
Sleep Apnea
Sleep Apnea
Last Section Update: 07/2025
Contributor(s): Shayna Sandhaus, PhD; Stephen Tapanes, PhD
1 Overview
Summary and Quick Facts for Sleep Apnea
- Sleep apnea is a disorder characterized by disruptions in normal breathing during sleep. These breathing interruptions often cause sleep disturbances throughout the night, including frequent waking and snoring. Sleep apnea is associated with increased risk for hypertension, heart disease, diabetes, poor cognitive function, and many other health concerns.
- In this protocol, you will learn about the different causes of sleep apnea and why it is important to have the condition properly diagnosed and treated. You will also learn about lifestyle and dietary habits that can supplement traditional treatment of sleep apnea. Dietary supplements that target some of the factors that contribute to sleep apnea will be reviewed as well.
- Effective sleep apnea treatments are available, and they can greatly improve quality of life. But many people either do not seek treatment or give up quickly due to inconvenience or discomfort. It is important to work with a qualified sleep specialist to find the right solution for you, which may take trial and error.
What is Sleep Apnea?
Sleep apnea is an increasingly common but underdiagnosed disorder characterized by repeated interruptions in normal breathing during sleep. It is associated with increased risk for several serious health problems, including:
- hypertension
- cardiovascular disease
- type 2 diabetes
- cancer
- gout
- cognitive dysfunction
- autoimmune disorders
- pregnancy complications
Fortunately, effective treatment options are available for sleep apnea, and dietary and lifestyle changes can reduce the severity of disease. Emerging research also suggests that integrative interventions, such as antioxidants, can improve sleep quality and respiratory function in patients with sleep apnea.
What are Risk Factors for Developing Sleep Apnea?
- Overweight and obesity
- Male sex
- Asian or Hispanic ethnicity
- Allergies
- Older age
- Anatomical abnormalities in the airway
What Signs and Symptoms are Associated with Sleep Apnea?
- Snoring/gasping/choking sounds while sleeping
- Pauses in breathing while sleeping
- Intermittent waking and insomnia
- Nocturia (frequent waking to use the restroom)
- Daytime sleepiness or fatigue
- Dry mouth, sore threat, or headache upon waking
- Difficulty concentrating or remembering things
- Irritability or depression
- Sexual dysfunction
- Refer to the STOP-BANG questionnaire in the Screening section of this protocol
What are Conventional Medical Treatments for Sleep Apnea?
- CPAP (continuous positive airway pressure) and positive airway pressure variations
- Oral appliances to help keep the upper airway open
- Surgery for when the obstruction is due to an anatomical problem, such as enlarged tonsils or adenoids
- Pharmacotherapy
- Hypoglossal nerve stimulation
What are Novel and Emerging Therapies for Sleep Apnea?
- Acetazolamide
- Renal denervation
- Nasal expiratory positive airway pressure therapy
- Oral negative pressure therapy
- Hormone replacement therapy (for women)
What Dietary and Lifestyle Changes Can Be Beneficial for Sleep Apnea?
- Weight loss
- Diet (Mediterranean diet, low-sodium diet)
- Exercise
- Alcohol avoidance
- Side sleeping or at least turning the head to the side while sleeping on your back
- Acupuncture
- Smoking cessation
What Integrative Interventions May Be Beneficial for Sleep Apnea?
- N-acetylcysteine (NAC): Supplementation with the antioxidant NAC has been shown to ameliorate signs and symptoms of sleep apnea, including apnea-related arousals, daytime sleepiness, and snoring, among others.
- Coenzyme Q10 (CoQ10): CoQ10, an antioxidant with beneficial effects on the cardiovascular system and metabolism, may benefit sleep apnea patients at increased risk for cardiovascular disease, hypertension, and impaired glucose tolerance.
- Vitamin D: Studies have found low vitamin D levels are associated with increased risk for insulin resistance, diabetes, and metabolic syndrome among patients with obstructive sleep apnea.
- Omega-3 fatty acids: Sufficient levels of omega-3 fatty acids are associated with a host of cardiovascular and anti-inflammatory benefits, which may be important for individuals with sleep apnea.
- B vitamins: Supplementation with B vitamins has been shown to reduce levels of serum homocysteine, an amino acid associated with increased risk of cardiovascular and neurogenerative diseases and shown to be elevated in patients with obstructive sleep apnea.
- Probiotics: Studies involving animal models of sleep apnea and intermittent hypoxia have found that probiotics can improve some side effects of systemic inflammation in the heart and brain, as well as hypertension.
2 Introduction
Sleep apnea is characterized by repeated disruptions in normal breathing during sleep.1 These frequent respiratory interruptions result in periods of low blood oxygen levels, nervous system dysfunction, and sleep fragmentation. People with sleep apnea often stop breathing periodically then suddenly gasp for air during sleep; loud snoring is also common in people with sleep apnea and is often reported by bed partners of those affected.
There are two main types of sleep apnea: obstructive and central. Obstructive sleep apnea is much more common and is associated with increased risk of several major health problems and greater mortality risk.2
Epidemiological studies have historically estimated the prevalence of obstructive sleep apnea to be around 3% to 7% in men and 2% to 5% in women, but it is believed to be vastly underdiagnosed.3 The National Healthy Sleep Awareness Project estimates that at least 25 million adults in the United States are living with obstructive sleep apnea.4
Obstructive sleep apnea can lead to several health concerns as a result of chronic sleep deprivation: daytime sleepiness, cognitive difficulties, depression, and increased risk of accidents and injuries.1,5 In addition, a significant and growing list of serious chronic diseases have been associated with sleep apnea, including cardiovascular and neurological diseases, obesity, polycystic ovary syndrome, and insulin resistance and type 2 diabetes.1,5,6
The first-line treatment approach for obstructive sleep apnea is a device that provides continuous positive airway pressure, known as CPAP.5-7 The device is worn during sleep and introduces mildly pressurized air into the airways to keep them open. CPAP therapy can be cumbersome and uncomfortable, so recommendations for CPAP are generally reserved for patients who experience excessive daytime sleepiness, those with comorbid hypertension, and those for whom their sleep disruption affects their quality of life.7 Research suggests CPAP use might improve blood pressure and cognitive function for those with sleep apnea.8-11
Some patients are unable to tolerate CPAP, in which case other therapies may be considered. Dental devices and surgical procedures to open the airway may be beneficial in some people with sleep apnea. Other methods sometimes recommended include variations of positive airway pressure and negative pressure devices.7
Dietary and lifestyle interventions can have a significant impact on the severity and consequences of sleep apnea as well. Weight loss is the most common and well-supported adjunct to CPAP, and can provide additional benefits by reducing inflammation.12 Exercise, a lateral sleep position, avoidance of alcohol and sedatives, and a generally healthy diet may also help manage symptoms and severity of sleep apnea. Integrative interventions may both reduce the severity of sleep apnea and help minimize the consequences of some of the serious conditions associated with sleep apnea.
