nov 2011;70(11):242-244.
Murray JA, McLachlan S, Adams PC, Eckfeldt JH, Garner CP, Vulpe CD, . . . McLaren CE. Association between celiac disease and iron deficiency in Caucasians, but not non-Caucasians. Clinical gastroenterology and hepatology: the official clinical practice journal of the American Gastroenterological Association. Jul 2013;11(7):808-814.
NIDDK. National Institute of Diabetes and Digestive and Kidney Disorders. Health Information page. Celiac Disease. Available at: http://www.niddk.nih.gov/health-information/health-topics/digestive-diseases/celiac-disease/Pages/facts.aspx. Last updated 1/27/2012. Accessed 3/2/2015.
Nikpour S. Neurological manifestations, diagnosis, and treatment of celiac disease: A comprehensive review. Iranian Journal of Neurology. 2012;11(2):59-64.
Nilsen EM, Jahnsen FL, Lundkin KE, et al. Gluten induces an intestinal cytokine response strongly dominated by interferon gamma in patients with celiac disease. Gastroenterology. 1998;Sep;115(3):551-63.
NLM. US National Library of Medicine. Compound Summary for CID 124886. Glutathione. Pharmacology and Biochemistry. Available at: http://pubchem.ncbi.nlm.nih.gov/compound/glutathione#section=Pharmacology-and-Biochemistry. Accessed 3/5/2015.
Nordström C, Dahlqvist A, Josefsson L. Quantitative determination of enzymes in different parts of the villi and crypts of rat small intestine. Comparison of alkaline phosphatase, disaccharidases and dipepeptidases. J Histochem Cytochem. 1967;15(12):713-21.
Odetti P, Valentini S, Aragno I, et al. Oxidative stress in subjects affected by celiac disease. Free Radic Res. 1998;Jul;29(1):17-24.
Olaussen RW, Karlsson MR, Lundin KEA, et al. Reduced chemokine receptor 9 on intraepithelial lymphocytes in celiac disease suggests persistent epithelial activation. Gastroenterology. 2007;132:2371-2382.
Packer L, Cadenas E. Lipoic acid: energy metabolism and redox regulation of transcription and cell signaling. J Clin Biochem Nutr. 2011;Jan;48:26-32.
Pantaleoni S, Luchino M, Adriani A, et al. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. Scientific World Journal. 2014;2014:173082.
Paterson BM, Lammers KM, Arrieta C, et al. The safety, tolerance, pharmacokinetic and pharmacodynamic effects of single doses of AT-1001 in coeliac disease subjects: a proof of concept study. Aliment Pharmacol Ther. 2007;26:757-766.
Pena AS, Garrote JA, Crusius JB. Advances in the immunogenetics of coeliac disease. Clues for understanding the pathogenesis and disease heterogeneity. Scandinavian journal of gastroenterology. Supplement. 1998;225:56-58.
Peters SL, Biesiekierski JR, Yelland GW, et al. Randomized clinical trial: gluten may cause depression in subjects with non-coeliac gluten sensitivity – an exploratory clinical study. 2014;May;39(10):1104-12.
Pischon T, Hankinson SE, Hotamisligil GS, et al. Habitual dietary intake of n-3 and n-6 fatty acids in relation to inflammatory markers among US men and women. Circulation. 2003;108:155-160.
Pozo-Rubio T, Olivares M, Nova E, et al. Immune development and intestinal microbiota in celiac disease. Clin Dev Immunol. 2012;2012:654143.
PR Newswire. Alba Therapeutics Announces Positive Results of Phase IIb Trial in Celiac Disease. [News Release]. Available at: http://www.prnewswire.com/news-releases/alba-therapeutics-announces-positive-results-of-phase-iib-trial-in-celiac-disease-244877451.html. 2/11/2014. Accessed 3/27/2015.
Presutti, RJ, Cangemi JR, Cassidy HD, et al. Celiac disease. Am Fam Physician. 2007;Dec15;76(12):1795-1802.
Prietl B, Treiber G, Pieber TR, et al. Vitamin D and immune function. Nutrients. 2013;5:2502-2521.
Pushpakumar S, Kundu S, Sen U. Endothelial dysfunction: the link between homocysteine and hydrogen sulfide. Current medicinal chemistry. 2014;21(32):3662-3672.
