Chronic Kidney Disease
Chronic Kidney Disease
Last Section Update: 12/2025
Contributor(s): Maureen Williams, ND; Shayna Sandhaus, PhD; Stephen Tapanes, PhD; Chancellor Faloon, Health & Wellness Author
Table of Contents
- What Is Chronic Kidney Disease?
- Kidney Function and the Development of Chronic Kidney Disease
- Most Common Chronic Kidney Disease Causes and Contributors
- Symptoms of Chronic Kidney Disease
- How Is Chronic Kidney Disease Diagnosed and Monitored?
- The Many Complications of Chronic Kidney Disease
- Management of Chronic Kidney Disease
- Novel & Emerging Treatment Approaches
- Dietary Changes for Chronic Kidney Disease
- Lifestyle Changes for Chronic Kidney Disease
- Nutrients to Support Kidney Function
- Frequently Asked Questions
- Update History
- References
1 What Is Chronic Kidney Disease?
Summary and Quick Facts for Chronic Kidney Disease
- About 15% of adults in the United States, or 37 million people, suffer from chronic kidney disease (CKD). However, 90% of these individuals do not know that they have this potentially deadly condition.
- Diabetes and high blood pressure are the leading causes of CKD, so people with high blood pressure or blood glucose levels need to be particularly vigilant about monitoring their kidney function.
- Timely lab testing can help detect declining kidney function before it becomes advanced.
- Treatment for CKD has progressed considerably in recent years. Highly effective blood pressure medications are usually the first-line therapy for CKD.
- A relatively new class of drugs—called sodium-glucose cotransporter-2 (SGLT2) inhibitors—have proven beneficial for both diabetic and non-diabetic CKD and appear poised to become a mainstay treatment. Both classes of drugs prevent kidney disease progression and lower the risk of common cardiovascular complications.
- A dietary pattern based on the Dietary Approaches to Stop Hypertension (DASH) and Mediterranean diets may help protect against CKD and its complications.
- Once people have developed advanced CKD, additional dietary changes that may be recommended include diets with reduced sodium, phosphorus, and sometimes protein.
- Some evidence suggests that supplements such as probiotics and prebiotics, vitamins D and K, the minerals selenium and zinc, as well as omega-3 fatty acids from fish may help support kidney health.
Chronic kidney disease, or CKD, is defined as abnormalities in kidney function or structure lasting three months or longer and impacting overall health.
Other categories of kidney disease include1:
- Acute kidney disease (AKD): Kidney damage or dysfunction with a duration of less than three months. AKD may progress to CKD or occur during the course of CKD.
- Acute kidney injury (AKI): A subset of AKD characterized by kidney damage or dysfunction that occurs for seven days or less.
This protocol focuses on CKD.
Over the last two decades, the number of people with CKD has increased in the United States and worldwide.2 About 15% of American adults, or 37 million people, are believed to have CKD. CKD generally does not cause symptoms until the kidneys are severely damaged. As a result, an estimated nine out of 10 adults with CKD are unaware that they have the condition.3 This means regular checkups and lab testing are essential to make sure those with major risk factors for CKD can catch any problems early.4
Diabetes and high blood pressure are the leading causes of CKD, accounting for 75% of new cases in the United States.5 Therefore, people with elevated blood pressure or blood glucose levels should have their kidney function evaluated at least annually. Kidney function can be easily assessed with simple blood and urine tests.6
CKD is a long-term condition that progresses slowly over time7; thus, dietary and lifestyle changes, particularly early in the course of CKD, can be very helpful in preserving kidney function and overall health.
Furthermore, CKD treatment has come a long way in the last few decades. For instance, angiotensin-converting enzyme (ACE) inhibitor and angiotensin receptor blocker (ARB) drugs are used as an effective first-line therapy. In addition, a newer class of drugs called sodium-glucose cotransporter-2 (SGLT2) inhibitors demonstrated cardioprotective properties in trials with individuals who have diabetes and CKD, as well as people with CKD who do not have diabetes. Both classes of drugs slow disease progression and help prevent complications.7,8 Moreover, eating a healthy diet, such as the Dietary Approaches to Stop Hypertension (DASH) or Mediterranean diet, and lowering the intake of sodium, phosphorus and sometimes protein may help protect kidney function. Preliminary evidence suggests supplementation with nutrients such as probiotics and prebiotics, omega-3 fatty acids from fish, vitamins D and K, and the minerals zinc and selenium may support kidney health as well.
2 Kidney Function and the Development of Chronic Kidney Disease
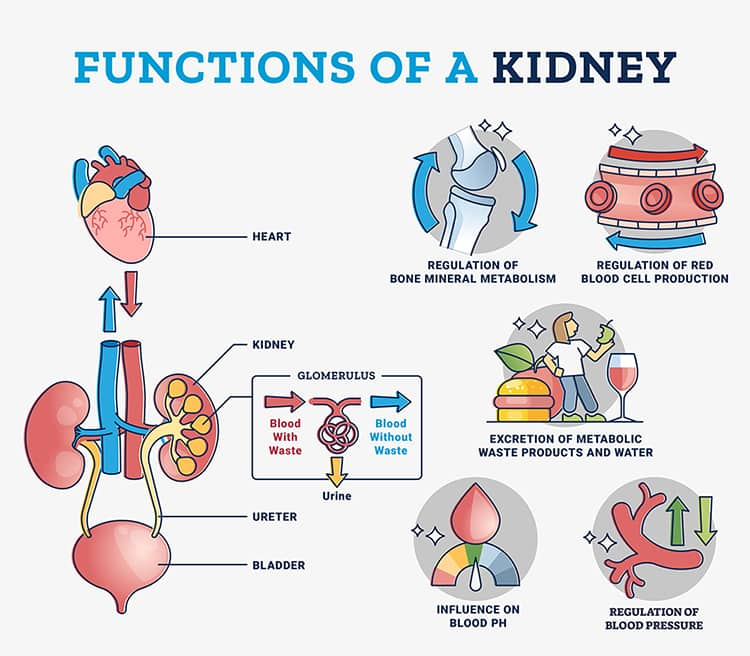
The main role of the kidneys is to filter water and waste out of the blood. Additional functions include9,10:
- Balancing blood pH
- Facilitating the conversion of vitamin D into its active form (ie, 1,25-dihydroxycholicalciferol)
- Producing hormones that help control blood pressure and red blood cell production
- Maintaining normal blood concentrations of electrolytes (eg, potassium, sodium, phosphorus, and calcium)
Kidney damage and the loss of function may gradually lead to CKD. Besides diabetes and high blood pressure, the following additional health conditions may also contribute to CKD1:
- Cardiovascular disease
- Systemic or kidney infections
- Immune system conditions (eg, lupus)
- Inflammation of the glomeruli (tiny filters) in the kidneys
- Inherited kidney disease (eg, polycystic kidney disease)
- Congenital defects (eg, kidney malformation, urinary tract obstruction)
- Medications and toxins (eg, prolonged medication, chemical, or street drug exposure)
Factors such as those described above and various others can contribute to the decline in kidney function characteristic of CKD. As these harmful factors damage the kidneys, the kidneys adapt in an effort to continue to meet the body’s needs for filtration. When some nephrons are injured or lost, the remaining healthy nephrons undergo a process called adaptive hyperfiltration. This process involves an increase in single-nephron glomerular filtration rate in the remaining nephrons to compensate for the loss of function. While initially beneficial, adaptive hyperfiltration can lead to further injury and long-term decline in kidney function.7,11
3 Most Common Chronic Kidney Disease Causes and Contributors
Diabetes
Diabetic kidney disease, an extremely common complication of type 1 or type 2 diabetes, is the leading cause of kidney failure in the world. Roughly 40% of individuals with type 2 diabetes and one-third with type 1 diabetes will develop CKD.12,128 More importantly, the high mortality risk associated with types 1 and 2 diabetes occurs primarily in those with CKD.13
Diabetic kidney disease begins with damage to the small blood vessels that supply the kidneys. Uncontrolled high blood sugar induces oxidative stress, which is accompanied by an increase in inflammatory cytokines (proteins) and advanced glycation end products (glycotoxins). Damage to the protective lining of the blood vessels ensues, as well as pathologic changes to kidney cells and kidney function. Diabetes is often accompanied by high blood pressure, which damages the kidneys.13-16
High Blood Pressure
High blood pressure is the second most common cause of kidney failure after diabetes.17 About 60–90% of CKD patients have high blood pressure, with uncontrolled high blood pressure being damaging to the kidneys and kidney disease also being a primary cause of hypertension. Multiple mechanisms contribute to this close relationship, including18,19:
- Endothelial dysfunction
- Volume overload and salt retention
- Overactivity of the sympathetic (“fight-or-flight”) nervous system
- Adverse changes in the hormonal regulation of blood pressure
- Dysfunction of the renin-angiotensin-aldosterone system
Ultimately, in people with CKD and diabetes, kidney tissue and blood vessels are much more sensitive to pressure damage than those with uncomplicated mild-to-moderate high blood pressure.17
Obesity
Obesity is a major contributing factor for CKD, explained in part by the observation that obesity predisposes individuals to diabetes and hypertension. However, obesity also appears to play an independent role in CKD risk and progression by triggering increased inflammatory signaling, oxidative stress, and gut microbiome disturbance, as well as premature cellular aging and dysfunction.20
In addition, aspects of body composition that are not efficiently accounted for by simple measures of weight or body mass index (BMI) also appear to be important modifiers of mortality risk in CKD. For instance, emerging research suggests that elevated body fat percentage in conjunction with low lean muscle mass—called sarcopenic obesity—is associated with poor kidney-related health outcomes. This phenomenon may help explain the “obesity paradox,” which suggests that, even though people who are obese are at greater risk of developing heart issues, once heart disease develops, individuals with higher BMIs appear to have a lower mortality risk than people with normal BMIs. However, obesity, as assessed by body composition, has successfully predicted mortality through repeated studies, highlighting the importance of maintaining adequate muscle mass in addition to avoiding excess body fat.22,23 Nonetheless, it is unknown whether losing weight will improve outcomes in people with obesity and CKD.
Non-Alcoholic Fatty Liver Disease (NAFLD)
Non-alcoholic fatty liver disease (NAFLD) and CKD are associated with multiple overlapping risk factors, including type 2 diabetes, high blood pressure, and obesity. These two conditions also involve many of the same mechanistic causes, such as oxidative stress, inflammation, and gut dysbiosis. Despite these similarities, it is unclear whether NAFLD causes or leads to CKD.24,25 Nonetheless, there is evidence that NAFLD is associated with a higher risk of CKD, even after ruling out contributing factors such as obesity and diabetes. It is thought that portal hypertension (high blood pressure in the liver’s venous system) due to NAFLD may impair the kidneys’ ability to regulate sodium levels and blood pressure, triggering dysfunctional compensatory mechanisms and setting the stage for CKD. In time, progression from NAFLD to fibrosis (scarring) and cirrhosis (scarring of the liver) can result in hepatorenal syndrome, a complication marked by severe kidney malfunction with reduced kidney blood flow and abdominal edema (ascites).26
High Homocysteine Levels
High homocysteine levels occur in over 80% of CKD patients, although it is typically described as a complication of poor kidney function rather than a cause. This is because impaired kidneys do not effectively metabolize or excrete homocysteine, resulting in the buildup of this amino acid in the bloodstream.27 However, there is evidence that elevated homocysteine is a risk factor for kidney damage and disease. In particular, one study examined the association between CKD and homocysteine levels in the context of genetic patterns and found that homocysteine was likely to play a causative role in CKD.28 In addition, homocysteine is known to cause endothelial (protective lining) damage, a recognized contributor to the progressive loss of kidney function.29 There is no evidence that homocysteine-lowering interventions are beneficial for people with or at risk of CKD.30
Additional Contributors
Other conditions that can lead to CKD include31-33:
- Kidney damage caused by the long-term use of certain medications (see section titled “What Medications Are Linked to Chronic Kidney Disease?” below)
- Polycystic kidney disease, a genetic condition in which multiple large cysts form in the kidneys
- Autoimmune disorders such as lupus and Goodpasture’s disease, in which the kidneys undergo inflammatory damage
- Recurring or persistent kidney or systemic infections
- Long-term heavy metal exposure (eg, lead poisoning)
- Gout and high uric acid (bi-directional relationship)
4 Symptoms of Chronic Kidney Disease
A major challenge with CKD is that it rarely causes symptoms before stages 4 and 5. When symptoms do occur, they may include7,46,47:
- Debilitating fatigue
- Itching and numbness
- Headaches
- Muscle cramps
- Weight loss
- Sleep disturbances
- Lack of appetite
- Nausea and vomiting
- Shortness of breath
- Seizures
- Edema (swelling) of the extremities
CKD is often discovered in people who do not have significant symptoms when they either attend their yearly physical and undergo lab testing or have blood work done for other reasons.
Estimated Glomerular Filtration Rate (eGFR) and Disease Stage48
- In stage 1, the eGFR is within normal limits (≥90 mL/min/1.73 m2) and the kidneys are still working adequately, but the presence of protein in the urine is indicative of mild kidney damage.49
- In stage 2, eGFR is 60–89. Even though there is mild, progressive damage to the kidneys, most people will not experience any symptoms.
-
Stage 3 is characterized by an eGFR range of 30–59.
This stage is generally considered the threshold for a CKD diagnosis,
and it falls into two subcategories: 3a and 3b. Mild-to-moderately
compromised kidney function falls within the eGFR range of 45–59 and is
referred to as stage 3a. An eGFR of 30–44 indicates the presence of
moderate-to-severe kidney disease, which is known as stage 3b.
An eGFR just below 60 typically indicates the presence of early-stage CKD and is also associated with a significantly increased risk of cardiovascular and any-cause mortality, kidney failure, acute kidney injury, or the progression of CKD. With healthy lifestyle changes and effective treatment, many people may be able to prevent advanced-stage CKD.
- In stage 4, kidney damage and loss of function are severe, as reflected by a low eGFR of 15–29. Symptoms such as fatigue, weakness, nausea, vomiting, and swelling of the hands and feet are increasingly probable at this stage, but some patients have no symptoms whatsoever.
- In stage 5, the eGFR is less than 15. This measurement reflects kidney failure. The symptoms associated with this stage reflect uremia, the syndrome in which failing kidneys are no longer able to manage the many biochemical and physiological processes for which they are responsible.50 Once symptoms develop or become inevitable, dialysis or a kidney transplant is necessary.
Albuminuria
Consideration of albuminuria together with eGFR helps predict the risk of mortality, as well as the risks of progressive kidney function loss and kidney failure.51
|
Prognosis of CKD by GFR & Albuminuria Categories |
Albuminuria Categories & Range | |||||
|---|---|---|---|---|---|---|
| A1 | A2 | A3 | ||||
| Normal to mildly increased | Moderately increased | Severely increased | ||||
|
<30 mg/g <3 mg/mmol |
30–200 mg/g 3–29 mg/mmol |
≥300 mg/g ≥30 mg/mmol |
||||
| GFR Categories (mL/min/1.73m2) Description & Range | G1 | Normal to high | ≥90 | |||
| G2 | Mildly decreased | 60-89 | ||||
| G3a | Mildly to moderately decreased | 45-59 | ||||
| G3b | Moderately to severely decreased | 30-44 | ||||
| G4 | Severely decreased | 15-29 | ||||
| G5 | Kidney failure | <15 | ||||
|
Green: Low risk (if no other markers of kidney disease,
no CKD)
Table recreated based on information from the National Kidney Foundation’s website52 |
||||||
5 How Is Chronic Kidney Disease Diagnosed and Monitored?
Blood and urine tests are used to assess and monitor kidney function and diagnose CKD.51,53
Urine Albumin
Albumin is an important blood protein. When the kidneys are functioning normally, there is very little or no albumin in the urine. Thus, albumin in urine (“albuminuria,” a type of proteinuria) may indicate abnormal kidney function or kidney damage from a wide range of causes. Clinically, albumin is often the earliest detected sign of kidney damage.7 Reducing albumin in the urine is an important consideration in CKD management that is usually accomplished with blood pressure-lowering therapies. A reduction in albuminuria often represents a better long-term prognosis and delayed disease progression.54
Estimated Glomerular Filtration Rate (eGFR)
The estimated glomerular filtration rate (eGFR) test measures how well the kidneys are filtering and excreting certain substances in the blood (eg, creatinine or cystatin C). This test is the predominant way physicians assess overall kidney function, as well as check for and monitor CKD. The eGFR value is expressed in units of mL/min/1.73 m2. The level of eGFR can help detect early CKD, and decreasing levels of eGFR are considered the most characteristic sign of progressive kidney disease.55,56
Usually, eGFR is estimated using the blood creatinine level. Creatinine is a waste product that is released during the normal breakdown of muscle tissue. Elevated creatinine levels are also observed with high-protein diets. For people with CKD, creatinine is not excreted efficiently, leading to low creatinine levels in the urine and high creatinine levels in the blood.51,53
Furthermore, eGFR can be estimated using levels of a blood protein called cystatin C, which is produced at a constant rate by all nucleus-containing cells. Since it is primarily broken down by the kidneys, the levels of cystatin C in the blood reflect kidney function. As there is less variation in the production and breakdown of cystatin C compared to creatinine across populations, and because it is less influenced by body composition, cystatin C is considered a more accurate marker of kidney function.57,58 Cystatin C-based eGFR (eGFRcys) is increasingly being adopted for detecting and staging CKD.57 The National Kidney Foundation and the American Society of Nephrology have recommended the use of both creatinine and cystatin C to most accurately estimate GFR.59
Another important advantage of eGFR estimates based on cystatin C is that they may better correlate with the risk of developing cardiovascular disease and premature mortality.56
Urine Albumin-to-Creatinine Ratio
The urine albumin-to-creatinine ratio (UACR) is another test used to assess kidney function. In CKD, urine albumin levels increase and, as a result, the ratio of albumin to creatinine increases. A normal UACR is <30 mg/g. In early-stage CKD, the UACR is between 30 and 300 mg/g; in later stages, it is >300 mg/g.60-62
Blood Urea Nitrogen (BUN)
Blood urea nitrogen (BUN) is an end product of protein metabolism that is measured in the blood. A BUN level of 7–20 mg/dL is considered normal. In AKD and CKD, BUN levels are typically elevated. A high-protein diet, certain drugs, and different medical conditions can also elevate BUN.51,53,63 BUN used to be commonly used in the assessment of kidney disease but in recent years has been de-emphasized in favor of eGFR, serum creatinine, and UACR.
6 The Many Complications of Chronic Kidney Disease
CKD is associated with a wide range of significant health complications, including64:
Cardiovascular Disease
Cardiovascular disease, particularly advanced cardiovascular disease, is associated with a significantly elevated risk of various cardiovascular problems, such as hypertension (high blood pressure), arterial calcification (calcium buildup in the arteries), heart failure, valvular disease (damaged heart valves), and atherosclerosis. Cardiovascular disease is the leading cause of death in patients with CKD.64,65 In particular, because CKD contributes to the progression of cardiovascular disease through many pathways, the authors of a 2021 review published in Circulationstated that “CKD mimics an accelerated aging of the cardiovascular system.”66
Blood Clot and Bleeding Risks
Chronic kidney disease increases the risk of bleeding events as well as blood clots (thrombosis) and related health problems.67,68 Accumulated toxins in the blood due to poor kidney filtration are thought to contribute to thrombosis risk by stimulating platelet (cell fragments responsible for blood clotting) and clotting factor activation. Furthermore, platelet activation leads to more inflammatory signaling, which contributes to the progression of kidney damage.68,69 Conversely, CKD in its later stages also increases the risk of bleeding due to platelet dysfunction.70 One large study found that CKD patients had a 35% higher risk of a bleeding event than individuals without CKD. Additional risk factors included older age, high blood pressure, smoking, and the use of aspirin.71
Anemia
Anemia is a common complication of CKD. The main cause of CKD-induced anemia is reduced output of erythropoietin—a protein that stimulates red blood cell production—from the kidneys. Another significant cause of CKD-related anemia is a shortened lifespan of red blood cells. Deficiencies of iron, vitamin B12, and folate can also cause anemia in CKD. Blood loss, especially from dialysis, infection, inflammation, and malnutrition, is another contributing factor.7,72
Electrolyte Imbalance and Metabolic Acidosis
The kidneys are responsible for controlling a broad range of essential mineral and fluid balances, including tightly regulating the retention and excretion of sodium and potassium while maintaining normal pH by balancing the acidity of urine, which impacts the blood and the whole body. When these functions break down in advanced CKD, multiple biochemical disturbances may develop.7,64
For instance, CKD patients may develop low or high sodium levels due to imbalances in sodium excretion and retention. As kidney disease progresses, imbalanced sodium levels become more likely; low sodium appears to be more common than high sodium in advancing CKD. Low and high sodium levels can cause different problems. For example, decreased sodium excretion (increasing levels) results in water retention and a condition known as volume overload, which causes swelling of tissues (edema). On the other hand, low sodium levels can cause fatigue, mood changes, and nausea; severely low sodium levels can cause confusion, seizures, and coma. Doctors managing patients with CKD will take into account sodium levels when determining appropriate treatment approaches.73-76 Hyperkalemia, a potentially dangerous condition in which potassium builds up in the bloodstream when potassium excretion is insufficient, is another possible consequence. A serious systemic disturbance called metabolic acidosis can occur in CKD when acidic compounds, especially nitrogen-containing waste from dietary protein metabolism, accumulate in the blood. Metabolic acidosis is a contributing factor in bone and muscle loss, potassium retention, and the further progression of CKD.7,77
Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD)
Chronic kidney disease-mineral and bone disorder (CKD-MBD) is a common complication in which one or more of the following occur due to the loss of proper regulation by the kidneys:
- Abnormal bone mineralization, strength, growth, and turnover
- Calcification of non-bone tissues, including blood vessels
- Irregularities in vitamin D, calcium, phosphorus, fibroblast growth factor-23, and parathyroid hormone metabolism
Each of these abnormalities is associated with a high risk of mortality, primarily attributable to the cardiovascular consequences. Most features of CKD-MBD are usually present by the time eGFR falls below 45 (stage 3b), though bone disease can begin as soon as stage 2 (eGFR 60 to 89). CKD-MBD occurs in nearly every patient with stage 5 CKD (eGFR <15). The onset of blood vessel calcification can also begin before CKD becomes advanced, though its prevalence increases with diminishing kidney function. An overwhelming majority of dialysis patients are found to have coronary artery calcification.78
Uremic Syndrome
Uremic syndrome refers to a cluster of non-specific symptoms often seen in CKD patients, including itching, low appetite, nausea, unexplained weight and muscle loss, fatigue, restless legs syndrome, sleep disorders, and sexual dysfunction. These symptoms are thought to be due in part to the accumulation of toxins of varying chemical properties and sizes in the blood that would ordinarily be cleared by the kidneys. The toxins can range from small molecules (eg, phenol or urea) to larger compounds (eg, beta2-microglobulin). Uremic symptoms can contribute to depression and poor quality of life in CKD patients.50,64
Cognitive Impairment
CKD is associated with an increased risk of cognitive impairment and dementia.79 Impaired kidney function can affect the brain and lead to neuropathy (nerve dysfunction) through a pathway that is described as the kidney–brain axis. Uremia, nerve inflammation, high blood pressure, depression, and disordered sleep may all play a role in this pathway. Oxidative stress and disruption of the blood–brain barrier are also suspected of diminishing cognitive function in CKD.46
Dysbiosis and Gut Barrier Dysfunction
Changes in gut microbiota and impaired gut barrier function have been observed in CKD. Gut dysbiosis (imbalance) and barrier dysfunction (“leaky gut”) may contribute to chronic inflammation, increased cardiovascular events, and other chronic disease risks.80-82
Immune System Dysfunction
CKD has been linked to impaired immune system function. Increased T-cell dysfunction and immune senescence (age-related immune impairment) have been reported in CKD patients, which can lead to an increased risk of infection and cancer as well as diminished vaccine responsiveness.83-85
Cancer
Research shows that advanced CKD—when patients need dialysis—is associated with an increased risk of multiple cancers by anywhere from 10–80%.86
Premature Aging and Mortality
Chronic kidney disease is a leading cause of death in the United States. In men and women aged 66 years and older, CKD is associated with twice the risk of dying compared to those without the disease.87 In particular, early death from CKD is more common than progression to kidney failure.88 One research group has published extensive research about the remarkable parallels between pathologic characteristics that are induced by or associated with progressive kidney disease and features that characterize the premature aging phenotype. These commonalities include accelerated vascular aging (blood vessel aging), sarcopenia (age-related muscle loss), cellular senescence (cell aging), and the impairment of mitochondrial biogenesis (an adaptive energy-producing process). Additional similarities are DNA and mitochondrial damage, persistent low-grade inflammation, stem cell exhaustion, telomere shortening (organismal aging), and an increase in the generation of reactive oxygen species.89-92
7 Management of Chronic Kidney Disease
Management of CKD must address several interrelated concerns to best slow its progression and attempt to preserve kidney function as well as patient quality of life. The following considerations guide this approach:
- Treat conditions that cause kidney damage, such as diabetes, high blood pressure, and autoimmune disease
- Discontinue nephrotoxic (toxic to kidneys) drugs, if possible
- Halt or slow disease progression
- Treat complications of CKD
- Adjust drug dosing in accordance with eGFR level
- Prepare for kidney replacement therapy in patients who are susceptible to kidney failure
CKD management usually involves targeting high blood pressure and diabetes. Dietary changes and smoking cessation are important lifestyle aspects of the approach.7,93
Medications
Clinical guidelines for CKD generally focus first on anti-hypertensive therapy, as high blood pressure is both a cause and consequence of kidney damage and disease. This means that uncontrolled blood pressure can lead to a vicious cycle for the kidneys. The management of high blood pressure in CKD is complex, with different considerations depending on the level of eGFR, presence and degree of albuminuria, various drug-specific effects and side effects, and presence of comorbidities (eg, diabetes or heart failure). Treatment of diabetes and elevated blood sugar (hyperglycemia) is also a high priority in CKD.93
All standard treatment medications slow the progression of kidney disease, prevent cardiovascular disease complications, and reduce the risk of early death. However, the medical management of CKD requires considerable expertise.