In this protocol, you will learn about the causes of sleep apnea and factors that increase sleep apnea risk. Conventional treatment approaches will be reviewed, as will several novel and emerging treatment strategies. A number of lifestyle considerations and natural, integrative interventions that may lessen the impact of sleep apnea will be discussed as well.
3 Background
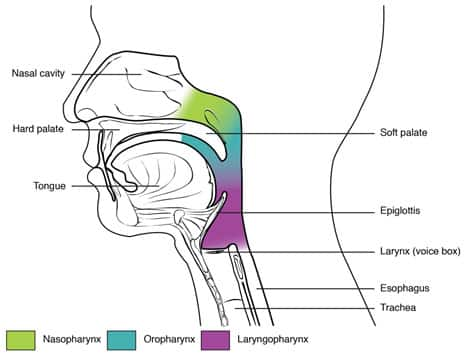
Obstructive sleep apnea is innately an anatomical disorder driven by abnormalities in the upper airway that increase collapsibility and reduce airway function. Enlargement of soft tissues within and around the airway, including the tongue, soft palate, tonsils, and fat deposits, can cause narrowing of the airway.14 The epiglottis, which prevents food from entering the airway, normally remains open during breathing, but may collapse during sleep and is also a frequent source of obstruction and snoring.15 Many groups have attempted to correlate the anatomical sources of obstruction with obstructive sleep apnea severity with limited success.16 However, the nature of the obstruction may have significant implications for treatment strategies, and a comprehensive assessment of the underlying pathophysiology of obstructive sleep apnea may facilitate an individualized approach to treatment.17
Types of Sleep Apnea
There are two main types of sleep apnea: obstructive and central, with obstructive being much more common.18 In obstructive sleep apnea, the upper airway intermittently becomes fully or partially blocked during sleep. In central sleep apnea, the respiratory control centers in the brain do not provide sufficient stimulation to maintain regular breathing during sleep. In some cases, referred to as complex sleep apnea, elements of both obstructive and central sleep apnea occur simultaneously.19,20
Obstructive sleep apnea. In obstructive sleep apnea, anatomical abnormalities and sleep-associated neuromuscular dysfunction coalesce to result in repeated airway blockages.21 The temporary pause in airflow results in frequent arousal as the brain reacts to reinitiate breathing. Obstructive sleep apnea is a state-dependent condition that occurs only during periods of sleep, primarily due to the positioning; when in the supine position (on the back), gravity promotes collapsibility of the airway and blocks airflow.22 Obstructive sleep apnea is very common among adults. A community-based study of over 5,800 adults 40 years or older found that over 46% of subjects had some degree of obstructive sleep apnea, suggesting that the condition is vastly underdiagnosed.18
Central sleep apnea. The natural breathing reflex is regulated by the autonomic nervous system and responds automatically to changes in blood carbon dioxide levels.23 In central sleep apnea, however, the brain does not adequately respond to the excitatory signals that trigger respiration, including carbon dioxide levels, and regulation of breathing is disrupted.24 Disordered breathing is thought to occur only during sleep because sleep diminishes the brain’s response to excitatory signals.25 Central sleep apnea occurs primarily in infants (both preterm and term) in the first weeks of life, adults abruptly transitioning to high altitudes, and patients with heart failure, but it may also be idiopathic in nature (no clear cause).24 Compared with obstructive sleep apnea, central sleep apnea is much less common, occurring in less than 1% of adults.18
Mixed or complex sleep apnea. Mixed sleep apnea, also referred to as complex sleep apnea, involves both obstructive and central components. Approximately 5.5% of people with sleep-disordered breathing—the broad category that includes apneas as well as less severe hypopneas (shallow and/or slow breathing)—have complex sleep apnea.18,24 In many cases, central apnea is revealed during treatment of the obstructive aspect of sleep apnea.20
4 Association with Other Health Conditions
Sleep apnea is associated with several serious health conditions, and at least some of these associations appear to be causal to some degree. This means sleep apnea may directly contribute to the development of the associated health condition, although this may not always be clearly delineated in each individual patient.
Conditions Associated with Sleep Apnea
High blood pressure (hypertension). Historically, there has been conflicting evidence regarding an association between sleep apnea and blood pressure. For example, in a cross-sectional analysis of over 6,000 participants in the Sleep Heart Health Study, participants with severe sleep apnea were 37% more likely to experience hypertension than those without evidence of sleep apnea.26 However, in a 5-year follow-up of a subset of these patients without hypertension at the start of the study, there was no association between the development of hypertension and sleep apnea after accounting for body mass index (BMI), suggesting this association may instead be caused by obesity.27
A direct association between obstructive sleep apnea and hypertension is most strongly supported by studies that demonstrate that treatment of obstructive sleep apnea also lowers blood pressure. Meta-analyses of clinical trials have found that, on average, treatment of obstructive sleep apnea using CPAP results in a 2.5‒7.2 mm Hg reduction in systolic blood pressure and a 2.0‒4.99 mm Hg reduction in diastolic blood pressure.8-10 These effects are small, but may have significant implications for heart health, as 1‒2 mm Hg reductions in blood pressure have previously been shown to decrease the risk for cardiovascular diseases, including stroke and heart failure.28 The association between sleep apnea and blood pressure is likely bidirectional and driven by a number of factors, including overactivation of the sympathetic nervous system, oxidative stress resulting from intermittent hypoxia (periods of low oxygen levels), and disruption of hormone levels.29
Cardiovascular disease. Sleep apnea, and particularly obstructive sleep apnea, is highly prevalent in patients with cardiovascular disease (CVD), including heart failure, stroke, atrial fibrillation, and end-stage renal disease.30 Intermittent hypoxia is thought to cause endothelial damage, inflammation, and metabolic dysfunction, which contribute to an increased risk for CVD.31 There has been some conflicting evidence, however, on whether treatment of sleep apnea improves cardiovascular outcomes. In a clinical trial of 2,717 adults with moderate-to-severe obstructive sleep apnea and CVD, management with CPAP did not reduce the frequency of CVD events.32 Similar results were seen in a smaller clinical trial of 244 patients with obstructive sleep apnea and coronary artery disease; however, when results were adjusted for adherence to treatment (ie, comparing those who used CPAP for <4 hours per night with those who used it for ≥4 hours per night), CPAP reduced CVD risk by 71%.33 A meta-analysis of eight studies on obstructive sleep apnea found CPAP reduced the risk for atrial fibrillation by 42%, providing further support that CPAP, when used properly, reduces CVD risk in patients with obstructive sleep apnea.34 The cardiovascular benefits of CPAP and other sleep apnea treatments in patients with central sleep apnea and heart failure, however, are less clear.35