Rabelink NM, Westgeest HM, Bravenboer N, Jacobs MA, Lips P. Bone pain and extremely low bone mineral density due to severe vitamin D deficiency in celiac disease. Archives of osteoporosis. Dec 2011;6(1-2):209-213.
Rajani S, Huynh HQ, Turner J. The changing frequency of celiac disease diagnosed at the Stollery Children’s Hospital. Can J Gastroenterol. 2010;24(2):109-112.
Rashtak S, Murray A. Review article: celiac disease, new approaches to therapy. Aliment Pharmacol Ther. 2012;35:768-781.
Rastogi A, Bhadada SK, Bhansali A, et al. Celiac disease: a missed cause of metabolic bone disease. Indian J Endocrinol Metab. 2012;Sep-Oct;16(5):780-785.
Real A, Comino I, de Lorenzo L, et al. Molecular and Immunological Characterization of Gluten Proteins Isolated from Oat Cultivars That Differ in Toxicity for Celiac Disease. PLoS One. 2012;7(12):e48365.
Regan PT, DiMagno EP. Exocrine pancreatic insufficiency in celiac sprue: a cause of treatment failure. Gastroenterology. Mar 1980;78(3):484-487.
Reinken L, Zieglauer H. Vitamin B-6 absorption in children with acute celiac disease and in control subjects. J Nutr. 1978;108:1562-1565.
Richards JB, Valdes AM, Gardner JP, Paximadas D, Kimura M, Nessa A, . . . Aviv A. Higher serum vitamin D concentrations are associated with longer leukocyte telomere length in women. The American journal of clinical nutrition. Nov 2007;86(5):1420-1425.
Richie JPJ, Nichenametla S, Neidig W, Calcagnotto A, Haley JS, Schell TD, Muscat JE. Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. European journal of nutrition. 2014:1-13.
Rodrigo L, Blanco I, Bobes J, et al. Remarkable prevalence of coeliac disease in patients with Irritable bowel syndrome plus fibromyalgia in comparison with those with isolated Irritable bowel syndrome: a case-finding study. Arthritis Res Ther. 2013;15:R201.
Rodrigo L, Hernandez-Lahoz C, Fuentes D, et al. Prevalence of celiac disease in multiple sclerosis. BMS Neurology, 2011;11:31.
Roma E, Panayiotou J, Karantana H, et al. Changing pattern in the clinical presentation of pediatric celiac disease: a 30-year study. Digestion. 2009;80:185-191.
Round JL, Mazmanian SK. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009;May;9:313-323.
Rubio-Tapia A, Hill ID, Kelly CP, et al. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol. 2013;108:656-676.
Rubio-Tapia A, Kyle RA, Kaplan EL, et al. Increased prevalence and mortality in undiagnosed celiac disease. Gastroenterology. 2009;137:88-93.
Rude RK, Olerich M. Magnesium deficiency: possible role in osteoporosis associated with gluten-sensitive enteropathy. Osteoporos Int. 1996;6(6):453-61.
Rujner J, Socha J, Syczewska M, Wojtasik A, Kunachowicz H, Stolarczyk A. Magnesium status in children and adolescents with coeliac disease without malabsorption symptoms. Clinical nutrition (Edinburgh, Scotland). Oct 2004;23(5):1074-1079.
Rujner J, Socha J, Wojtasik A, et al. Magnesium status in children and adolescents with celiac disease. Wiad Lek. 2001;54(5-6):277-285.
Saibeni S, Lecchi A, Meucci G, et al. Prevalence of hyperhomocysteinemia in adult gluten-sensitive enteropathy at diagnosis: role of B12, folate, and genetics. Clin Gastroenterol Hepatol. 2005;3:574-580.
Salcedo G, Quirce S, Diaz-Perales A. Wheat allergens associated with Baker's asthma. J Investig Allergol Clin Immunol. 2011;21(2):81-92.
Sanders DS, Carter MJ, Hurlstone DP, et al. Association of adult coeliac disease with Irritable bowel syndrome: a case-control study in patients fulfilling ROME II criteria referred to secondary care. Lancet. 2001;358:1504-08.
Sanmukhani J, Satodia V, Trivedi J, et al. Efficacy and safety of curcumin in major depressive disorder: a randomized controlled trial. Phytother Res. 2014;Apr;28(4):579-85.