Sodium-glucose cotransporter-2 inhibitors. In recent years, several clinical trials have revealed the remarkable benefits of the relatively new class of diabetes drugs—called sodium-glucose cotransporter-2 (SGLT2) inhibitors—for chronic kidney disease. These medications treat both diabetes and diabetic CKD, as well as non-diabetic CKD. The use of SGLT2 inhibitors has also been associated with weight loss, suggesting they could help address obesity, a difficult and important cause of CKD. Furthermore, SGLT2 inhibitors is an important treatment for heart failure, which is both a cause and consequence of CKD.94-96
SGLT2 inhibitor drugs, such as dapagliflozin (Farxiga), canagliflozin (Invokana), and empagliflozin (Jardiance), are used to treat type 2 diabetes. Their mechanism of action involves the reduction of sodium and glucose reabsorption in the kidneys, resulting in increased glucose and sodium excretion in the urine.97,98
In large clinical trials, SGLT2 inhibitors have been shown to improve kidney-related outcomes in people with and without diabetes. These benefits appear to be independent of the drugs’ glucose-lowering effects.7,99-101 Among the proposed mechanisms for these benefits are a reduction in kidney workload and improvement in oxygen dynamics, along with the lowering of blood pressure in the kidneys. These changes lead to reduced hyperfiltration, improved vascular health, anti-inflammatory and antifibrotic effects within the kidneys, and diuretic effects.97,102,103
A large randomized controlled trial including 4,304 participants with stage 2–4 eGFR, with or without type 2 diabetes, and with A2-A3 albumin-to-creatinine ratio examined the effect of dapagliflozin (10 mg once daily) or placebo on kidney function over a median period of 2.4 years.104 In comparison to the placebo, participants treated with dapagliflozin had a 39% lower risk of experiencing substantial disease progression (defined as 50% or greater sustained decline in eGFR, progression to kidney failure, or death from kidney or cardiovascular causes). More importantly, these benefits were similarly robust in people with and without type 2 diabetes, and interestingly, more pronounced in non-diabetics. Participants with type 2 diabetes experienced a risk reduction of 36% versus 50% in those without diabetes. Furthermore, participants treated with dapagliflozin had a 31% lower risk of death from any cause compared with placebo, and the rate of serious adverse side effects was similar in the treated and placebo groups. This trial was discontinued early due to clear evidence of the treatment benefits. Based on these findings, the Food and Drug Administration (FDA) approved the use of dapagliflozin in CKD patients with a high risk of disease progression.105
A 2023 study reported the effects of empagliflozin in CKD patients with regard to disease progression and death from cardiovascular causes. In this randomized controlled trial, 6,609 men and women received either 10 mg empagliflozin daily or placebo. During a median of 2-year follow-up visits, patients in the empagliflozin group had a 28% lower risk of progression to worsening kidney disease or cardiovascular death compared with the placebo group. In addition, patients in the empagliflozin group were less likely to be hospitalized for any cause.106 Another randomized controlled trial compared the effect of 100 mg canagliflozin with placebo in 4,401 patients with type 2 diabetes and CKD (stage 2–3 eGFR and stage A2 to A3 albumin-to-creatinine ratio). After a median follow-up of 2.62 years, the relative risk of progressing to kidney failure (dialysis, transplantation, or a sustained eGFR of <15), a two-fold increase of serum creatinine levels, or death from kidney-related or cardiovascular causes was 30% lower in the canagliflozin-treated group.107, 108 Based on these results, the FDA approved the use of canagliflozin for treating diabetic kidney disease.109
Although SGLT2 inhibitors have demonstrated impressive efficacy and a robust safety profile in clinical trials as well as in practice, they are not without adverse effects—some of which vary by medication. Among these effects are potentially serious dehydration, low blood volume, urinary tract infections, low blood pressure, and genital fungal infections.110,111 A 2023 study in individuals 65 years and older with type 2 diabetes also assessed the safety of SGLT2 inhibitor treatment through a meta-analysis of randomized controlled trials that reported safety outcomes for SGLT2 inhibitors compared with placebo. A total of 19,986 participants were included in the assessment. The findings showed that the risk of acute kidney injury was significantly lower in the SGLT2 inhibitor group compared with placebo. However, the SGLT2 inhibitor discontinuation rate was 20%, and SGLT2 inhibitor treatment was associated with a six-fold increased risk of genital tract infections. An increased incidence of amputations was also observed, but only among individuals who took canagliflozin. Furthermore, the risk of urinary tract infection, fractures, volume depletion, hypoglycemia (low blood glucose levels), and diabetic ketoacidosis was similar between SGLT2 inhibitors and placebo.112 Similar research shows SGLT2 inhibitor treatment is also associated with an increased risk of diabetic ketoacidosis, a relatively uncommon but potentially life-threatening condition.113,114
For people with diabetes who take other blood glucose-lowering drugs, adding an SGLT2 inhibitor to the treatment regimen may increase the risk of hypoglycemia, but SGLT2 inhibitors generally do not cause hypoglycemia on their own.115 Although additional side effects have been observed and reported, rigorous studies and analyses have failed to confirm that they are elevated in SGLT2 inhibitor users.110,111
Renin-angiotensin-aldosterone system (RAAS) inhibitors.This class of drugs, which includes ACE inhibitors and ARBs, is central to the treatment of high blood pressure and CKD.116 RAAS inhibitors can reduce albumin excretion in the urine, which is associated with disease progression. This benefit occurs, regardless of the cause of kidney disease, in people with diabetes and even in patients without high blood pressure.54
Numerous randomized controlled trials have demonstrated the ability of these medications to slow CKD progression.117 Although the benefit is clear for individuals with stage 2 and 3 eGFR, the efficacy of this drug class in more advanced kidney disease is less certain. One reason for this uncertainty is that a drop in blood pressure in response to treatment results in an initial decrease in eGFR. This lowers the workload of the kidneys, but clinically resembles progressive loss of kidney function. As long as kidney function remains relatively stable following this initial decline, continuing the RAAS inhibitor is not only safe but beneficial. Another difficulty is that few clinical trials recruit participants with advanced kidney disease,118,119 although some evidence suggests RAAS inhibition remains beneficial even in people with kidney failure or after initiation of dialysis.
Results from multiple observational studies have confirmed the benefits demonstrated in controlled clinical trials, including an association between RAAS inhibition and the preservation of kidney function, as well as lowered risk of kidney failure, need for a kidney transplant, or death.120
Mineralocorticoid receptor antagonists. Mineralocorticoid receptor antagonists (MRAs) are medications used most often to treat patients with congestive heart failure and high blood pressure.121 These drugs work by blocking the body’s aldosterone (mineralocorticoid) receptor, which helps prevent sodium and water retention by the kidneys (so more sodium is excreted and less is retained).349
There are two types of MRAs: older medications such as spironolactone (Aldactone) and eplerenone (Inspra) are steroidal compounds, while newer MRAs, most notably finerenone (Kerendia), are non-steroidal. In a large multi-center heart failure cohort, non-steroidal MRAs were found to be associated with a significantly lower risk of worsening heart failure and all‐cause hospitalization and mortality compared to steroidal MRAs.350 Non-steroidal MRAs are generally used as add-on therapy to maximally tolerated treatment with ACE inhibitors or ARB medications in select patients with CKD and type 2 diabetes and albuminuria.351
A growing body of research indicates finerenone is safe and effective for treating diabetes-related CKD, even in the absence of heart failure. The FIDELITY study pooled findings from two large randomized controlled trials involving a total of 13,026 participants with type 2 diabetes and CKD who were being treated with a maximal dose of an ACE inhibitor or ARB. The study showed adding finerenone to treatment for a median of three years improved cardiovascular and renal outcomes compared with placebo.352 In fact, the addition of finerenone was linked to a 14% reduction in the risk of cardiovascular events and a 23% reduction in the risk of kidney disease events relative to placebo.353 Secondary analyses of FIDELITY study data have found finerenone use was also associated with lower risk of arrhythmia diagnosis or events and reduced all-cause and cardiovascular disease-related deaths compared with placebo.354,355 In addition to these benefits, a meta-analysis of four randomized controlled trials with a total of 13,943 participants with CKD and type 2 diabetes found the inclusion of finerenone in treatment reduced the risk of new-onset hypertension by 29%356 and was as safe as placebo.357
A number of studies later analyzed subgroup data from the FIDELITY study finding finerenone was effective in men and women of all ages358 and was protective against kidney and cardiovascular disease in those with other additional chronic conditions, including:
- abnormal liver tests359
- anemia360
- left ventricular hypertrophy (which is associated with heart failure)361
- a history of atherosclerotic cardiovascular disease362
- frailty363
Finerenone was effective across all baseline CKD severities364,365 and levels of insulin sensitivity.366 In addition, finerenone appeared to be effective in patients from various ethnicities and racial backgrounds.367-370
Secondary analyses of FIDELITY’s findings also showed that finerenone’s benefits were the same regardless of whether patients were also using diuretics or glucagon-like peptide 1 (GLP-1) receptor agonists (a class of anti-diabetes and anti-obesity medications).371,372 A comparison of clinical evidence found SGLT2 inhibitors and finerenone were likely to have similar cardiovascular and mortality benefits, but SGLT2 inhibitors may reduce CKD events and heart failure more strongly than finerenone and GLP-1 receptor agonists.373 More recently, a randomized controlled trial in 800 patients with CKD and type 2 diabetes found a combination of finerenone plus the SGLT2 inhibitor empagliflozin was more effective at reducing urinary albumin-to-creatinine ratio than either medication plus placebo.374
The most important side effect of finerenone is hyperkalemia (high potassium levels), and the risk increases as kidney function declines.375 A real-world observational study that analyzed results from 504 patients with CKD and type 2 diabetes reported hyperkalemia occurred in 5% of participants during seven months of finerenone use and did not lead to any cases of dialysis, hospitalization, or death.376
Clinical practice guidelines issued in 2024 by the American Diabetes Association and Kidney Disease: Improving Global Outcomes (KDIGO) recommend finerenone in eligible patients with diabetic kidney disease who are already receiving treatment with antihypertensives and glucose-lowering medication.351
Diuretics. Diuretics play a role in CKD management to help control volume overload, which refers to excess salt and water retained in tissues and the bloodstream, leading to swelling (edema) and often elevated blood pressure. By increasing urine production and excretion of sodium, diuretics can lower blood pressure, reduce edema, and increase the effectiveness of other blood pressure drugs. Some diuretics can also be used to treat hyperkalemia.7,129 Volume overload that necessitates diuretic therapy is more common in advanced kidney disease. Three of the most common types of diuretics are:
- Loop diuretics (eg, furosemide [Lasix])
- Thiazides including hydrochlorothiazide and chlorthalidone (Thalitone)
- Potassium-sparing diuretics such as spironolactone and eplerenone
A recommendation for dietary salt restriction usually accompanies a diuretic prescription.7,129
Generally, loop diuretics such as furosemide and torsemide (Demadex) are first-line therapies for fluid overload in stage 4–5 CKD. Unfortunately, loop diuretics can also stimulate the sympathetic nervous system and RAAS activity, countering some of their benefits on progressive kidney function loss. Although these types of diuretics reduce symptoms related to fluid overload, such as leg swelling and shortness of breath, loop diuretics do not improve outcomes in the advanced stages of CKD.130
Thiazide diuretics, such as hydrochlorothiazide (HCTZ), Aquazide, Microzide, and others, as well as chlorthalidone, may be used to help manage high blood pressure in CKD patients.131 A randomized placebo-controlled trial published in late 2021 showed that patients with stage 4 CKD and poorly controlled hypertension who were treated with chlorthalidone for 12 weeks experienced lowered blood pressure and urinary albumin-to-creatinine ratio compared with placebo. However, there was a higher incidence of adverse effects, including low potassium and high blood sugar.132
Electrolyte disturbances are also common in patients using diuretics. Sodium, potassium, calcium, and magnesium levels all decrease with diuretic use (especially loop diuretics), leading to an increased risk of serious heart arrhythmias. Therefore, blood electrolyte levels should be carefully monitored in CKD patients being treated with diuretics. In addition, diuretics can cause fluid depletion that can cause potentially dangerously low blood pressure.131
Furthermore, patients with CKD who take diuretics are prone to several additional complications. These include decreased kidney perfusion, kidney congestion, reduced diuretic secretion from renal tubules, abnormal ion transporter transport, neuroendocrine (nerve and hormone) system abnormalities, electrolyte disorders, drug interactions, and hypoproteinemia (low protein in the blood). These factors can contribute to diuretic resistance (ie, when the patient takes the recommended dosage of diuretics but the regimen is not effective) as well as increased readmission rates and mortality. The main causes of diuretic resistance are excess sodium intake and poor drug compliance. Additional contributing factors include changes in pharmacodynamics and pharmacokinetics, decreased kidney blood flow, changes in renal adaptation, and functional nephron reduction. Common treatment approaches for diuretic resistance involve increasing diuretic dose, changing the dosing frequency, switching to a new diuretic, sequential nephron blockage, and ultrafiltration treatment (which is similar to dialysis but removes only salt and water rather than toxins).133
Treatment of Kidney Failure
Note: Kidney failure used to be often described as “end-stage kidney disease.” However, this terminology is no longer used because it is potentially misleading since people with kidney failure often live for a long time with ideal management. “End-stage” implied imminent demise, but that is not always true.
When kidney disease has progressed to eGFR below 30 mL/min/1.73 m2, patients and their providers should initiate a discussion about kidney replacement therapy since, at this stage, progression to kidney failure is more likely. Hemodialysis, peritoneal dialysis, or kidney transplantation are the available treatments in these cases. Not all patients choose or have access to kidney replacement therapy for medical or personal reasons, instead entering what is called kidney palliative care or conservative management.7,134
Dialysis. Dialysis is a method of filtering excess fluid and waste from the blood when the kidneys are no longer able to perform this function.
In hemodialysis, blood is removed from circulation through the placement of a needle in the forearm that passes through a dialyzer (a kind of filter) and is returned to the body through another needle in the same arm. A hemodialysis session typically takes about four hours and is performed three times per week at a dialysis center. Some dialysis centers offer a nighttime option, which allows patients to undergo longer dialysis therapy while they sleep. This option has the advantages of requiring fewer restrictions on diet and fluid intake, better quality of life, and longer treatment sessions that may reduce symptoms. Alternatively, some patients are able to have dialysis at home, in which sessions can be longer or shorter and more frequent, along with a more flexible schedule and more consistent removal of toxins, salt, and water.135
Peritoneal dialysis is a type of kidney replacement therapy that uses the lining of the abdomen (peritoneum) as a filter for the blood. The first step in peritoneal dialysis is the placement of a tube (catheter) in the abdominal cavity a few weeks before treatment commences. The treatment involves the use of dialysis solution and a combination of water, salts, and additives that flow from a bag, through the catheter, and into the abdomen where it absorbs excess fluid and waste products while being retained in the abdomen for several hours. After this step is complete, the solution is drained into another bag. This process is generally repeated four to six times per day. In some cases, an automated system that performs three to five cycles during the night can be used.136
Kidney transplant. Some people with kidney failure are candidates for a kidney transplant, a surgical procedure in which a kidney from another person (a healthy donor or a person who has died) is placed into the patient. A donated kidney is more effective at replacing kidney function than dialysis and is the preferred treatment whenever possible. However, there are several obstacles to this treatment option. First, a kidney recipient must be physically, mentally, and emotionally healthy enough to have a high likelihood of a good outcome. Second, an appropriate donor, matched for the lowest risk of transplant rejection, must be identified. If a living donor is not found among family or friends, the patient is placed on a waiting list to receive a kidney, usually from a deceased donor. The waiting time for a donated kidney can be months to years.137
A kidney transplant also has potential complications, including bleeding, infections, herniation, and pain or numbness. Transplant rejection by the immune system that results in the loss of function in the donated kidney is also possible. Long-term immunosuppressant therapy is used to lower the risk of transplant rejection.137
8 Novel & Emerging Treatment Approaches
Glucagon-like Peptide-1 Receptor Agonists (GLP-1RAs)
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are a class of drugs used to treat type 2 diabetes and obesity. There are multiple FDA-approved GLP-1RAs, including the well-known semaglutide (Ozempic, Wegovy). Tirzepatide (Mounjaro, Zepbound), a dual GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonist, is also well known. Due to their wide-ranging metabolic benefits, GLP-1RAs have also been explored for their effect on kidney health and function.317,318 This class of drugs may protect kidney function via improving traditional CKD risk factors, such as lowering glucose levels, improving insulin signaling, and weight reduction. However, GLP-1RAs appear to exert protective effects on the kidneys beyond glucose metabolism; they have been shown to decrease inflammation and oxidative stress, lower elevated albumin, and improve histological damage scores in animal models.319,320
In a randomized controlled trial that included 3,533 participants with diabetes and CKD published in May 2024, 1 mg subcutaneous semaglutide administered weekly reduced the incidence of major kidney disease events by 24% compared with placebo.321 The primary endpoint of this trial focused on kidney outcomes. The trial was planned to run for 4–5 years but was halted at around 3.5 years because the evidence of benefit at an interim analysis was very clear. As of June 2024, this trial represents some of the most robust evidence available showing benefit for use of a GLP-1RA drug for improving kidney outcomes in people with diabetes and CKD. The trial findings were in line with those of observational studies.317
Other GLP-1RA drugs have shown kidney benefits as well, but the evidence is generally not as robust as that for semaglutide as revealed in the trial described above. Several randomized controlled trials have shown that liraglutide (Victoza, Saxenda) improves hemoglobin A1C and glucose levels in people with diabetes undergoing dialysis.317 Dulaglutide (Trulicity) has been shown to improve kidney outcomes in people with diabetes and kidney disease as well.322-324 Tirzepatide has also shown promise for improving kidney outcomes in people with diabetes and high cardiovascular risk. In a post-hoc prespecified exploratory analysis of the SURPASS-4 randomized controlled trial, tirzepatide was shown to slow eGFR decline and prevent an increase in the urinary albumin-creatinine ratio in clinically meaningful ways compared with insulin glargine (ie, an FDA-approved synthetic version of human insulin). Tirzepatide also reduced the occurrence of a composite of unfavorable kidney-related outcomes.325
The majority of clinical trials have only included people with diabetes; however, a meta-analysis of 13 randomized controlled trials including over 83,000 people found that GLP-1RAs reduced the risk of mortality by 13% and composite kidney outcomes by 24% in people both with and without diabetes.326 As of June 2024, GLP-1RAs have not yet been FDA-approved for CKD without diabetes.
Colchicine
Colchicine (Mitigare, Colcrys) is an anti-inflammatory medication mainly used to treat gout, a type of inflammatory arthritis often associated with high blood uric acid levels.138 Gout and CKD frequently co-occur. Indeed, gout may be a risk factor for CKD. A correlational study that compared data from over 41,000 individuals with gout matched to data from an equal number of gout-free controls found that those with gout were 78% more likely to develop stage 3 or higher CKD during a median of 6-year follow-up visits.139
In addition, colchicine has demonstrated multiple anti-fibrotic and anti-inflammatory effects in preclinical studies. As kidney fibrosis (tissue scarring) is an important feature of advanced CKD, there remains great interest in colchicine as a potential therapy for CKD. Colchicine has also been found to reduce the severity of kidney damage in preclinical models of diabetic nephropathy, a leading cause of CKD, by reducing inflammation. Preliminary clinical data appears promising as well.140 Moreover, a multicenter, nested, case-control study in Korea investigated the effect of colchicine on CKD progression in patients treated for hyperuricemia or chronic gout. The study included CKD patients aged 19 years and older, comparing those who had CKD progression with matched controls. Results indicated patients with long-term colchicine use (≥90 cumulative daily doses) had a lower risk of CKD progression, and the reduced risk of progression was more pronounced in patients without diabetes or hypertension and those with CKD stage G3.141 However, prospective randomized controlled trials are needed to clarify whether colchicine is beneficial for CKD patients. Individuals with CKD receiving colchicine to manage gout have reportedly experienced increased drug accumulation due to reduced renal clearance, which increases the risk of drug interactions and other serious adverse effects related to colchicine toxicity.140,142
Pentoxifylline
Pentoxifylline is a xanthine-derived phosphodiesterase inhibitor drug that decreases blood viscosity, and has anti-inflammatory, antifibrotic, and antioxidant properties. Its main indication is for peripheral arterial disease, though accumulating evidence suggests pentoxifylline may benefit individuals with diabetes and CKD.327,328
A meta-analysis of 11 randomized controlled trials including 705 patients with CKD found that pentoxifylline (when used in combination with an ACE-inhibitor and ARB) resulted in a significant reduction in proteinuria within six months. The treatment also slowed the expected decline in eGFR in patients with stage 3–5 CKD after six months, and significantly reduced albuminuria at 9–12 months. Dosages in the included trials ranged from 400 to 1,200 mg/day.329
A cohort study analyzed 10 years of records for over 7,000 people with advanced CKD who took pentoxifylline, and an equal number of matched participants who did not receive pentoxifylline. Pentoxifylline treatment was associated with a reduced risk of end-stage kidney disease, to a similar extent as treatment with combination renin-angiotensin-aldosterone system blockade, compared to RAAS inhibitor monotherapy. The authors speculated that pentoxifylline may be effective for preventing progression to end-stage kidney disease in those with pre-dialysis stage 5 CKD.330
Follow-up (up to seven years) of participants who had participated in a randomized controlled trial of 400 mg pentoxifylline twice daily versus standard of care for CKD found that adding pentoxifylline to standard of care conferred significant protection against kidney function decline and cardiovascular mortality.331,332
Pentoxifylline is generally well-tolerated overall, with the most common adverse effects being gastrointestinal distress or dizziness. Gastrointestinal side effects are generally self-limited and respond to dosage adjustments.333 However, research from Taiwan has indicated that pentoxifylline use in CKD may increase the risk of major bleeding events, especially in females, individuals with hyperlipidemia or a history of ischemic stroke, those taking higher doses of pentoxifylline, and in aspirin users.334
As of June 2024, the United States Veterans Affairs Administration is recruiting for a multicenter, randomized, controlled trial of 400 mg/day of extended-release pentoxifylline, ascending to a maximum of 800 mg/day after five weeks, in individuals with diabetic kidney disease.335,336 This study will primarily test whether pentoxifylline, added to usual care and compared with placebo, delays progression to end stage kidney disease or death, and will also evaluate multiple secondary endpoints.
9 Dietary Changes for Chronic Kidney Disease
Overall Dietary Patterns
A growing body of evidence shows the potential importance of diets that emphasize plant foods and de-emphasize red meats, salt, and sweets for individuals who have or are at risk of CKD.143-145 Two diets that represent this dietary pattern and have a high degree of research substantiation are the Dietary Approaches to Stop Hypertension (DASH) diet and Mediterranean dietary pattern.
The DASH diet is a plant-focused eating plan that emphasizes fresh, non-processed vegetables, fruits, whole grains, legumes, nuts, low-fat dairy, fish, and poultry, but limits red meat, salt, and sugars. This diet is based on research from the US National Heart, Lung, and Blood Institute. The DASH diet has been shown to lower blood pressure and cardiovascular risk, and possibly diabetes, in the general population.146-148
An influential observational study with 14,882 participants that measured the effect of the degree of DASH diet adherence on the risk of developing kidney disease found that the participants in the lowest one-third of adherence to the DASH diet, compared to those in the highest one-third, had a 16% higher chance of developing kidney disease.149 A similar study in 1,110 adults with CKD (eGFR 30–59) and high blood pressure who were enrolled in the observational study known as the National Health and Nutrition Examination Survey (NHANES) and followed for 7.8 years also reported that participants whose diet had poor resemblance to a DASH diet were significantly more likely to progress to kidney failure.150
Multiple observational studies have found that diets higher in plant foods or lower in animal foods are associated with improved kidney outcomes. One such study, known as the Multiethnic Study of Atherosclerosis, showed that in 5,042 patients aged 45–84 years, the participants who consumed a diet rich in whole grains, vegetables, fruits, and low-fat dairy had a 20% lower UACR. In another study that compared diets high in animal protein to diets high in vegetable protein, results indicated that people who consumed larger amounts of plant protein had 30% lower risk of CKD while animal protein conferred 37% higher odds of CKD. Another small study in people with diabetes showed that the participants in the highest third of vegetable protein intake had a 53% lower risk of developing CKD compared with those in the lowest third of intake.143
Another observational study evaluated the association of four different healthy diets, including DASH and a variety of the Mediterranean diet, with the risk of disease progression and all-cause mortality among people with CKD. About 2,403 men and women aged 21 to 74 years with eGFRs of 20–70 were followed over a 14-year period. The participants with the highest adherence to a healthy diet had a significantly lower risk of CKD progression and all-cause mortality, with the Mediterranean diet yielding the strongest protection as reflected by a 25% lower risk of disease progression.151
The Mediterranean diet is based on traditional foods from countries bordering the Mediterranean Sea, such as Spain and Greece. The Mediterranean diet is high in plant foods, including fresh vegetables, grains, fruits, legumes, and nuts, and includes moderate amounts of fish and poultry, with olive oil being the main source of additional fat. As in the DASH diet, red meat, sweets, butterfat, and highly processed foods are eaten infrequently.
A prospective cohort study in 975 men and women aged 60 years and older also reported that participants with the highest degree of adherence to a Mediterranean diet pattern had approximately half the risk of developing declining kidney function compared to those with the lowest adherence.152 In addition, a randomized controlled trial compared a standard low-fat diet to a Mediterranean diet high in extra virgin olive oil over a 5-year period to evaluate the effect on the preservation of kidney function in patients with coronary heart disease. The results showed eGFR declined significantly less in the participants assigned to the Mediterranean diet group, an effect observed more clearly in participants with mild loss of kidney function.153
Several individual parts of these diets have been studied for kidney benefits. In people with CKD, nut consumption 1–6 times per week was associated with a 37% lower risk of dying from any cause. In healthy individuals, the same level of nut consumption was associated with a 33% lower likelihood of being diagnosed with CKD.154 Other studies have shown that walnut or Brazil nut consumption favorably influences biomarkers of cardiovascular risk, inflammation, and oxidative stress, including in people undergoing dialysis.155-157
Furthermore, an observational study that used data from 157 CKD patients found that adequate fiber intake (25 grams per day or more) was associated with a less progressive decline in eGFR over a period of 18 months compared with lower intake. Fiber adequacy was also linked to lower blood levels of pro-inflammatory factors and a gut-derived toxin. Overall, high dietary fiber intake was associated with a 46% reduction in estimated cardiovascular risk.158 This finding is consistent with the hypothesis that fiber exerts multiple beneficial effects for CKD patients, including decreasing inflammation, supporting the presence of beneficial microbiota, and lowering the production of uremic toxins.159
Extra virgin olive oil is a key component of the Mediterranean dietary pattern and has been studied for its role in protecting against CKD and related conditions. In one study, 40 CKD patients consumed about 2.7 tablespoons of extra virgin olive oil per day for nine weeks: eGFR, albuminuria, and markers of oxidative stress and inflammation all improved.160
Alcohol
The kidneys are second only to the liver in importance with regards to metabolizing alcohol. About 10% of ingested alcohol is excreted from the kidneys in an unmetabolized form. Excessive alcohol consumption places an undue strain on normal kidney metabolism, including the production of free radical stress. Several lines of evidence indicate excessive alcohol consumption directly and significantly damages the kidneys.161 However, moderate drinking does not raise the chances of CKD progression compared with not drinking, and CKD guidelines only recommend that CKD patients adhere to alcohol intake guidance for the general population. Individualized decisions about drinking must take into account co-morbidities (eg, diabetes).162 Binge drinking, by contrast, does increase the risk of CKD progression.163
Balancing Specific Nutrients
Eating a healthy overall diet—high in fruits and vegetables and other nutrient-dense foods—is recommended for patients with stage 1–4 CKD.164 However, as kidney disease gradually progresses, it becomes increasingly important to protect against protein loss and to balance mineral and electrolyte levels. This can require careful management of the dietary intake of specific nutrients.165 In some cases, this can mean limiting consumption of foods that are normally considered healthy.165 More importantly, the nutritional needs of each individual can vary depending on the extent of kidney dysfunction and other factors. Therefore, it is advisable to work directly with a qualified dietitian nutritionist who has experience in CKD to optimize your nutrition.