Obesity and type 2 diabetes. Obesity is a well-established risk factor for obstructive sleep apnea, and the relationship between obesity and sleep apnea is likely bidirectional. Disordered sleep has been found to increase the risk for weight gain through physiological and hormonal changes, as well as behavioral changes.36 Obstructive sleep apnea is also closely associated with type 2 diabetes. Approximately 54% to 75% of patients with type 2 diabetes have obstructive sleep apnea, and type 2 diabetes patients are 48% more likely to have obstructive sleep apnea than individuals without diabetes.37-39 Additionally, type 2 diabetes patients with obstructive sleep apnea are over three times more likely to develop diabetes-related complications than those without.38 This relationship likely reflects the complicated interplay among metabolic changes, weight gain, and hypertension.38,39 Intermittent hypoxia may also cause insulin resistance.40 There have been conflicting results regarding the ability of CPAP treatment to improve glycemic control in patients with type 2 diabetes.41-44 However, preliminary results suggest treatment of type 2 diabetes with dapagliflozin (Farxiga) (a sodium-glucose cotransporter-2 (SGLT-2) inhibitor, a class of medicine that lowers high blood glucose levels in people with type 2 diabetes) can improve sleep and decrease oxygen desaturation events in patients with type 2 diabetes and sleep-disordered breathing, suggesting that treatment of comorbid type 2 diabetes may improve sleep apnea symptoms as well.45,46
Obesity hypoventilation syndrome is a condition characterized by BMI ≥30 kg/m2, elevated daytime carbon dioxide levels, and sleep-disordered breathing.47 While this disorder is often closely associated with the presence of sleep apnea, and particularly obstructive sleep apnea, it is a separate clinical entity.
Cognitive deficits and mood disorders. Studies suggest obstructive sleep apnea may be associated with the development of cognitive deficits in areas including attention, executive function, visuospatial reasoning, and both short- and long-term memory.48 This may be driven by a number of factors, including chronic sleep deprivation and hypertension.49 An estimated 27% of individuals with mild cognitive impairment (MCI) have obstructive sleep apnea, which may accelerate the progression from MCI to Alzheimer disease in some affected individuals.50 Results from a small clinical trial involving 54 individuals with MCI and obstructive sleep apnea suggest proper adherence to CPAP treatment is associated with small but significant improvements in memory, attention, and everyday function.11
Obstructive sleep apnea has also been linked to several affective disorders, including anxiety and depression.51-53
Evolving Concepts of Disease Association with Sleep Apnea
Cancer. The incidences of certain cancers, including kidney, melanoma, and breast cancers, are higher in patients with obstructive sleep apnea than in the general population.66,67 In a study that followed 390 adults over 20 years, moderate-to-severe obstructive sleep apnea was associated with a 2.5-fold increased risk for cancer diagnosis and a 3.4-fold increased risk for cancer death.68 In another study, which followed over 1,500 individuals for up to 22 years, those with moderate sleep-disordered breathing had a 2-fold higher risk of dying from cancer, and those with severe sleep-disordered breathing had a 4.8-fold higher risk, compared with healthy subjects.69 Intermittent hypoxia has been proposed as a driver for cancer development in obstructive sleep apnea patients, but the exact cause of this association remains unclear.70
Gout and hyperuricemia. Gout is a painful arthritic condition caused by an excess of uric acid in the blood (hyperuricemia), which accumulates as crystals in the joints and causes inflammation.71 People with gout appear to be at increased risk of sleep apnea; an analysis of US Medicare beneficiaries found that among adults 65 years or older, individuals with gout were over twice as likely to develop new obstructive sleep apnea.72 This association is likely bidirectional, as several studies have found that patients with obstructive sleep apnea experience incident cases of gout up to 64% more frequently than adults without sleep apnea.73,74 Furthermore, individuals with high uric acid levels have been shown to be more likely to snore more than five nights per week and to experience daytime sleepiness.75 Serum uric acid levels have been found to be elevated in individuals with obstructive sleep apnea, and increasing uric acid levels are strongly associated with the presence of obstructive sleep apnea and sleep apnea risk factors.76,77 In addition to gout, hyperuricemia in obstructive sleep apnea is also associated with an increased risk for CVD.78 Further research is needed on the implications of this relationship, though, as studies suggest CPAP therapy does not reduce uric acid levels.79
Pregnancy complications. Sleep-disordered breathing, including obstructive sleep apnea, is common in pregnancy, often characterized by snoring, fatigue, and daytime sleepiness as pregnancy progresses.80 A growing body of evidence suggests that in pregnant women, sleep-disordered breathing may have effects on the health of both the mother and fetus, but this remains controversial.80-82 However, in a study of 3,705 pregnant women, sleep-disordered breathing was associated with increased risk of gestational hypertension, gestational diabetes, and preeclampsia.83
Low testosterone levels and erectile dysfunction. Several studies have confirmed that serum testosterone levels are often lower in men with obstructive sleep apnea.84 One study found that men with obstructive sleep apnea had 17% lower serum testosterone levels than controls.85 Another study, which compared 36 men with obstructive sleep apnea with healthy controls, found that lower serum testosterone levels were associated with greater sleep apnea severity and increased BMI.86 Decreased testosterone levels may also explain the observation that men with obstructive sleep apnea have a 55% increased risk for erectile dysfunction than men without obstructive sleep apnea.87 This effect is likely driven by other factors as well, such as endothelial dysfunction, as CPAP has been shown to improve some measures of erectile dysfunction but is not associated with elevations in testosterone levels.88,89
Other conditions. Obstructive sleep apnea has also been associated with glaucoma,90 Parkinson’s disease,91 polycystic ovary syndrome (PCOS),92 hypothyroidism,93 kidney stones,94 and chronic kidney disease.95
5 Risk Factors
Obesity
Obesity is the number one risk factor for obstructive sleep apnea. Obesity is so strongly associated with obstructive sleep apnea that, compared with CPAP management, weight loss alone improved measures of inflammation and insulin resistance in patients with obstructive sleep apnea.12 Obesity is thought to contribute to increased risk for obstructive sleep apnea by compromising the airway and decreasing lung volume. Excess fat deposits in the upper airway, including the pharynx, tongue, soft palate, and uvula, can lead to narrowing of the airway and may promote collapsibility.14 Studies of individuals with a family history of obstructive sleep apnea suggest that, while genetics may play a role in the heritability of the disease, familial aggregation of obstructive sleep apnea is primarily driven by patterns of obesity within families.99