Sapone A, Bai JC, Ciacci C, et al. Spectrum of gluten-related disorders: consensus on new nomenclature and classification. BMC Med. 2012;Feb 7;10:13.
Sapone A, Lammers KM, Casolaro V, et al. Divergence of gut permeability and mucosal immune gene expression in two gluten-associated conditions: celiac disease and gluten sensitivity. BMC Med. 2011;9:23.
Sekhar RV, Patel SG, Guthikonda AP, Reid M, Balasubramanyam A, Taffet GE, Jahoor F. Deficient synthesis of glutathione underlies oxidative stress in aging and can be corrected by dietary cysteine and glycine supplementation. The American journal of clinical nutrition. Sep 2011;94(3):847-853.
Sevastiadou S, Malamitsi-Puchner A, Costalos C, et al. The impact of oral glutamine supplementation on the intestinal permeability and incidence of necrotizing enterocolitis/septicemia in premature neonates. J Matern Fetal Neonatal Med. 2011;24(10):1294-1300.
Shan L, Molberg O, Parrot I, et al. Structural basis for gluten intolerance in celiac sprue. Science. 2002;297:2275-2279.
SHC. Stanford Hospital & Clinics. The Low FODMAP Diet. Available at: https://stanfordhealthcare.org/content/dam/SHC/for-patients-component/programs-services/clinical-nutrition-services/docs/pdf-lowfodmapdiet.pdf. 1/2014. Accessed 2/27/2015.
Shepherd SJ, Gibson PR. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J Hum Nutr Diet. 2013;Aug;26(4):349-58.
Shewry PR, Halford NG. Cereal seed storage proteins: structures, properties and role in grain utilization. J Exp Bot. 2002;Apr;53(370):947-58.
Siegel M, Bethune MT, Gass J, et al. Rational design of combination enzyme therapy for celiac sprue. Chem Biol. 2006;13:649-658.
Siegel M, Garber ME, Spencer AG, Botwick W, Kumar P, Williams RN, . . . Adelman DC. Safety, tolerability, and activity of ALV003: results from two phase 1 single, escalating-dose clinical trials. Digestive diseases and sciences. Feb 2012;57(2):440-450.
Siegel M, Khosla C. Transglutaminase 2 inhibitors and their therapeutic role in disease states. Pharmacol Ther. 2007;115:232-245.
Silvester J, Duerksen D. Five things to know about...celiac disease. CMAJ : Canadian Medical Association journal = journal de l'Association medicale canadienne. Jan 8 2013;185(1):60.
Skovbjerg H. Immunoelectrophoretic studies on human small intestinal brush border proteins – the longitudinal distribution of peptidases and disaccharidases. Clin Chim Acta. 1981;May 5;112(2):205-12.
Skulas-Ray AC. Omega-3 fatty acids and inflammation: A perspective on the challenges of evaluating efficacy in clinical research. Prostaglandins & other lipid mediators. Feb 16 2015.
Smecuol E, Hwang HJ, Sugai E, et al. Exploratory, randomized, double-blind, placebo-controlled study on the effects of Bifidobacterium infantis natren life start strain super strain in active celiac disease. J Clin Gastroenterol. 2013;47:139-147.
Smith DF, Gerdes LU. Meta-analysis on anxiety and depression in adult celiac disease. Acta Psychiatr Scand. 2012;Mar;125(3):189-93.
Snyder CL, O Young D, Green PHR, et al. Celiac Disease. In: GeneReviews. Pagon RA, Adam MP, Amemiya A, et al. eds. Seattle WA: University of Washington, Seattle; 2015. <http://www.ncbi.nlm.nih.gov/books/NBK1727/>
Solakivi T, Kaukinen K, Kunnas T, et al. Serum fatty acid profile in celiac disease patients before and after a gluten-free diet. Scand J Gastrolenterol. 2009;44:826-830.
Sollid LM, Khosla C. Novel therapies for coeliac disease. Journal of Internal Medicine. 2011;269(6):604-613.
Spaenij-Dekking L, Kooy-Winkelaar Y, Van Veelen P, et al. Natural variation in toxicity of wheat: potential for selection of nontoxic varieties for celiac disease patients. Gastroenterology. 2005;129:797-806.