Limit sodium intake. Excessive sodium intake can cause high blood pressure and contribute to CKD. Most packaged, processed, and canned foods, as well as table salt, are high in sodium.165 The 2020 National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines recommend adults with stage 3–5 CKD limit sodium intake to <2.3 grams daily to help control blood pressure and reduce urinary protein loss. The guidelines further acknowledge that maintaining a low sodium-to-potassium intake ratio may be more important than restricting sodium intake alone.164 Studies have shown that very high consumption of dietary sodium can blunt the effectiveness of ACE inhibitors in patients with CKD. Furthermore, an analysis of data from the Ramipril Efficacy in Nephropathy trial showed that high daily salt intake (>14 grams) decreased the effectiveness of ACE inhibitor therapy and increased the risk for kidney failure.166 For a clearer perspective, 14 grams is equivalent to more than 14 standard-sized ham and egg biscuits, which is one of the highest-sodium processed foods.167
Low- and very-low-protein diets. Limiting portions of high-protein foods such as red meat, poultry, eggs, dairy products, and fish is generally recommended to help preserve kidney function.165 Although protein is an essential component of a healthy diet, too much can be problematic for people with CKD. Many studies have shown that a high-protein diet (typical of Western-style diets) can lead to progressive kidney damage and albumin loss in individuals with suboptimal kidney function. On the other hand, protein restriction reduces nitrogen waste and helps decrease kidney workload by lowering pressure in the capillaries of the kidney’s filtration system.168
The 2020 KDOQI guidelines for the management of kidney diseases recommends that people with stage 3–5 CKD who are not receiving dialysis eat a low-protein diet (0.55–0.60 grams per kilogram of body weight per day [g/kg/day]) or a very-low-protein diet (0.28–0.43 g/kg/day) supplemented with 7–15 grams of keto-analogues of essential amino acids. Multiple meta-analyses and pooled analyses of the published literature have found that low-protein diets and very-low-protein diets supplemented with nitrogen-free derivatives of essential amino acids (eg, compounds called keto-analogues or keto acids) have a range of benefits in slowing disease progression. These potential benefits include preserved eGFR, reversal of bone-mineral disorder, decreased risk of progression to dialysis, reduced urinary protein loss, decreased blood phosphate, and reduced all-cause mortality.169-172 Ketoacid analogues provide the materials for protein synthesis without the nitrogenous waste products.172
A higher protein intake of 0.60–0.80 g/kg/day is recommended for those who also have diabetes in order to maintain blood glucose control. Protein intake may need to be temporarily boosted during illness or other times of increased protein need. For those with stage 5 CKD that is managed with hemodialysis or peritoneal dialysis, a diet providing 1.0–1.2 g/kg/day of protein is recommended.164,173
Adherence to protein-restricted diets can be very challenging and may be more effective when supported with nutritional counseling.174 Interestingly, some emerging evidence suggests protein restriction may not be as important for patients being treated with RAAS inhibitors and SGLT2 inhibitors, and several leading guidelines do not recommend any protein restriction for people with CKD, although excess protein intake is often discouraged.175
Limit phosphorus intake. Reducing the consumption of foods that are high in phosphorus (phosphate), such as meat, poultry, fish, dairy products, chocolate, beans, lentils, nuts, some whole grains, baking powder, and soft drinks (especially colas and other dark-colored sodas), becomes increasingly important as kidney function declines.165
Phosphorus occurs naturally in protein-rich foods, and it is added to many processed foods. In people with CKD, phosphorus can build up to unhealthy levels and deleteriously affect bones, making them weak and prone to fracture. A systematic review and meta-analysis of serum phosphorous levels and the progression of CKD showed an association between elevated phosphorous levels and an increased risk of kidney failure, along with higher mortality in patients with CKD.176 The 2020 KDOQI guidelines recommend that adults with stage 3–5 CKD limit dietary phosphorus intake to maintain normal blood phosphate levels (2.5–4.5 mg/dL).164
Balance potassium intake. The regulation of potassium levels is disrupted by kidney disease.177 People with CKD have more difficulty balancing potassium levels in the blood, particularly when they are managing their condition with medications or dialysis. Some CKD patients will need to monitor their potassium levels closely and adjust their intake of high-potassium foods, such as oranges, bananas, avocados, potatoes, tomatoes, beans, lentils, nuts, and some whole grains, as well as many herbal medicines and salt substitutes.165
Some medications used in CKD (eg, ACE inhibitors, ARBs, and MRAs) can raise or lower potassium levels, making it challenging to keep the dosage in a safe range. In advanced CKD, potassium accumulates in the blood, which can lead to dangerous arrhythmias and other serious health problems.178,179 Conversely, low potassium levels can contribute to arterial calcification, high blood pressure, and stroke.177 It is recommended that patients with stage 3–5 CKD adjust their potassium intake, most often by decreasing intake, to maintain normal blood levels.164
Although potassium restriction generally requires limiting healthy foods that are also high in vitamins, other minerals, phytonutrients, and fiber, some experts advocate for the use of potassium-binding medications to lower high potassium levels without restricting fruit and vegetable consumption.178
Reduce calcium intake. Similar to potassium intake, calcium levels are harder to regulate when the kidneys malfunction. Moderate dietary calcium is required for different processes, but consuming too much calcium may be problematic. Therefore, monitoring the intake of high-calcium foods, such as dairy products, rhubarb, spinach, and canned fish with bones, may be advised. CKD patients may also be instructed to avoid calcium-based antacids.165
Consuming excess or insufficient amounts of calcium can cause significant health issues, and the kidneys play a vital role in maintaining calcium balance. In CKD, this balance can be disrupted, leaving people with low calcium levels and an increased risk of osteoporosis or fractures. Elevated calcium also increases the risk of blood vessel calcification and heart disease.180 However, studies have shown that in patients with CKD, calcium intake of 800–1,000 mg daily can help maintain healthy calcium levels.181 Therefore, the 2020 KDOQI guidelines recommend 800–1,000 mg daily as a target for total calcium intake from food, supplements, and medications in adults with stage 3 and 4 CKD.164
10 Lifestyle Changes for Chronic Kidney Disease
Exercise
Maintaining a physically active lifestyle may be one of the keys to slowing the progression of CKD. Data from 5,916 CKD patients participating in the NHANES study indicated that those who adhered to physical activity guidelines (≥150 minutes of moderate-intensity physical activity per week) had better albumin-to-creatinine ratios and a 51% lower risk of death from any cause during 10 years of follow-up. In addition, those who were more sedentary had worse eGFRs and were more likely to die from cardiovascular disease or any cause.182 Other observational studies have also found a sedentary lifestyle to be associated with impaired kidney function and increased CKD risk.183,184 For patients with CKD, physical activity may slow kidney function decline and improve survival.185,186 Furthermore, regular physical activity improves overall health and quality of life and reduces the risk of related conditions, such as diabetes, obesity, and hypertension, that are associated with more rapid progression of CKD.187,188
Multiple clinical trials have found that exercise therapy may slow the rate of kidney function loss or, in some cases, improve kidney function. A meta-analysis of 13 clinical trials that included 421 patients with stage 2–5 CKD who did not require dialysis found that up to three months of exercise therapy resulted in a significant increase in eGFR compared with no exercise intervention. Exercise also reduced systolic and diastolic blood pressure and led to decreased BMI in those who undertook more than six months of exercise.189 More recently, an analysis of data from the LIFE clinical trial, which enrolled 1,199 sedentary adults aged 70–89 years, examined the effect of a moderate-intensity physical activity plus strength and flexibility exercise intervention on kidney function. The exercise intervention resulted in a significantly slower decline of eGFR (calculated using cystatin-C levels) over two years compared with health education alone. In addition, participants in the exercise group were found to have 21% lower odds of rapid eGFR decline (defined as a decline of 6.7% per year), suggesting this exercise program may help preserve kidney function.190
In a separate systematic review and meta-analysis of 18 clinical trials involving 848 patients with CKD who did not require dialysis, the effects of exercise on all-cause mortality and eGFR were not significantly different from those of usual care.191 Nevertheless, regular exercise confers many benefits, and clinicians, as well as professional societies, recommend regular physical activity for all CKD patients unless there is a compelling reason not to do so.162
Quit Smoking
The risk of CKD progression is higher in current and past smokers than in people who never smoked. Furthermore, individuals who continue to smoke after a CKD diagnosis have a higher risk of disease progression and dying than those who quit or those who never smoked. The all-cause mortality may be more than twice as high in current smokers with CKD compared with those who never smoked.192,193 Although the nature of the relationship is not entirely understood, it is believed that smoking causes blood vessel damage and dysfunction that leads to poor kidney blood flow and decreased kidney function.193
11 Nutrients to Support Kidney Function
Note: People with CKD should consult a qualified healthcare provider with expertise in the management of CKD before initiating supplementation. CKD can affect the metabolism of some nutrients, and supplementation may not be advisable in those with advanced CKD.
Probiotics & Prebiotics
Probiotics are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host.”194 Prebiotics are defined as “a substrate that is selectively utilized by host microorganisms conferring a health benefit.”195 Probiotics use prebiotics as a source of nourishment for growth and reproduction. Synbiotics are supplements that combine the two.
Both probiotics and prebiotics have a variety of biological actions, including outcompeting pathogenic microorganisms in the digestive tract, inactivating harmful metabolites, producing powerful bioactive molecules (short-chain fatty acids), modulating immune and metabolic signaling, improving overall digestive tract function, and enhancing central nervous system effects through the gut–brain axis.196 Both probiotics and prebiotics can have favorable impacts on the composition of the gut microbiome, the community of microorganisms that normally reside in the digestive tract and may have benefits for systemic health.197
Gut microbiome composition is usually altered in CKD patients. It is believed that imbalanced gut microflora may contribute to CKD progression, but CKD also appears to disrupt the intestinal environment and affect microbiome composition.197 For instance, increased blood urea concentrations can alter the microbiome, resulting in the increased generation of toxic metabolites from the gastrointestinal tract as well as an increased tendency for toxins to cross the gut barrier, which can promote further kidney injury.198
A randomized placebo-controlled trial in 50 patients on hemodialysis found that supplementing with a probiotic formulation containing Lactobacillus sporogenes and fructo-oligosaccharides (FOS) (ie, fermentable prebiotic fibers) for eight weeks led to increased hematocrit, hemoglobin, and red blood cell counts, as well as improved markers of iron status.199 Iron status was also improved in stage 3a CKD patients treated for nearly four months with mixtures of Bifidobacterium and Lactobacillus species in a randomized placebo-controlled trial with 28 participants. The sequential probiotics used in this trial were: a combination of Enterococcus faecium UBEF-41, L. acidophilus LA-14, and Saccharomyces cerevisiae ssp. Boulardii MTCC-5375 for one week, followed by B. brevis BB03, B. bifidum BB06, and B. longum BL05, as well as L. rhamnosus HN-001, L. rhamnosus LR-32, and L. acidophilus LA-14 taken three times daily for two weeks, then twice daily for three months.200
Another trial involving hemodialysis patients suffering from constipation demonstrated that a synbiotic formula afforded digestive benefits. The formulation consisted of 10 billion colony forming units (CFUs) of L. acidophilus plus B. longum and 120 mg of FOS. When taken daily for 60 days, the synbiotic formula relieved constipation symptoms, improved bowel movement frequency, and improved quality of life scores more than placebo. The formulation did not lower levels of a uremic toxin related to CKD symptoms.201
A meta-analysis that reported findings from 23 randomized controlled trials involving 842 participants with stage 3–5 CKD indicated that probiotic, prebiotic, and synbiotic supplements improved creatinine and BUN levels, heightened markers of oxidative stress, reduced some markers of inflammation, and decreased uremic toxin levels. However, these supplements did not improve eGFR or blood albumin levels.202 Additional meta-analyses have also concluded that probiotics, prebiotics, and synbiotics can reduce oxidative stress, decrease inflammation, improve lipid levels, and heighten glucose metabolism in CKD patients.203-205
Another open-label controlled trial in 24 stable stage 3–4 CKD patients found that a low-protein diet supplemented with a synbiotic formula for six months resulted in a slower decline in GFR than a low-protein diet alone during one year of monitoring.206 In a placebo-controlled trial, 60 patients with stage 4 CKD who had been eating a low-protein diet for two months were assigned to add probiotic supplements providing 1 billion CFUs L. reuteri and 5 billion CFUs B. longum or placebo twice daily for one month and then once daily for another two months. The probiotic supplement was found to mitigate increased lipid levels, reduce urine protein levels, lower loop-diuretic and anti-hypertensive medication use, and improve quality of life. In addition, a trend toward delayed progression to kidney failure and the need for dialysis was observed in the probiotic group compared with the placebo group at a 3-year follow-up.207 However, whether synbiotic or probiotic treatment has clinical benefits for people with CKD has not been conclusively demonstrated in clinical trials.
Omega-3 Fats
Omega-3 fatty acids, such as eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) from fish oil, have been extensively studied in the context of cardiovascular health. Accumulating evidence suggests EPA and DHA may be associated with benefits for those who have CKD. For instance, a meta-analysis of 19 observational studies with a combined total of 25,570 participants found that those in the highest 20% of intake of omega-3 fats from seafood had a 13% lower risk of developing CKD than those in the lowest 20% during a median of 11.3 years of monitoring.208
Omega-3 fatty acid supplements may be especially helpful in CKD patients undergoing hemodialysis. An observational study that followed 336 hemodialysis patients for five years found lower levels of EPA plus DHA were correlated with higher risks of cardiovascular events, peripheral vascular disease, and all-cause mortality.343 According to a 2020 systematic review and meta-analysis that included 60 randomized controlled trials with a total of 4,129 patients, omega-3 fatty acid supplementation appeared to decrease the incidence of cardiovascular death by 55% in CKD patients on hemodialysis, though it did not appear to reduce mortality overall. Omega-3 fatty acid intake may have also reduced the risk of progression to kidney failure in participants who did not receive a kidney transplant, but there was not enough data to draw a solid conclusion.213
More recent clinical trials have added to the evidence that omega-3 fatty acids have important benefits in CKD patients requiring hemodialysis. A randomized placebo-controlled trial in 1,228 CKD patients receiving maintenance hemodialysis found that 4 grams of fish oil (providing 1.6 grams of EPA and 0.8 grams of DHA) daily for 3.5 years reduced the rate of serious cardiovascular events by 43%. This protective effect was similar in those with and without a history of a previous cardiovascular event. Specifically, heart attack rate was 44% lower, stroke rate was 63% lower, rate of cardiac death was 45% lower, and rate of peripheral artery disease requiring amputation was 43% lower with omega-3 fatty acids than placebo (corn oil).344 Another randomized controlled trial included 120 CKD patients receiving hemodialysis and found 3 grams of omega-3 fatty acids per day for two months improved BUN and creatinine levels compared with control (medium-chain triglyceride oil).345 Other trials have shown omega-3 fatty acid supplementation can increase skin hydration, reduce skin itching, decrease depression symptom scores, and reduce levels of markers of oxidative stress and inflammation in CKD patients receiving hemodialysis.210,346-348
Clinical trials investigating the effects of omega-3 fats in non-hemodialysis patients with or at high risk of CKD have yielded less-promising findings. A randomized placebo-controlled trial that included 1,312 individuals with type 2 diabetes demonstrated that 1 gram of EPA and DHA, plus 50 mcg (2,000 IU) of vitamin D, taken daily for five years had no effect on the progressive loss of kidney function.211 A randomized controlled trial involving 100 CKD patients reported that consuming 3,666 mg EPA and DHA daily was not more effective than corn oil at reducing urine albumin levels, but the formulation did improve blood vessel function and lower triglyceride levels more than corn oil.212
Coenzyme Q10
Coenzyme Q10 (CoQ10), a potent antioxidant molecule made in the body that plays a vital role in cellular metabolism and energy production, is especially important for maintaining healthy function of the heart muscle. Blood levels of CoQ10 have been shown to be lower in people with mild kidney disease as well as those with advanced kidney disease undergoing dialysis.214,215 A systematic review of 12 clinical trials found that CoQ10 supplementation in patients with CKD significantly reduced blood levels of malonaldehyde, a marker of oxidative stress, and high-sensitivity C-reactive protein (hs-CRP), which is a marker of systemic inflammation.216 Furthermore, a randomized, double-blind, placebo-controlled trial in 65 hemodialysis patients reported that taking 1,200 mg, but not 600 mg, of CoQ10 daily for four months decreased oxidative stress (as indicated by a decrease in F2-isoprostanes). However, CoQ10 did not improve markers of cardiac damage or stress more than placebo.217 In addition, a double-blind, placebo-controlled, crossover trial with 23 participants reported that taking 200 mg CoQ10 daily did not affect exercise performance or markers of exercise-induced oxidative stress in hemodialysis patients.218 Therefore, it is uncertain whether CoQ10 has clinically important benefits for people with or at risk for CKD.
Vitamin D
Low vitamin D levels are common in the United States. One study that analyzed data from a representative sample of the U.S. population found that roughly 70% of the non-institutionalized population had 25-hydroxyvitamin D levels below about 30 ng/mL.219 Low vitamin D levels are associated with multiple health issues, including cardiovascular disease, which is a leading cause of death in patients with CKD. A 2-year observational study that included 168 CKD patients demonstrated that participants with the lowest vitamin D levels had a faster rate of CKD progression and shorter time to kidney failure and death.220 These findings were confirmed in a meta-analysis of 10 observational studies involving a total of 6,853 participants with CKD, which reported that higher vitamin D levels were associated with lower mortality.221
Although the effects of vitamin D supplementation on the course of CKD are still unknown, it may play a role in mitigating certain CKD complications. A meta-analysis that reported findings from seven randomized controlled trials indicated that vitamin D supplementation may improve blood vessel function in individuals with CKD, and this vitamin may be more effective during the earlier stage of CKD and when high doses are consumed.222 In one randomized placebo-controlled trial, 120 nondiabetic men and women aged 18–70 years with stage 3–4 CKD and low (<20 ng/mL) vitamin D levels were assigned to receive two oral doses of vitamin D (7,500 mcg [300,000 IU] each) or a placebo at baseline (day 0) and again at eight weeks. Blood vessel function improved in participants given vitamin D, but this parameter did not change in the placebo group.223 Findings from another meta-analysis that included 17 randomized controlled trials in patients with CKD indicated that vitamin D supplementation may also improve markers of cardiometabolic health, including fasting blood glucose, triglyceride levels, and cholesterol levels, as well as a marker of insulin resistance.224 The effect of vitamin D supplementation on bone loss and fracture risk in CKD patients is still uncertain.225-227
Note: High doses of vitamin D can affect calcium and phosphate balance. Therefore, people with severe CKD should contact their physician or a dietitian nutritionist before commencing vitamin D supplementation.
Menaquinone-7/Vitamin K-2
Vitamin K is a fat-soluble vitamin that is present in food in two forms: K1 (phylloquinone) and K2 (menaquinone). Vitamin K1 is well known for its role in blood clotting, whereas K2 appears to be important for diverse processes, including bone preservation, cardiovascular and kidney protection, healthy cognitive function, blood glucose regulation, and cancer prevention.228,229
Observational studies indicate that CKD patients are prone to inadequate vitamin K intake and low vitamin K status, partly due to dietary restrictions. Low vitamin K status is thought to be a factor in vascular calcification that leads to complications and the progression of CKD.228 An uncontrolled trial also reported that in 60 patients who had undergone kidney transplant surgery, supplementation with 360 mcg menaquinone-7 (a form of vitamin K2) for eight weeks reduced a measure of arterial stiffness.230 More than half of the participants had subclinical vitamin K2 deficiency at study recruitment.
Another trial that included 53 long-term dialysis patients and 50 healthy controls demonstrated that vitamin K2 supplementation, at daily doses of 45 mcg, 135 mcg, or 360 mcg for six weeks, improved a marker of vitamin K2 status and blood vessel calcification risk. The effect of K2 supplementation on this marker increased at higher doses.231
Low vitamin K levels may also contribute to increased bone loss and fracture risk in CKD patients.229 An observational study in 387 patients on hemodialysis showed that vitamin K1 deficiency was strongly correlated with vertebral fractures.232 Another study that included data from 468 subjects with kidney failure awaiting transplant reported that 90% of participants had low vitamin K status, and lower vitamin K levels were associated with lower bone mineral density and increased fracture risk over five years of follow-up visits.233 Future studies should further explore the potential benefits of vitamin K supplementation in people with CKD.
Selenium & Vitamin E
Selenium is an essential trace mineral that the body needs for the internal synthesis of a range of proteins and enzymes, including the glutathione antioxidant system.234 An observational study found that higher selenium levels are associated with greater preservation of kidney function in CKD patients.235 In a randomized placebo-controlled trial in 170 subjects with type 2 diabetes at high risk of CKD, supplementation with 20 mcg selenium and 400 IUs vitamin E (form not specified) per day for 12 months also improved kidney blood flow and eGFR. Improvement in glomerular filtration paralleled the increase in glutathione activity.236 Another randomized placebo-controlled trial in 215 elderly individuals who did not have kidney disease—but presented with low selenium and CoQ10 levels—demonstrated that daily supplementation with 200 mcg selenium and 200 mg CoQ10 for 5.1 years led to improved creatinine and cystatin-C levels, indicating improved kidney function.237 Whether these benefits will translate into stability of kidney function over the long-term remains to be shown by future studies.
An observational study analyzed data from 20,295 participants in the NHANES study from 2009 to 2016 to investigate the relationship between vitamin E intake and CKD prevalence and risk. The researchers found a negative association between high vitamin E intake and CKD prevalence, and the effect was more pronounced in very high-risk CKD patients. The authors concluded that higher vitamin E intake was protective against CKD across different risk strata, and subgroup analysis confirmed the stability of these findings across populations.238
Resveratrol
Resveratrol is a polyphenol (antioxidant) that helps attenuate oxidative stress and promotes anti-inflammatory activity. In addition, resveratrol has demonstrated anti-aging properties in preclinical research.239
In a double-blind, randomized, placebo-controlled trial, 72 CKD patients aged 43–67 years being treated with peritoneal dialysis were randomized to receive 150 mg/day resveratrol, 450 mg/day resveratrol, or a placebo for 12 weeks. Patients in both resveratrol groups experienced significant improvement in their ultrafiltration rate and volume (measures of the effectiveness of dialysis) compared with participants in the placebo group. Furthermore, the participants who received high-dose resveratrol were less likely to show markers of angiogenesis (new blood vessel formation) compared with placebo.240 Excessive angiogenesis can be a problem in patients undergoing peritoneal dialysis. In a randomized, double-blind, placebo-controlled, crossover study investigating the effects of resveratrol supplementation on endothelial function in 28 adults with stage 3 CKD and diabetes, six weeks of supplementation with 400 mg resveratrol daily significantly increased brachial artery flow-mediated dilation. However, eGFR, hemoglobin A1C levels, blood pressure, and nitroglycerin-mediated dilation remained unchanged.241 Overall, more research is needed to establish whether resveratrol can improve outcomes in people with kidney disease.
Grape Seed Extract
Grape seeds contain proanthocyanidins, which are flavonoid polyphenols that are considered to be among the strongest plant antioxidants.242 In a placebo-controlled trial involving 43 CKD patients, 23 participants were given 2 grams grape seed extract daily, while 10 were given a placebo. After six months, the participants who took grape seed extract experienced an improvement in GFR levels and an increase in antioxidant enzyme activity compared with the placebo group.243 In another randomized placebo-controlled trial, 30 CKD patients were given grape seed flour at a dose of 1 g/kg of body weight daily, while 10 CKD patients were given a starch placebo. Blood and urine samples were collected at baseline and six months. As in the previous study, the patients who consumed grape seed demonstrated improved GFR levels and antioxidant status compared with placebo.244 Additional studies are needed to further elucidate the potential of grape seed extract to support kidney health.
Zinc
Zinc is an essential trace mineral with a myriad of functions in the body. Zinc deficiency can lead to skin problems, immune dysfunction, neurological disorders, and metabolic disturbances.245 A correlational study with nearly 160,000 participants demonstrated that people with lower dietary zinc intake were more likely to have CKD.246 Zinc deficiency has also been associated with CKD risk, progression, and complications such as anemia and cardiovascular disease.247,248
Observational evidence has shown that individuals with CKD have lower zinc levels than healthy people and that lower zinc levels are associated with lower eGFR.249-251 In addition, the relationship between zinc and CKD progression may be bidirectional since compromised kidney function may contribute to urinary zinc loss and low baseline blood zinc levels are linked to faster CKD progression.252 However, in a double-blind randomized trial involving 150 hemodialysis patients in Alberta, Canada, researchers evaluated the effects of adding zinc, selenium, and vitamin E to standard oral renal vitamins (B and C) over 90 and 180 days. Two dosage groups were compared to controls: a medium-dose group received 50 mg zinc, 75 mcg selenium, and 250 IU vitamin E (form not specified) daily; a low-dose group received 25 mg zinc, 50 mcg selenium, and 250 IU vitamin E daily. The study found no significant differences in correcting low levels of zinc or selenium in blood between the control and intervention groups. Limitations include a relatively small sample size and conservative doses of the supplements, which might have reduced the ability to detect benefits. Since these doses of zinc and selenium did not effectively correct deficiencies in hemodialysis patients, future studies exploring higher doses may be warranted.253
Other research shows that zinc supplementation at 50 mg/day for six months helped resolve CKD-related anemia in 21 hemodialysis patients with zinc deficiency.254 Furthermore, a controlled trial in 70 hemodialysis patients with zinc deficiency demonstrated that taking 34 mg zinc daily for 12 months decreased the required dosage of treatments that increase red blood cell production. This study provided evidence that zinc supplementation in people undergoing hemodialysis and who were zinc deficient improved their bodies’ ability to produce red blood cells.255 In addition, a randomized controlled trial in 40 patients receiving hemodialysis for kidney failure showed that taking 100 mg zinc (as 440 mg zinc sulfate) daily for two months reduced skin itching better than placebo.256
B Vitamins
Elevated levels of homocysteine, a nonessential amino acid, can impair blood vessel function and increase oxidative stress and inflammation. Homocysteine elevation is a known risk factor for cardiovascular disease, the leading cause of death in CKD patients, and decreased glomerular filtration tends to elevate homocysteine levels.30,257,258 Supplementation with B vitamins, such as folic acid, B6, and B12, effectively lowers high homocysteine levels.259
A meta-analysis of randomized controlled trials found that the use of folic acid to lower high homocysteine levels resulted in a 10% lower risk of stroke and a 4% lower risk of cardiovascular disease.260 However, the precise role of high homocysteine in the development of cardiovascular disease in CKD patients remains unclear, as does the ability of B vitamins to influence risk or outcomes.258 Furthermore, high doses of B vitamins may be contraindicated for some patients with CKD.