Sex and Hormone Levels
Male sex is a significant risk factor for obstructive sleep apnea, with males being twice as likely to develop obstructive sleep apnea as females.1 The increased prevalence of disease in males, coupled with the observations that postmenopausal women and women with PCOS are at increased risk for obstructive sleep apnea relative to their healthy premenopausal counterparts, has led some to suggest that estrogen may play a protective role against the development of obstructive sleep apnea.100,101 However, experimental evidence to support this theory is lacking, and more information is needed on estrogen’s role in the development of sleep apnea.102
Ethnicity
Results of a multiethnic study involving 2,230 adults aged 54 years or older found that individuals of Chinese and Hispanic ethnicities were at increased risk for sleep-disordered breathing compared with White and Black individuals. Additionally, Chinese individuals were 37% more likely to develop severe sleep-disordered breathing. Some of this disparity may be due to differences in access to care, though, as individuals of Chinese ethnicity were 49% less likely to receive a diagnosis of sleep apnea from their doctor compared with their White counterparts.103
Age
The risk for sleep apnea increases with age. Individuals over 50 years of age are considered to be at increased risk for obstructive sleep apnea.1 This may be due to factors such as diminished efficiency of muscles and soft tissues in the upper airway or reduced sleep quality.104
Anatomy
Individuals with anatomical abnormalities of the bony and soft tissues in the head or neck are at high risk for obstructive sleep apnea. Additionally, a neck circumference greater than 40 cm is considered a risk factor for obstructive sleep apnea.1
Nasal Allergies (Allergic Rhinitis)
In a study in over 1,900 individuals with asthma, allergic rhinitis was associated with 1.44-fold higher odds of obstructive sleep apnea compared with those with asthma alone. When allergic rhinitis duration and severity were taken into account, the odds rose to almost 2-fold.105 Allergic (and non-allergic) rhinitis is thought to increase the risk for obstructive sleep apnea by a combination of increased airway resistance due to inflammation and a reduction in pharyngeal diameter resulting from increased mouth breathing.106
Dietary Habits
Among individuals with sleep apnea, certain dietary habits have been linked to poorer quality of sleep. An analysis of a multiethnic cohort of over 1,800 individuals found obstructive sleep apnea was associated with lower intake of whole grains, increased consumption of processed red meats, and lower overall quality of diet. This association was at least partially driven by a decrease in slow-wave sleep with red meat consumption.107 Additionally, studies have linked later evening meals with the presence and severity of obstructive sleep apnea.108,109
6 Signs and Symptoms
Sleep apnea often goes undiagnosed largely because the direct symptoms are witnessed only by bedmates, and the indirect symptoms, such as those related to sleep deprivation, can be vague or attributable to many possible causes.1 The most common sleep apnea symptoms include1,6,110:
- Snoring. Loud or irregular snoring often indicates the presence of sleep apnea, and it is more pronounced in obstructive sleep apnea. Apneas may also interrupt snoring and cause the patient to snort.
- Pauses in breathing. Sleep apnea often causes pauses in breathing during sleep that are noticed by another person. Someone with sleep apnea may pause breathing for a period during sleep then gasp for air.
- Frequent nighttime waking and insomnia. It may be difficult to remain asleep. Patients may also awaken suddenly with shortness of breath. Poor sleep and insomnia are often the primary symptoms of central sleep apnea. Nocturia, or frequently waking to go to the bathroom, is also common.
- Dry mouth, sore throat, and headache upon waking.
- Fatigue and excessive daytime drowsiness. Patients with sleep apnea are susceptible to falling asleep at work and while driving.
- “Brain fog.” This can include difficulty concentrating, reasoning, and paying attention.
- Feeling moody, depressed, or irritable.
- Lack of libido or sexual dysfunction.
7 Sleep Apnea Diagnosis & Sleep Monitoring
Screening
Despite the high prevalence of sleep apnea and its association with serious health conditions, obstructive sleep apnea is underdiagnosed and many patients remain untreated.111 Women are disproportionately underdiagnosed; despite a 2:1 ratio in the prevalence of sleep apnea in men versus women, women are 70% less likely to receive treatment than men, even with evidence of sleep-disordered breathing.112 Therefore, simple clinical screening for obstructive sleep apnea, particularly in high-risk groups, is important.
Several screening tools have been developed and validated to identify patients at high risk for obstructive sleep apnea, as well as the likelihood of clinical complications of obstructive sleep apnea in pre-surgical and hospital settings. One of the most widely used screening tools is STOP-BANG, which stands for “Snoring, Tiredness during daytime, Observed apnea, high blood Pressure, BMI >35 kg/m2, Age >50 years, Neck circumference >40 cm, and male Gender.”1 The STOP-BANG test is graded on a yes/no basis, with each “yes” being equal to 1 point. A score of 3 or greater indicates high risk for obstructive sleep apnea, and a score of 5 or more indicates high risk for moderate-to-severe obstructive sleep apnea.1,113
Table 1: The STOP-BANG Obstructive Sleep Apnea Questionnaire*
|
|
Yes |
No |
|
Snoring: Do you snore loudly (louder than talking or loud enough to be heard through closed doors)? |
||
|
Tired: Do you often feel tired, fatigued, or sleepy during daytime? |
||
|
Observed: Has anyone observed you stop breathing during your sleep? |
||
|
Blood Pressure: Do you have or are you being treated for high blood pressure? |
||
|
BMI: BMI greater than 35 kg/m2? |
||
|
Age over 50 years old? |
||
|
Neck circumference greater than 40 cm (approx. 16 in.)? |
||
|
Gender male? |
||
|
Scoring criteria: |
||
Low risk of obstructive sleep apnea: Yes to 0 to 2 questions |
||
Intermediate risk of obstructive sleep apnea: Yes to 3 to 4 questions |
||
High risk of obstructive sleep apnea: Yes to 5 to 8 questions |
||
|
*Adapted from: Jaspal Singh MD, Nicole M. Screening Tools for the Obstructive Sleep Apnea for the Cardiovascular Clinician - American College of Cardiology114 and Kline LR. Clinical presentation and diagnosis of obstructive sleep apnea in adults. UpToDate.115 |
||
Although screening tools are often highly sensitive for prediction of obstructive sleep apnea, they are not a replacement for formal testing. Additionally, before diagnosing sleep apnea, physicians should rule out a number of other conditions that can cause similar symptoms, including restless leg syndrome, gastroesophageal reflux disease, narcolepsy, swallowing disorders, mood disorders, or obesity hypoventilation.
