Life Extension Magazine®

Life Extension® has an advantage in identifying modern causes of premature aging and death. That’s because we have direct access to tens of thousands of our members’ blood test results.
Our review of this real-world data enables us to uncover disease risk factors that are overlooked by the mainstream media. We then alert members about simple steps they can take to mitigate these hazards.
Earlier this year, we analyzed fasting insulin and hemoglobin A1c blood levels in over 10,000 members. A startling 66% had higher than desired fasting insulin. Twenty-two percent had hemoglobin A1c levels that placed them in a pre-diabetic state.
Hemoglobin A1c measures the percentage of glycated hemoglobin in one’s blood. Hemoglobin A1c levels should be below 5.6%,1 yet more than one in five people we tested had a reading over 6%.
Gaining early access to this kind of data can spare aging humans the ravages of degenerative illness. Armed with this knowledge, Foundation members can slash their risk of cancer, vascular occlusion, and other complications before frank diabetes is diagnosed.
This article will describe the dangers of elevated fasting insulin and hemoglobin A1c, our recent analysis of member blood tests, and a novel way to protect against glycemic overload.
What Is Insulin Supposed to Do?

Insulin is a hormone that regulates carbohydrate and fat metabolism.2 Insulin enables liver and muscle cells to take up blood sugar (glucose) for energy production or storage.2 Insulin also facilitates the packing of glucose into fat cells as triglycerides.2
A burst of insulin is released in response to food ingestion. Once glucose has been safely shuttled into energy producing cells or stored, insulin levels should drop below 5 µIU/mL.3 Only a tiny amount of residual insulin should be needed to maintain glucose homeostasis.
When fasting insulin is over 5 µIU/mL, this indicates a metabolic problem such as pre-diabetes, which sharply increases risk for degenerative diseases.3 Some medical texts state that insulin should virtually vanish from the blood once glucose levels reach 83 mg/dL.4
In people suffering from metabolic disorders and/or obesity, insulin levels remain stubbornly high. This not only generates damaging reactions throughout the body, but precludes weight loss as glucose is forced into fat cell storage.
Life Extension’s analysis of over 10,000 fasting insulin blood test results showed that 66% were over 5 µIU/mL. Many of these subjects were overweight, which partially explains why such a high percentage had high fasting insulin levels. Insulin control is an important component for successful weight loss.
The Dark Side of Insulin
Those afflicted with type I diabetes do not produce enough insulin. For these individuals, insulin injections become a lifesaving therapy.
Insulin, however, has an insidious dark side. High levels are associated with virtually every aging-related disease, thus making insulin control essential if one is to achieve optimal longevity.
Aging, poor diet, and other factors deprive cells of insulin sensitivity.5-7 Loss of insulin sensitivity contributes to excess insulin release as the body seeks to force serum glucose into cells.
The most immediate and noticeable effect of excess insulin production is unwanted weight gain.8-10 Insulin drives fat into cells, prevents fat from being released from cells, and can result in chronic hunger.11,12 High insulin levels contribute not only to obesity, but also to the disease states associated with being overweight.
Some degenerative disorders associated with too much insulin include heart attack13-16 and cancer.17-19
Insulin saves the lives of type I diabetics who are dependent on it, but becomes a toxic hormone in aging people who secrete too much. Reducing excess serum insulin is a critical component of any scientific program designed to facilitate fat loss and extend life span.
Hyperinsulinemia
Insulin is produced in the pancreas to stimulate uptake of glucose from blood into the body’s cells. The inability of the body’s cells to utilize insulin is called insulin resistance.
As a result of insulin resistance and other factors, the pancreas produces more insulin than normal so there are higher levels of insulin circulating in the bloodstream. This is called hyperinsulinemia.
In a normal person, one unit of insulin might be needed to help 10 mg of glucose go into a cell group. In hyperinsulinemia, ten units of insulin might be needed to get the same 10 mg of glucose into the cell group.20 Hyperinsulinemia and insulin resistance create myriad problems, including elevated triglycerides,21-24 low HDL,25,26 type II diabetes,27-29 and obesity.14,30-32
Figure 1 on this page shows the pathways to obesity and several degenerative diseases associated with insulin resistance and hyperinsulinemia.
Risks Associated with Insulin Dysregulation
Excess insulin and insulin resistance cause deleterious changes in many biochemical pathways that can lead to the development of a number of degenerative diseases and potentially life-threatening metabolic consequences.14,17-19,21-32
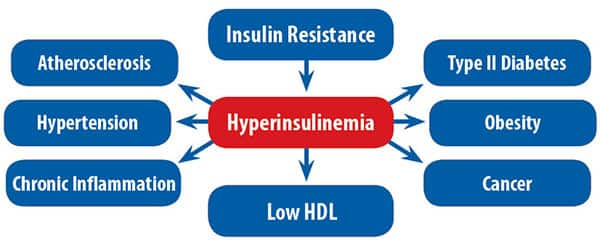
Insulin and Age-Related Disease

An enormous volume of peer-reviewed published studies reveal that excess serum insulin (hyperinsulinemia) is a major health problem. Regrettably, this danger still is not recognized by the medical mainstream.
High serum insulin promotes hypertension by impairing sodium balance.33,34 Too much insulin harms the kidneys.35 The vascular system is severely damaged by prolonged exposure to excess insulin.36,37
By acting as a catalyst in promoting cell growth, excess insulin increases the risk and progression of certain cancers.17,19,38-40 High insulin promotes the formation of beta-amyloid in brain cells and may contribute to the development of Alzheimer’s disease.41 Overproduction of insulin is even a contributory factor to prostate enlargement because of its effects in promoting the overgrowth of prostate cells.42
High serum insulin is associated with the development of abdominal obesity, which exacerbates the many problems induced by insulin resistance and increased belly fat, including atherosclerosis 14,43-45 and impotence.46-50 Obesity is associated with excess insulin and reduced insulin sensitivity, both risk factors for type II diabetes.51
Perhaps the simplest way to evaluate the toxic effects of excess insulin is by examining its effects on human mortality. One study showed that over a 10-year period, the risk of dying was almost twice as great for those with the highest levels of insulin compared to those with the lowest.52 The study authors stated that hyperinsulinemia is associated with increased all-cause and cardiovascular mortality independent of other risk factors.