Previous research involving the assessment of B vitamin supplementation on kidney outcomes showed that high daily doses of B vitamins (42.5 mg DFE folic acid, 25 mg B6, and 1 mg B12) appeared to be associated with a greater decrease in GFR during a median follow-up of 32 months compared with placebo.261 Additional outcomes of stroke (ie, revascularization, myocardial infarction, and all-cause mortality) also developed twice as often with B vitamin treatment compared with placebo. These findings raise safety concerns regarding high doses of B vitamins for the treatment of diabetic kidney disease.
With regard to advanced CKD and kidney failure, the Homocysteine Study (HOST) also reported that all-cause mortality did not decrease in the group of participants who received high daily doses of B vitamins (68 mg DFE folic acid, 100 mg B6, and 2 mg B12) versus placebo.262 Furthermore, treatment with high doses of B vitamins did not delay the initiation of dialysis.
In another double-blind placebo-controlled trial that examined the daily use of folic acid (68 mg DFE), vitamin B6 (100 mg), and vitamin B12 (2 mg) in 2,056 men and women with advanced CKD or kidney failure requiring kidney replacement surgery, similar findings were observed. After 3.2 years, the authors reported a significant reduction in homocysteine levels but no reduction in all-cause mortality or cardiovascular death.263
Conversely, a large study in 15,104 patients with high blood pressure in China found that the combination of enalapril (Vasotec) (an ACE inhibitor, 10 mg daily) and folic acid (1,360 mcg DFE daily), compared to enalapril alone, significantly improved eGFR and reduced CKD progression.264 However, hyperhomocysteinemia (elevated homocysteine in the blood) and folic acid deficiency are widespread in China, a country that does not fortify its food supply with folic acid (as of 2023). Therefore, these potential benefits of folate supplementation may not apply to people living in other countries.
Individuals with CKD should be tested for homocysteine, folate, and vitamin B12 levels before initiating B vitamin treatment.
Vitamin C
Hemodialysis substantially reduces vitamin C blood (plasma) levels.265,266 This is a serious problem because foods rich in vitamin C are often rich in potassium as well, and thus are frequently avoided by people undergoing dialysis for advanced kidney disease since dialysis does not remove much potassium.267 Moreover, vitamin C increases iron absorption, and anemia is a common problem in advanced CKD. Increased oxidative stress and inflammation are also common in advanced kidney disease, and vitamin C supplementation may help counteract these effects. As such, vitamin C supplementation has been studied in patients with CKD and shown to provide various benefits, although studies have often been small. Generally, vitamin C dosages ranging from about 250 mg daily to 500 mg every other day have been studied in patients with CKD.268
Importantly, vitamin C supplementation in CKD, particularly in the context of advanced kidney disease, is controversial. People with advanced CKD are prone to developing high levels of a compound called oxalate in their blood, and high oxalate levels can cause health problems. Vitamin C in high dosages may increase oxalate levels in people with kidney disease. Therefore, high-dosage vitamin C, and high-dosage intravenous vitamin C in particular, should be avoided by people with CKD. Overall, research is inconclusive as to the optimal vitamin C dosage in people with CKD, particularly those with advanced CKD.269,270 Experts in the nutritional needs of CKD patients have stated that daily vitamin C supplementation as necessary to meet the recommended daily intake of 90 mg for men and 75 mg for women may be reasonable.164
Melatonin
Melatonin, a hormone produced mainly in the pineal gland, is best known for its role in regulating circadian rhythms. This hormone has a range of health-promoting effects, including sleep induction and antioxidant activity.271,272 Melatonin also appears to protect kidney function through anti-inflammatory, oxidative stress-reducing, anti-fibrotic, and mitochondria-supportive mechanisms, as well as by suppressing renin-angiotensin system activation.273-275 Observational research in CKD patients has indicated that impaired melatonin production is associated with nighttime renin-angiotensin system activation, increased kidney damage, and lower GFR.276,277
Furthermore, sleep disturbances—such as insomnia, sleep apnea, and restless legs syndrome—frequently accompany CKD and may contribute to its complications and progression.278
In a pilot trial, 60 stage 5 CKD patients awaiting kidney replacement therapy who also had impaired melatonin production were treated with 3 mg melatonin nightly for eight weeks. Following melatonin intervention, the need for anti-hypertensive medication decreased.279 A similar observational study in 117 kidney failure patients receiving hemodialysis compared the effects of alprazolam (Xanax) to melatonin supplementation. The patients had been taking either alprazolam (0.5 mg nightly) or melatonin (3 mg nightly) to treat sleep disturbances for at least three months, and it was noted that those who took melatonin had lower insomnia severity, less daytime sleepiness, and better sleep quality scores than alprazolam users.280
Melatonin may also have benefits beyond sleep improvement in individuals with CKD. In a controlled trial in 41 CKD patients with anemia, adding 5 mg melatonin nightly for 12 weeks to standard therapies increased hemoglobin levels, improved markers of iron status, and decreased levels of inflammatory markers compared with standard therapies alone.281 However, the efficacy of melatonin has not been conclusively demonstrated in CKD patients and further studies appear advisable, especially in patients with kidney failure.
Lipoic Acid
Lipoic acid, also known as alpha-lipoic acid, is a sulfur-containing compound that is made naturally by humans as well as plants and other animals. It acts as a powerful antioxidant and plays a vital role in cellular energy production and mitochondrial health. The body also needs lipoic acid for normal blood vessel function.282 A randomized controlled trial with 59 participants examined the effects of 1.6 grams of lipoic acid taken daily for six months on markers of systemic inflammation, blood vessel dysfunction, and atherosclerosis in patients with stage 2–3 CKD due to polycystic kidney disease. Participants who took lipoic acid showed a significant reduction in levels of blood glucose and C-reactive protein (CRP), a marker of systemic inflammation. However, markers of atherosclerosis and blood vessel function were unchanged in participants who did not take lipoic acid.283 In a double-blind, randomized, placebo-controlled trial that included 63 CKD hemodialysis patients, 600 mg lipoic acid taken daily for eight weeks decreased levels of hs-CRP, but this dose did not affect oxidative stress or blood lipid levels.284 Future studies are needed.
Carnosine
Carnosine, a dipeptide made from the amino acids alanine and histidine, is found in skeletal muscles as well as the nervous system and other tissues. Carnosine has demonstrated anti-inflammatory and antioxidant properties, inhibits the formation of advanced glycation end products (AGEs; biomolecules that have been damaged by exposure to sugars), and supports healthy mitochondrial function.285-287 In addition, carnosine is a natural inhibitor of ACE. These combined actions protect the kidneys from stressors such as low blood flow, high glucose levels, and kidney toxins.288 Increased levels of carnosinase-1 (the enzyme that breaks down carnosine) due to a variation in the CNDP1 gene have been linked to urinary and blood carnosine depletion, increased kidney damage, and the progressive loss of kidney function in CKD patients with diabetes.289-291
A trial involving 40 adult patients with diabetic kidney disease reported that 2 grams L-carnosine, taken daily for 12 weeks, decreased urinary levels of transforming growth factor (TGF)-beta, a cytokine related to kidney damage, but had no significant effect on BUN, blood creatinine levels, or GFR.292 Baseline carnosinase-1 enzyme levels may explain the absence of an effect in some patients following carnosine supplementation. In particular, a clinical trial in subjects with diabetic kidney disease found that 8 out of 25 participants experienced no measurable increase in carnosine levels after taking a single dose of carnosine at 60 mg/kg of body weight. These non-responsive individuals were found to have circulating carnosinase-1 levels that were 2-fold higher and carnosinase-1 activity that was 1.5-fold higher than participants whose carnosine levels increased after supplementation.293 Whether carnosine has any clinical benefits for people with CKD remains to be determined.
Magnesium
Magnesium, an essential mineral, is a cofactor for a vast number of enzymatic reactions. In advanced CKD, magnesium loss can occur as the kidney loses the ability to retain this mineral, resulting in low serum magnesium levels. Decreased magnesium levels then hasten disease progression, as magnesium can no longer balance phosphate. This process also leads to kidney toxicity. Low serum magnesium and elevated phosphate also appear to contribute to calcification of the arteries, thus compounding cardiovascular disease—the primary cause of death in CKD patients.294,295
Magnesium has a number of nutritional effects of direct relevance to many CKD patients. It is cardioprotective,296 involved in the regulation of blood pressure,297 provides protection against diabetes and prediabetes,298,299 and is recognized as a vital mineral for bone health.300
There is also considerable evidence that low serum magnesium levels are associated with poor outcomes in CKD patients. Conversely, higher serum magnesium levels are associated with health benefits, and magnesium supplementation may improve some outcomes for CKD patients.
A large meta-analysis that included data from over 205,000 CKD patients concluded that low serum magnesium, compared with normal levels, was associated with nearly double the risk of death from any cause in patients with kidney failure. The risk was similar when all hemodialysis patients were included. However, in this large dataset, higher magnesium levels were associated with protection from cardiovascular and all-cause death.301 A similar large meta-analysis involving 22 observational studies of CKD patients reported that low magnesium levels were associated with an increased risk of cardiovascular events (eg, heart attack and stroke), death due to cardiovascular disease, and all-cause mortality.302
Another meta-analysis that included findings from nine clinical trials involving magnesium supplementation in CKD patients suggested magnesium therapy reduced calcium and parathyroid levels and reduced atherosclerosis (measured as inner wall thickness) of the carotid arteries, but did not reduce vascular calcification.303 In a 2-year, open-label, randomized, controlled trial that enrolled 125 patients with stage 3–4 CKD, one group received an ascending daily dose of oral magnesium oxide, eventually reaching a daily dose of 507 mg. The trial was terminated early due to the dramatic benefit observed in the magnesium oxide group, in which the rate of increase in coronary artery calcium was 71.4% lower than the controls. No differences were observed for the increase in thoracic artery calcification.304 However, in a randomized placebo-controlled trial with 148 participants with stage 3–4 CKD, 15 millimoles of oral magnesium hydroxide (about 875 mg, providing approximately 368 mg elemental magnesium) taken twice daily for 12 months did not prevent the advancement of coronary artery calcification, despite an increase in plasma magnesium.305
Improved magnesium status has also been correlated with lower hip fracture risk in hemodialysis patients.306 In particular, a meta-analysis of eight clinical trials reported that magnesium supplementation improved markers of calcium metabolism, potentially leading to better bone mineral density in patients on hemodialysis.307
Terminalia bellerica
Terminalia bellerica (T. bellerica), a tree whose fruit has been used in traditional medicine for centuries, may have uric acid-lowering properties. Preclinical research suggests T. bellerica may reduce uric acid via inhibition of the enzyme xanthine oxidase, which is involved in the synthesis of uric acid.340 This evidence raises the intriguing possibility that T. bellerica may have therapeutic effects in the context of CKD, since high uric acid can promote inflammation and contribute to CKD progression, and since the kidneys’ ability to secrete uric acid decreases as CKD advances.341
In a randomized controlled trial, 88 people with high uric acid were given one of several interventions, including 500 mg twice daily of a T. bellerica extract called Ayuric (standardized to at least 15% tannins). After 24 weeks, subjects taking the T. bellerica extract had a mean reduction in uric acid of about 28%—from 8 to 5.8 mg/dL.341
In a separate 24-week, randomized, controlled trial conducted by the same research group, the effects of T. bellerica were compared with the uric acid-lowering drug febuxostat (Uloric) in 55 people with CKD. Participants received either 500 or 1,000 mg of the same T. bellerica extract twice daily or a once-daily 40 mg dose of febuxostat. All treatments decreased uric acid, with febuxostat being most effective. The drug and the high dosage of T. bellerica lowered serum creatinine equally, by about 24%. The lower dosage of T. bellerica reduced creatinine by about half as much. In terms of their effect on eGFR, febuxostat and high-dose T. bellerica performed similarly, resulting in an increase of about 40–41 mL/min/1.73m2. The low-dose T. bellerica was far less effective. Nitric oxide, a critical factor in endothelial function, increased similarly in both the febuxostat and high-dose T. bellerica groups.342
12 Frequently Asked Questions
How common is chronic kidney disease?
Chronic kidney disease (CKD) is relatively common among U.S. adults. According to recent estimates, about 15% of the U.S. adult population, or 37 million people, have CKD. As many as nine out of 10 of these people do not realize they have CKD, and about two out of five people do not know they have advanced CKD.308
Does chronic kidney disease lead to other health conditions?
As CKD progresses, other serious health conditions can develop or worsen, including heart disease, stroke, anemia, high blood pressure, cognitive impairment, and bone loss. CKD can also lead to early death.64
Does chronic kidney disease make you immunocompromised?
CKD can lead to a decreased immune response and predispose individuals to more infections, cancers, and a diminished response to vaccines.309,310
Is chronic kidney disease hereditary?
Chronic kidney disease can be hereditary, meaning that this condition may run in families. However, many times a family history of CKD is due to social factors, which doctors call social determinants of health. These factors can trigger other health conditions that contribute to the onset of CKD.311
What is hypertensive chronic kidney disease?
Hypertensive CKD is the co-occurrence of chronic high blood pressure and CKD. Declining kidney function triggers the kidneys to release hormones that cause salt and water retention through a process that leads to or aggravates high blood pressure. Due to the cyclic relationship between these conditions, it is not typically possible to know whether kidney dysfunction is the cause or result of increased blood pressure.312
How long can someone live with chronic kidney disease?
The life expectancy for someone with CKD depends on multiple factors, including age and disease stage at diagnosis, presence of other health conditions, and their response to treatment. As most CKD-related deaths are due to cardiovascular events, managing cardiovascular risk factors, such as high blood pressure and imbalanced lipid levels, is paramount for delaying mortality.313
Can a person who donates a kidney live a normal life with only one kidney?
Yes! People who donate a kidney generally have a normal life after their donation. There are some risks to consider, such as a very small risk of complications during surgery and a slightly increased risk of kidney disease over the long-term. The magnitude of these risks is generally thought to be small,314,315 and research is ongoing to better understand long-term outcomes for living donors.316 Donating a kidney to improve the quality of life for someone you care about can be a tremendously rewarding experience. Of course, donating a kidney is a major life decision requiring ample introspection and access to complete, accurate information upon which to base your decision. For more information on living kidney donation, speak with your healthcare provider(s) and consult authoritative resources such as the National Kidney Foundation’s information for living donors.
2025
- Dec: Updated sections on mineralocorticoid receptor antagonists in Management of Chronic Kidney Disease and omega-3 fats in Nutrients to Support Kidney Function
- Jan: Added section on Terminalia bellerica to Nutrients to Support Kidney Function
2024
- Dec: Added section on Terminalia bellerica to Nutrients to Support Kidney Function
- Jun: Added section on glucagon-like peptide-1 receptor agonists (GLP-1RAs) to Novel & Emerging Treatment Approaches
- Jun: Added section on pentoxifylline to Novel & Emerging Treatment Approaches
2023
- Dec: Comprehensive update & review
2022
- May: Added Colchicine & Chronic Kidney Disease
- May: Added Exercise & Chronic Kidney Disease
2021
- Nov: Added section on overcoming CKD-related fatigue to SGLT2 Inhibitors and Chronic Kidney Disease
2012
- Mar: Comprehensive update & review
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
- Levey AS. Defining AKD: The Spectrum of AKI, AKD, and CKD. Nephron . 2022;146(3):302-305. doi:10.1159/000516647. https://karger.com/nef/article-pdf/146/3/302/3752785/000516647.pdf
- Kovesdy CP. Epidemiology of chronic kidney disease: an update 2022. Kidney Int Suppl (2011) . Apr 2022;12(1):7-11. doi:10.1016/j.kisu.2021.11.003. https://www.ncbi.nlm.nih.gov/pubmed/35529086
- CDC. Chronic Kidney Disease in the United States, 2023. CDC. Accessed Dec. 6, 2023, https://www.cdc.gov/kidney-disease/php/data-research
- Berns JS. Patient education: Chronic kidney disease (Beyond the Basics). UpToDate. Updated 7/8/2021. Accessed 11/17/2022, https://www.uptodate.com/contents/chronic-kidney-disease-beyond-the-basics
- CDC. Chronic Kidney Disease Basics. Updated 02/28/2022. Accessed 11/10/2022, https://www.cdc.gov/kidney-disease/about/
- Levey AS, Grams ME, Inker LA. Uses of GFR and Albuminuria Level in Acute and Chronic Kidney Disease. N Engl J Med. Jun 2 2022;386(22):2120-2128. doi:10.1056/NEJMra2201153. https://www.ncbi.nlm.nih.gov/pubmed/35648704
- Rosenberg M. Overview of the management of chronic kidney disease in adults. UpToDate. Updated 11/10/2022. Accessed 11/17/2022, https://www.uptodate.com/contents/overview-of-the-management-of-chronic-kidney-disease-in-adults
- Mende CW. Chronic Kidney Disease and SGLT2 Inhibitors: A Review of the Evolving Treatment Landscape. Adv Ther. Jan 2022;39(1):148-164. doi:10.1007/s12325-021-01994-2. https://www.ncbi.nlm.nih.gov/pubmed/34846711
- National Institute of Diabetes and Digestive and Kidney Diseases. Chronic Kidney Disease (CKD). National Institutes of Health. Accessed 10/25/2022, https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kidney-disease-ckd/what-is-chronic-kidney-disease
- Kumar R, Tebben PJ, Thompson JR. Vitamin D and the kidney. Arch Biochem Biophys . Jul 1 2012;523(1):77-86. doi:10.1016/j.abb.2012.03.003. https://www.ncbi.nlm.nih.gov/pubmed/22426203
- Cortinovis M, Perico N, Ruggenenti P, Remuzzi A, Remuzzi G. Glomerular hyperfiltration. Nature Reviews Nephrology. 2022/07/01 2022;18(7):435-451. doi:10.1038/s41581-022-00559-y. https://doi.org/10.1038/s41581-022-00559-y https://www.nature.com/articles/s41581-022-00559-y
- Alicic RZ, Neumiller JJ, Galindo RJ, Tuttle KR. Use of Glucose-Lowering Agents in Diabetes and CKD. Kidney Int Rep. Dec 2022;7(12):2589-2607. doi:10.1016/j.ekir.2022.09.018. https://www.ncbi.nlm.nih.gov/pubmed/36506243 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9727535/pdf/main.pdf
- Thomas MC, Brownlee M, Susztak K, et al. Diabetic kidney disease. Nature Reviews Disease Primers . 2015/07/30 2015;1(1):15018. doi:10.1038/nrdp.2015.18. https://doi.org/10.1038/nrdp.2015.18 https://www.nature.com/articles/nrdp201518.pdf
- Rabbani N, Thornalley PJ. Advanced glycation end products in the pathogenesis of chronic kidney disease. Kidney Int. Apr 2018;93(4):803-813. doi:10.1016/j.kint.2017.11.034. https://www.ncbi.nlm.nih.gov/pubmed/29477239 https://www.kidney-international.org/article/S0085-2538(18)30051-6/pdf
- Varghese RT, Jialal I. Diabetic Nephropathy. StatPearls. StatPearls Publishing Copyright © 2023, StatPearls Publishing LLC.; 2023.
- O'Brien F. Focal Segmental Glomerulosclerosis. Merck Manual Professional Version. Updated 9/2022. Accessed 6/14/2023, https://www.merckmanuals.com/professional/genitourinary-disorders/glomerular-disorders/focal-segmental-glomerulosclerosis
- Griffin KA. Hypertensive Kidney Injury and the Progression of Chronic Kidney Disease. Hypertension. Oct 2017;70(4):687-694. doi:10.1161/hypertensionaha.117.08314.
- Ku E, Lee BJ, Wei J, Weir MR. Hypertension in CKD: Core Curriculum 2019. American Journal of Kidney Diseases. 2019;74(1):120-131. doi:10.1053/j.ajkd.2018.12.044. https://doi.org/10.1053/j.ajkd.2018.12.044
- Pugh D, Gallacher PJ, Dhaun N. Management of Hypertension in Chronic Kidney Disease. Drugs. Mar 2019;79(4):365-379. doi:10.1007/s40265-019-1064-1. https://www.ncbi.nlm.nih.gov/pubmed/30758803
- Arabi T, Shafqat A, Sabbah BN, et al. Obesity-related kidney disease: Beyond hypertension and insulin-resistance. Front Endocrinol (Lausanne) . 2022;13:1095211. doi:10.3389/fendo.2022.1095211. https://www.ncbi.nlm.nih.gov/pubmed/36726470
- Barreto Silva MI, Picard K, Klein M. Sarcopenia and sarcopenic obesity in chronic kidney disease: update on prevalence, outcomes, risk factors and nutrition treatment. Curr Opin Clin Nutr Metab Care. Nov 1 2022;25(6):371-377. doi:10.1097/MCO.0000000000000871. https://www.ncbi.nlm.nih.gov/pubmed/36039925
- Lin TY, Lim PS, Hung SC. Impact of Misclassification of Obesity by Body Mass Index on Mortality in Patients With CKD. Kidney Int Rep. Mar 2018;3(2):447-455. doi:10.1016/j.ekir.2017.12.009. https://www.ncbi.nlm.nih.gov/pubmed/29725649
- Theofilis P, Vordoni A, Kalaitzidis RG. Interplay between metabolic dysfunction-associated fatty liver disease and chronic kidney disease: Epidemiology, pathophysiologic mechanisms, and treatment considerations. World J Gastroenterol. Oct 21 2022;28(39):5691-5706. doi:10.3748/wjg.v28.i39.5691. https://www.ncbi.nlm.nih.gov/pubmed/36338895 https://f6publishing.blob.core.windows.net/ccfc92d8-d29c-43a0-bdbc-cde80838e108/WJG-28-5691.pdf
- Tao Z, Li Y, Cheng B, Zhou T, Gao Y. Influence of Nonalcoholic Fatty Liver Disease on the Occurrence and Severity of Chronic Kidney Disease. J Clin Transl Hepatol . Feb 28 2022;10(1):164-173. doi:10.14218/JCTH.2021.00171. https://www.ncbi.nlm.nih.gov/pubmed/35233386
- Lonardo A, Mantovani A, Targher G, Baffy G. Nonalcoholic Fatty Liver Disease and Chronic Kidney Disease: Epidemiology, Pathogenesis, and Clinical and Research Implications. Int J Mol Sci. Nov 1 2022;23(21)doi:10.3390/ijms232113320. https://www.ncbi.nlm.nih.gov/pubmed/36362108
- Cianciolo G, De Pascalis A, Di Lullo L, Ronco C, Zannini C, La Manna G. Folic Acid and Homocysteine in Chronic Kidney Disease and Cardiovascular Disease Progression: Which Comes First. Cardiorenal Medicine. 2017;7(4):255-266. doi:10.1159/000471813. https://doi.org/10.1159/000471813
- Xiong Y, Zhang Y, Zhang F, et al. Genetic Evidence Supporting the Causal Role of Homocysteine in Chronic Kidney Disease: A Mendelian Randomization Study. Front Nutr. 2022;9:843534. doi:10.3389/fnut.2022.843534. https://www.ncbi.nlm.nih.gov/pubmed/35495913
- Long Y, Nie J. Homocysteine in Renal Injury. Kidney Dis (Basel). Jun 2016;2(2):80-7. doi:10.1159/000444900.
- Badri S, Vahdat S, Seirafian S, Pourfarzam M, Gholipur-Shahraki T, Ataei S. Homocysteine-Lowering Interventions in Chronic Kidney Disease. Journal of research in pharmacy practice . Jul-Sep 2021;10(3):114-124. doi:10.4103/jrpp.jrpp_75_21. https://www.ncbi.nlm.nih.gov/pubmed/35198504
- National Institute of Diabetes and Digestive and Kidney Diseases. Causes of Chronic Kidney Disease. Available at https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kidney-disease-ckd/causes Last update 10/2016. Accessed 03/15/2023. 2016;
- National Kidney Foundation. Gout and Kidney Disease. Accessed 12/15/2023, https://www.kidney.org/atoz/content/gout/gout-kidney-disease
- American Kidney Fund. Gout and kidney disease. Updated 11/13/2023. Accessed 12/15/2023, https://www.kidneyfund.org/living-kidney-disease/health-problems-caused-kidney-disease/gout/gout-and-kidney-disease
- Dobrek L. A Synopsis of Current Theories on Drug-Induced Nephrotoxicity. Life (Basel). Jan 24 2023;13(2)doi:10.3390/life13020325. https://www.ncbi.nlm.nih.gov/pubmed/36836682
- Lucas GNC, Leitao ACC, Alencar RL, Xavier RMF, Daher EF, Silva Junior GBD. Pathophysiological aspects of nephropathy caused by non-steroidal anti-inflammatory drugs. J Bras Nefrol. Jan-Mar 2019;41(1):124-130. doi:10.1590/2175-8239-JBN-2018-0107. https://www.ncbi.nlm.nih.gov/pubmed/30281062
- Wan EYF, Yu EYT, Chan L, et al. Comparative Risks of Nonsteroidal Anti-Inflammatory Drugs on CKD. Clinical journal of the American Society of Nephrology : CJASN . Jun 2021;16(6):898-907. doi:10.2215/CJN.18501120. https://www.ncbi.nlm.nih.gov/pubmed/33910887
- Al-Aly Z, Maddukuri G, Xie Y. Proton Pump Inhibitors and the Kidney: Implications of Current Evidence for Clinical Practice and When and How to Deprescribe. American journal of kidney diseases : the official journal of the National Kidney Foundation . Apr 2020;75(4):497-507. doi:10.1053/j.ajkd.2019.07.012. https://www.ncbi.nlm.nih.gov/pubmed/31606235
- Lazarus B, Chen Y, Wilson FP, et al. Proton Pump Inhibitor Use and the Risk of Chronic Kidney Disease. JAMA Intern Med. Feb 2016;176(2):238-46. doi:10.1001/jamainternmed.2015.7193. https://www.ncbi.nlm.nih.gov/pubmed/26752337
- Wu B, Li D, Xu T, Luo M, He Z, Li Y. Proton pump inhibitors associated acute kidney injury and chronic kidney disease: data mining of US FDA adverse event reporting system. Sci Rep. Feb 11 2021;11(1):3690. doi:10.1038/s41598-021-83099-y. https://www.ncbi.nlm.nih.gov/pubmed/33574396
- Zhang XY, He QS, Jing Z, He JX, Yuan JQ, Dai XY. Effect of proton pump inhibitors on the risk of chronic kidney disease: A propensity score-based overlap weight analysis using the United Kingdom Biobank. Front Pharmacol . 2022;13:949699. doi:10.3389/fphar.2022.949699. https://www.ncbi.nlm.nih.gov/pubmed/36438798 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9685407/pdf/fphar-13-949699.pdf
- Nochaiwong S, Ruengorn C, Awiphan R, et al. The association between proton pump inhibitor use and the risk of adverse kidney outcomes: a systematic review and meta-analysis. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Feb 1 2018;33(2):331-342. doi:10.1093/ndt/gfw470. https://www.ncbi.nlm.nih.gov/pubmed/28339835
- Sun J, Sun H, Cui M, et al. The use of anti-ulcer agents and the risk of chronic kidney disease: a meta-analysis. International urology and nephrology . Oct 2018;50(10):1835-1843. doi:10.1007/s11255-018-1908-8. https://www.ncbi.nlm.nih.gov/pubmed/29948864
- Wu CC, Liao MH, Kung WM, Wang YC. Proton Pump Inhibitors and Risk of Chronic Kidney Disease: Evidence from Observational Studies. J Clin Med . Mar 15 2023;12(6)doi:10.3390/jcm12062262. https://www.ncbi.nlm.nih.gov/pubmed/36983271
- De Broe ME. Epidemiology and pathogenesis of analgesic-related chronic kidney disease. UpToDate. Updated 10/13/2022. Accessed 6/14/2023, https://www.uptodate.com/contents/epidemiology-and-pathogenesis-of-analgesic-related-chronic-kidney-disease
- Park WY. Controversies in acetaminophen nephrotoxicity. Kidney Res Clin Pract . Mar 31 2020;39(1):4-6. doi:10.23876/j.krcp.20.027. https://www.ncbi.nlm.nih.gov/pubmed/32176973
- Kelly DM, Rothwell PM. Disentangling the Relationship Between Chronic Kidney Disease and Cognitive Disorders. Front Neurol. 2022;13:830064. doi:10.3389/fneur.2022.830064. https://www.ncbi.nlm.nih.gov/pubmed/35280286 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8914950/pdf/fneur-13-830064.pdf
- Vaidya SR, Aeddula NR. Chronic Renal Failure. StatPearls. 2023.