Testing & Monitoring
There are two main ways to assess for sleep apnea: polysomnography (a sleep study—the traditional, “gold-standard” method) and FDA-cleared home sleep apnea tests (HSATs).231 Newer consumer devices with only one point of contact, such as rings or wristbands (including smartwatches), may be designed to monitor some sleep parameters but cannot definitively diagnose sleep apnea or substitute for evaluation and diagnosis by a clinician.232 Polysomnography and HSATs both have their own advantages and drawbacks; therefore, the preferred approach may vary depending on the person and the practitioner’s clinical judgement. Alternatives to polysomnography or HSATs are not recommended for sleep apnea testing.
Polysomnography. The definitive test for sleep apnea is an overnight polysomnography, often called a “sleep study.” This test is done in a clinical laboratory setting and monitors the occurrence of apneas (paused breathing) and hypopneas (shallow and/or slow breathing) by measuring respiratory effort, breathing patterns, and blood oxygen saturation.6 Polysomnography also includes neurological testing to monitor limb movements, body position, and the sleep-wake state.116 It may also include electroencephalography (EEG), electrooculography (EOG), electromyography (EMG), and airflow measurements.233 The American Academy of Sleep Medicine (AASM) considers polysomnography to be the “gold standard” for evaluating sleep and sleep-related breathing disorders; however, it has limitations including expense, availability, and the intrusiveness of the test and test environment, the latter of which can have a deleterious effect on the patients’ ability to sleep during the evaluation.234-236
When people sleep in a new environment for the first time, they often experience the “first night effect,” which is a phenomenon characterized by overall poorer sleep quality and altered sleep architecture.237,238 In an observational study involving 852 individuals who underwent polysomnography, about half reported experiencing the first night effect, roughly 20% showed a “reverse first night effect” (meaning they slept better than usual), and the remaining 30% reported no significant change in their sleep.239
At-home tests. Home sleep apnea tests (HSATs) are designed to be alternatives to polysomnography for people suspected of sleep apnea that do not have significant comorbidities such as cardiopulmonary disease, neuromuscular conditions, or chronic opioid use. If the HSAT is negative, and sleep apnea is still suspected, a polysomnography is recommended to ensure accuracy of the results.235 Examples of FDA-cleared, clinically validated HSATs include the WatchPAT, NightOwl, Sunrise, and ARES devices.233 These devices measure various respiratory parameters at one or more points of contact such as the fingertip, chin, or chest.240-242 According to the AASM, HSATs should incorporate a minimum of three parameters: either oximetry (blood oxygen concentration), nasal pressure, and chest and abdominal respiratory inductance plethysmography, or, peripheral arterial tonometry (measurement of pulse changes because of narrowing or widening of blood vessels), oximetry, and actigraphy (rest vs. activity).241
A meta-analysis published in 2022 found a significant number of false negatives and false positives in studies using HSATs that measure peripheral arterial tonometry.243
The WatchPAT device is one of the more intensively studied HSATs. A meta-analysis of 24 observational studies, including a total of 1,644 people, found no significant differences in predictive correlations between the WatchPAT 100 or 200 and polysomnography for apnea-hypopnea index, oxygen saturation, oxygen desaturation index (how often blood oxygen levels drop), respiratory disturbance index (abnormal breathing events per hour), and lowest oxyhemoglobin saturation (lowest point of oxygen desaturation). Oxygen saturation was the only measure that did not significantly correlate between the two tests.244 As of mid-2025, the WatchPAT 300 and the WatchPAT ONE are the latest models (both launched in 2019). Compared with the WatchPAT 200, the 300 is more compact; includes a chest sensor; and can measure sleep architecture (REM/non-REM), sleep efficiency, sleep latency, and REM latency.245 The WatchPAT One is the world’s first fully disposable HSAT.246
Multi-purpose wearable devices. Sleep devices with only one point of contact, that are designed to do more than just measure parameters related to sleep, are less accurate than HSATs or polysomnography.
A meta-analysis published in 2025 evaluated 24 studies, totaling 798 people, that compared at least one of the following wrist-worn devices to polysomnography: Fitbit, Jawbone, myCadian watch, WHOOP strap, Garmin, Basis B1, Zulu Watch, Huami Arc, E4 wristband, Fatigue Science Readiband, Apple Watch (Series 8), or Xiaomi Mi Band 5. The results showed a significant difference in measured total sleep time, sleep efficiency, sleep latency, and wake after sleep onset between wrist-worn devices and polysomnography. The wrist-worn sleep devices were concluded to not be as reliable as polysomnography in measuring key sleep parameters.247 A clinical study compared three popular devices (Oura Ring Gen3, Fitbit Sense 2, and Apple Watch Series 8) to polysomnography. A total of 35 healthy people without a sleeping disorder were enrolled in a single night polysomnography during which they wore all three devices at once. The Oura Ring was not different from the polysomnography in wake, light sleep, deep sleep, or REM sleep estimation. The Fitbit overestimated light sleep and underestimated deep sleep. The Apple Watch underestimated the duration of deep sleep and overestimated light sleep.248
Multi-purpose wearable devices that have received FDA clearance for use in monitoring sleep parameters intended to inform users of potential sleep apnea risk include the Samsung Galaxy Watch and certain models of the Apple Watch.249,250
Classification
The criteria for diagnosis of sleep apnea are based on the frequency of apneas and hypopneas per hour that last ≥10 seconds, known as the apnea-hypopnea index (AHI).6 An AHI of 5 or greater is considered clinically definitive for a diagnosis of sleep apnea.122 Classification of mild, moderate, or severe sleep apnea is based on a combination of clinical presentation and sleep study results.
Table 2: American Academy of Sleep Medicine Obstructive Sleep Apnea Classifications122
|
Classification |
AHI |
Clinical Presentation and Associated Risks |
|
Mild* |
5–15 episodes per hour |
|
|
Moderate |
15–30 episodes per hour |
|
|
Severe |
>30 episodes per hour |
|
|
AHI, apnea-hypopnea index
| ||
Obstructive sleep apnea may also be classified based on a combination of phenotype and endotype.17,123 The phenotype of the disease represents the observable characteristics of the disease or patient (such as obstructive sleep apnea in the elderly, men, or Black patients), whereas the endotype refers to the underlying biological mechanisms which drive disease (such as fat distribution, hyporesponsive muscles of the airway, or abnormal craniofacial structures). These classifications are not based on the severity of disease, although certain phenotypes may correlate with increased risk for more severe disease.