Sperandeo MP, Tosco A, Izzo V, Tucci F, Troncone R, Auricchio R, . . . Greco L. Potential celiac patients: a model of celiac disease pathogenesis. PloS one. 2011;6(7):e21281.
Spiegel BMR, DeRosa VP, Gralnek IM, et al. Testing for celiac sprue in Irritable bowel syndrome with predominant diarrhea: a cost-effective analysis. Gastroenterology. 2004;126:1721-1732.
Staudacher HM, Irving PM, Lomer MC, Whelan K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat Rev Gastroenterol Hepatol. Apr 2014;11(4):256-266.
Stazi AV, Trinti B. Selenium deficiency in celiac disease: risk of autoimmune thyroid diseases. Minerva Med. 2008;Dec;99(6):643-53.
Stazi AV, Trinti B. Selenium status and over-expression of interleukin-15 in celiac disease and autoimmune thyroid diseases. Ann Ist Super Sanita. 2010;46(4):389-399.
Stenman SM, Lindfors K, Venalainen JI, Hautala A, Mannisto PT, Garcia-Horsman JA, . . . Kaukinen K. Degradation of coeliac disease-inducing rye secalin by germinating cereal enzymes: diminishing toxic effects in intestinal epithelial cells. Clinical and experimental immunology. Aug 2010;161(2):242-249.
Stepniak D, Spaenij-Dekking L, Mitea C, et al. Highly efficient gluten degradation with a newly identified prolyl endoprotease: implications for celiac disease. Am J Physiol Gastrointest Liver Physiol. 2006;291:G621-G629.
Stern M. Gluten effects, analysis, and limits in celiac disease. J Pediatr Gastroenterol Nutr. 2000;Nov;31(5):468.
Stojiljkovic V, Pejic S, Kasapovic J, et al. Glutathione redox cycle in small intestinal mucosa and peripheral blood of pediatric celiac disease patients. An Acad Bras Cienc. 2012;84(1):175-184.
Stojiljkovic V, Todorovic A, Pejic S, et al. Antioxidant status and lipid peroxidation in small intestinal mucosa of children with celiac disease. Clin Biochem. 2009;Sep;42(13-14):1432-7.
Stojiljkovic V, Todorovic A, Radlovic N, et al. Antioxidant enzymes, glutathione and lipid peroxidation in peripheral blood of children affected by coeliac disease. Ann Clin Biochem. 2007;44:537-543.
Stoven S, Murray JA, Marietta EV. Latest in vitro and in vivo models of celiac disease. Expert opinion on drug discovery. Apr 2013;8(4):445-457.
Suh JH, Wang H, Liu RM, et al. (R)-alpha-lipoic acid reverses the age-related loss in GSH redox status in post-mitotic tissues: evidence for increased cysteine requirement for GSH synthesis. Arch Biochem Biophys. 2004;423:126-135.
SUMC. Stanford University Medical Center. The Low FODMAP Diet. Available at: https://stanfordhealthcare.org/content/dam/SHC/for-patients-component/programs-services/clinical-nutrition-services/docs/pdf-lowfodmapdiet.pdf. 1/2014. Accessed 4/29/2015.
Surette ME. The science behind dietary omega-3 fatty acids. CMAJ : Canadian Medical Association journal = journal de l'Association medicale canadienne. Jan 15 2008;178(2):177-180.
Szymczak J, Bohdanowicz-Pawlak A, Waszczuk E, et al. Low bone mineral density in adult coeliac disease. Endokrynol Pol. 2012;63(4):270-6.
Tarnok A, Marosvolgyi T, Szabo E, Gyorei E, Decsi T. Low n-3 Long-Chain Polyunsaturated Fatty Acids in Newly Diagnosed Celiac Disease in Children With Preexisting Type 1 Diabetes Mellitus. Journal of pediatric gastroenterology and nutrition. Feb 2015;60(2):255-258.
Tatham AS, Gilbert SM, Fido RJ, et al. Extraction, separation, and purification of wheat gluten proteins and related proteins of barley, rye, and oats. Methods Mol Med. 2000;41:55-73.
Telega G, Bennet TR, Werlin S. Emerging new clinical patterns in the presentation of celiac disease. Arch Pediatr Adolesc Med. 2008;162(2):164-168.