- American Kidney Fund. Stages of kidney disease. Accessed 10/25/2022, https://www.kidneyfund.org/all-about-kidneys/stages-kidney-disease
- American Kidney Fund. Stage 1 of chronic kidney disease CKD: Causes, symptoms and treatment. Accessed 6/14/2023, https://www.kidneyfund.org/all-about-kidneys/stages-kidney-disease/stage-1-chronic-kidney-disease
- Vanholder R. Uremic toxins. UpToDate. Updated 11/29/2021. Accessed 6/14/2023, https://www.uptodate.com/contents/uremic-toxins
- KDIGO. CKD Evaluation and Management. KDIGO. Accessed Dec. 7, 2023, https://kdigo.org/guidelines/ckd-evaluation-and-management/
- National Kidney Foundation. Quick Reference Guide on Kidney Disease Screening. National Kidney Foundation. Accessed Jun. 28, 2023, https://www.kidney.org/kidneydisease/siemens_hcp_quickreference
- Levey AS, Coresh J, Balk E, et al. National Kidney Foundation practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Ann Intern Med. Jul 15 2003;139(2):137-47. doi:10.7326/0003-4819-139-2-200307150-00013.
- Mann J, L. BG. Antihypertensive therapy and progression of nondiabetic chronic kidney disease in adults. UpToDate. Updated 12/16/2022. Accessed 5/12/2023, https://www.uptodate.com/contents/antihypertensive-therapy-and-progression-of-nondiabetic-chronic-kidney-disease-in-adults#H594278
- Gounden V, Bhatt H, Jialal I. Renal Function Tests. StatPearls. StatPearls Publishing Copyright © 2022, StatPearls Publishing LLC.; 2023.
- Levey AS, Inker LA. Definition and staging of chronic kidney disease in adults. UpToDate. Updated 10/17/2022. Accessed 5/9/2023, https://www.uptodate.com/contents/definition-and-staging-of-chronic-kidney-disease-in-adults?topicRef=7172
- Chen DC, Potok OA, Rifkin D, Estrella MM. Advantages, Limitations, and Clinical Considerations in Using Cystatin C to Estimate GFR. Kidney360 . Oct 27 2022;3(10):1807-1814. doi:10.34067/KID.0003202022. https://www.ncbi.nlm.nih.gov/pubmed/36514729
- Coll E, Botey A, Alvarez L, et al. Serum cystatin C as a new marker for noninvasive estimation of glomerular filtration rate and as a marker for early renal impairment. American journal of kidney diseases : the official journal of the National Kidney Foundation . Jul 2000;36(1):29-34. doi:10.1053/ajkd.2000.8237. https://www.ncbi.nlm.nih.gov/pubmed/10873868
- Delgado C, Baweja M, Crews DC, et al. A Unifying Approach for GFR Estimation: Recommendations of the NKF-ASN Task Force on Reassessing the Inclusion of Race in Diagnosing Kidney Disease. American journal of kidney diseases : the official journal of the National Kidney Foundation . Feb 2022;79(2):268-288 e1. doi:10.1053/j.ajkd.2021.08.003. https://www.ncbi.nlm.nih.gov/pubmed/34563581
- National Kidney Foundation. Kidney Failure Risk Factor: Urine Albumin-Creatinine Ratio (uACR). Accessed 10/25/2022, https://www.kidney.org/content/kidney-failure-risk-factor-urine-albumin-to-creatinine-ration-uacr
- NKF. National Kidney Foundation. Tests to Measure Kidney Function, Damage, and Detect Abnormalities. Available at https://www.kidney.org/atoz/content/kidneytests Accessed 03/11/2023.
- Ren F, Li M, Xu H, Qin X, Teng Y. Urine albumin-to-creatinine ratio within the normal range and risk of hypertension in the general population: A meta-analysis. Journal of clinical hypertension (Greenwich, Conn). Jul 2021;23(7):1284-1290. doi:10.1111/jch.14263. https://www.ncbi.nlm.nih.gov/pubmed/34089300
- American Kidney Fund. Blood urea nitrogen (BUN) test. Updated 1/5/2022. Accessed 5/12/2023, https://www.kidneyfund.org/all-about-kidneys/tests/blood-urea-nitrogen-bun-test
- Bello AK, Alrukhaimi M, Ashuntantang GE, et al. Complications of chronic kidney disease: current state, knowledge gaps, and strategy for action. Kidney Int Suppl (2011). Oct 2017;7(2):122-129. doi:10.1016/j.kisu.2017.07.007. https://www.ncbi.nlm.nih.gov/pubmed/30675426
- Matsushita K, Ballew SH, Wang AY, Kalyesubula R, Schaeffner E, Agarwal R. Epidemiology and risk of cardiovascular disease in populations with chronic kidney disease. Nat Rev Nephrol. Nov 2022;18(11):696-707. doi:10.1038/s41581-022-00616-6. https://www.ncbi.nlm.nih.gov/pubmed/36104509
- Jankowski J, Floege J, Fliser D, Bohm M, Marx N. Cardiovascular Disease in Chronic Kidney Disease: Pathophysiological Insights and Therapeutic Options. Circulation. Mar 16 2021;143(11):1157-1172. doi:10.1161/CIRCULATIONAHA.120.050686. https://www.ncbi.nlm.nih.gov/pubmed/33720773
- Ocak G, Rookmaaker MB, Algra A, et al. Chronic kidney disease and bleeding risk in patients at high cardiovascular risk: a cohort study. J Thromb Haemost . Jan 2018;16(1):65-73. doi:10.1111/jth.13904. https://www.ncbi.nlm.nih.gov/pubmed/29125709
- Oe Y, Takahashi N. Tissue Factor, Thrombosis, and Chronic Kidney Disease. Biomedicines. Oct 28 2022;10(11)doi:10.3390/biomedicines10112737. https://www.ncbi.nlm.nih.gov/pubmed/36359257
- Gong S, Wang C, Xiong J, Zhao J, Yang K. Activated Platelets, the Booster of Chronic Kidney Disease and Cardiovascular Complications. Kidney Dis (Basel) . Jul 2022;8(4):297-307. doi:10.1159/000525090. https://www.ncbi.nlm.nih.gov/pubmed/36157264
- Baaten C, Schroer JR, Floege J, et al. Platelet Abnormalities in CKD and Their Implications for Antiplatelet Therapy. Clinical journal of the American Society of Nephrology : CJASN . Jan 2022;17(1):155-170. doi:10.2215/CJN.04100321. https://www.ncbi.nlm.nih.gov/pubmed/34750169
- Mahady SE, Polekhina G, Woods RL, et al. Association Between CKD and Major Hemorrhage in Older Persons: Data From the Aspirin in Reducing Events in the Elderly Randomized Trial. Kidney Int Rep. Apr 2023;8(4):737-745. doi:10.1016/j.ekir.2023.01.012. https://www.ncbi.nlm.nih.gov/pubmed/37069989
- National Institute of Diabetes and Digestive and Kidney Diseases. Anemia in Chronic Kidney Disease. Updated 9/2020. Accessed 6/14/2023, https://www.niddk.nih.gov/health-information/kidney-disease/anemia
- Adrogué HJ, Madias NE. Hyponatremia. New England Journal of Medicine . 2000;342(21):1581-1589. doi:10.1056/nejm200005253422107. https://www.nejm.org/doi/full/10.1056/NEJM200005253422107
- Adrogué HJ, Tucker BM, Madias NE. Diagnosis and Management of Hyponatremia: A Review. Jama. Jul 19 2022;328(3):280-291. doi:10.1001/jama.2022.11176. https://jamanetwork.com/journals/jama/article-abstract/2794358
- Arzhan S, Lew SQ, Ing TS, Tzamaloukas AH, Unruh ML. Dysnatremias in Chronic Kidney Disease: Pathophysiology, Manifestations, and Treatment. Front Med (Lausanne) . 2021;8:769287. doi:10.3389/fmed.2021.769287. https://www.ncbi.nlm.nih.gov/pubmed/34938749
- Combs S, Berl T. Dysnatremias in patients with kidney disease. American journal of kidney diseases : the official journal of the National Kidney Foundation . Feb 2014;63(2):294-303. doi:10.1053/j.ajkd.2013.09.017. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5325671/pdf/nihms607280.pdf
- Vincent-Johnson A, Scialla JJ. Importance of Metabolic Acidosis as a Health Risk in Chronic Kidney Disease. Adv Chronic Kidney Dis. Jul 2022;29(4):329-336. doi:10.1053/j.ackd.2022.05.002. https://www.ncbi.nlm.nih.gov/pubmed/36175070
- Qunibi WY. Overview of chronic kidney disease-mineral and bone disorder (CKD-MBD). UpToDate. Updated 6/22/2022. Accessed 6/14/2023, https://www.uptodate.com/contents/overview-of-chronic-kidney-disease-mineral-and-bone-disorder-ckd-mbd
- Viggiano D, Wagner CA, Martino G, et al. Mechanisms of cognitive dysfunction in CKD. Nat Rev Nephrol. Aug 2020;16(8):452-469. doi:10.1038/s41581-020-0266-9. https://www.ncbi.nlm.nih.gov/pubmed/32235904
- Noce A, Marchetti M, Marrone G, et al. Link between gut microbiota dysbiosis and chronic kidney disease. Eur Rev Med Pharmacol Sci. Mar 2022;26(6):2057-2074. doi:10.26355/eurrev_202203_28354. https://www.ncbi.nlm.nih.gov/pubmed/35363356
- Feng Z, Wang T, Dong S, et al. Association between gut dysbiosis and chronic kidney disease: a narrative review of the literature. J Int Med Res . Oct 2021;49(10):3000605211053276. doi:10.1177/03000605211053276. https://www.ncbi.nlm.nih.gov/pubmed/34704483
- Hernandez L, Ward LJ, Arefin S, et al. Blood-brain barrier and gut barrier dysfunction in chronic kidney disease with a focus on circulating biomarkers and tight junction proteins. Sci Rep. Mar 15 2022;12(1):4414. doi:10.1038/s41598-022-08387-7. https://www.ncbi.nlm.nih.gov/pubmed/35292710
- Syed-Ahmed M, Narayanan M. Immune Dysfunction and Risk of Infection in Chronic Kidney Disease. Adv Chronic Kidney Dis. Jan 2019;26(1):8-15. doi:10.1053/j.ackd.2019.01.004. https://www.ncbi.nlm.nih.gov/pubmed/30876622
- Crepin T, Legendre M, Courivaud C, et al. [Premature immune senescence and chronic kidney disease: Update and perspectives]. Nephrol Ther. Feb 2020;16(1):9-18. Maladie renale chronique et immunosenescence prematuree : donnees et perspectives. doi:10.1016/j.nephro.2019.04.005. https://www.ncbi.nlm.nih.gov/pubmed/31848067
- Lioulios G, Fylaktou A, Papagianni A, Stangou M. T cell markers recount the course of immunosenescence in healthy individuals and chronic kidney disease. Clinical immunology (Orlando, Fla). Apr 2021;225:108685. doi:10.1016/j.clim.2021.108685. https://www.ncbi.nlm.nih.gov/pubmed/33549833
- Stengel B. Chronic kidney disease and cancer: a troubling connection. Journal of nephrology . May-Jun 2010;23(3):253-62. https://www.ncbi.nlm.nih.gov/pubmed/20349418
- National Institute of Diabetes and Digestive and Kidney Diseases. Kidney Disease Statistics for the United States. National Institutes of Health. Updated 9/2021. Accessed 10/25/2022, https://www.niddk.nih.gov/health-information/health-statistics/kidney-disease
- Ortiz A, Mattace-Raso F, Soler MJ, Fouque D. Ageing meets kidney disease. Clinical Kidney Journal. 2022;15(10):1793-1796. doi:10.1093/ckj/sfac151. https://doi.org/10.1093/ckj/sfac151
- Stenvinkel P, Larsson TE. Chronic kidney disease: a clinical model of premature aging. American journal of kidney diseases : the official journal of the National Kidney Foundation . Aug 2013;62(2):339-51. doi:10.1053/j.ajkd.2012.11.051.
- Shiels P, Tran N, McCavitt J, Neytchev O, Stenvinkel P. Chronic Kidney Disease and the Exposome of Ageing. In: Harris JR, Korolchuk VI, eds. Biochemistry and Cell Biology of Ageing: Part IV, Clinical Science . Springer International Publishing; 2023:79-94.
- Ebert T, Pawelzik SC, Witasp A, et al. Inflammation and Premature Ageing in Chronic Kidney Disease. Toxins. Apr 4 2020;12(4)doi:10.3390/toxins12040227.
- Kooman JP, Kotanko P, Schols AMWJ, Shiels PG, Stenvinkel P. Chronic kidney disease and premature ageing. Nature Reviews Nephrology. 2014/12/01 2014;10(12):732-742. doi:10.1038/nrneph.2014.185. https://doi.org/10.1038/nrneph.2014.185
- Chen TK, Knicely DH, Grams ME. Chronic Kidney Disease Diagnosis and Management: A Review. JAMA. 2019;322(13):1294-1304. doi:10.1001/jama.2019.14745. https://doi.org/10.1001/jama.2019.14745 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7015670/pdf/nihms-1069237.pdf
- Vaduganathan M, Docherty KF, Claggett BL, et al. SGLT2 inhibitors in patients with heart failure: a comprehensive meta-analysis of five randomised controlled trials. The Lancet. 2022;400(10354):757-767. doi:10.1016/S0140-6736(22)01429-5. https://doi.org/10.1016/S0140-6736(22)01429-5
- Zheng H, Liu M, Li S, et al. Sodium-Glucose Co-Transporter-2 Inhibitors in Non-Diabetic Adults With Overweight or Obesity: A Systematic Review and Meta-Analysis. Systematic Review. Frontiers in Endocrinology. 2021-August-16 2021;12doi:10.3389/fendo.2021.706914. https://www.frontiersin.org/articles/10.3389/fendo.2021.706914
- National Kidney Foundation. Heart Failure and CKD: What You Need to Know. Accessed 6/14/2023, https://www.kidney.org/sites/default/files/Heart_Failure_and_CKD_2018.pdf
- Tuttle KR, Brosius FC, 3rd, Cavender MA, et al. SGLT2 Inhibition for CKD and Cardiovascular Disease in Type 2 Diabetes: Report of a Scientific Workshop Sponsored by the National Kidney Foundation. Diabetes. Jan 2021;70(1):1-16. doi:10.2337/dbi20-0040. https://www.ncbi.nlm.nih.gov/pubmed/33106255
- Layton AT, Vallon V. SGLT2 inhibition in a kidney with reduced nephron number: modeling and analysis of solute transport and metabolism. Am J Physiol Renal Physiol . May 1 2018;314(5):F969-F984. doi:10.1152/ajprenal.00551.2017. https://www.ncbi.nlm.nih.gov/pubmed/29361669
- Heerspink HJ, Desai M, Jardine M, Balis D, Meininger G, Perkovic V. Canagliflozin Slows Progression of Renal Function Decline Independently of Glycemic Effects. Journal of the American Society of Nephrology : JASN . Jan 2017;28(1):368-375. doi:10.1681/ASN.2016030278. https://www.ncbi.nlm.nih.gov/pubmed/27539604
- Fernandez-Fernandez B, Sarafidis P, Kanbay M, et al. SGLT2 inhibitors for non-diabetic kidney disease: drugs to treat CKD that also improve glycaemia. Clin Kidney J. Oct 2020;13(5):728-733. doi:10.1093/ckj/sfaa198. https://www.ncbi.nlm.nih.gov/pubmed/33123352
- Zelniker TA, Wiviott SD, Raz I, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. Jan 5 2019;393(10166):31-39. doi:10.1016/S0140-6736(18)32590-X. https://www.ncbi.nlm.nih.gov/pubmed/30424892
- Miyata KN, Zhang SL, Chan JSD. The Rationale and Evidence for SGLT2 Inhibitors as a Treatment for Nondiabetic Glomerular Disease. Glomerular Dis . Apr 2021;1(1):21-33. doi:10.1159/000513659. https://www.ncbi.nlm.nih.gov/pubmed/36751486
- Tian B, Deng Y, Cai Y, Han M, Xu G. Efficacy and safety of combination therapy with sodium-glucose cotransporter 2 inhibitors and renin-angiotensin system blockers in patients with type 2 diabetes: a systematic review and meta-analysis. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Mar 25 2022;37(4):720-729. doi:10.1093/ndt/gfab048. https://www.ncbi.nlm.nih.gov/pubmed/33605424
- Heerspink HJL, Stefansson BV, Correa-Rotter R, et al. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. Oct 8 2020;383(15):1436-1446. doi:10.1056/NEJMoa2024816. https://www.ncbi.nlm.nih.gov/pubmed/32970396 https://www.nejm.org/doi/pdf/10.1056/NEJMoa2024816?articleTools=true
- FDA. FDA Approves Treatment for Chronic Kidney Disease. Updated 04/30/2021. Accessed 11/10/2022, https://www.fda.gov/news-events/press-announcements/fda-approves-treatment-chronic-kidney-disease
- The E-KCG, Herrington WG, Staplin N, et al. Empagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. Jan 12 2023;388(2):117-127. doi:10.1056/NEJMoa2204233. https://www.ncbi.nlm.nih.gov/pubmed/36331190
- Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N Engl J Med. Jun 13 2019;380(24):2295-2306. doi:10.1056/NEJMoa1811744.
- Yi TW, Smyth B, Di Tanna GL, et al. Kidney and Cardiovascular Effects of Canagliflozin According to Age and Sex: A Post Hoc Analysis of the CREDENCE Randomized Clinical Trial. American journal of kidney diseases : the official journal of the National Kidney Foundation . Mar 7 2023;doi:10.1053/j.ajkd.2022.12.015. https://www.ncbi.nlm.nih.gov/pubmed/36889425
- Caffrey M. FDA Approves Canagliflozin to Prevent Kidney Failure, Hospitalization for Heart Failure. The American Journal of Managed Care. Updated 9/30/2019. Accessed 6/20/2023, https://www.ajmc.com/view/fda-approves-canagliflozin-to-prevent-kidney-failure-hospitalization-for-heart-failure
- Pelletier R, Ng K, Alkabbani W, Labib Y, Mourad N, Gamble J-M. Adverse events associated with sodium glucose co-transporter 2 inhibitors: an overview of quantitative systematic reviews. Therapeutic advances in drug safety . 2021;12:2042098621989134. doi:10.1177/2042098621989134. https://journals.sagepub.com/doi/abs/10.1177/2042098621989134
- Ramaswamy K. Side Effects of SGLT2 Inhibitors. Renal Fellow Network. Updated 11/10/2022. Accessed 6/20/2023, https://www.renalfellow.org/2022/11/10/side-effects-of-sglt2-inhibitors/
- Rigato M, Fadini GP, Avogaro A. Safety of sodium-glucose cotransporter 2 inhibitors in elderly patients with type 2 diabetes: A meta-analysis of randomized controlled trials. Diabetes, obesity & metabolism. Jul 4 2023;doi:10.1111/dom.15193. https://dom-pubs.pericles-prod.literatumonline.com/doi/10.1111/dom.15193
- Wang KM, Isom RT. SGLT2 Inhibitor-Induced Euglycemic Diabetic Ketoacidosis: A Case Report. Kidney Med. Mar-Apr 2020;2(2):218-221. doi:10.1016/j.xkme.2019.12.006. https://www.ncbi.nlm.nih.gov/pubmed/32734242
- Menghoum N, Oriot P, Hermans MP. Clinical and biochemical characteristics and analysis of risk factors for euglycaemic diabetic ketoacidosis in type 2 diabetic individuals treated with SGLT2 inhibitors: A review of 72 cases over a 4.5-year period. Diabetes Metab Syndr. Nov-Dec 2021;15(6):102275. doi:10.1016/j.dsx.2021.102275. https://www.ncbi.nlm.nih.gov/pubmed/34562870
- DeSantis A. Sodium-glucose cotransporter 2 inhibitors for the treatment of hyperglycemia in type 2 diabetes mellitus. UpToDate. Updated 6/1/2023. Accessed 6/20/2023, https://www.uptodate.com/contents/sodium-glucose-cotransporter-2-inhibitors-for-the-treatment-of-hyperglycemia-in-type-2-diabetes-mellitus
- Banerjee D, Winocour P, Chowdhury TA, et al. Management of hypertension and renin-angiotensin-aldosterone system blockade in adults with diabetic kidney disease: Association of British Clinical Diabetologists and the Renal Association UK guideline update 2021. BMC Nephrology. 2022/01/03 2022;23(1):9. doi:10.1186/s12882-021-02587-5. https://doi.org/10.1186/s12882-021-02587-5 https://bmcnephrol.biomedcentral.com/counter/pdf/10.1186/s12882-021-02587-5.pdf
- Panagiotis IG, Elias VB, Pantelis EZ. RAAS Blockade as First-Line Antihypertensive Therapy among People with CKD. In: Anna Naidenova T, ed. Renin-Angiotensin System. IntechOpen; 2017:Ch. 3.
- Weir MR, Lakkis JI, Jaar B, et al. Use of Renin-Angiotensin System Blockade in Advanced CKD: An NKF-KDOQI Controversies Report. American Journal of Kidney Diseases . 2018/12/01/ 2018;72(6):873-884. doi:https://doi.org/10.1053/j.ajkd.2018.06.010. https://www.sciencedirect.com/science/article/pii/S0272638618307960
- Gaudreault-Tremblay M-M, Foster BJ. Benefits of Continuing RAAS Inhibitors in Advanced CKD. Clinical Journal of the American Society of Nephrology . 2020;15(5):592-593. doi:10.2215/cjn.02920320. https://journals.lww.com/cjasn/Fulltext/2020/05000/Benefits_of_Continuing_RAAS_Inhibitors_in_Advanced.4.aspx
- Murakami T, Iwamoto T, Yasuda G, et al. Role of renin-angiotensin system inhibitors in retardation of progression of end-stage renal failure: a retrospective study. Clin Exp Nephrol. Aug 2016;20(4):603-610. doi:10.1007/s10157-015-1191-2. https://www.ncbi.nlm.nih.gov/pubmed/26519376
- Folkerts K, Millier A, Smela B, et al. Real-world evidence for steroidal mineralocorticoid receptor antagonists in patients with chronic kidney disease. Journal of nephrology. Nov 23 2022;doi:10.1007/s40620-022-01492-w. https://link.springer.com/content/pdf/10.1007/s40620-022-01492-w.pdf
- de Boer IH, Khunti K, Sadusky T, et al. Diabetes Management in Chronic Kidney Disease: A Consensus Report by the American Diabetes Association (ADA) and Kidney Disease: Improving Global Outcomes (KDIGO). Diabetes Care . 2022;45(12):3075-3090. doi:10.2337/dci22-0027. https://doi.org/10.2337/dci22-0027
- Shah PB, Soundararajan P, Sathiyasekaran BWC, Hegde SC. Diuretics for people with chronic kidney disease. Review. Cochrane Database Syst Rev . Oct Oct 26 2017;2017(10):CD011339. doi: 10.1002/14651858.CD011339.pub2. eCollection 2017 Oct.
- Szymczak A, Kusztal M, Krajewska M. Overhydration: A cause or an effect of kidney damage and how to treat it. Adv Clin Exp Med. Feb 2021;30(2):219-227. doi:10.17219/acem/132035. https://www.ncbi.nlm.nih.gov/pubmed/33636058
- Minutolo R, De Nicola L, Mallamaci F, Zoccali C. Thiazide diuretics are back in CKD: the case of chlorthalidone. Clin Kidney J. Jan 2023;16(1):41-51. doi:10.1093/ckj/sfac198. https://www.ncbi.nlm.nih.gov/pubmed/36726437 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9871852/pdf/sfac198.pdf
- Agarwal R, Sinha AD, Cramer AE, et al. Chlorthalidone for Hypertension in Advanced Chronic Kidney Disease. New England Journal of Medicine. 2021;385(27):2507-2519. doi:10.1056/NEJMoa2110730. https://www.nejm.org/doi/full/10.1056/NEJMoa2110730
- Guo L, Fu B, Liu Y, Hao N, Ji Y, Yang H. Diuretic resistance in patients with kidney disease: Challenges and opportunities. Biomed Pharmacother . Jan 2023;157:114058. doi:10.1016/j.biopha.2022.114058. https://www.sciencedirect.com/science/article/pii/S0753332222014470?via%3Dihub
- Schell JO, Arnold RM, Davison SN. Kidney pallative care: Conservative kidney management. UpToDate. Updated 1/30/2023. Accessed 6/20/2023, https://www.uptodate.com/contents/kidney-palliative-care-conservative-kidney-management
- National Institute of Diabetes and Digestive and Kidney Diseases. Hemodialysis. Accessed 05/18/23, https://www.niddk.nih.gov/health-information/kidney-disease/kidney-failure/hemodialysis
- National Institute of Diabetes and Digestive and Kidney Diseases. Peritoneal Dialysis. Accessed 05/18/23, https://www.niddk.nih.gov/health-information/kidney-disease/kidney-failure/peritoneal-dialysis
- National Institute of Diabetes and Digestive and Kidney Diseases. Kidney Transplant. Accessed 05/18/23, https://www.niddk.nih.gov/health-information/kidney-disease/kidney-failure/kidney-transplant
- Sadiq NM, Robinson KJ, Terrell JM. Colchicine. StatPearls. 2023.