Why Aging People Gain Weight
A review of the published literature indicates that a significant percentage of degenerative diseases are attributed to excess body fat. Those who are overweight face a significant risk of developing type II diabetes.53-55 The treatments for obesity and type II diabetes are interrelated. By effectively treating either one of these diseases, doctors can mitigate or control the other.
Poor diet, obesity, and aging result in excessive secretion of insulin.5-7 Suppressing the overproduction of insulin is a crucial component of a medically supervised weight-loss program.
A noticeable effect of surplus serum insulin can be constant hunger that results in a vicious cycle in which overeating causes more body fat to accumulate, which in turn causes even greater amounts of unwanted insulin to be secreted from the pancreas.12,56 We now know that hyperinsulinemia predicts diabetes mellitus.51 Even in children, serum insulin levels are far higher in obese than in non-obese children of the same age.57
The effects of consuming high-glycemic foods and the subsequent hyperinsulinemia, hunger, and weight gain that occur were the subject of an article published in the Journal of the American Medical Association titled “The Glycemic Index.”58 The article’s authors summarized their position as follows:
“It is possible that the hunger incident to hyperinsulinemia may be a cause of overeating, and therefore, the obesity that so often precedes diabetes.”58
Dangerous After-Meal Sugar Levels

The excess elevation of blood sugar after eating wreaks havoc in the body via multiple pathological mechanisms. Elevations in postprandial (after-meal) blood sugar, along with the accompanying insulin surge, are major contributors to the development of diabetic and age-related disorders such as heart disease, as well as diseases of the microvasculature (small blood vessels within the eyes, kidneys, and those supplying the nerves).59-67
People who have normal fasting glucose levels but whose average after-meal glucose level exceeds 194 mg/dL are three times more likely to suffer diabetic retino-pathy than those who do not.68
Growing evidence indicates that severe spikes in after-meal blood sugar are a major problem for non-diabetics as well.59,69 The two primary mechanisms by which post-meal hyperglycemia causes such problems are formation of advanced glycation end products (the binding of glucose to body proteins) and increased production of free radicals that lead to arterial wall damage.70,71
To investigate the relationship between glucose metabolism and the severity of heart disease, one study measured how many coronary arteries were blocked in relation to after-meal glucose/insulin blood levels and other atherogenic risk factors. All of the men participating in the study had normal fasting glucose blood readings, but in response to a glucose challenge, demonstrated significantly different rates of glucose-insulin spikes. Men with the highest levels of post-load glucose, insulin, and other measurements of glycemic imbalance had the greatest number of blocked coronary arteries.72
These studies make it abundantly clear that blunting the after-meal increase in blood sugar and insulin is an important goal for those seeking optimal longevity.
Fasting Insulin Blood Levels in Life Extension Members

Since 1996, members of the Life Extension Foundation® have been able request their own blood tests. This has resulted in a significant increase for unique tests that are not routinely ordered by physicians. One of these tests is for fasting insulin. Since it was added to the Male and Female Weight Loss Panels several years ago, the number of fasting insulin tests has surged.
In the most recent analysis, data was collected from January 2010 to February 2013, a period spanning more than three years. More than 10,000 tests of fasting insulin were reviewed.
An upper limit for fasting insulin of 5 µIU/mL (micro international unit per milliliter) was established for this analysis.3 Only 34% of these blood tests had a fasting insulin level of 5 µIU/mL or lower. A startling 66% showed fasting insulin over 5 µIU/mL, with some in dangerously high ranges of 10-30 µIU/mL.
There is an important confounding factor in this analysis that skewed the results upwards. Most of the people who ordered their fasting insulin blood test did so as part of the Male or Female Weight Loss Panel. Since insulin levels are higher in overweight and obese individuals, it is logical to assume that many of the 66% whose fasting insulin was over 5 µIU/mL also were overweight or obese. Thinner people tend to have lower fasting insulin levels.
Latest Studies on Dangers of Excess Insulin
From 1999 to 2005, Life Extension spent millions of dollars evaluating published studies on the mechanisms and dangers of excess fasting insulin and conducting our own clinical trials on a compound that suppressed pancreatic release of surplus insulin.
We were never able to synthesize sufficient quantities of our insulin-suppressing compound, but another research team has developed an approach to reduce glucose/insulin levels that may revolutionize the way glycemic control disorders are treated.
Just to make sure we were still on the right track, we went back and looked at the peer-reviewed published literature to confirm the deadly effects of hyperinsulinemia.
Liver Diseases Caused by Hyperinsulinemia
As a greater percentage of the public becomes overweight or obese, an illness called non-alcoholic fatty liver disease is becoming prevalent. It is characterized by infiltration of fat into the liver that impairs normal functions.
In 2013, two studies identified hyperinsulinemia as a major causative factor of non-alcoholic fatty liver disease, with fasting insulin being significantly elevated compared to controls.73,74 Two studies published in 2012 also connected hyperinsulinemia with this liver disease.75,76
A 2013 study looked at patients affected with primary liver cancer and found a high prevalence of liver cancer in type II diabetics. Researchers noticed that liver cancer developed in these patients within the first five years after diagnosis of type II diabetes, which is when insulin levels are extremely high.77
Hyperinsulinemia in College Students
A 2012 study looked at college students and found 8 out of 22 subjects were hyperinsulinemic with fasting insulin levels greater than 19 µIU/mL.78
The study concluded by stating that the high prevalence of hyperinsulinemia in this population merits further investigation and intervention.78
Studies of older people often show these high insulin readings, but this study reveals damaging processes occurring early in life. Remember that any fasting insulin reading above 5 µIU/mL is cause for concern and many of these college students were already almost four times as high!
Kidney Failure Linked to Hyperinsulinemia
In the US, a startling 40% of people over age 65 show some sign of kidney failure.79 Between 1980 and 2009, the prevalence of end stage renal disease increased nearly 600%.80
A 2012 study found a strong correlation between hyperinsulinemia and chronic kidney disease in metabolic syndrome patients. Insulin resistance was shown to be an independent risk factor for kidney disease in this patient group.81
High Insulin Initiates Cancer

People with hyperinsulinemia suffer higher rates of deadly malignancies.