8 Conventional Treatments
CPAP: Continuous Positive Airway Pressure
CPAP is first-line treatment for obstructive sleep apnea.7 Numerous studies show that this method of applying constant mild air pressure to the airway during sleep reduces apneic and hypopneic episodes and helps relieve the signs and symptoms of sleep loss, such as fatigue, drowsiness, and cognitive and mood changes.124 Studies suggest these benefits are associated with significant improvements in quality of life, particularly for those with moderate-to-severe obstructive sleep apnea.124,125 The more hours per night a CPAP device is used, the greater the benefit.126
One barrier to consistent CPAP use is comfort, which may affect adherence. It is estimated that only about 66% of people who are prescribed CPAP are properly adherent, which has remained largely unchanged over the past two decades.127 In addition to the cumbersome nature of the masks, CPAP treatment is associated with dry mouth, nasal discharge, congestion, bloody nose, headache, sore throat, hoarse voice, and reduced sense of smell.7 People who tolerate the CPAP mask and straps but find the constant air pressure uncomfortable may prefer one of two other options for positive airway pressure. The first is called bilevel positive airway pressure, or BiPAP. With BiPAP therapy, a lower level of air pressure is used during expiration, and a higher level during inspiration. This may improve comfort, especially for people who use higher pressure settings with CPAP and have trouble exhaling against the pressure, and is considered an effective strategy for treatment of obstructive sleep apnea. The second is called auto-adjusting positive airway pressure, or APAP. With APAP therapy, the device automatically adjusts its pressure output to the pressures it senses during the breathing cycle and reduces the pressure applied when no events are detected over a period of time. APAP may be more comfortable for people whose apnea occurs mainly in certain sleep positions or in response to variable triggers, such as alcohol consumption, allergies, and upper respiratory infections.124 Clinical guidelines recommend the use of CPAP or APAP over BiPAP when possible, as lower pressure during expiration may not prevent the occurrence of airway collapse. BiPAP therapy is also more expensive than CPAP or APAP which may represent a barrier for many patients.7
The cardiovascular benefits of CPAP have been somewhat controversial. Meta-analyses of clinical trials have collectively found that CPAP therapy is associated with small but significant reductions in both systolic and diastolic blood pressure, particularly in those with moderate-to-severe obstructive sleep apnea.8-10,124 However, the effects on the frequency of cardiovascular events such as stroke or heart attack remain less clear. Some retrospective studies suggest CPAP treatment may reduce the risk for cardiovascular events, but other observational studies and, most importantly, randomized clinical trials have not found a significant reduction in the frequency of cardiovascular events and CVD in obstructive sleep apnea with CPAP use.32,128-131 However, cardiovascular benefits may be greater in patients with more severe obstructive sleep apnea, who are often excluded from clinical trials.124 Additionally, in a small study of 244 patients with coronary artery disease and obstructive sleep apnea, a significant reduction in cardiovascular events was found when adjusting for adherence, suggesting increased use of CPAP may confer better cardiovascular benefits.33
Clinical trials have demonstrated that CPAP improves insulin sensitivity in patients with severe obstructive sleep apnea,132,133 but the effects of CPAP on blood glucose control are less convincing.124 A 6-month clinical trial examining the use of CPAP in individuals with obstructive sleep apnea and type 2 diabetes found that patients treated with CPAP experienced greater reductions in glycated hemoglobin levels compared with control patients.41 However, many other trials and studies suggest CPAP use has no effect on glycemic control, and the American Academy of Sleep Medicine has concluded that there is insufficient evidence to support the use of CPAP for glycemic control in adults with obstructive sleep apnea.42-44,124,133
Oral Appliances
Oral appliances, the most common of which is the mandibular advancement device (MAD), are used during sleep to physically keep the upper airway open by either holding the lower jaw forward or moving the tongue forward.134 These devices are usually custom-fitted by a dentist and are more likely to help people who have mild-to-moderate sleep apnea and are female, younger, and carry less excess weight.134,135 They are particularly effective for patients who complain of snoring, regardless of apnea status.135 Although not considered a first-line treatment for obstructive sleep apnea, oral appliances are sometimes used in those who are unable to tolerate CPAP.124
Upper Airway Surgery
Surgery can be an important part of the treatment approach for sleep apnea when the obstruction is due to an obvious anatomical problem, such as enlarged tonsils or adenoids, or a deviated septum. The goal of surgical intervention is to open the airway and decrease collapsibility. There is considerable variability in the success rate of sleep apnea surgery, with estimates of benefit ranging from as low as 35% to as high as 98.8%.136-138 There are no major studies that successfully identify which patients will most likely benefit from surgery, but it is generally considered an appropriate option for patients with a severe, correctable anatomic abnormality.
Pharmacotherapy
Given the heterogeneity of obstructive sleep apnea, effective pharmacologic treatment has been challenging, despite significant clinical trial efforts.139 Central sleep apnea, however, may respond to medications that stimulate breathing or promote sedation. Modafinil (Provigil) has been approved for daytime sleepiness in patients with sleep apnea. Other medications that are sometimes prescribed, but are not FDA-approved for sleep apnea, include acetazolamide (Diamox) and theophylline, to stimulate breathing, as well as the sedatives triazolam (Halcion) and zolpidem (Ambien).140
For people with obstructive sleep apnea who continue to suffer from excessive daytime sleepiness despite CPAP therapy, modafinil (or armodafinil) can reduce daytime sleepiness and improve wakefulness. Common side effects of modafinil include anxiety, headache, nausea, and nervousness, which lead to high rates of discontinuation.141 In 2019, the FDA approved solriamfetol (Sunosi) for the treatment of excessive daytime sleepiness in adults with obstructive sleep apnea, which represents an effective option with a more tolerable safety profile.142
Tirzepatide. Tirzepatide (Mounjaro, Zepbound) is a long-acting glucose-dependent insulinotropic polypeptide (GIP) receptor and glucagon-like peptide-1 (GLP-1) receptor agonist that has previously been approved by the FDA for the treatment of type 2 diabetes and chronic weight management.251 Tirzepatide received FDA approval for the treatment of obstructive sleep apnea in December 2024.252 Evidence suggests that tirzepatide improves obstructive sleep apnea primarily through a reduction of body weight, with improvements proportional to the degree of weight loss achieved. Current evidence does not support such an effect independent of weight loss.253
The SURMOUNT-OSA phase 3 clinical trials were two multicenter, randomized, double-blind, placebo-controlled trials. These trials were designed to evaluate the safety and efficacy of tirzepatide, administered once weekly at the maximum tolerated dose of 10 or 15 mg, in individuals with obesity (BMI ≥30 kg/m2) and moderate-to-severe obstructive sleep apnea.253 The first trial enrolled 234 participants (mean age 48 years) who were unwilling or unable to use positive airway pressure (PAP) therapy, and the second group consisted of 235 participants (mean age 52 years) who had been on consistent PAP therapy for at least three months prior to screening and were expected to continue therapy during the trial. Both groups received treatment over a 52-week period. The study cohorts were approximately 70% male.