- Roughley M, Sultan AA, Clarson L, et al. Risk of chronic kidney disease in patients with gout and the impact of urate lowering therapy: a population-based cohort study. Arthritis research & therapy. Oct 30 2018;20(1):243. doi:10.1186/s13075-018-1746-1. https://www.ncbi.nlm.nih.gov/pubmed/30376864
- Solak Y, Siriopol D, Yildiz A, et al. Colchicine in Renal Medicine: New Virtues of an Ancient Friend. Blood purification. 2017;43(1-3):125-135. doi:10.1159/000454669. https://www.ncbi.nlm.nih.gov/pubmed/27951538
- Kim HW, Joo YS, Yun HR, et al. Colchicine use and the risk of CKD progression: a multicentre nested case-control study. Rheumatology (Oxford) . Nov 2 2022;61(11):4314-4323. doi:10.1093/rheumatology/keac077. https://www.ncbi.nlm.nih.gov/pubmed/35139160
- Pisaniello HL, Fisher MC, Farquhar H, et al. Efficacy and safety of gout flare prophylaxis and therapy use in people with chronic kidney disease: a Gout, Hyperuricemia and Crystal-Associated Disease Network (G-CAN)-initiated literature review. Arthritis research & therapy. Apr 28 2021;23(1):130. doi:10.1186/s13075-021-02416-y. https://www.ncbi.nlm.nih.gov/pubmed/33910619
- Joshi S, Hashmi S, Shah S, Kalantar-Zadeh K. Plant-based diets for prevention and management of chronic kidney disease. Current opinion in nephrology and hypertension . Jan 2020;29(1):16-21. doi:10.1097/MNH.0000000000000574. https://www.ncbi.nlm.nih.gov/pubmed/31725014
- Carrero JJ, González-Ortiz A, Avesani CM, et al. Plant-based diets to manage the risks and complications of chronic kidney disease. Nat Rev Nephrol . Sep 2020;16(9):525-542. doi:10.1038/s41581-020-0297-2.
- Kalantar-Zadeh K, Joshi S, Schlueter R, et al. Plant-Dominant Low-Protein Diet for Conservative Management of Chronic Kidney Disease. Nutrients . Jun 29 2020;12(7)doi:10.3390/nu12071931.
- HARVARD T.H. CHAN. Diet Review: DASH. Harvard. Accessed 7/14/2021, https://www.hsph.harvard.edu/nutritionsource/healthy-weight/diet-reviews/dash-diet/
- Song Y, Lobene AJ, Wang Y, Hill Gallant KM. The DASH Diet and Cardiometabolic Health and Chronic Kidney Disease: A Narrative Review of the Evidence in East Asian Countries. Nutrients. Mar 18 2021;13(3)doi:10.3390/nu13030984. https://www.ncbi.nlm.nih.gov/pubmed/33803731
- MedlinePlus. DASH Eating Plan. National Library of Medicine. Updated 10/1/2021. Accessed 12/3/2021, https://medlineplus.gov/dasheatingplan.html
- Rebholz CM, Crews DC, Grams ME, et al. DASH (Dietary Approaches to Stop Hypertension) Diet and Risk of Subsequent Kidney Disease. American journal of kidney diseases : the official journal of the National Kidney Foundation . Dec 2016;68(6):853-861. doi:10.1053/j.ajkd.2016.05.019. https://www.ncbi.nlm.nih.gov/pubmed/27519166
- Banerjee T, Crews DC, Tuot DS, et al. Poor accordance to a DASH dietary pattern is associated with higher risk of ESRD among adults with moderate chronic kidney disease and hypertension. Kidney Int. Jun 2019;95(6):1433-1442. doi:10.1016/j.kint.2018.12.027. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6602537/pdf/nihms-1037886.pdf
- Hu EA, Coresh J, Anderson CAM, et al. Adherence to Healthy Dietary Patterns and Risk of CKD Progression and All-Cause Mortality: Findings From the CRIC (Chronic Renal Insufficiency Cohort) Study. American journal of kidney diseases : the official journal of the National Kidney Foundation . Feb 2021;77(2):235-244. doi:10.1053/j.ajkd.2020.04.019. https://www.ncbi.nlm.nih.gov/pubmed/32768632
- Bayan-Bravo A, Banegas JR, Donat-Vargas C, et al. The Mediterranean Diet Protects Renal Function in Older Adults: A Prospective Cohort Study. Nutrients . Jan 19 2022;14(3)doi:10.3390/nu14030432. https://www.ncbi.nlm.nih.gov/pubmed/35276791
- Podadera-Herreros A, Alcala-Diaz JF, Gutierrez-Mariscal FM, et al. Long-term consumption of a mediterranean diet or a low-fat diet on kidney function in coronary heart disease patients: The CORDIOPREV randomized controlled trial. Clin Nutr. Feb 2022;41(2):552-559. doi:10.1016/j.clnu.2021.12.041.
- Wang K, Qian D, Hu Y, Cheng Y, Ge S, Yao Y. Nut Consumption and Effects on Chronic Kidney Disease and Mortality in the United States. American journal of nephrology . 2022;53(6):503-512. doi:10.1159/000524382. https://www.ncbi.nlm.nih.gov/pubmed/35609522
- Sanchis P, Molina M, Berga F, et al. A Pilot Randomized Crossover Trial Assessing the Safety and Short-Term Effects of Walnut Consumption by Patients with Chronic Kidney Disease. Nutrients. Dec 25 2019;12(1)doi:10.3390/nu12010063. https://www.ncbi.nlm.nih.gov/pubmed/31881702
- Cardozo LF, Stockler-Pinto MB, Mafra D. Brazil nut consumption modulates Nrf2 expression in hemodialysis patients: A pilot study. Mol Nutr Food Res . Jul 2016;60(7):1719-24. doi:10.1002/mnfr.201500658. https://www.ncbi.nlm.nih.gov/pubmed/26992136
- Stockler-Pinto MB, Mafra D, Moraes C, et al. Brazil nut (Bertholletia excelsa, H.B.K.) improves oxidative stress and inflammation biomarkers in hemodialysis patients. Biol Trace Elem Res. Apr 2014;158(1):105-12. doi:10.1007/s12011-014-9904-z. https://www.ncbi.nlm.nih.gov/pubmed/24504745
- Lu L, Huang YF, Wang MQ, et al. Dietary fiber intake is associated with chronic kidney disease (CKD) progression and cardiovascular risk, but not protein nutritional status, in adults with CKD. Asia Pac J Clin Nutr . 2017;26(4):598-605. doi:10.6133/apjcn.072016.08. https://www.ncbi.nlm.nih.gov/pubmed/28582807
- Su G, Qin X, Yang C, et al. Fiber intake and health in people with chronic kidney disease. Clin Kidney J. Feb 2022;15(2):213-225. doi:10.1093/ckj/sfab169.
- Noce A, Marrone G, Urciuoli S, et al. Usefulness of Extra Virgin Olive Oil Minor Polar Compounds in the Management of Chronic Kidney Disease Patients. Nutrients. Feb 10 2021;13(2)doi:10.3390/nu13020581. https://mdpi-res.com/d_attachment/nutrients/nutrients-13-00581/article_deploy/nutrients-13-00581.pdf?version=1612950728
- Fan Z, Yun J, Yu S, Yang Q, Song L. Alcohol Consumption Can be a "Double-Edged Sword" for Chronic Kidney Disease Patients. Med Sci Monit . Sep 20 2019;25:7059-7072. doi:10.12659/MSM.916121. https://www.ncbi.nlm.nih.gov/pubmed/31538630
- Baker LA, March DS, Wilkinson TJ, et al. Clinical practice guideline exercise and lifestyle in chronic kidney disease. BMC Nephrol. Feb 22 2022;23(1):75. doi:10.1186/s12882-021-02618-1. https://www.ncbi.nlm.nih.gov/pubmed/35193515
- Alkhatib L, Velez Diaz LA, Varma S, et al. Lifestyle Modifications and Nutritional and Therapeutic Interventions in Delaying the Progression of Chronic Kidney Disease: A Review. Cureus. Feb 2023;15(2):e34572. doi:10.7759/cureus.34572. https://www.ncbi.nlm.nih.gov/pubmed/36874334
- Ikizler TA, Burrowes JD, Byham-Gray LD, et al. KDOQI Clinical Practice Guideline for Nutrition in CKD: 2020 Update. American journal of kidney diseases : the official journal of the National Kidney Foundation . Sep 2020;76(3 Suppl 1):S1-S107. doi:10.1053/j.ajkd.2020.05.006. https://www.ncbi.nlm.nih.gov/pubmed/32829751
- National Institute of Diabetes and Digestive and Kidney Diseases. Eating Right for Chronic Kidney DIsease. Available at https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kidney-disease-ckd/eating-nutrition Last updated 10/2016. Accessed 03/19/2023. 2016;
- Vegter S, Perna A, Postma MJ, Navis G, Remuzzi G, Ruggenenti P. Sodium intake, ACE inhibition, and progression to ESRD. Journal of the American Society of Nephrology : JASN . Jan 2012;23(1):165-73. doi:10.1681/ASN.2011040430. https://www.ncbi.nlm.nih.gov/pubmed/22135311
- Whitbread D. Top 10 Foods Highest in Sodium. Updated 5/29/2023. Accessed 6/20/2023, https://www.myfooddata.com/articles/what-foods-high-sodium.php#high-sodium-foods
- Ko GJ, Obi Y, Tortorici AR, Kalantar-Zadeh K. Dietary protein intake and chronic kidney disease. Curr Opin Clin Nutr Metab Care. Jan 2017;20(1):77-85. doi:10.1097/MCO.0000000000000342. https://www.ncbi.nlm.nih.gov/pubmed/27801685 https://escholarship.org/content/qt02q3f8n9/qt02q3f8n9.pdf?t=pzxozc
- Chewcharat A, Takkavatakarn K, Wongrattanagorn S, et al. The Effects of Restricted Protein Diet Supplemented With Ketoanalogue on Renal Function, Blood Pressure, Nutritional Status, and Chronic Kidney Disease-Mineral and Bone Disorder in Chronic Kidney Disease Patients: A Systematic Review and Meta-Analysis. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . May 2020;30(3):189-199. doi:10.1053/j.jrn.2019.07.005. https://www.ncbi.nlm.nih.gov/pubmed/31607548
- Li A, Lee HY, Lin YC. The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis. Nutrients. Apr 26 2019;11(5)doi:10.3390/nu11050957.
- Wu C-H, Yang Y-W, Hung S-C, et al. Ketoanalogues supplementation decreases dialysis and mortality risk in patients with anemic advanced chronic kidney disease. PLoS One. 2017;12(5):e0176847. doi:10.1371/journal.pone.0176847. https://doi.org/10.1371/journal.pone.0176847
- Bellizzi V, Garofalo C, Ferrara C, Calella P. Ketoanalogue Supplementation in Patients with Non-Dialysis Diabetic Kidney Disease: A Systematic Review and Meta-Analysis. Nutrients. Jan 19 2022;14(3):441. doi:10.3390/nu14030441. https://www.ncbi.nlm.nih.gov/pubmed/35276799
- Molina P, Gavela E, Vizcaino B, Huarte E, Carrero JJ. Optimizing Diet to Slow CKD Progression. Mini Review. Front Med (Lausanne). 2021-June-25 2021;8:654250. doi:10.3389/fmed.2021.654250. https://www.ncbi.nlm.nih.gov/pubmed/34249961
- Hamidianshirazi M, Shafiee M, Ekramzadeh M, Torabi Jahromi M, Nikaein F. Diet therapy along with nutrition education can improve renal function in people with stages 3-4 chronic kidney disease who do not have diabetes: a randomised controlled trial. Br J Nutr. Jul 7 2022:1-11. doi:10.1017/S0007114522002094. https://www.ncbi.nlm.nih.gov/pubmed/35795914
- Obeid W, Hiremath S, Topf JM. Protein Restriction for CKD: Time to Move On. Kidney360. Sep 29 2022;3(9):1611-1615. doi:10.34067/KID.0001002022. https://www.ncbi.nlm.nih.gov/pubmed/36245656
- Da J, Xie X, Wolf M, et al. Serum Phosphorus and Progression of CKD and Mortality: A Meta-analysis of Cohort Studies. American journal of kidney diseases : the official journal of the National Kidney Foundation . Aug 2015;66(2):258-65. doi:10.1053/j.ajkd.2015.01.009. https://www.ncbi.nlm.nih.gov/pubmed/25804679
- DuBose TD, Jr. Regulation of Potassium Homeostasis in CKD. Adv Chronic Kidney Dis . Sep 2017;24(5):305-314. doi:10.1053/j.ackd.2017.06.002. https://www.ncbi.nlm.nih.gov/pubmed/29031357
- Clegg DJ, Headley SA, Germain MJ. Impact of Dietary Potassium Restrictions in CKD on Clinical Outcomes: Benefits of a Plant-Based Diet. Kidney Med . Jul-Aug 2020;2(4):476-487. doi:10.1016/j.xkme.2020.04.007. https://www.ncbi.nlm.nih.gov/pubmed/32775988
- Hunter RW, Bailey MA. Hyperkalemia: pathophysiology, risk factors and consequences. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Dec 1 2019;34(Suppl 3):iii2-iii11. doi:10.1093/ndt/gfz206.
- Hill Gallant KM, Spiegel DM. Calcium Balance in Chronic Kidney Disease. Current osteoporosis reports. Jun 2017;15(3):214-221. doi:10.1007/s11914-017-0368-x. https://www.ncbi.nlm.nih.gov/pubmed/28474258
- Moe SM. Rationale to reduce calcium intake in adult patients with chronic kidney disease. Current opinion in nephrology and hypertension. Jul 2018;27(4):251-257. doi:10.1097/MNH.0000000000000416. https://www.ncbi.nlm.nih.gov/pubmed/29702490
- Qu X, Tong X, Hou X, Zhang J, Hou L, Chen J. Trends in Adherence to Recommended Physical Activity and Its Association with Mortality and Disease Progression among US Adults with Chronic Kidney Disease. American journal of nephrology . 2022;53(8-9):591-602. doi:10.1159/000526956. https://www.ncbi.nlm.nih.gov/pubmed/36349764
- Concha AT, Mendoza FAR. Sedentarism, A Modifiable Risk Factor for Developing Chronic Kidney Disease in Healthy People. Korean J Fam Med . Jan 2022;43(1):27-36. doi:10.4082/kjfm.20.0172. https://www.ncbi.nlm.nih.gov/pubmed/35130637
- Volaklis K, Mamadjanov T, Meisinger C. Sedentary behavior and kidney function in adults: a narrative review. Wiener klinische Wochenschrift . Feb 2021;133(3-4):144-152. doi:10.1007/s00508-020-01673-2. https://www.ncbi.nlm.nih.gov/pubmed/32435869
- MacKinnon HJ, Wilkinson TJ, Clarke AL, et al. The association of physical function and physical activity with all-cause mortality and adverse clinical outcomes in nondialysis chronic kidney disease: a systematic review. Therapeutic advances in chronic disease. Nov 2018;9(11):209-226. doi:10.1177/2040622318785575. https://www.ncbi.nlm.nih.gov/pubmed/30364521
- Clyne N, Anding-Rost K. Exercise training in chronic kidney disease-effects, expectations and adherence. Clin Kidney J. Apr 2021;14(Suppl 2):ii3-ii14. doi:10.1093/ckj/sfab012. https://www.ncbi.nlm.nih.gov/pubmed/33981415
- Schrauben SJ, Apple BJ, Chang AR. Modifiable Lifestyle Behaviors and CKD Progression: A Narrative Review. Kidney360. Apr 28 2022;3(4):752-778. doi:10.34067/KID.0003122021. https://www.ncbi.nlm.nih.gov/pubmed/35721622
- Baker LA, March DS, Wilkinson TJ, et al. Clinical practice guideline exercise and lifestyle in chronic kidney disease. BMC Nephrology. 2022/02/22 2022;23(1):75. doi:10.1186/s12882-021-02618-1. https://doi.org/10.1186/s12882-021-02618-1
- Zhang L, Wang Y, Xiong L, Luo Y, Huang Z, Yi B. Exercise therapy improves eGFR, and reduces blood pressure and BMI in non-dialysis CKD patients: evidence from a meta-analysis. BMC Nephrol. Oct 29 2019;20(1):398. doi:10.1186/s12882-019-1586-5. https://www.ncbi.nlm.nih.gov/pubmed/31664943
- Shlipak MG, Sheshadri A, Hsu FC, et al. Effect of Structured, Moderate Exercise on Kidney Function Decline in Sedentary Older Adults: An Ancillary Analysis of the LIFE Study Randomized Clinical Trial. JAMA Intern Med . Jun 1 2022;182(6):650-659. doi:10.1001/jamainternmed.2022.1449. https://www.ncbi.nlm.nih.gov/pubmed/35499834
- Nakamura K, Sasaki T, Yamamoto S, Hayashi H, Ako S, Tanaka Y. Effects of exercise on kidney and physical function in patients with non-dialysis chronic kidney disease: a systematic review and meta-analysis. Sci Rep . Oct 23 2020;10(1):18195. doi:10.1038/s41598-020-75405-x. https://www.ncbi.nlm.nih.gov/pubmed/33097801
- Oliveira Coelho F, Andrade L. Smoking and Kidney Disease: Risk Factors, Challenges, and Preventive Strategies. Contributions to nephrology . 2021;199:179-187. doi:10.1159/000517749. https://www.ncbi.nlm.nih.gov/pubmed/34344004
- Formanek P, Salisbury-Afshar E, Afshar M. Helping Patients With ESRD and Earlier Stages of CKD to Quit Smoking. American journal of kidney diseases : the official journal of the National Kidney Foundation . Aug 2018;72(2):255-266. doi:10.1053/j.ajkd.2018.01.057. https://www.ncbi.nlm.nih.gov/pubmed/29661542
- Hill C, Guarner F, Reid G, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol . Aug 2014;11(8):506-14. doi:10.1038/nrgastro.2014.66. https://www.ncbi.nlm.nih.gov/pubmed/24912386
- Gibson GR, Hutkins R, Sanders ME, et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat Rev Gastroenterol Hepatol . Aug 2017;14(8):491-502. doi:10.1038/nrgastro.2017.75. https://www.ncbi.nlm.nih.gov/pubmed/28611480
- Plaza-Diaz J, Ruiz-Ojeda FJ, Gil-Campos M, Gil A. Mechanisms of Action of Probiotics. Adv Nutr. Jan 1 2019;10(suppl_1):S49-s66. doi:10.1093/advances/nmy063.
- Voroneanu L, Burlacu A, Brinza C, et al. Gut Microbiota in Chronic Kidney Disease: From Composition to Modulation towards Better Outcomes-A Systematic Review. J Clin Med. Mar 1 2023;12(5)doi:10.3390/jcm12051948. https://www.ncbi.nlm.nih.gov/pubmed/36902734
- Hobby GP, Karaduta O, Dusio GF, Singh M, Zybailov BL, Arthur JM. Chronic kidney disease and the gut microbiome. Am J Physiol Renal Physiol. Jun 1 2019;316(6):F1211-F1217. doi:10.1152/ajprenal.00298.2018. https://www.ncbi.nlm.nih.gov/pubmed/30864840
- Kooshki A, Akbarzadeh R, Amin B, Tofighiyan T, Foroumandi E. Synbiotic supplement for treatment of iron deficiency anaemia in haemodialysis patients: A randomized controlled trial. Nephrology (Carlton). Apr 2023;28(4):234-239. doi:10.1111/nep.14149. https://www.ncbi.nlm.nih.gov/pubmed/36745046
- Simeoni M, Citraro ML, Cerantonio A, et al. An open-label, randomized, placebo-controlled study on the effectiveness of a novel probiotics administration protocol (ProbiotiCKD) in patients with mild renal insufficiency (stage 3a of CKD). Eur J Nutr. Aug 2019;58(5):2145-2156. doi:10.1007/s00394-018-1785-z. https://www.ncbi.nlm.nih.gov/pubmed/30076458
- Lydia A, Indra TA, Rizka A, Abdullah M. The effects of synbiotics on indoxyl sulphate level, constipation, and quality of life associated with constipation in chronic haemodialysis patients: a randomized controlled trial. BMC Nephrol. Jul 22 2022;23(1):259. doi:10.1186/s12882-022-02890-9. https://www.ncbi.nlm.nih.gov/pubmed/35869437
- Liu J, Zhong J, Yang H, et al. Biotic Supplements in Patients With Chronic Kidney Disease: Meta-Analysis of Randomized Controlled Trials. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . Jan 2022;32(1):10-21. doi:10.1053/j.jrn.2021.08.005. https://www.ncbi.nlm.nih.gov/pubmed/34666930
- Zheng HJ, Guo J, Wang Q, et al. Probiotics, prebiotics, and synbiotics for the improvement of metabolic profiles in patients with chronic kidney disease: A systematic review and meta-analysis of randomized controlled trials. Crit Rev Food Sci Nutr. 2021;61(4):577-598. doi:10.1080/10408398.2020.1740645. https://www.ncbi.nlm.nih.gov/pubmed/32329633
- Liu T, Wang X, Li R, Zhang ZY, Fang J, Zhang X. Effects of Probiotic Preparations on Inflammatory Cytokines in Chronic Kidney Disease Patients: A Systematic Review and Meta-Analysis. Current pharmaceutical biotechnology . 2021;22(10):1338-1349. doi:10.2174/1389201021666201119124058. https://www.ncbi.nlm.nih.gov/pubmed/33213338
- Bakhtiary M, Morvaridzadeh M, Agah S, et al. Effect of Probiotic, Prebiotic, and Synbiotic Supplementation on Cardiometabolic and Oxidative Stress Parameters in Patients With Chronic Kidney Disease: A Systematic Review and Meta-analysis. Clin Ther. Mar 2021;43(3):e71-e96. doi:10.1016/j.clinthera.2020.12.021. https://www.ncbi.nlm.nih.gov/pubmed/33526314
- Pavan M. Influence of prebiotic and probiotic supplementation on the progression of chronic kidney disease. Minerva urologica e nefrologica = The Italian journal of urology and nephrology . Apr 2016;68(2):222-6. https://www.ncbi.nlm.nih.gov/pubmed/24990390
- De Mauri A, Carrera D, Bagnati M, et al. Probiotics-Supplemented Low-Protein Diet for Microbiota Modulation in Patients with Advanced Chronic Kidney Disease (ProLowCKD): Results from a Placebo-Controlled Randomized Trial. Nutrients. Apr 14 2022;14(8)doi:10.3390/nu14081637. https://www.ncbi.nlm.nih.gov/pubmed/35458199
- Ong KL, Marklund M, Huang L, et al. Association of omega 3 polyunsaturated fatty acids with incident chronic kidney disease: pooled analysis of 19 cohorts. BMJ. Jan 18 2023;380:e072909. doi:10.1136/bmj-2022-072909. https://www.ncbi.nlm.nih.gov/pubmed/36653033
- Fazelian S, Moradi F, Agah S, et al. Effect of omega-3 fatty acids supplementation on cardio-metabolic and oxidative stress parameters in patients with chronic kidney disease: a systematic review and meta-analysis. BMC Nephrol. May 1 2021;22(1):160. doi:10.1186/s12882-021-02351-9. https://www.ncbi.nlm.nih.gov/pubmed/33933009
- de Boer IH, Zelnick LR, Ruzinski J, et al. Effect of Vitamin D and Omega-3 Fatty Acid Supplementation on Kidney Function in Patients With Type 2 Diabetes: A Randomized Clinical Trial. JAMA. Nov 19 2019;322(19):1899-1909. doi:10.1001/jama.2019.17380. https://www.ncbi.nlm.nih.gov/pubmed/31703120
- Bunout D, Barrera G, Hirsch S, Lorca E. A Randomized, Double-Blind, Placebo-Controlled Clinical Trial of an Omega-3 Fatty Acid Supplement in Patients With Predialysis Chronic Kidney Disease. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . Jan 2021;31(1):64-72. doi:10.1053/j.jrn.2020.04.004. https://www.ncbi.nlm.nih.gov/pubmed/32732154
- Saglimbene VM, Wong G, van Zwieten A, et al. Effects of omega-3 polyunsaturated fatty acid intake in patients with chronic kidney disease: Systematic review and meta-analysis of randomized controlled trials. Clin Nutr . Feb 2020;39(2):358-368. doi:10.1016/j.clnu.2019.02.041. https://www.ncbi.nlm.nih.gov/pubmed/30905498
- Gazdíková K, Gvozdjáková A, Kucharská J, Spustová V, Braunová Z, Dzúrik R. Oxidative Stress and Plasma Concentrations of Coenzyme Q10, α-Tocopherol, and β-Carotene in Patients with a Mild to Moderate Decrease of Kidney Function. Nephron. 2001;88(3):285-285. doi:10.1159/000046007. https://doi.org/10.1159/000046007 https://karger.com/nef/article-abstract/88/3/285/219697/Oxidative-Stress-and-Plasma-Concentrations-of?redirectedFrom=fulltext
- Mehmetoglu I, Yerlikaya FH, Kurban S, Erdem SS, Tonbul Z. Oxidative stress markers in hemodialysis and peritoneal dialysis patients, including coenzyme Q10 and ischemia-modified albumin. Int J Artif Organs. Mar 2012;35(3):226-32. doi:10.5301/ijao.5000078.