A 2013 study showed over a ten-year period that type II diabetics treated with any kind of insulin-augmenting drug had an up to 80% increased risk of experiencing cancer, an adverse cardiac event, or death from any cause compared to patients who only received the drug metformin, which lowers insulin levels.82 Metformin reduces risk of many cancers,83-91 and some of its mechanisms include reducing hepatic production of glucose92-94 and improving insulin sensitivity,95-100 both of which reduce insulin levels.
A drug class known as sulfonylureas stimulates pancreatic insulin secretion and temporarily reduces glucose. After the pancreas fails, these patients often resort to insulin injections to keep glucose under control. By giving type II diabetics insulin-augmenting therapies instead of lifestyle changes, nutrients, and metformin, doctors have unwittingly condemned huge segments of their patient population to higher risks of weight gain, neuropathy, renal failure, atherosclerosis, and cancer.101-109
Not every diabetic patient needs insulin-augmenting drugs. Some people can produce all the surplus insulin they need in their pancreas to keep glucose down. Their chronic hyperinsulinemic condition, however, predisposes them to greater cancer incidences as insulin provides a strong signal for malignant cells to proliferate.17 One study found that individuals with the highest insulin levels had a 62% increased risk of cancer mortality.110
Hyperinsulinemic individuals, including type II diabetics and the obese, suffer far greater rates of cancer. Hyperinsulinemia creates chronic inflammation and the generation of free radicals, both of which damage DNA genes needed to regulate healthy cellular proliferation.16,19,38,111,112
A number of studies published in 2012-2013 describe the mechanisms by which hyperinsulinemia increases cancer risk and suggests that understanding and circumventing these pathways may lead to targeted prevention.113-125
Controlling Hyperinsulinemia Critical for Cancer Patients
Once a person is diagnosed with cancer, it becomes imperative to suppress excess insulin secretion. That’s because in addition to initiating cancer, insulin stimulates the proliferation of malignant cells.126
A 2013 review evaluated the scientific literature on the role of hyperinsulinemia in promoting existing cancers. It summarized by stating that the death rate of pancreatic and other cancers can be reduced by an aggressive approach to reversing obesity and hyperinsulinemia and achieving good glycemic control.113
When we consult with oncologists about comprehensive cancer treatments, a central element is for the patient to make significant lifestyle changes and be prescribed the drug metformin to reduce levels of tumor-promoting insulin.
Role in Alzheimer’s Disease
Alzheimer’s disease is the fastest growing threat to health in the United States, according to a report released in 2013 from a team of researchers at the University of Washington in Seattle.127
We know of multiple underlying causes for Alzheimer’s, including mitochondrial dysfunction, oxidative stress, and chronic inflammation.128-135 A study published in 2012 described how insulin is involved in the metabolism of beta-amyloid and concluded that insulin resistance is involved in the pathogenesis of neurodegenerative diseases including Alzheimer’s.136
Other studies published in 2012-2013 describe the toxic effects of hyperinsulinemia in the development of neurodegenerative diseases that result in senility.137-141
Mainstream Medicine’s Lethal Misconception

We don’t blame doctors who desperately sought out therapies to lower surging glucose levels in their diabetic patients. They often had no choice but to administer insulin-augmenting drugs, which temporarily dropped their patient’s glucose to survivable ranges.
A side effect quickly observed was weight gain in those using insulin-augmentation to control blood glucose. As these patients gained weight, they became more insulin-resistant and had to rely on greater quantities of insulin-augmented drugs. Many of these drug labels started carrying warnings of increased risk of heart attack and early death.
Research initiated by Life Extension in 1999 relating to the toxic role of excess insulin in virtually every age-related disease has since been corroborated by numerous independent studies. Hyperinsulinemia is a killer worse than cholesterol, since high insulin levels not only increase vascular risk,36,37,141 but also cancer,17-19,126,142 liver and kidney disease,143-147 neurodegeneration,148,149 and obesity.8,30-32
The scientific literature is unanimous in recognizing the lethal impact of hyperinsulinemia. An analysis of blood test results from Life Extension members reveals that 66% have higher than desired fasting insulin levels (above 5 µIU/mL). Virtually all of these members should be asking their doctors to prescribe metformin, as this drug reduces glucose and insulin blood levels via multiple mechanisms including curbing excessive glucose production (gluconeogenesis) in liver.93
Twenty-two percent of our members had hemoglobin A1c levels over 6%, which places them in a pre-diabetic state. Clearly more has to be done to gain control of these glycemic measurements of future disease risks.
Breakthrough in Preventing Dietary Starches from Converting to Glucose
The modern American diet contains excessive amounts of starches.
What most people don’t realize is that starches are broken down in the intestines and produce rapid elevation of after-meal glucose and insulin blood levels. Even people with “normal” fasting blood sugar levels are at increased cardiovascular risk if their after-meal glucose/insulin levels rise too high, too fast.150,151
A natural enzyme has been discovered that helps reduce after-meal blood sugar and insulin spikes in a totally unique way.152 This enzyme converts rapidly digestible starch in the intestines into a fiber that is not as readily absorbed as glucose.153,154
Based on the evidence you will read in this month’s issue, widespread use of this enzyme before starch-containing meals could help reduce the epidemic of glucose intolerance and hyperinsulinemia that is plaguing the modern world.
For longer life,
William Faloon
References
- Geberhiwot T, Haddon A, Labib M. HbA1c predicts the likelihood of having impaired glucose tolerance in high-risk patients with normal fasting plasma glucose. Ann Clin Biochem. 2005 May;42(Pt 3):193-5.
- Available at: http://www.princeton.edu/~achaney/tmve/wiki100k/docs/insulin.html. Accessed April 23, 2013.
- Available at: http://www.a4m.com/assets/pdf/bookstore/thera6_ch4.pdf?session_main=dap3mjgnb96luhbje8tcklgp75. Accessed April 29, 2013.
- Isselbacher KJ, Braunwald E, Wilson JD, Martin JB, & Fauci AS. (Eds.). (1994). Harrison’s Principles of Internal Medicine (13th Ed.), McGraw-Hill.
- Tzima N, Pitsavos C, Panagiotakos DB, et al. Mediterranean diet and insulin sensitivity, lipid profile and blood pressure levels, in overweight and obese people; the Attica study. Lipids Health Dis. 2007 Sep 19;6:22.