Thomas H, Beck K, Adamczyk M, Aeschlimann P, Langley M, Oita RC, . . . Aeschlimann D. Transglutaminase 6: a protein associated with central nervous system development and motor function. Amino acids. Jan 2013;44(1):161-177.
Thompson T. Folate, iron, and dietary fiber contents of the gluten-free diet. Journal of the American Dietetic Association. Nov 2000;100(11):1389-1396.
Tikkakoski S, Savilahti E, Kolho KL. Undiagnosed coeliac disease and nutritional deficiencies in adults screened in primary health care. Scand J Gastroenterol. 2007;Jan;’42(1):60-65.
Tilley KA, Benjamin RE, Bagorogoza KE, et al. Tyrosine cross-links: molecular basis of gluten structure and function. J Agric Food Chem. 2001;49:2627-2632.
Toouli J, Blankin AV, Oliver MR, et al. Management of pancreatic exocrine insufficiency: Australasian Pancreatic Club recommendations. Med J Aust. 2010;193(8):461-7.
Tripathi A, Lammers KM, Goldblum S, Shea-Donohue T, Netzel-Arnett S, Buzza MS, . . . Fasano A. Identification of human zonulin, a physiological modulator of tight junctions, as prehaptoglobin-2. Proceedings of the National Academy of Sciences of the United States of America. Sep 29 2009;106(39):16799-16804.
Turner J, Pellerin G, Mager D. Prevalence of metabolic bone disease in children with celiac disease is independent of symptoms at diagnosis. J Pediatr Gastroenterol Nutr. 2009;49:589-593.
UCLA Health. Celiac Disease Program. For Patients page. Celiac vs Gluten-Sensitivity vs Wheat Allergies. Available at: http://gastro.ucla.edu/site.cfm?id=281. Accessed 3/6/2015.
UCMC. University of Chicago Medical Center. The University of Chicago Celiac Disease Center. Celiac disease facts and figures. Available at: http://www.cureceliacdisease.org/wp-content/uploads/2014/06/CeliacDiseaseFactsAndFigures0614.pdf. Updated 2014. Accessed 10/14/2014.
UCMC. University of Chicago Medical Center. The University of Chicago Celiac Disease Center. Jump start your gluten-free diet. Available at: http://www.cureceliacdisease.org/living-with-celiac/resources/jump-start-your-gluten-free-diet-ebook. Updated 2013. Accessed 10/17/2014.
Vahedi K, Mascart F, Mary JY, et al. Reliability of antitransglutaminase antibodies as predictors of gluten-free diet compliance in adult celiac disease. Am J Gastroenterol. 2003;98(5):1079-1087.
van den Broeck HC, van Herpen TWJM, Schuit C, et al. Removing celiac disease-related gluten proteins from bread wheat while retaining technological properties: a study with chinese spring deletion lines. BMC Plant Biol. 2009;9:41.
van Elburg RM, Uil JJ, Mulder CJJ, et al. Intestinal permeability in patients with coeliac disease and relatives of patients with coeliac disease. Gut. 1993;34:354-357.
Vaynshtein G, Rosenbaum H, Groisman GM, Markel A. Celiac sprue presenting as severe hemorrhagic diathesis due to vitamin K deficiency. The Israel Medical Association journal : IMAJ. Dec 2004;6(12):781-783.
Vazquez-Roque MI, Camilleri M, Smyrk T, et al. A controlled trial of gluten-free diet in patients with Irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology. 2013;144:903-911.
Ventura A, Magazzu G, Greco L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Gastroenterology. Aug 1999;117(2):297-303
Veraverbeke WS, Delcour JA. Wheat protein composition and properties of wheat glutenin in relation to breadmaking functionality. Critical reviews in food science and nutrition. 2002;42(3):179-208.
Verdu EF, Armstrong D, Murray JA. Between celiac disease and irritable bowel syndrome: the "no man's land" of gluten sensitivity. The American journal of gastroenterology. Jun 2009;104(6):1587-1594.
Vilela RM, Lands LC, Chan HM, et al. High hydrostatic pressure enhances whey protein digestibility to generate whey peptides that improve glutathione status in CFTR-deficient lung epithelial cells. Mol Nutr Food Res. 2006;50:1013-1029.