- Xu Y, Yang G, Zuo X, et al. A systematic review for the efficacy of coenzyme Q10 in patients with chronic kidney disease. International urology and nephrology . Jan 2022;54(1):173-184. doi:10.1007/s11255-021-02838-2. https://www.ncbi.nlm.nih.gov/pubmed/33782820
- Rivara MB, Yeung CK, Robinson-Cohen C, et al. Effect of Coenzyme Q(10) on Biomarkers of Oxidative Stress and Cardiac Function in Hemodialysis Patients: The CoQ(10) Biomarker Trial. American journal of kidney diseases : the official journal of the National Kidney Foundation . Mar 2017;69(3):389-399. doi:10.1053/j.ajkd.2016.08.041. https://www.ncbi.nlm.nih.gov/pubmed/27927588
- Gokbel H, Turk S, Okudan N, et al. Effects of Coenzyme Q10 Supplementation on Exercise Performance and Markers of Oxidative Stress in Hemodialysis Patients: A Double-Blind Placebo-Controlled Crossover Trial. Am J Ther . Nov/Dec 2016;23(6):e1736-e1743. doi:10.1097/MJT.0000000000000166. https://www.ncbi.nlm.nih.gov/pubmed/26844861
- Liu X, Baylin A, Levy PD. Vitamin D deficiency and insufficiency among US adults: prevalence, predictors and clinical implications. Br J Nutr . Apr 2018;119(8):928-936. doi:10.1017/s0007114518000491. https://www.cambridge.org/core/services/aop-cambridge-core/content/view/44E436843510FE6BDE856D5BCB9C651F/S0007114518000491a.pdf/div-class-title-vitamin-d-deficiency-and-insufficiency-among-us-adults-prevalence-predictors-and-clinical-implications-div.pdf
- Ravani P, Malberti F, Tripepi G, et al. Vitamin D levels and patient outcome in chronic kidney disease. Kidney Int. Jan 2009;75(1):88-95. doi:10.1038/ki.2008.501. https://www.ncbi.nlm.nih.gov/pubmed/18843258
- Pilz S, Iodice S, Zittermann A, Grant WB, Gandini S. Vitamin D status and mortality risk in CKD: a meta-analysis of prospective studies. American journal of kidney diseases : the official journal of the National Kidney Foundation . Sep 2011;58(3):374-82. doi:10.1053/j.ajkd.2011.03.020. https://www.ncbi.nlm.nih.gov/pubmed/21636193
- Dou D, Yang B, Gan H, Xie D, Lei H, Ye N. Vitamin D supplementation for the improvement of vascular function in patients with chronic kidney disease: a meta-analysis of randomized controlled trials. International urology and nephrology . May 2019;51(5):851-858. doi:10.1007/s11255-019-02088-3. https://www.ncbi.nlm.nih.gov/pubmed/30737643
- Kumar V, Yadav AK, Lal A, et al. A Randomized Trial of Vitamin D Supplementation on Vascular Function in CKD. Journal of the American Society of Nephrology : JASN . Oct 2017;28(10):3100-3108. doi:10.1681/ASN.2017010003. https://www.ncbi.nlm.nih.gov/pubmed/28667080
- Milajerdi A, Ostadmohammadi V, Amirjani S, Kolahdooz F, Asemi Z. The effects of vitamin D treatment on glycemic control, serum lipid profiles, and C-reactive protein in patients with chronic kidney disease: a systematic review and meta-analysis of randomized controlled trials. International urology and nephrology . Sep 2019;51(9):1567-1580. doi:10.1007/s11255-019-02236-9. https://www.ncbi.nlm.nih.gov/pubmed/31338797
- Urena Torres PA, Souberbielle JC, Solal MC. Bone Fragility in Chronic Kidney Disease Stage 3 to 5: The Use of Vitamin D Supplementation. Metabolites . Mar 20 2022;12(3)doi:10.3390/metabo12030266. https://www.ncbi.nlm.nih.gov/pubmed/35323709
- Khelifi N, Desbiens LC, Sidibe A, Mac-Way F. Vitamin D Analogues and Fracture Risk in Chronic Kidney Disease: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. JBMR Plus. Apr 2022;6(4):e10611. doi:10.1002/jbm4.10611. https://www.ncbi.nlm.nih.gov/pubmed/35434454
- Christodoulou M, Aspray TJ, Schoenmakers I. Vitamin D Supplementation for Patients with Chronic Kidney Disease: A Systematic Review and Meta-analyses of Trials Investigating the Response to Supplementation and an Overview of Guidelines. Calcified tissue international. Aug 2021;109(2):157-178. doi:10.1007/s00223-021-00844-1. https://www.ncbi.nlm.nih.gov/pubmed/33895867
- Lin YL, Hsu BG. Vitamin K and vascular calcification in chronic kidney disease: An update of current evidence. Tzu Chi Med J. Jan-Mar 2023;35(1):44-50. doi:10.4103/tcmj.tcmj_100_22. https://www.ncbi.nlm.nih.gov/pubmed/36866348
- Bellone F, Cinquegrani M, Nicotera R, et al. Role of Vitamin K in Chronic Kidney Disease: A Focus on Bone and Cardiovascular Health. Int J Mol Sci . May 9 2022;23(9)doi:10.3390/ijms23095282. https://www.ncbi.nlm.nih.gov/pubmed/35563672
- Mansour AG, Hariri E, Daaboul Y, et al. Vitamin K2 supplementation and arterial stiffness among renal transplant recipients-a single-arm, single-center clinical trial. J Am Soc Hypertens. Sep 2017;11(9):589-597. doi:10.1016/j.jash.2017.07.001. https://www.ncbi.nlm.nih.gov/pubmed/28756183
- Westenfeld R, Krueger T, Schlieper G, et al. Effect of vitamin K2 supplementation on functional vitamin K deficiency in hemodialysis patients: a randomized trial. American journal of kidney diseases : the official journal of the National Kidney Foundation . Feb 2012;59(2):186-95. doi:10.1053/j.ajkd.2011.10.041. https://www.ncbi.nlm.nih.gov/pubmed/22169620
- Fusaro M, Noale M, Viola V, et al. Vitamin K, vertebral fractures, vascular calcifications, and mortality: VItamin K Italian (VIKI) dialysis study. J Bone Miner Res. Nov 2012;27(11):2271-8. doi:10.1002/jbmr.1677. https://asbmr.onlinelibrary.wiley.com/doi/pdfdirect/10.1002/jbmr.1677?download=true
- Evenepoel P, Claes K, Meijers B, et al. Poor Vitamin K Status Is Associated With Low Bone Mineral Density and Increased Fracture Risk in End-Stage Renal Disease. J Bone Miner Res. Feb 2019;34(2):262-269. doi:10.1002/jbmr.3608. https://www.ncbi.nlm.nih.gov/pubmed/30427544
- Higdon J. Selenium. Oregon State University. Data on file.
- Chen CY, Chiu CH, Wu IW, et al. Micronutrients and Renal Outcomes: A Prospective Cohort Study. Nutrients. Jul 26 2022;14(15)doi:10.3390/nu14153063. https://www.ncbi.nlm.nih.gov/pubmed/35893916
- Zitouni K, Steyn M, Lyka E, et al. Derepression of glomerular filtration, renal blood flow and antioxidant defence in patients with type 2 diabetes at high-risk of cardiorenal disease. Free Radic Biol Med. Dec 2020;161:283-289. doi:10.1016/j.freeradbiomed.2020.10.005. https://www.ncbi.nlm.nih.gov/pubmed/33039650
- Alehagen U, Aaseth J, Alexander J, Brismar K, Larsson A. Selenium and Coenzyme Q10 Supplementation Improves Renal Function in Elderly Deficient in Selenium: Observational Results and Results from a Subgroup Analysis of a Prospective Randomised Double-Blind Placebo-Controlled Trial. Nutrients . Dec 9 2020;12(12)doi:10.3390/nu12123780. https://www.ncbi.nlm.nih.gov/pubmed/33317156
- Li J, Liu Z, Pu Y, Dai H, Peng F. Association between dietary vitamin E intake and chronic kidney disease events in US adults: a cross-sectional study from NHANES 2009–2016. Clinical Kidney Journal. 2023:sfad162. doi:10.1093/ckj/sfad162. https://doi.org/10.1093/ckj/sfad162
- Saldanha JF, Leal Vde O, Stenvinkel P, Carraro-Eduardo JC, Mafra D. Resveratrol: why is it a promising therapy for chronic kidney disease patients? Oxid Med Cell Longev. 2013;2013:963217. doi:10.1155/2013/963217. https://www.ncbi.nlm.nih.gov/pubmed/24489988 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3893857/pdf/OXIMED2013-963217.pdf
- Lin CT, Sun XY, Lin AX. Supplementation with high-dose trans-resveratrol improves ultrafiltration in peritoneal dialysis patients: a prospective, randomized, double-blind study. Renal failure. 2016;38(2):214-21. doi:10.3109/0886022X.2015.1128236. https://www.ncbi.nlm.nih.gov/pubmed/26727506
- Gimblet CJ, Kruse NT, Geasland K, et al. Effect of Resveratrol on Endothelial Function in Patients with CKD and Diabetes: A Randomized Controlled Trial. Clinical journal of the American Society of Nephrology : CJASN . Oct 16 2023;doi:10.2215/cjn.0000000000000337.
- Liu M, Yun P, Hu Y, Yang J, Khadka Rim B, Peng X. Effects of Grape Seed Proanthocyanidin Extract on Obesity. Obes Facts. 2020;13(2):279-291. doi:10.1159/000502235. https://doi.org/10.1159/000502235
- Turki K, Charradi K, Boukhalfa H, Belhaj M, Limam F, Aouani E. Grape seed powder improves renal failure of chronic kidney disease patients. EXCLI J . 2016;15:424-433. doi:10.17179/excli2016-363. https://www.ncbi.nlm.nih.gov/pubmed/27822171
- Bejaoui W, Mahmoudi M, Charradi K, et al. Preventive and healing effect of high dosing grape seed flour on CKD patients of various stages and aetiologies. Biomarkers. Dec 2022;27(8):795-801. doi:10.1080/1354750X.2022.2125580. https://www.ncbi.nlm.nih.gov/pubmed/36103212
- Higdon J. Zinc. Oregon State University. Data on file.
- Kim J, Lee J, Kim KN, et al. Association between Dietary Mineral Intake and Chronic Kidney Disease: The Health Examinees (HEXA) Study. Int J Environ Res Public Health . May 24 2018;15(6)doi:10.3390/ijerph15061070. https://www.ncbi.nlm.nih.gov/pubmed/29795052
- Fukasawa H, Furuya R, Kaneko M, et al. Clinical Significance of Trace Element Zinc in Patients with Chronic Kidney Disease. J Clin Med. Feb 20 2023;12(4)doi:10.3390/jcm12041667. https://www.ncbi.nlm.nih.gov/pubmed/36836202
- Abdollahi A, Ghahramani A, Ghahramani N. Zinc and Kidney Disease: A Review. Iran J Kidney Dis. Mar 2022;16(2):79-87. https://www.ncbi.nlm.nih.gov/pubmed/35489076
- Elgenidy A, Amin MA, Awad AK, Husain-Syed F, Aly MG. Serum Zinc Levels in Chronic Kidney Disease Patients, Hemodialysis Patients, and Healthy Controls: Systematic Review and Meta-Analysis. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . Jan 2023;33(1):103-115. doi:10.1053/j.jrn.2022.04.004. https://www.ncbi.nlm.nih.gov/pubmed/35472507
- Mohtashamian A, Soleimani A, Gilasi HR, Kheiripour N, Moeini Taba SM, Sharifi N. Association of Zinc Status with Matrix Metalloproteinases, Advanced Glycation End-Products, and Blood Pressure in Patients with Chronic Kidney Disease. Biol Trace Elem Res. Dec 14 2022;doi:10.1007/s12011-022-03524-9. https://www.ncbi.nlm.nih.gov/pubmed/36515817
- Jiang C, Ye H, Cui L, Pai P, Wang G. Relationship of serum copper and zinc with kidney function and urinary albumin to creatinine ratio: Cross-sectional data from the NHANES 2011-2016. Eur J Clin Nutr. Dec 2022;76(12):1748-1754. doi:10.1038/s41430-022-01181-8. https://www.ncbi.nlm.nih.gov/pubmed/35906329
- Damianaki K, Lourenco JM, Braconnier P, et al. Renal handling of zinc in chronic kidney disease patients and the role of circulating zinc levels in renal function decline. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Jul 1 2020;35(7):1163-1170. doi:10.1093/ndt/gfz065. https://www.ncbi.nlm.nih.gov/pubmed/31006015
- Tonelli M, Wiebe N, Thompson S, et al. Trace element supplementation in hemodialysis patients: a randomized controlled trial. BMC Nephrology . 2015/04/11 2015;16(1):52. doi:10.1186/s12882-015-0042-4. https://doi.org/10.1186/s12882-015-0042-4 https://bmcnephrol.biomedcentral.com/counter/pdf/10.1186/s12882-015-0042-4.pdf
- Sato E, Sato S, Degawa M, et al. Effects of Zinc Acetate Hydrate Supplementation on Renal Anemia with Hypozincemia in Hemodialysis Patients. Toxins. Oct 31 2022;14(11)doi:10.3390/toxins14110746. https://www.ncbi.nlm.nih.gov/pubmed/36355996
- Kobayashi H, Abe M, Okada K, et al. Oral zinc supplementation reduces the erythropoietin responsiveness index in patients on hemodialysis. Nutrients . May 15 2015;7(5):3783-95. doi:10.3390/nu7053783. https://www.ncbi.nlm.nih.gov/pubmed/25988769 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4446779/pdf/nutrients-07-03783.pdf
- Najafabadi MM, Faghihi G, Emami A, et al. Zinc sulfate for relief of pruritus in patients on maintenance hemodialysis. Therapeutic apheresis and dialysis : official peer-reviewed journal of the International Society for Apheresis, the Japanese Society for Apheresis, the Japanese Society for Dialysis Therapy . Apr 2012;16(2):142-5. doi:10.1111/j.1744-9987.2011.01032.x. https://www.ncbi.nlm.nih.gov/pubmed/22458392
- Guieu R, Ruf J, Mottola G. Hyperhomocysteinemia and cardiovascular diseases. Ann Biol Clin (Paris). Feb 1 2022;80(1):7-14. doi:10.1684/abc.2021.1694. https://www.ncbi.nlm.nih.gov/pubmed/35129442
- Capelli I, Cianciolo G, Gasperoni L, et al. Folic Acid and Vitamin B12 Administration in CKD, Why Not? Nutrients. Feb 13 2019;11(2)doi:10.3390/nu11020383. https://www.ncbi.nlm.nih.gov/pubmed/30781775
- Kaye AD, Jeha GM, Pham AD, et al. Folic Acid Supplementation in Patients with Elevated Homocysteine Levels. Adv Ther. Oct 2020;37(10):4149-4164. doi:10.1007/s12325-020-01474-z.
- Li Y, Huang T, Zheng Y, Muka T, Troup J, Hu FB. Folic Acid Supplementation and the Risk of Cardiovascular Diseases: A Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc. Aug 15 2016;5(8)doi:10.1161/JAHA.116.003768. https://www.ncbi.nlm.nih.gov/pubmed/27528407 https://www.ahajournals.org/doi/pdf/10.1161/JAHA.116.003768?download=true
- House AA, Eliasziw M, Cattran DC, et al. Effect of B-Vitamin Therapy on Progression of Diabetic Nephropathy: A Randomized Controlled Trial. JAMA . 2010;303(16):1603-1609. doi:10.1001/jama.2010.490. https://doi.org/10.1001/jama.2010.490 https://jamanetwork.com/journals/jama/articlepdf/185758/joc05040_1603_1609.pdf
- Jamison RL, Hartigan P, Kaufman JS, et al. Effect of Homocysteine Lowering on Mortality and Vascular Disease in Advanced Chronic Kidney Disease and End-stage Renal DiseaseA Randomized Controlled Trial. JAMA. 2007;298(10):1163-1170. doi:10.1001/jama.298.10.1163. https://doi.org/10.1001/jama.298.10.1163 https://jamanetwork.com/journals/jama/articlepdf/208754/joc70095_1163_1170.pdf
- Jamison RL, Hartigan P, Kaufman JS, et al. Effect of homocysteine lowering on mortality and vascular disease in advanced chronic kidney disease and end-stage renal disease: a randomized controlled trial. JAMA. Sep 12 2007;298(10):1163-70. doi:10.1001/jama.298.10.1163. https://www.ncbi.nlm.nih.gov/pubmed/17848650
- Xu X, Qin X, Li Y, et al. Efficacy of Folic Acid Therapy on the Progression of Chronic Kidney Disease: The Renal Substudy of the China Stroke Primary Prevention Trial. JAMA Intern Med. Oct 1 2016;176(10):1443-1450. doi:10.1001/jamainternmed.2016.4687.
- Wang S, Eide TC, Sogn EM, Berg KJ, Sund RB. Plasma ascorbic acid in patients undergoing chronic haemodialysis. Eur J Clin Pharmacol. Sep 1999;55(7):527-32. doi:10.1007/s002280050668. https://link.springer.com/article/10.1007/s002280050668
- Ke G, Huang J, Zhu Y, et al. Effect of Ascorbic Acid on Mineral and Bone Disorders in Hemodialysis Patients: a Systematic Review and Meta-Analysis. Kidney & blood pressure research. 2018;43(5):1459-1471. doi:10.1159/000493661. https://karger.com/kbr/article-pdf/43/5/1459/3051869/000493661.pdf
- Handelman GJ. Vitamin C deficiency in dialysis patients—are we perceiving the tip of an iceberg? Nephrology Dialysis Transplantation. 2007;22(2):328-331. doi:10.1093/ndt/gfl534. https://doi.org/10.1093/ndt/gfl534
- Kędzierska-Kapuza K, Szczuko U, Stolińska H, Bakaloudi DR, Wierzba W, Szczuko M. Demand for Water-Soluble Vitamins in a Group of Patients with CKD versus Interventions and Supplementation-A Systematic Review. Nutrients . Feb 8 2023;15(4)doi:10.3390/nu15040860. https://mdpi-res.com/d_attachment/nutrients/nutrients-15-00860/article_deploy/nutrients-15-00860.pdf?version=1675847457
- Juszczak AB, Kupczak M, Konecki T. Does Vitamin Supplementation Play a Role in Chronic Kidney Disease? Nutrients. Jun 23 2023;15(13)doi:10.3390/nu15132847. https://mdpi-res.com/d_attachment/nutrients/nutrients-15-02847/article_deploy/nutrients-15-02847.pdf?version=1687509217
- Chaghouri P, Maalouf N, Peters SL, et al. Two Faces of Vitamin C in Hemodialysis Patients: Relation to Oxidative Stress and Inflammation. Nutrients . Feb 27 2021;13(3)doi:10.3390/nu13030791. https://mdpi-res.com/d_attachment/nutrients/nutrients-13-00791/article_deploy/nutrients-13-00791-v2.pdf?version=1615452951
- Reiter RJ, Tan DX, Rosales-Corral S, Galano A, Zhou XJ, Xu B. Mitochondria: Central Organelles for Melatonin's Antioxidant and Anti-Aging Actions. Molecules. Feb 24 2018;23(2):509. doi:10.3390/molecules23020509. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6017324/
- Fatemeh G, Sajjad M, Niloufar R, Neda S, Leila S, Khadijeh M. Effect of melatonin supplementation on sleep quality: a systematic review and meta-analysis of randomized controlled trials. J Neurol. Jan 8 2021;doi:10.1007/s00415-020-10381-w. https://www.ncbi.nlm.nih.gov/pubmed/33417003
- Theofilis P, Vordoni A, Kalaitzidis RG. The Role of Melatonin in Chronic Kidney Disease and Its Associated Risk Factors: A New Tool in Our Arsenal? American journal of nephrology. 2022;53(7):565-574. doi:10.1159/000525441. https://www.ncbi.nlm.nih.gov/pubmed/35767942
- Movahhed SMM. Possible benefits of exogenous melatonin for individuals on dialysis: a narrative review on potential mechanisms and clinical implications. Naunyn-Schmiedeberg's archives of pharmacology. Aug 2021;394(8):1599-1611. doi:10.1007/s00210-021-02099-x. https://www.ncbi.nlm.nih.gov/pubmed/34097094
- Ohashi N, Ishigaki S, Isobe S. The pivotal role of melatonin in ameliorating chronic kidney disease by suppression of the renin-angiotensin system in the kidney. Hypertension research : official journal of the Japanese Society of Hypertension . Jun 2019;42(6):761-768. doi:10.1038/s41440-018-0186-2. https://www.ncbi.nlm.nih.gov/pubmed/30610209
- Ishigaki S, Ohashi N, Isobe S, et al. Impaired endogenous nighttime melatonin secretion relates to intrarenal renin-angiotensin system activation and renal damage in patients with chronic kidney disease. Clin Exp Nephrol . Dec 2016;20(6):878-884. doi:10.1007/s10157-015-1224-x. https://www.ncbi.nlm.nih.gov/pubmed/26743744
- Koch BC, van der Putten K, Van Someren EJ, et al. Impairment of endogenous melatonin rhythm is related to the degree of chronic kidney disease (CREAM study). Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Feb 2010;25(2):513-9. doi:10.1093/ndt/gfp493. https://www.ncbi.nlm.nih.gov/pubmed/19767630
- Maung SC, El Sara A, Chapman C, Cohen D, Cukor D. Sleep disorders and chronic kidney disease. World J Nephrol. May 6 2016;5(3):224-32. doi:10.5527/wjn.v5.i3.224. https://www.ncbi.nlm.nih.gov/pubmed/27152260
- Petrova A, Kondratiuk V, Karpenko O, Ostashevska T, Krasiuk E. The Effectiveness of Melatonin in the Complex Treatment of Hypertension in Patients with Stage 5 Chronic Kidney Disease. Georgian Med News. Feb 2020;(299):87-93. https://www.ncbi.nlm.nih.gov/pubmed/32242852
- Asghar MS, Ahsan MN, Jawed R, et al. A Comparative Study on the Use of Alprazolam and Melatonin for Sleep Disturbances in Hemodialysis Patients. Cureus. Nov 28 2020;12(11):e11754. doi:10.7759/cureus.11754. https://www.ncbi.nlm.nih.gov/pubmed/33403184
- Hameed EN, Hadi Al Tukmagi HF, Allami HCA. Melatonin Improves Erythropoietin Hyporesponsiveness via Suppression of Inflammation. Reviews on recent clinical trials . 2019;14(3):203-208. doi:10.2174/1574887114666190528120357. https://www.ncbi.nlm.nih.gov/pubmed/31132978
- Tripathi AK, Ray AK, Mishra SK, Bishen SM, Mishra H, Khurana A. Molecular and Therapeutic Insights of Alpha-Lipoic Acid as a Potential Molecule for Disease Prevention. Rev Bras Farmacogn. 2023;33(2):272-287. doi:10.1007/s43450-023-00370-1. https://www.ncbi.nlm.nih.gov/pubmed/36778891
- Lai S, Petramala L, Muscaritoli M, et al. alpha-lipoic acid in patients with autosomal dominant polycystic kidney disease. Nutrition. Mar 2020;71:110594. doi:10.1016/j.nut.2019.110594. https://www.ncbi.nlm.nih.gov/pubmed/31790890
- Khabbazi T, Mahdavi R, Safa J, Pour-Abdollahi P. Effects of alpha-lipoic acid supplementation on inflammation, oxidative stress, and serum lipid profile levels in patients with end-stage renal disease on hemodialysis. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . Mar 2012;22(2):244-250. doi:10.1053/j.jrn.2011.06.005. https://www.ncbi.nlm.nih.gov/pubmed/21908204
- Diniz FC, Hipkiss AR, Ferreira GC. The Potential Use of Carnosine in Diabetes and Other Afflictions Reported in Long COVID Patients. Front Neurosci . 2022;16:898735. doi:10.3389/fnins.2022.898735. https://www.ncbi.nlm.nih.gov/pubmed/35812220
- Calabrese V, Scuto M, Salinaro AT, et al. Hydrogen Sulfide and Carnosine: Modulation of Oxidative Stress and Inflammation in Kidney and Brain Axis. Antioxidants (Basel). Dec 18 2020;9(12)doi:10.3390/antiox9121303. https://www.ncbi.nlm.nih.gov/pubmed/33353117
- Aldini G, de Courten B, Regazzoni L, et al. Understanding the antioxidant and carbonyl sequestering activity of carnosine: direct and indirect mechanisms. Free Radic Res. Apr 2021;55(4):321-330. doi:10.1080/10715762.2020.1856830. https://www.ncbi.nlm.nih.gov/pubmed/33302741
- Kilis-Pstrusinska K. Carnosine and Kidney Diseases: What We Currently Know? Curr Med Chem. 2020;27(11):1764-1781. doi:10.2174/0929867326666190730130024. https://www.ncbi.nlm.nih.gov/pubmed/31362685
- Zhou Z, Liu XQ, Zhang SQ, et al. Correlation between serum carnosinase concentration and renal damage in diabetic nephropathy patients. Amino Acids . May 2021;53(5):687-700. doi:10.1007/s00726-021-02975-z. https://www.ncbi.nlm.nih.gov/pubmed/33811534
- Zhang S, Cui D, Tang M, et al. Serum and urinary carnosinase-1 correlate with kidney function and inflammation. Amino Acids. Jan 2023;55(1):89-100. doi:10.1007/s00726-022-03206-9. https://www.ncbi.nlm.nih.gov/pubmed/36319874
- Qiu J, Yard BA, Kramer BK, van Goor H, van Dijk P, Kannt A. Association Between Serum Carnosinase Concentration and Activity and Renal Function Impairment in a Type-2 Diabetes Cohort. Original Research. Front Pharmacol . 2022-July-08 2022;13:899057. doi:10.3389/fphar.2022.899057. https://www.ncbi.nlm.nih.gov/pubmed/35873562
- Siriwattanasit N, Satirapoj B, Supasyndh O. Effect of Oral carnosine supplementation on urinary TGF-beta in diabetic nephropathy: a randomized controlled trial. BMC Nephrol. Jun 26 2021;22(1):236. doi:10.1186/s12882-021-02434-7. https://www.ncbi.nlm.nih.gov/pubmed/34174842
- Everaert I, Taes Y, De Heer E, et al. Low plasma carnosinase activity promotes carnosinemia after carnosine ingestion in humans. Am J Physiol Renal Physiol . Jun 15 2012;302(12):F1537-44. doi:10.1152/ajprenal.00084.2012. https://www.ncbi.nlm.nih.gov/pubmed/22496410
- Sakaguchi Y. The emerging role of magnesium in CKD. Clin Exp Nephrol . May 2022;26(5):379-384. doi:10.1007/s10157-022-02182-4. https://www.ncbi.nlm.nih.gov/pubmed/35076791 https://link.springer.com/content/pdf/10.1007/s10157-022-02182-4.pdf
- Ding Z, Chen W, Zhang C, Wang H, Ma X. Correlation between Serum Magnesium Level and Cardiac Valve Calcification in Patients with Chronic Kidney Disease. Clin Lab. Apr 1 2022;68(4)doi:10.7754/Clin.Lab.2021.210533. https://www.ncbi.nlm.nih.gov/pubmed/35443599
- Tangvoraphonkchai K, Davenport A. Magnesium and Cardiovascular Disease. Adv Chronic Kidney Dis. May 2018;25(3):251-260. doi:10.1053/j.ackd.2018.02.010. https://www.ncbi.nlm.nih.gov/pubmed/29793664
- Zhang X, Li Y, Gobbo LCD, et al. Effects of Magnesium Supplementation on Blood Pressure. Hypertension. 2016;68(2):324-333. doi:doi:10.1161/HYPERTENSIONAHA.116.07664. https://www.ahajournals.org/doi/abs/10.1161/HYPERTENSIONAHA.116.07664
- Chen S, Jin X, Liu J, et al. Association of Plasma Magnesium with Prediabetes and Type 2 Diabetes Mellitus in Adults. Scientific Reports . 2017/10/06 2017;7(1):12763. doi:10.1038/s41598-017-13050-7. https://doi.org/10.1038/s41598-017-13050-7
- Ebrahimi Mousavi S, Ghoreishy SM, Hemmati A, Mohammadi H. Association between magnesium concentrations and prediabetes: a systematic review and meta-analysis. Scientific Reports. 2021/12/22 2021;11(1):24388. doi:10.1038/s41598-021-03915-3. https://doi.org/10.1038/s41598-021-03915-3
- Rondanelli M, Faliva MA, Tartara A, et al. An update on magnesium and bone health. Biometals : an international journal on the role of metal ions in biology, biochemistry, and medicine . Aug 2021;34(4):715-736. doi:10.1007/s10534-021-00305-0.