- Ryan AS. Insulin resistance with aging: effects of diet and exercise. Sports Med. 2000 Nov;30(5):327-46.
- Campos G, Ryder E, Diez-Ewald M, et al. Prevalence of obesity and hyperinsulinemia: its association with serum lipid and lipoprotein concentrations in healthy individuals from Maracaibo, Venezuela. Invest Clin. 2003 Mar;44(1):5-19.
- Mehran AE, Templeman NM, Brigidi GS, et al. Hyperinsulinemia drives diet-induced obesity independently of brain insulin production. Cell Metab. 2012 Dec 5;16(6):723-37.
- Lemay A, Turcot L, Déchêne F, Dodin S, Forest JC. Hyperinsulinemia in nonobese women reporting a moderate weight gain at the beginning of menopause: a useful early measure of susceptibility to insulin resistance. Menopause. 2010 Mar;17(2):321-5.
- Odeleye OE, de Courten M, Pettitt DJ, Ravussin E. Fasting hyperinsulinemia is a predictor of increased body weight gain and obesity in Pima Indian children. Diabetes. 1997 Aug;46(8):1341-5.
- Coomans CP, Geerling JJ, Guigas B, et al. Circulating insulin stimulates fatty acid retention in white adipose tissue via KATP channel activation in the central nervous system only in insulin-sensitive mice. J Lipid Res. 2011 Sep;52(9):1712-22.
- Heller RF. Hyperinsulinemic obesity and carbohydrate addiction: the missing link is the carbohydrate frequency factor. Med Hypotheses. 1994 May;42(5):307-12.
- Després JP, Lamarche B, Mauriège P, et al. Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med. 1996 Apr 11;334(15):952-7.
- Chu N, Spiegelman D, Hotamisligil GS, Rifai N, Stampler M, Rimm EB. Plasma insulin, leptin, and soluble TNF receptors levels in relation to obesity-related atherogenic and thrombogenic cardiovascular disease risk factors among men. Atherosclerosis. 2001 Aug;157(2):495-503.
- Cersosimo E, DeFronzo RA. Insulin resistance and endothelial dysfunction: the road map to cardiovascular diseases. Diabetes Metab Res Rev. 2006 Nov-Dec;22(6):423-36.
- Eschwège E. The dysmetabolic syndrome, insulin resistance and increased cardiovascular (CV) morbidity and mortality in type 2 diabetes: aetiological factors in the development of CV complications. Diabetes Metab. 2003 Sep;29(4 Pt 2):6S19-27.
- Burnol AF, Morzyglod L, Popineau L. Cross-talk between insulin signaling and cell proliferation pathways. Ann Endocrinol (Paris). 2013 Apr 9. [Epub ahead of print]
- Nilsen TI, Vatten LJ. Prospective study of colorectal cancer risk and physical activity, diabetes, blood glucose and BMI: exploring the hyperinsulinaemia hypothesis. Br J Cancer. 2001 Feb 2;84(3):417-22.
- Balkau B, Kahn HS, Courbon D, Eschwege E, Ducimetier P. Hyperinsulinemia predicts fatal liver cancer but is inversely associated with fatal cancer at some other sites. Diabetes Care. 2001 May;24(5):843-9.
- Cochran E, Musso C, Gorden P. The use of U-500 in patients with extreme insulin resistance. Diabetes Care. 2005 May;28(5):1240-4.
- Godsland IF, Crook D, Walton C, Wynn V, Oliver MF. Influence of insulin resistance, secretion, and clearance on serum cholesterol, triglycerides, lipoprotein cholesterol, and blood pressure in healthy men. Arterioscler Thromb. 1992 Sep;12(9):1030-5.
- Salonen JT, Lakka TA, Lakka HM, Valkonen VP, Everson SA, Kaplan GA. Hyperinsulinemia is associated with the incidence of hypertension and dyslipidemia in middle-aged men. Diabetes. 1998 Feb;47(2):270-5.
- Stannard SR, Johnson NA. Insulin resistance and elevated triglyceride in muscle: more important for survival than “thrifty” genes? J Physiol. 2004 Feb 1;554(Pt 3):595-607.
- Mykkänen L, Kuusisto J, Haffner SM, Pyörälä K, Laakso M. Hyperinsulinemia predicts multiple atherogenic changes in lipoproteins in elderly subjects. Arterioscler Thromb. 1994 Apr;14(4):518-26.
- Karhapää P, Malkki M, Laakso M. Isolated low HDL cholesterol. An insulin-resistant state. Diabetes. 1994 Mar;43(3):411-7.
- Ko GT, Cockram CS, Woo J, Chan JC. Obesity, insulin resistance and isolated low high-density-lipoprotein cholesterol in Chinese subjects. Diabet Med. 2001 Aug;18(8):663-6.
- Goldstein BJ. Insulin resistance as the core defect in type 2 diabetes mellitus. Am J Cardiol. 2002 Sep 5; 90(5A):3G-10G.
- Haffner SM, Stern MP, Mitchell BD, Hazuda HP, Patterson JK. Incidence of type II diabetes in Mexican Americans predicted by fasting insulin and glucose levels, obesity, and body-fat distribution. Diabetes. 1990 Mar;39(3):283-9.
- Hansen J, Rinnov A, Krogh-Madsen R, et al. Plasma follistatin is elevated in patients with type 2 diabetes: relationship to hyperglycemia, hyperinsulinemia, and systemic low-grade inflammation. Diabetes Metab Res Rev. 2013 Apr 6.
- Despres JP, Pascot A, Lemieux I. Risk factors associated with obesity: a metabolic perspective. Ann Endocrinol. 2000 Dec;61 Suppl 6:31-8.
- Modan M, Halkin H, Almog S, et al. Hyperinsulinemia. A link between hypertension obesity and glucose intolerance. J Clin Invest. 1985 Mar;75(3):809-17.
- Feuers RJ, Desai VG, Chen FX, Hunter JD, Duffy PH, Oriaku ET. Effects of dietary restriction on insulin resistance in obese mice. J Am Aging Assoc. 2000 Apr;23(2):95-101.
- Hano T, Nishio I. Treatment of hypertension in the patients with obesity. Nippon Rinsho. 2001 May;59(5):973-7.