Vincentini O, Quaranta MG, Viora M, et al. Docosahexaenoic acid modulates in vitro the inflammation of celiac disease in intestinal epithelial cells via the inhibition of cPLA2. Clin Nutr. 2011;30:541-546.
Volta U, Caio G, Tovoli F, et al. Non-celiac gluten sensitivity: questions still to be answered despite increasing awareness. Cell Mol Immunol. 2013;10:383-392.
Volta U, De Giorgio R. New understanding of gluten sensitivity. Nat Rev Gastroenterol Hepatol. 2012;9:295-299.
Volta U, Tovoli F, Cicola R, Parisi C, Fabbri A, Piscaglia M, . . . Caio G. Serological tests in gluten sensitivity (nonceliac gluten intolerance). Journal of clinical gastroenterology. Sep 2012;46(8):680-685.
Watson JL, Ansari S, Cameron H, et al. Green tea polyphenol (-)-epigallocatechin gallate blocks epithelial barrier dysfunction provoked by IFN-gamma but not byIL-4. Am J Physiol Gastrointest Liver Physiol. 2004;Nov;287(5):G954-61.
Watts RE, Siegel M, Khosla C. Structure – activity relationship analysis of the selective inhibition of transglutaminase 2 by dihydroisoxazoles. J Med Chem. 2006;49:7493-7501.
Weizman Z, Hamilton JR, Kopelman HR, Cleghorn G, Durie PR. Treatment failure in celiac disease due to coexistent exocrine pancreatic insufficiency. Pediatrics. Dec 1987;80(6):924-926.
Westphal C, Konkel A, Schunck WH. CYP-eicosanoids – a new link between omega-3 fatty acids and cardiac disease? Prostaglandins Other Lipid Mediat. 2011;96:99-108.
Wierdsma NJ, van Bokhorst-de van der Schueren MA, Berkenpas M, et al. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients. 2013;Sep 30;5(10):3975-92.
Woodward J. The management of refractory coeliac disease. Therapeutic advances in chronic disease. Mar 2013;4(2):77-90.
Wu D, Wang J, Pae M, et al. Green tea EGCG, T cells, and T cell-mediated autoimmune diseases. Mol Aspects Med. 2012;33:107-118.
Xia J, Bergseng E, Fleckenstein B, et al. Cyclic and dimeric gluten peptide analogues inhibiting DQ2-mediated antigen presentation in celiac disease. Bioorg Med Chem. 2007;15:6565-6573.
Yan Y, Jiang W, Spinetti T, Tardivel A, Castillo R, Bourquin C, . . . Zhou R. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity. Jun 27 2013;38(6):1154-1163.
Yokoyama S, Watanabe N, Sato N, et al. Antibody-mediated blockade of Il-15 reverses the autoimmune intestinal damage in transgenic mice that overexpress Il-15 in enterocytes. Proc Natl Acad Sci USA. 2009;Sep 15;106(37):15849-15854
Yuce A, Demir H, Temizel INS, et al. Serum carnitine and selenium levels in children with celiac disease. Indian J Gastroenterol. 2004;May-June;23:87-88.
Zapata-Gonzalez F, Rueda F, Petriz J, et al. Human dendritic cell activities are modulated by the omega-3 fatty acid, docosahexaenoic acid, mainly through PPARgamma: RXR heterodimers: comparison with other polyunsaturated fatty acids. J Leukoc Biol. 2008;84:1172-1182.
Zavorsky GS, Kubow S, Grey V, et al. An open-label dose-response study of lymphocyte glutathione levels in healthy men and women receiving pressurized whey protein isolate supplements. Int J Food Sci Nutr. 2007;58(6):429-436.
Zingone F, Capone P, Ciacci C. Celiac disease: Alternatives to a gluten free diet. World Journal of Gastrointestinal Pharmacology and Therapeutics. 2010;1(1):36-39.
Zittermann A. Effects of vitamin K on calcium and bone metabolism. Current opinion in clinical nutrition and metabolic care. Nov 2001;4(6):483-487.
Zofkova I, Kancheva RL. The relationship between magnesium and calciotropic hormones. Magnes Res. 1995;Mar;8(1):77-84.
Zwolińska-Wcisło M, Galicka-Latala D, Rozpondek P, et al. [Frequency of celiac disease and Irritable bowel syndrome coexistence and its influence on the disease course].Przegl Lek. 2009;66(3):126-9.