- Liu H, Wang R. Associations between the serum magnesium and all-cause or cardiovascular mortality in chronic kidney disease and end-stage renal disease patients: A meta-analysis. Medicine (Baltimore). Nov 12 2021;100(45):e27486. doi:10.1097/MD.0000000000027486. https://www.ncbi.nlm.nih.gov/pubmed/34766558 https://docserver.ingentaconnect.com/deliver/connect/wk/00257974/v100n45/s3.pdf?expires=1685998935&id=0000&titleid=99002871&checksum=B491A1D254456D2BB1D43C02D51BA023&host=https://www.ingentaconnect.com
- Leenders NHJ, Vermeulen EA, van Ballegooijen AJ, et al. The association between circulating magnesium and clinically relevant outcomes in patients with chronic kidney disease: A systematic review and meta-analysis. Clin Nutr . May 2021;40(5):3133-3147. doi:10.1016/j.clnu.2020.12.015. https://www.ncbi.nlm.nih.gov/pubmed/33419615
- Zhan Y, Zhang R, Li G. Effect of magnesium on vascular calcification in chronic kidney disease patients: a systematic review and meta-analysis. Renal failure. Dec 2023;45(1):2182603. doi:10.1080/0886022X.2023.2182603. https://www.ncbi.nlm.nih.gov/pubmed/36856310
- Sakaguchi Y, Hamano T, Obi Y, et al. A Randomized Trial of Magnesium Oxide and Oral Carbon Adsorbent for Coronary Artery Calcification in Predialysis CKD. Journal of the American Society of Nephrology : JASN. Jun 2019;30(6):1073-1085. doi:10.1681/ASN.2018111150. https://www.ncbi.nlm.nih.gov/pubmed/31036759
- Bressendorff I, Hansen D, Schou M, et al. The Effect of Magnesium Supplementation on Vascular Calcification in CKD: A Randomized Clinical Trial (MAGiCAL-CKD). Journal of the American Society of Nephrology : JASN . May 1 2023;34(5):886-894. doi:10.1681/ASN.0000000000000092. https://www.ncbi.nlm.nih.gov/pubmed/36749131
- Cowan AC, Clemens KK, Sontrop JM, et al. Magnesium and Fracture Risk in the General Population and Patients Receiving Dialysis: A Narrative Review. Canadian journal of kidney health and disease. 2023;10:20543581231154183. doi:10.1177/20543581231154183. https://www.ncbi.nlm.nih.gov/pubmed/36814964
- Guo G, Zhou J, Xu T, et al. Effect of Magnesium Supplementation on Chronic Kidney Disease-Mineral and Bone Disorder in Hemodialysis Patients: A Meta-Analysis of Randomized Controlled Trials. Journal of renal nutrition : the official journal of the Council on Renal Nutrition of the National Kidney Foundation . Jan 2022;32(1):102-111. doi:10.1053/j.jrn.2021.07.009. https://www.ncbi.nlm.nih.gov/pubmed/34531112
- CDC. Centers for Disease Control and Prevention. Chronic Kidney Disease Initiative. Chronic Kidney Disease in the United States, 2021. Updated 7/12/2022. Accessed 10/25/2022, https://www.cdc.gov/kidneydisease/publications-resources/ckd-national-facts.html
- Ishigami J, Matsushita K. Clinical epidemiology of infectious disease among patients with chronic kidney disease. Clin Exp Nephrol. Apr 2019;23(4):437-447. doi:10.1007/s10157-018-1641-8. https://www.ncbi.nlm.nih.gov/pubmed/30178234
- Lees JS, Elyan BMP, Herrmann SM, Lang NN, Jones RJ, Mark PB. The 'other' big complication: how chronic kidney disease impacts on cancer risks and outcomes. Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association . Jan 28 2022;doi:10.1093/ndt/gfac011. https://www.ncbi.nlm.nih.gov/pubmed/35090037
- Torra R, Furlano M, Ortiz A, Ars E. Genetic kidney diseases as an underrecognized cause of chronic kidney disease: the key role of international registry reports. Clin Kidney J. Aug 2021;14(8):1879-1885. doi:10.1093/ckj/sfab056. https://www.ncbi.nlm.nih.gov/pubmed/34345410
- Maranduca MA, Clim A, Pinzariu AC, et al. Role of arterial hypertension and angiotensin II in chronic kidney disease (Review). Exp Ther Med. Apr 2023;25(4):153. doi:10.3892/etm.2023.11852. https://www.ncbi.nlm.nih.gov/pubmed/36911375
- Neild GH. Life expectancy with chronic kidney disease: an educational review. Pediatr Nephrol. Feb 2017;32(2):243-248. doi:10.1007/s00467-016-3383-8. https://www.ncbi.nlm.nih.gov/pubmed/27115888
- Muzaale AD, Massie AB, Wang M-C, et al. Risk of End-Stage Renal Disease Following Live Kidney Donation. JAMA. 2014;311(6):579-586. doi:10.1001/jama.2013.285141. https://doi.org/10.1001/jama.2013.285141 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4411956/pdf/nihms676797.pdf
- Matas AJ, Rule AD. Long-term Medical Outcomes of Living Kidney Donors. Mayo Clin Proc . Nov 2022;97(11):2107-2122. doi:10.1016/j.mayocp.2022.06.013. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9747133/pdf/nihms-1856044.pdf
- Vock DM, Helgeson ES, Mullan AF, et al. The Minnesota attributable risk of kidney donation (MARKD) study: a retrospective cohort study of long-term (> 50 year) outcomes after kidney donation compared to well-matched healthy controls. BMC Nephrol. May 1 2023;24(1):121. doi:10.1186/s12882-023-03149-7. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10152793/pdf/12882_2023_Article_3149.pdf
- Clemens KK, Ernst J, Khan T, et al. Glucagon-like peptide 1 receptor agonists in end-staged kidney disease and kidney transplantation: A narrative review. Nutr Metab Cardiovasc Dis. Jun 2023;33(6):1111-1120. doi:10.1016/j.numecd.2023.03.023. https://www.ncbi.nlm.nih.gov/pubmed/37100640
- Greco EV, Russo G, Giandalia A, Viazzi F, Pontremoli R, De Cosmo S. GLP-1 Receptor Agonists and Kidney Protection. Medicina (Kaunas, Lithuania). May 31 2019;55(6)doi:10.3390/medicina55060233. https://www.ncbi.nlm.nih.gov/pubmed/31159279
- Winiarska A, Knysak M, Nabrdalik K, Gumprecht J, Stompor T. Inflammation and Oxidative Stress in Diabetic Kidney Disease: The Targets for SGLT2 Inhibitors and GLP-1 Receptor Agonists. International journal of molecular sciences. Oct 6 2021;22(19)doi:10.3390/ijms221910822. https://www.ncbi.nlm.nih.gov/pubmed/34639160
- Wang N, Zhang C. Oxidative Stress: A Culprit in the Progression of Diabetic Kidney Disease. Antioxidants (Basel, Switzerland). Apr 12 2024;13(4)doi:10.3390/antiox13040455. https://www.ncbi.nlm.nih.gov/pubmed/38671903
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. The New England journal of medicine. May 24 2024;doi:10.1056/NEJMoa2403347. https://www.ncbi.nlm.nih.gov/pubmed/38785209
- Tuttle KR, Lakshmanan MC, Rayner B, et al. Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate-to-severe chronic kidney disease (AWARD-7): a multicentre, open-label, randomised trial. The lancet Diabetes & endocrinology. Aug 2018;6(8):605-617. doi:10.1016/s2213-8587(18)30104-9. https://pubmed.ncbi.nlm.nih.gov/29910024/
- Kim S, An JN, Song YR, et al. Effect of once-weekly dulaglutide on renal function in patients with chronic kidney disease. PLoS One. 2022;17(8):e0273004. doi:10.1371/journal.pone.0273004. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9374206/pdf/pone.0273004.pdf
- Tsuchida KI, Taneda S, Yokota I, et al. The renoprotective effect of once-weekly GLP-1 receptor agonist dulaglutide on progression of nephropathy in Japanese patients with type 2 diabetes and moderate to severe chronic kidney disease (JDDM67). J Diabetes Investig. Nov 2022;13(11):1834-1841. doi:10.1111/jdi.13877. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9623516/pdf/JDI-13-1834.pdf
- Heerspink HJL, Sattar N, Pavo I, et al. Effects of tirzepatide versus insulin glargine on kidney outcomes in type 2 diabetes in the SURPASS-4 trial: post-hoc analysis of an open-label, randomised, phase 3 trial. The lancet Diabetes & endocrinology. Nov 2022;10(11):774-785. doi:10.1016/S2213-8587(22)00243-1. https://www.ncbi.nlm.nih.gov/pubmed/36152639
- Rivera FB, Cruz LLA, Magalong JV, et al. Cardiovascular and renal outcomes of glucagon-like peptide 1 receptor agonists among patients with and without type 2 diabetes mellitus: A meta-analysis of randomized placebo-controlled trials. Am J Prev Cardiol. Jun 2024;18:100679. doi:10.1016/j.ajpc.2024.100679. https://www.ncbi.nlm.nih.gov/pubmed/38779187
- DRUGBANK Online. Pentoxifylline. University of Alberta. Accessed 06/03/2024, https://go.drugbank.com/drugs/DB00806
- Donate-Correa J, Sanchez-Nino MD, Gonzalez-Luis A, et al. Repurposing drugs for highly prevalent diseases: pentoxifylline, an old drug and a new opportunity for diabetic kidney disease. Clin Kidney J. Dec 2022;15(12):2200-2213. doi:10.1093/ckj/sfac143. https://www.ncbi.nlm.nih.gov/pubmed/36381364
- Liu D, Wang LN, Li HX, Huang P, Qu LB, Chen FY. Pentoxifylline plus ACEIs/ARBs for proteinuria and kidney function in chronic kidney disease: a meta-analysis. The Journal of international medical research. Apr 2017;45(2):383-398. doi:10.1177/0300060516663094. https://www.ncbi.nlm.nih.gov/pubmed/28415944
- Wu PC, Wu CJ, Lin CJ, et al. Pentoxifylline Decreases Dialysis Risk in Patients With Advanced Chronic Kidney Disease. Clin Pharmacol Ther. Oct 2015;98(4):442-9. doi:10.1002/cpt.173. https://www.ncbi.nlm.nih.gov/pubmed/26082272
- de Morales AM, Goicoechea M, Verde E, et al. Pentoxifylline, progression of chronic kidney disease (CKD) and cardiovascular mortality: long-term follow-up of a randomized clinical trial. Journal of nephrology. Aug 2019;32(4):581-587. doi:10.1007/s40620-019-00607-0. https://www.ncbi.nlm.nih.gov/pubmed/30949987
- Goicoechea M, Garcia de Vinuesa S, Quiroga B, et al. Effects of pentoxifylline on inflammatory parameters in chronic kidney disease patients: a randomized trial. Journal of nephrology. Nov-Dec 2012;25(6):969-75. doi:10.5301/jn.5000077. https://www.ncbi.nlm.nih.gov/pubmed/22241639
- Chen YM, Chiang WC, Lin SL, Tsai TJ. Therapeutic efficacy of pentoxifylline on proteinuria and renal progression: an update. J Biomed Sci. Nov 13 2017;24(1):84. doi:10.1186/s12929-017-0390-4. https://pubmed.ncbi.nlm.nih.gov/29132351/
- Fang JH, Chen YC, Ho CH, et al. The risk of major bleeding event in patients with chronic kidney disease on pentoxifylline treatment. Sci Rep. Jun 29 2021;11(1):13521. doi:10.1038/s41598-021-92753-4. https://www.ncbi.nlm.nih.gov/pubmed/34188087
- Leehey DJ, Carlson K, Reda DJ, et al. Pentoxifylline in diabetic kidney disease (VA PTXRx): protocol for a pragmatic randomised controlled trial. BMJ open. Aug 16 2021;11(8):e053019. doi:10.1136/bmjopen-2021-053019. https://www.ncbi.nlm.nih.gov/pubmed/34400461
- Leehey DJ, Hines Jr., EH. Pentoxifylline in Diabetic Kidney Disease (PTXRx). ClinicalTrials.gov identifier: NCT03625648. National Library of Medicine. Updated 05/01/2024. Accessed 06/03/2024. https://clinicaltrials.gov/study/NCT03625648
- Prud'homme GJ, Kurt M, Wang Q. Pathobiology of the Klotho Antiaging Protein and Therapeutic Considerations. Front Aging. 2022;3:931331. doi:10.3389/fragi.2022.931331. https://www.ncbi.nlm.nih.gov/pubmed/35903083
- Abraham CR, Li A. Aging-suppressor Klotho: Prospects in diagnostics and therapeutics. Ageing Res Rev. Dec 2022;82:101766. doi:10.1016/j.arr.2022.101766. https://www.ncbi.nlm.nih.gov/pubmed/36283617
- Navarro-Gonzalez JF, Sanchez-Nino MD, Donate-Correa J, et al. Effects of Pentoxifylline on Soluble Klotho Concentrations and Renal Tubular Cell Expression in Diabetic Kidney Disease. Diabetes Care. Aug 2018;41(8):1817-1820. doi:10.2337/dc18-0078. https://www.ncbi.nlm.nih.gov/pubmed/29866645
- Sandhya T, Lathika KM, Pandey BN, et al. Protection against radiation oxidative damage in mice by Triphala. Mutat Res. Oct 10 2006;609(1):17-25. doi:10.1016/j.mrgentox.2006.05.006. https://pubmed.ncbi.nlm.nih.gov/16860592/
- Usharani P, Nutalapati C, Pokuri VK, Kumar CU, Taduri G. A randomized, double-blind, placebo-, and positive-controlled clinical pilot study to evaluate the efficacy and tolerability of standardized aqueous extracts of Terminalia chebula and Terminalia bellerica in subjects with hyperuricemia. Clinical pharmacology : advances and applications. 2016;8:51-9. doi:10.2147/CPAA.S100521. https://pubmed.ncbi.nlm.nih.gov/27382337/
- Pingali U, Nutalapati C, Koilagundla N, Taduri G. A randomized, double-blind, positive-controlled, prospective, dose-response clinical study to evaluate the efficacy and tolerability of an aqueous extract of Terminalia bellerica in lowering uric acid and creatinine levels in chronic kidney disease subjects with hyperuricemia. BMC Complement Med Ther. Sep 15 2020;20(1):281. doi:10.1186/s12906-020-03071-7. https://pubmed.ncbi.nlm.nih.gov/32933504/
- Liboriussen C, Nygaard L, Jensen JD, Schmidt EB, Glerup RI, Svensson MHS. Low Plasma Marine N-3 Polyunsaturated Fatty Acids are Associated With Increased Risk of Cardiovascular Events in Patients Treated With Maintenance Hemodialysis. Journal of renal nutrition: the official journal of the Council on Renal Nutrition of the National Kidney Foundation. Jul 2025;35(4):531-538. doi:10.1053/j.jrn.2025.02.001. https://www.ncbi.nlm.nih.gov/pubmed/40020764
- Lok CE, Farkouh M, Hemmelgarn BR, et al. Fish-Oil Supplementation and Cardiovascular Events in Patients Receiving Hemodialysis. The New England journal of medicine. Nov 7 2025;doi:10.1056/NEJMoa2513032. https://www.nejm.org/doi/10.1056/NEJMoa2513032
- Zeinalabedini M, Shapouri M, Mirzaee P, et al. The Effect of Omega-3 Supplements on Renal Function Indices in Chronic Kidney Patients Undergoing Hemodialysis. Nutr Metab Insights. 2025;18:11786388251345518. doi:10.1177/11786388251345518. https://pmc.ncbi.nlm.nih.gov/articles/PMC12206260/
- Lin YL, Wang CL, Chiang TI. Eicosapentaenoic acid supplementation alleviates pruritus, enhances skin moisture, and mitigates depression in maintenance hemodialysis patients. Front Nephrol. 2024;4:1365809. doi:10.3389/fneph.2024.1365809. https://pubmed.ncbi.nlm.nih.gov/39139799/
- Borbe JBC, Elvambuena BF, Eugenio FAD, Tan RJM. Effect of Omega-3 Fatty Acid Supplementation in the Treatment of Uremic Pruritus among Dialytic Chronic Kidney Disease Patients: A Meta-analysis. Acta Med Philipp. 2024;58(8):125-131. doi:10.47895/amp.vi0.7015. https://pubmed.ncbi.nlm.nih.gov/38812761/
- Fatima K, Mahmood A, Sayeed FZ, et al. Effectiveness of fish oil in controlling inflammation in adult patients undergoing hemodialysis: A systematic review and meta-analysis. SAGE open medicine. 2024;12:20503121241275467. doi:10.1177/20503121241275467. https://pubmed.ncbi.nlm.nih.gov/39286404/
- Kolkhof P, Bärfacker L. 30 YEARS OF THE MINERALOCORTICOID RECEPTOR: Mineralocorticoid receptor antagonists: 60 years of research and development. The Journal of endocrinology. Jul 2017;234(1):T125-t140. doi:10.1530/joe-16-0600. https://pubmed.ncbi.nlm.nih.gov/28634268/
- Wu JY, Tseng KJ, Lin YM. Nonsteroidal Versus Steroidal Mineralocorticoid Receptor Antagonists in Heart Failure: Real‐World Evidence From a Multicenter Cohort Study. Journal of the American Heart Association. 2025;14(21):e043373. doi:doi:10.1161/JAHA.125.043373. https://www.ahajournals.org/doi/abs/10.1161/JAHA.125.043373
- Group KCW. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney international. Apr 2024;105(4s):S117-s314. doi:10.1016/j.kint.2023.10.018. https://www.kidney-international.org/article/S0085-2538(23)00766-4/fulltext
- Agarwal R, Filippatos G, Pitt B, et al. Cardiovascular and kidney outcomes with finerenone in patients with type 2 diabetes and chronic kidney disease: the FIDELITY pooled analysis. Eur Heart J. Feb 10 2022;43(6):474-484. doi:10.1093/eurheartj/ehab777. https://pubmed.ncbi.nlm.nih.gov/35023547/
- Humle K, Klanger B, Kolkhof P, et al. Summary of Research: Cardiovascular and Kidney Outcomes with Finerenone in Patients with Type 2 Diabetes and Chronic Kidney Disease-The FIDELITY Pooled Analysis. Diabetes Ther. Sep 2024;15(9):1861-1864. doi:10.1007/s13300-024-01591-w. https://link.springer.com/content/pdf/10.1007/s13300-024-01591-w.pdf
- Filippatos G, Anker SD, August P, et al. Finerenone and effects on mortality in chronic kidney disease and type 2 diabetes: a FIDELITY analysis. European heart journal Cardiovascular pharmacotherapy. Feb 2 2023;9(2):183-191. doi:10.1093/ehjcvp/pvad001. https://pubmed.ncbi.nlm.nih.gov/36639130/
- Pitt B, Agarwal R, Anker SD, et al. Hypokalaemia in patients with type 2 diabetes and chronic kidney disease: the effect of finerenone-a FIDELITY analysis. European heart journal Cardiovascular pharmacotherapy. Feb 8 2025;11(1):10-19. doi:10.1093/ehjcvp/pvae074. https://www.ncbi.nlm.nih.gov/pubmed/39380152
- Yang S, Shen W, Zhang HZ, Wang CX, Yu WQ, Wu QH. Efficacy and Safety of Finerenone for Prevention of Cardiovascular Events in Type 2 Diabetes Mellitus With Chronic Kidney Disease: A Meta-analysis of Randomized Controlled Trials. Journal of cardiovascular pharmacology. Jan 1 2023;81(1):55-62. doi:10.1097/fjc.0000000000001364. https://pubmed.ncbi.nlm.nih.gov/36027585/
- Ghosal S, Sinha B. Finerenone in type 2 diabetes and renal outcomes: A random-effects model meta-analysis. Frontiers in endocrinology. 2023;14:1114894. doi:10.3389/fendo.2023.1114894. https://pubmed.ncbi.nlm.nih.gov/36742404/
- Bansal S, Canziani MEF, Birne R, et al. Finerenone cardiovascular and kidney outcomes by age and sex: FIDELITY post hoc analysis of two phase 3, multicentre, double-blind trials. BMJ open. Mar 19 2024;14(3):e076444. doi:10.1136/bmjopen-2023-076444. https://bmjopen.bmj.com/content/bmjopen/14/3/e076444.full.pdf
- Perakakis N, Bornstein SR, Birkenfeld AL, et al. Efficacy of finerenone in patients with type 2 diabetes, chronic kidney disease and altered markers of liver steatosis and fibrosis: A FIDELITY subgroup analysis. Diabetes, obesity & metabolism. Jan 2024;26(1):191-200. doi:10.1111/dom.15305. https://www.ncbi.nlm.nih.gov/pubmed/37814928
- Singh AK, James G, Anker SD, et al. Finerenone Efficacy in Patients with Chronic Kidney Disease, Type 2 Diabetes, and Anemia in FIDELITY. JACC Adv. Feb 2025;4(2):101524. doi:10.1016/j.jacadv.2024.101524. https://pubmed.ncbi.nlm.nih.gov/40021271/
- Filippatos G, Anker SD, Bakris GL, et al. Finerenone and left ventricular hypertrophy in chronic kidney disease and type 2 diabetes. ESC Heart Fail. Feb 2025;12(1):185-188. doi:10.1002/ehf2.14962. https://pubmed.ncbi.nlm.nih.gov/39226189/
- Filippatos G, Anker SD, Pitt B, et al. Finerenone efficacy in patients with chronic kidney disease, type 2 diabetes and atherosclerotic cardiovascular disease. European heart journal Cardiovascular pharmacotherapy. Dec 15 2022;9(1):85-93. doi:10.1093/ehjcvp/pvac054. https://pmc.ncbi.nlm.nih.gov/articles/PMC9753093/
- Rossing P, Birkenfeld AL, Fioretto P, et al. Finerenone and Clinical Outcomes in CKD and Type 2 Diabetes by Frailty Index: FIDELITY Post Hoc Analysis. Clinical journal of the American Society of Nephrology : CJASN. May 2 2025;20(7):968-977. doi:10.2215/cjn.0000000700. https://pubmed.ncbi.nlm.nih.gov/40315020/
- Filippatos G, Anker SD, Pitt B, et al. Finerenone and Heart Failure Outcomes by Kidney Function/Albuminuria in Chronic Kidney Disease and Diabetes. JACC Heart failure. Nov 2022;10(11):860-870. doi:10.1016/j.jchf.2022.07.013. https://pubmed.ncbi.nlm.nih.gov/36328655/
- Sarafidis P, Agarwal R, Pitt B, et al. Outcomes with Finerenone in Participants with Stage 4 CKD and Type 2 Diabetes: A FIDELITY Subgroup Analysis. Clinical journal of the American Society of Nephrology : CJASN. May 1 2023;18(5):602-612. doi:10.2215/cjn.0000000000000149. https://pubmed.ncbi.nlm.nih.gov/36927680/
- Ebert T, Anker SD, Ruilope LM, et al. Outcomes With Finerenone in Patients With Chronic Kidney Disease and Type 2 Diabetes by Baseline Insulin Resistance. Diabetes Care. Mar 1 2024;47(3):362-370. doi:10.2337/dc23-1420. https://pubmed.ncbi.nlm.nih.gov/38151465/
- Rosas SE, Ruilope LM, Anker SD, et al. Finerenone in Hispanic Patients With CKD and Type 2 Diabetes: A Post Hoc FIDELITY Analysis. Kidney Med. Oct 2023;5(10):100704. doi:10.1016/j.xkme.2023.100704. https://pmc.ncbi.nlm.nih.gov/articles/PMC10514441/
- Flack JM, Agarwal R, Anker SD, et al. Finerenone in Black Patients With Type 2 Diabetes and CKD: A Post hoc Analysis of the Pooled FIDELIO-DKD and FIGARO-DKD Trials. Kidney Med. Dec 2023;5(12):100730. doi:10.1016/j.xkme.2023.100730. https://www.kidneymedicinejournal.org/article/S2590-0595(23)00148-6/pdf
- Wada T, Anker SD, Liu Z, et al. Efficacy and Safety of Finerenone in Asian Patients with Type 2 Diabetes and Chronic Kidney Disease: A FIDELITY Analysis. Kidney Dis (Basel). Jan-Dec 2025;11(1):402-415. doi:10.1159/000545415. https://karger.com/kdd/article-pdf/11/1/402/4379701/000545415.pdf
- Li P, Zheng H, Ma J, et al. Impact of finerenone on chronic kidney disease progression in Chinese patients with type 2 diabetes: a FIGARO-DKD subgroup analysis. Frontiers in endocrinology. 2025;16:1568438. doi:10.3389/fendo.2025.1568438. https://public-pages-files-2025.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2025.1568438/pdf
- Mentz RJ, Anker SD, Pitt B, et al. Efficacy and safety of finerenone in patients with chronic kidney disease and type 2 diabetes by diuretic use: A FIDELITY analysis. European journal of heart failure. May 2025;27(5):764-774. doi:10.1002/ejhf.3569. https://onlinelibrary.wiley.com/doi/10.1002/ejhf.3569
- Rossing P, Agarwal R, Anker SD, et al. Finerenone in patients across the spectrum of chronic kidney disease and type 2 diabetes by glucagon-like peptide-1 receptor agonist use. Diabetes, obesity & metabolism. Feb 2023;25(2):407-416. doi:10.1111/dom.14883. https://pubmed.ncbi.nlm.nih.gov/36193847/
- Zhang Y, Jiang L, Wang J, et al. Network meta-analysis on the effects of finerenone versus SGLT2 inhibitors and GLP-1 receptor agonists on cardiovascular and renal outcomes in patients with type 2 diabetes mellitus and chronic kidney disease. Cardiovasc Diabetol. Nov 5 2022;21(1):232. doi:10.1186/s12933-022-01676-5. https://link.springer.com/content/pdf/10.1186/s12933-022-01676-5.pdf
- Agarwal R, Green JB, Heerspink HJL, et al. Finerenone with Empagliflozin in Chronic Kidney Disease and Type 2 Diabetes. The New England journal of medicine. Aug 7 2025;393(6):533-543. doi:10.1056/NEJMoa2410659. https://pubmed.ncbi.nlm.nih.gov/40470996/
- Sato A, Nishimoto M. Real-world use of finerenone in patients with chronic kidney disease and type 2 diabetes based on large-scale clinical studies: FIDELIO-DKD and FIGARO-DKD. Hypertension research : official journal of the Japanese Society of Hypertension. Jun 2025;48(6):1929-1938. doi:10.1038/s41440-025-02175-2. https://www.nature.com/articles/s41440-025-02175-2
- Nicholas SB, Correa-Rotter R, Desai NR, et al. First interim results from FINE-REAL: a prospective, non-interventional, phase 4 study providing insights into the use and safety of finerenone in a routine clinical setting. Journal of nephrology. Nov 2024;37(8):2223-2232. doi:10.1007/s40620-024-02070-y. https://link.springer.com/content/pdf/10.1007/s40620-024-02070-y.pdf