- Kopf D, Muhlen I, Kroning G, Sendzik I, Huschke B, Lehnert H. Insulin sensitivity and sodium excretion in normotensive offspring and hypertensive patients. Metabolism. 2001 Aug;50(8):929-35.
- Noda M, Matsuo T, Nagano-Tsuge H, et al. Involvement of angiotensin II in progression of renal injury in rats with genetic non- insulin-dependent diabetes mellitus (Wistar fatty rats). Jpn J Pharmacol. 2001 Apr;85(4):416-22.
- Hegele RA. Premature atherosclerosis associated with monogenic insulin resistance. Circulation. 2001 May 8;103(18):2225-9.
- Katz AS, Goff DC, Feldman SR. Acanthosis nigricans in obese patients: Presentations and implications for prevention of atherosclerotic vascular disease. Dermatol Online J. 2000 Sep;6(1):1.
- Kaaks R. Plasma insulin, IGF-I and breast cancer. Gynecol Obstet Fertil. 2001 Mar;29(3):185-91.
- Czyzyk A, Szczepanik Z. Diabetes mellitus and cancer. Eur J Intern Med. 2000 Oct;11(5):245-52.
- Bruce WR, Wolever TM, Giacca A. Mechanisms linking diet and colorectal cancer: the possible role of insulin resistance. Nutr Cancer. 2000 37(1):19-26.
- Cook DG, Leverenz JB, McMillian PJ, et al. Reduced hippocampal insulin-degrading enzyme in late-onset Alzheimer’s disease is associated with the apolipoprotein E-epsilon4 allele. Am J Pathol. 2003 Jan;162(1):313-9.
- Hammarsten J, Hogstedt B. Hyperinsulinaemia as a risk factor for developing benign prostatic hyperplasia. Eur Urol. 2001 Feb;39(2):151-8.
- Nakamura T, Tokunaga K, Shimomura I, et al. Contribution of visceral fat accumulation to the development of coronary artery disease in non-obese men. Atherosclerosis. 1994 Jun;107(2):239-46.
- Lear SA, Humphries KH, Kohli S, Frohlich JJ, Birmingham CL, Mancini GB. Visceral adipose tissue, a potential risk factor for carotid atherosclerosis: results of the Multicultural Community Health Assessment Trial (M-CHAT). Stroke. 2007 Sep;38(9):2422-9.
- Chien KL, Lee YT, Sung FC, Hsu HC, Su TC, Lin RS. Hyperinsulinemia and related atherosclerotic risk factors in the population at cardiovascular risk: a community-based study. Clin Chem. 1999 Jun;45(6 Pt 1):838-46.
- Bansal TC, Guay AT, Jacobson J, Woods BO, Nesto RW. Incidence of metabolic syndrome and insulin resistance in a population with organic erectile dysfunction. J Sex Med. 2005 Jan;2(1):96-103.
- Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013 Jan 12;381(9861):153-65.
- Chitaley K, Kupelian V, Subak L, Wessells H. Diabetes, obesity and erectile dysfunction: field overview and research priorities. J Urol. 2009 Dec;182(6 Suppl):S45-50.
- Trussell JC, Legro RS. Erectile dysfunction: does insulin resistance play a part? Fertil Steril. 2007 Oct;88(4):771-8.
- Tamler R. Diabetes, obesity, and erectile dysfunction. Gend Med. 2009;6 Suppl 1:4-16.
- Johnson MS, Figueroa-Colon R, Huang TT, Dwyer JH, Goran MI. Longitudinal changes in body fat in African-American and caucasian children: influence of fasting insulin and insulin sensitivity. J Clin Endocrinol Metab. 2001 Jul;86(7):3182-7.
- Pyorala M, Miettinen H, Laakso M, Pyorala K. Plasma insulin and all-cause, cardiovascular, and noncardiovascular mortality: the 22-year follow-up results of the Helsinki Policemen Study. Diabetes Care. 2000 Aug;23(8):1097-102.
- Ye J. Mechanisms of insulin resistance in obesity. Front Med. 2013 Mar;7(1):14-24.
- van ‘t Riet E, Alssema M, Nijpels G, Dekker JM. Estimating the individual risk of diabetes: not on the grounds of overweight only. Ned Tijdschr Geneeskd. 2008 Nov 1;152(44):2385-8.
- Krishnan S, Rosenberg L, Djoussé L, Cupples LA, Palmer JR. Overall and central obesity and risk of type 2 diabetes in U.S. black women. Obesity (Silver Spring). 2007 Jul;15(7):1860-6.
- Rodin J. Insulin levels, hunger, and food intake: an example of feedback loops in body weight regulation. Health Psychol. 1985;4(1):1-24.
- Reinehr T, de Sousa G, Toschke AM, Andler W. Long-term follow-up of cardiovascular disease risk factors in children after an obesity intervention. Am J Clin Nutr. 2006 Sep;84(3):490-6.
- Ludwig DS. The glycemic index: physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA. 2002 May 8;287(18):2414-23.
- Ceriello A. The postprandial state and cardiovascular disease: relevance to diabetes mellitus. Diabetes Metab Res Rev. 2000 Mar-Apr;16(2):125-32.
- Coutinho M, Gerstein HC, Wang Y, Yusuf S. The relationship between glucose and incident cardiovascular events. A metaregression analysis of published data from 20 studies of 95,783 individuals followed for 12.4 years. Diabetes Care. 1999 Feb;22(2):233-40.
- Lin HJ, Lee BC, Ho YL, et al. Postprandial glucose improves the risk prediction of cardiovascular death beyond the metabolic syndrome in the nondiabetic population. Diabetes Care. 2009 Sep;32(9):1721-6.
- Mannucci E, Monami M, Lamanna C, Adalsteinsson JE. Post-prandial glucose and diabetic complications: systematic review of observational studies. Acta Diabetol. 2012 Aug;49(4):307-14.
- Xin Z, Yuan MX, Li HX, et al. Evaluation for fasting and 2-hour glucose and HbA1c for diagnosing diabetes based on prevalence of retinopathy in a Chinese population. PLoS One. 2012;7(7):e40610.
- Bash, LD, Selvin E, Steffes M, Coresh J, Astor BC. Poor glycemic control in diabetes and the risk of incident kidney disease even in the absence of albuminuria and retinopathy: atherosclerosis risk in communities (ARIC) study. Arch Intern Med. 2008 Dec 8;168(22):2440-7.