In Trial 1, the mean baseline AHI was 51.5 events/hour. At 52 weeks, tirzepatide led to a reduction of 25.3 events/hour, compared to a reduction of 5.3 events/hour with the placebo, for a treatment difference of –20.0 events/hour. In Trial 2, the mean AHI at baseline was 49.5 events/hour. Tirzepatide reduced AHI by 29.3 events/hour, compared to a reduction of 5.5 events/hour with the placebo, for a treatment difference of –23.8 events/hour. Among people with moderate-to-severe obstructive sleep apnea and obesity, tirzepatide reduced the AHI, body weight, systolic blood pressure, and improved sleep-related patient-reported outcomes.
Hypoglossal Nerve Stimulation
The hypoglossal nerve is one of 12 cranial nerves and is involved in control of the tongue.143 When stimulated, the hypoglossal nerve causes the base of the tongue to protrude forward, which results in opening of the airway.144 The hypoglossal nerve is stimulated by a device, which is surgically implanted under the skin on the chest. A pressure sensor in the device detects breathing patterns and delivers electrical stimulation to the hypoglossal nerve intermittently to trigger dilation of the airway during inspiration.143 In a meta-analysis of outcomes for 200 patients with obstructive sleep apnea, hypoglossal nerve stimulation reduced AHI by up to 57% and was associated with reduced daytime sleepiness.145 An analysis of 5-year outcomes for patients who received hypoglossal nerve stimulation found that 63% experienced a greater than 50% reduction in AHI, with sustained improvements in daytime sleepiness and overall quality of life.146 Hypoglossal nerve stimulation may be helpful for individuals with obstructive sleep apnea who fail to respond to CPAP, and greater improvements in AHI can be expected for those with more severe disease, older age, and lower BMI.147
9 Novel and Emerging Strategies
Acetazolamide
Acetazolamide is a carbonic anhydrase inhibitor that is thought to promote respiratory stimulation by improving the body’s natural response to fluctuations in carbon dioxide levels.148 A meta-analysis of 24 studies of acetazolamide use in sleep apnea found that the drug reduced AHI by about 38% and was effective in both central and obstructive sleep apnea.149 In addition to improving sleep-disordered breathing, studies suggest acetazolamide reduces blood pressure in hypertensive adults with obstructive sleep apnea.150,151 Acetazolamide may be a particularly useful option for patients with sleep apnea resulting from sudden transition to high altitudes.152
Nasal Expiratory Positive Airway Pressure and Oral Negative Pressure Therapy
Because many people find CPAP difficult to use, other devices based on the same principle—using air pressure to keep airways open—have been developed.
Nasal expiratory positive airway pressure (EPAP). This treatment creates positive pressure only in the expiratory phase of the breath cycle, rather than continuously. The pressure is generated by adhesive nasal patches that function as one-way valves that close during exhalation, providing air resistance pressure. These nasal patches may be more tolerable to some people than a CPAP facemask. A meta-analysis of 18 studies, which included a total of 920 patients with obstructive sleep apnea, found that nasal EPAP reduced AHI by an average 53.2% and was associated with improved oxygen levels and reduced daytime sleepiness. In studies that compared nasal EPAP with CPAP, EPAP was associated with smaller reductions in AHI, suggesting it may not be as efficacious as CPAP.161 There is limited evidence available demonstrating who is likely to respond well to EPAP, but it is generally not recommended for patients with severe obstructive sleep apnea, reduced airway size or lung volume, high risk for airway collapse, nasal obstruction, blood gas abnormalities, or for use after stroke.162,163
Oral negative pressure therapy. In this therapy, negative pressure is provided through an oral suction device that draws the tongue and soft palate forward, maintaining airway openness.164 In a pilot study, oral negative pressure therapy significantly reduced obstructive sleep apnea severity and improved wakefulness.165 Study results published in 2019 described significant reductions in AHI and oxygen desaturation associated with oral negative pressure therapy, with 64% of patients achieving a ≥50% reduction in AHI.164
10 Dietary and Lifestyle Considerations
Diet and Weight Loss
Obesity has been described as the most important risk factor for obstructive sleep apnea, and weight loss is recommended for most patients with obstructive sleep apnea and excess weight.172 A study in 690 adults found that a 10% weight loss was predictive of a 26% reduction in AHI, whereas a 10% weight gain predicted a 32% increase in AHI.173 Weight loss intervention has also been shown to improve measures of inflammation and insulin resistance in obstructive sleep apnea, an effect which was not observed with CPAP alone.12 For some patients with severe obstructive sleep apnea and obesity who struggle with conventional weight-loss strategies, bariatric surgery has been found to improve apnea severity, but up to 20% of patients may experience persistent moderate-to-severe obstructive sleep apnea after surgery.174
A 6-month randomized study in 40 subjects with a BMI ≥30 kg/m2 and moderate-to-severe obstructive sleep apnea found that a Mediterranean diet combined with increased physical activity was more effective for both weight loss and reduction in severity of sleep apnea than a prudent diet coupled with physical activity.175 Both the Mediterranean diet and prudent diet emphasize whole grains, legumes, fruits, vegetables, nuts, fish, and poultry, but the Mediterranean diet features more olive oil, suggesting an important role for heart-healthy fats in weight loss for sleep apnea management.176 Additionally, a low-sodium diet has been proposed to improve symptoms of obstructive sleep apnea by reducing fluid retention, but showed greater reductions in AHI than diuretics, suggesting other potential benefits of a low-sodium diet for adults with severe obstructive sleep apnea.177
A thorough discussion of weight loss strategies is included in the Weight Management protocol.