- Polhill TS, Saad S, Poronnik SSP, Fulcher GR, Pollock CA. Short-term peaks in glucose promote renal fibrogenesis independently of total glucose exposure. Am J Physiol Renal Physiol. 2004 Aug;287(2):F268-73.
- Sumner CJ, Sheth S, Griffin JW, Cornblath DR, Polydefkis M. The spectrum of neuropathy in diabetes and impaired glucose tolerance. Neurology. 2003 Jan 14;60(1):108-11.
- Hoffman-Snyder C, Smith BE, Ross MA, Hernandez J, Bosch EP. Value of the oral glucose tolerance test in the evaluation of chronic idiopathic axonal polyneuropathy. Arch Neurol. 2006 Aug;63(8):1075-9.
- Bell DS. Importance of postprandial glucose control. South Med J. 2001 Aug;94(8):804-9.
- Chien KL, Lee BC, Lin HJ, Hsu HC, Chen MF. Association of fasting and post-prandial hyperglycemia on the risk of cardiovascular and all-cause death among non-diabetic Chinese. Diabetes Res Clin Pract. 2009 Feb;83(2):e47-50.
- Ceriello A. Impaired glucose tolerance and cardiovascular disease: the possible role of postprandial hyperglycemia. Am Heart J. 2004 May;147(5):803-7.
- Lebovitz HE. Effect of the postprandial state on nontraditional risk factors. Am J Cardiol. 2001 Sep 20;88(6A):20H-5H.
- Sasso FC, Carbonara O, Nasti R, et al. Glucose metabolism and coronary heart disease in patients with normal glucose tolerance. JAMA. 2004 Apr 21;291(15):1858-63.
- Pirgon O, Bilgin H, Cekmez F, Kurku H, Dündar BN. Association between insulin resistance and oxidative stress parameters in obese adolescents with non-alcoholic Fatty liver disease. J Clin Res Pediatr Endocrinol. 2013 Mar 22;5(1):33-9.
- Kawano Y, Cohen DE. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J Gastroenterol. 2013 Feb 9.
- Hurjui DM, Niţă O, Graur LI, Mihalache L, Popescu DS, Graur M. The central role of the non alcoholic fatty liver disease in metabolic syndrome. Rev Med Chir Soc Med Nat Iasi. 2012 Apr-Jun;116(2):425-31.
- Titov VN, Ivanova KV, Malyshev PP, Kaba SI, Shiriaeva IuK. The unity of pathogenesis of insulin resistance syndrome and non-alcoholic fatty disease of liver. The metabolic disorder of fatty acids and triglycerides. Klin Lab Diagn. 2012 Nov;(11):3-12.
- Yang WS, Shu XO, Gao J, et al. Prospective evaluation of type 2 diabetes mellitus on the risk of primary liver cancer in Chinese men and women. Ann Oncol. 2013 Feb 13.
- Hopper MK, Brown GW, Funke KA, Pike Brown LR. Prevalence of hyperinsulinemia associated with body mass index, genetic predisposition, and lifestyle in college freshmen students. J Am Coll Health. 2012 60(1):27-36.
- Available at: http://www.cdc.gov/diabetes/pubs/factsheets/kidney.htm. Accessed April 25, 2013.
- Available at: http://kidney.niddk.nih.gov/kudiseases/pubs/kustats. Accessed April 25, 2013.
- Kutyrina IM, Kriachkova AA, Savel’eva SA. The role of hyperinsulinemia and insulin resistance in development of chronic kidney disease in patients with metabolic syndrome. Ter Arkh. 2012 84(6):32-6.
- Currie CJ, Poole CD, Evans M, Peters JR, Morgan CL. Mortality and other important diabetes-related outcomes with insulin vs other antihyperglycemic therapies in type 2 diabetes. J Clin Endocrinol Metab. 2013 Feb;98(2):668-77.
- Hirsch HA, Iliopoulos D, Tsichlis PN, Struhl K. Metformin selectively targets cancer stem cells, and acts together with chemotherapy to block tumor growth and prolong remission. Cancer Res. 2009 Oct 1;69(19):7507-11.
- Bodmer M, Meier C, Krähenbühl S, Jick SS, Meier CR. Long-term metformin use is associated with decreased risk of breast cancer. Diabetes Care. 2010 Jun;33(6):1304-8.
- Rizos CV, Elisaf MS. Metformin and cancer. Eur J Pharmacol. 2013 Apr 5;705(1-3):96-108.
- Cantrell LA, Zhou C, Mendivil A, Malloy KM, Gehrig PA, Bae-Jump VL. Metformin is a potent inhibitor of endometrial cancer cell proliferation--implications for a novel treatment strategy. Gynecol Oncol. 2010 Jan;116(1):92-8.
- Wang LW, Li ZS, Zou DW, Jin ZD, Gao J, Xu GM. Metformin induces apoptosis of pancreatic cancer cells. World J Gastroenterol. 2008 Dec 21;14(47):7192-8.
- Wright JL, Stanford JL. Metformin use and prostate cancer in Caucasian men: results from a population-based case-control study. Cancer Causes Control. 2009 Nov;20(9):1617-22.
- Hosono K, Endo H, Takahashi H, et al. Metformin suppresses colorectal aberrant crypt foci in a short-term clinical trial. Cancer Prev Res (Phila). 2010 Sep;3(9):1077-83.
- Libby G, Donnelly LA, Donnan PT, Alessi DR, Morris AD, Evans JM. New users of metformin are at low risk of incident cancer: a cohort study among people with type 2 diabetes. Diabetes Care. 2009 Sep;32(9):1620-5.
- Memmott RM, Mercado JR, Maier CR, Kawabata S, Fox SD, Dennis PA. Metformin prevents tobacco carcinogen--induced lung tumorigenesis. Cancer Prev Res (Phila). 2010 Sep;3(9):1066-76.
- Takahara M, Kaneto H, Katakami N, Matsuhisa M, Shimomura I. Effect of metformin on hepatic glucose production in Japanese patients with type 2 diabetes mellitus. Endocr J. 2012 Sep 30;59(9):845-7.
- Caton PW, Nayuni NK, Kieswich J, Khan NQ, Yaqoob MM, Corder R. Metformin suppresses hepatic gluconeogenesis through induction of SIRT1 and GCN5. J Endocrinol. 2010 Apr;205(1):97-106.
- Hundal RS, Krssak M, Dufour S, et al. Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes. 2000 Dec;49(12):2063-9.
- Moon RJ. The addition of metformin in type 1 diabetes improves insulin sensitivity, diabetic control, body composition and patient well-being. Diabetes Obes Metab. 2007 Jan;9(1):143-5.
- Bhansali A, Sridhar S. Insulin sensitizers and serum testosterone in men. Clin Endocrinol (Oxf). 2013 Apr 9.
- Sir T, Castillo T, Munoz S, Lopez G, Calvillan M. Effects of metformin on insulin resistance in obese and hyperandrogenic women. Rev Med Chil. 1997 Dec;125(12):1457-63.
- Goodwin PJ, Pritchard KI, Ennis M, Clemons M, Graham M, Fantus IG. Insulin-lowering effects of metformin in women with early breast cancer. Clin Breast Cancer. 2008 Dec;8(6):501-5.
- Rojas LB, Gomes MB. Metformin: an old but still the best treatment for type 2 diabetes. Diabetol Metab Syndr. 2013 Feb 15;5(1):6.
- Vaikkakara S, Al-Ozairi E, Lim E, et al. The investigation and management of severe hyperandrogenism pre- and postmenopause: non-tumor disease is strongly associated with metabolic syndrome and typically responds to insulin-sensitization with metformin. Gynecol Endocrinol. 2008 Feb;24(2):87-92.
- Gibbons CH, Freeman R. Treatment-induced diabetic neuropathy: a reversible painful autonomic neuropathy. Ann Neurol. 2010 Apr;67(4):534-41.
- Tesfaye S, Malik R, Harris N, et al. Arterio-venous shunting and proliferating new vessels in acute painful neuropathy of rapid glycaemic control (insulin neuritis). Diabetologia. 1996 Mar;39(3):329-35.
- Savage S, Estacio RO, Jeffers B, Schrier RW. Increased complications in noninsulin-dependent diabetic patients treated with insulin versus oral hypoglycemic agents: a population study. Proc Assoc Am Physicians. 1997 Mar;109(2):181-9.
- Donner T, Muñoz M. Update on insulin therapy for type 2 diabetes. J Clin Endocrinol Metab. 2012 May;97(5):1405-13.
- Russell-Jones D, Khan R. Insulin-associated weight gain in diabetes--causes, effects and coping strategies. Diabetes Obes Metab. 2007 Nov;9(6):799-812.
- Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med. 2007 Jun 14;356(24):2457-71.
- Sobenin IA, Maksumova MA, Slavina ES, Balabolkin MI, Orekhov AN. Sulfonylureas induce cholesterol accumulation in cultured human intimal cells and macrophages. Atherosclerosis. 1994 Feb;105(2):159-63.
- Li D, Yeung SC, Hassan MM, Konopleva M, Abbruzzese JL. Antidiabetic therapies affect risk of pancreatic cancer. Gastroenterology. 2009 Aug;137(2):482-8.
- Yang YX, Hennessy S, Lewis JD. Insulin therapy and colorectal cancer risk among type 2 diabetes mellitus patients. Gastroenterology. 2004 Oct;127(4):1044-50.
- Perseghin G, Calori G, Lattuada G, et al. Insulin resistance/hyperinsulinemia and cancer mortality: the Cremona study at the 15th year of follow-up. Acta Diabetol. 2012 Dec;49(6):421-8.
- Available at: http://edrv.endojournals.org/content/24/3/278.full. Accessed April 18, 2013.
- Ruskovska T, Bernlohr DA. Oxidative stress and protein carbonylation in adipose tissue - Implications for insulin resistance and diabetes mellitus. J Proteomics. 2013 Apr 11. [Epub ahead of print]
- Andersen DK. Diabetes and cancer: placing the association in perspective. Curr Opin Endocrinol Diabetes Obes. 2013 Apr;20(2):81-6.
- Aleksandrova K, Nimptsch K, Pischon T. Obesity and colorectal cancer. Front Biosci (Elite Ed). 2013 Jan 1;5:61-77.
- Gunter JH, Sarkar PL, Lubik AA, Nelson CC. New players for advanced prostate cancer and the rationalisation of insulin-sensitising medication. Int J Cell Biol. 2013;2013:834684.
- Shikata K, Ninomiya T, Kiyohara Y. Diabetes mellitus and cancer risk: review of the epidemiological evidence. Cancer Sci. 2013 Jan;104(1):9-14.
- Aleksandrova K, Nimptsch K, Pischon T. Influence of Obesity and Related Metabolic Alterations on Colorectal Cancer Risk. Curr Nutr Rep. 2013 Mar;2(1):1-9.
- Seim I, Lubik AA, Lehman ML, et al. Cloning of a novel insulin-regulated ghrelin transcript in prostate cancer. J Mol Endocrinol. 2013 Feb 15;50(2):179-91.
- Friedenreich CM, Langley AR, Speidel TP, et al. Case-control study of markers of insulin resistance and endometrial cancer risk. Endocr Relat Cancer. 2012 Nov 9;19(6):785-92.
- Berryhill GE, Gloviczki JM, Trott JF, et al. Diet-induced metabolic change induces estrogen-independent allometric mammary growth. Proc Natl Acad Sci U S A. 2012 Oct 2;109(40):16294-9.
- Vidal AC, Lund PK, Hoyo C, et al. Elevated C-peptide and insulin predict increased risk of colorectal adenomas in normal mucosa. BMC Cancer. 2012 Sep 5;12:389.
- Campagnoli C, Abbà C, Ambroggio S, Brucato T, Pasanisi P. Life-style and metformin for the prevention of endometrial pathology in postmenopausal women. Gynecol Endocrinol. 2013 Feb;29(2):119-24.
- Rose DP, Vona-Davis L. The cellular and molecular mechanisms by which insulin influences breast cancer risk and progression. Endocr Relat Cancer. 2012 Nov 9;19(6):R225-41.
- Cui Y, Andersen DK. Diabetes and pancreatic cancer. Endocr Relat Cancer. 2012 Sep 5;19(5):F9-F26.
- Arcidiacono B, Iiritano S, Nocera A, et al. Insulin resistance and cancer risk: an overview of the pathogenetic mechanisms. Exp Diabetes Res. 2012;2012:789174.
- Koenuma M, Yamori T, Tsuruo T. Insulin and insulin-like growth factor 1 stimulate proliferation of metastatic variants of colon carcinoma 26. Jpn J Cancer Res. 1989 Jan;80(1):51-8.
- Available at: http://www.huffingtonpost.com/2013/03/06/alzheimers-health-united-states-america-threat_n_2820887.html. Accessed April 18, 2013.
- Blass JP. The mitochondrial spiral. An adequate cause of dementia in the Alzheimer’s syndrome. Ann N Y Acad Sci. 2000;924:170-83.
- Chen JX, Yan SD. Amyloid-beta-induced mitochondrial dysfunction. J Alzheimers Dis. 2007 Sep;12(2):177-84.
- Lustbader JW, Cirilli M, Lin C, et al. ABAD directly links Abeta to mitochondrial toxicity in Alzheimer’s disease. Science. 2004 Apr 16;304(5669):448–52.
- Wang X, Su B, Lee HG, et al. Impaired balance of mitochondrial fission and fusion in Alzheimer’s disease. J Neurosci. 2009 Jul 15;29(28):9090–103.
- Muller WE, Eckert A, Kurz C, Eckert GP, Leuner K. Mitochondrial dysfunction: common final pathway in brain aging and Alzheimer’s disease--therapeutic aspects. Mol Neurobiol. 2010 Jun;41(2-3):159-71.
- Moreira PI, Harris PL, Zhu X, et al. Lipoic acid and N-acetyl cysteine decrease mitochondrial-related oxidative stress in Alzheimer disease patient fibroblasts. J Alzheimers Dis. 2007 Sep;12(2):195-206.
- Fishel MA, Watson GS, Montine TJ, et al. Hyperinsulinemia provokes synchronous increases in central inflammation and beta-amyloid in normal adults. Arch Neurol. 2005 Oct;62(10):1539-44.
- Available at: http://www.hindawi.com/journals/tswj/2012/756357. Accessed April 18, 2013.
- Jovanović Z. Mechanisms of neurodegeneration in Alzheimer’s disease. Med Pregl. 2012 Jul-Aug;65(7-8):301-7.
- Umegaki H. Neurodegeneration in diabetes mellitus. Adv Exp Med Biol. 2012;724:258-65.
- de la Monte SM. Contributions of brain insulin resistance and deficiency in amyloid-related neurodegeneration in Alzheimer’s disease. Drugs. 2012 Jan 1;72(1):49-66.
- Faivre E, Holscher C. Neuroprotective effects of D-Ala2GIP on Alzheimer’s disease biomarkers in an APP/PS1 mouse model. Alzheimers Res Ther. 2013 Apr 19;5(2):20.
- Ghasemi R, Dargahi L, Haeri A, Moosavi M, Mohamed Z, Ahmadiani A. Brain insulin dysregulation: Implication for neurological and neuropsychiatric disorders. Mol Neurobiol. 2013 Jan 20. [Epub ahead of print]
- Bianchi C, Miccoli R, Trombetta MJ. Elevated 1-hour postload plasma glucose levels identify subjects with normal glucose tolerance but impaired β-cell function, insulin resistance, and worse cardiovascular risk profile: The GENFIEV Study. Clin Endocrinol Metab. 2013 Mar 28. [Epub ahead of print]
- Othman EM, Leyh A, Stopper H. Insulin mediated DNA damage in mammalian colon cells and human lymphocytes in vitro. Mutat Res. 2013 Mar 21. [Epub ahead of print]
- Rhee EJ, Lee WY, Cho YK, Kim BI, Sung KC. Hyperinsulinemia and the development of nonalcoholic fatty liver disease in nondiabetic adults. Am J Med. 2011 Jan;124(1):69-76.
- Sechi LA, Catena C, Zingaro L, Melis A, De Marchi S. Abnormalities of glucose metabolism in patients with early renal failure. Diabetes. 2002 Apr;51(4):1226-32.
- Kobayashi S, Maesato K, Moriya H, Ohtake T, Ikeda T. Insulin resistance in patients with chronic kidney disease. Am J Kidney Dis. 2005 45(2):275-80.
- Kurella M, Lo JC, Chertow GM. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J Am Soc Nephrol. 2005;16(7):2134-40.
- Cheng HT, Huang JW, Chiang CK, Yen CJ, Hung KY, Wu KD. Metabolic syndrome and insulin resistance as risk factors for development of chronic kidney disease and rapid decline in renal function in elderly. J Clin Endocrin Metab. 2012;97(4):1268-76.
- de la Monte SM, Longato L, Tong M, Wands JR. Insulin resistance and neurodegeneration: roles of obesity, type 2 diabetes mellitus and non-alcoholic steatohepatitis. Curr Opin Investig Drugs. 2009 Oct;10(10):1049-60.
- S Roriz-Filho J, Sá-Roriz TM, Rosset I, et al. (Pre)diabetes, brain aging, and cognition. Biochim Biophys Acta. 2009 May;1792(5):432-43.
- Abdelmoneim SS, Hagen ME, Mendrick E, et al. Acute hyperglycemia reduces myocardial blood flow reserve and the magnitude of reduction is associated with insulin resistance: a study in nondiabetic humans using contrast echocardiography. Heart Vessels. 2012 Nov 23.
- Reaven G. Insulin resistance and coronary heart disease in nondiabetic individuals. Arterioscler Thromb Vasc Biol. 2012 Aug;32(8):1754-9.
- Sasaki M, Joh T, Koikeda S, et al. A novel strategy in production of oligosaccharides in digestive tract: prevention of postprandial hyperglycemia and hyperinsulinemia. J Clin Biochem Nutr. 2007 Nov;41(3):191-6.
- Ao Z, Simsek S, Zhang G, Venkatachalam M, Reuhs BL, Hamaker BR. Starch with a slow digestion property produced by altering its chain length, branch density, and crystalline structure. J Agric Food Chem. 2007 May 30;55(11):4540-7.
- Ota M, Okamoto T, Wakabayashi H. Action of transglucosidase from Aspergillus niger on maltoheptaose and [U-(13)C]maltose. Carbohydr Res. 2009 Mar 10;344(4):460-5.