Exercise
Exercise, even without weight loss, appears to reduce both the severity and symptoms of obstructive sleep apnea.178 A meta-analysis of 80 randomized controlled trials found that exercise training reduced AHI to a greater degree than MADs, and was comparable with CPAP for reducing daytime sleepiness.179 A study in 43 obese/overweight adults with obstructive sleep apnea found that a supervised exercise program involving 150 minutes per week of moderate-intensity aerobic exercise plus strength training led to reductions in sleep apnea severity, depressive symptoms, and fatigue, and also improved daytime functioning.180 In a randomized controlled trial, a 6-month graduated walking program, which increased in duration and intensity over the course of the program, improved not only obstructive sleep apnea severity and oxygen desaturation, but was associated with significant reductions in cholesterol levels.181 The well-established cardiovascular benefits of physical activity may thus have important implications for heart health in sleep apnea patients at risk for CVD.
Alcohol Avoidance
Studies suggest alcohol use is associated with a 25% to 33% increased risk for obstructive sleep apnea.182,183 Alcohol is a central nervous system depressant that can contribute to obstructive sleep apnea. Like other sedatives, it can decrease respiratory center activity and weaken the signals triggering inhalation. It may also cause weakness in the muscular structures of the pharynx, increasing the tendency for airway collapse, causing snoring and obstructive apnea. People with obstructive sleep apnea have also been found to be more susceptible to the effects of alcohol and may have a higher risk of alcohol-related automobile accidents compared with people without sleep apnea.184
Sleep Position
For some people, sleep position has a marked impact on severity of apneic and hypopneic breathing during sleep. Lying on one’s back (supine position) is associated with the occurrence of obstructive sleep apnea, as gravitational pull increases risk for airway collapse.1 For some individuals with obstructive sleep apnea, changing sleep position can improve the severity of disease. A study of 85 adults found that in patients with moderate obstructive sleep apnea, switching from sleeping on their backs to on their sides reduced the frequency of airway collapse by nearly 50%.22 Simply turning the head, so that the body remains supine but the head is rotated to a lateral position, also reduces AHI, although not as drastically as full-body side sleeping.185 Strategies for maintaining a non-supine position during sleep include using barriers such as firm pillows. Additionally, sleep-position trainers in the form of a vibrational device or smartphone app have been shown to effectively reduce supine sleeping, AHI, and daytime sleepiness in clinical trials, and are associated with >75% adherence.186,187
Acupuncture
Acupuncture has been explored as a treatment option for patients with obstructive sleep apnea with mixed results. Two small clinical trials found that acupuncture was associated with reduced AHI and respiratory events using both manual acupuncture and electroacupuncture.188,189 However, a more recent randomized controlled trial found that, after 10 acupuncture sessions, there was no significant change in AHI, blood pressure, or quality of life in hypertensive adults with obstructive sleep apnea.190 A meta-analysis of nine clinical trials published in 2020 found that acupuncture significantly improved AHI, daytime sleepiness, and oxygen saturation, particularly in patients with moderate obstructive sleep apnea.191 Importantly, all of these trials have been small and the quality of evidence supporting acupuncture as a treatment for obstructive sleep apnea is low to very low—larger, more rigorous controlled trials are needed.191
Smoking Cessation
The effects of smoking on risk for and severity of obstructive sleep apnea remain controversial.192-194 However, the combined effect of smoking and sleep apnea has been linked to worse cognitive impairment and metabolic parameters.195,196 Although further clinical trials are needed to demonstrate the benefits of smoking cessation for obstructive sleep apnea severity, current evidence suggests it may improve important comorbidities related to sleep apnea.
11 Nutrients
In addition to the interventions described in this protocol, readers should also review the Life Extension protocols on Weight Management, Cardiovascular Disease, Chronic Inflammation, and Diabetes, given the strong associations between sleep apnea and metabolic health. In addition, a number of strategies for improving sleep quality and sleep hygiene in general are described in the Insomnia protocol and may be of use for those with sleep apnea.
N-acetylcysteine
Oxidative stress is thought to cause many of the clinical signs and symptoms of sleep apnea, including cardiovascular, neurological, and immune dysfunction. N-acetylcysteine (NAC) is an antioxidant that reduces intermittent hypoxia resulting from excitation of the sympathetic nervous system.197 In an animal model of sleep apnea, supplementation with NAC mitigated oxidative stress and inflammation that occurred as a result of intermittent hypoxia.198 In rats, NAC in combination with an arginase inhibitor reduced systemic oxidative stress resulting from chronic intermittent hypoxia, including normalization of elevated blood pressure.199 In a randomized placebo-controlled trial in 20 adults with obstructive sleep apnea, 600 mg NAC given three times daily for 30 days resulted in significant reductions in AHI, apnea-related arousals, oxygen desaturation, daytime sleepiness, and snoring. These effects were also associated with significant reductions in measures of oxidative stress, including decreased lipid peroxidation and increased total reduced glutathione level.200 Although larger clinical trials are needed, these results suggest a promising role for NAC and antioxidants in the treatment of systemic symptoms associated with sleep apnea.
Vitamins C and E
Vitamins C and E are antioxidants that may have potential roles in the treatment of sleep apnea. In an animal model of obstructive sleep apnea, treatment with vitamins C and E was associated with reduction in markers of both oxidative and carbonyl stress resulting from intermittent hypoxia.201 In a preliminary study, 10 people with obstructive sleep apnea were found to have dysfunction of the delicate lining of blood vessels (the endothelium) compared with healthy controls, which improved to a level comparable with that of controls after a single 500-mg dose of intravenous vitamin C.202 In another study, a combination of vitamin C (100 mg) and vitamin E (400 IU), taken by mouth twice daily for 45 days, led to reductions in the frequency of apneic episodes and daytime sleepiness and improved sleep quality in 20 men with obstructive sleep apnea being treated with CPAP. Those who received the vitamin combination were also able to reduce pressure settings on their CPAP machines.203 In a clinical trial involving 26 men, treatment with an antioxidant cocktail containing both vitamins C and E, as well as coenzyme Q10, was associated with significant improvements in measures of respiratory function in patients with obstructive sleep apnea.204 These results support the increased emphasis on clinical trials examining the use of antioxidants in the treatment of sleep apnea.
Coenzyme Q10
Coenzyme Q10 (CoQ10) is involved in mitochondrial energy production and is best known for its beneficial effects on the cardiovascular system and metabolism.205 As demonstrated in the study mentioned previously, CoQ10 in combination with other antioxidants improved respiratory function in men with obstructive sleep apnea.204 Although specific trials examining the use of CoQ10 in patients with sleep apnea are still needed, patients with sleep apnea are at increased risk for CVD, hypertension, and impaired glucose tolerance, and supplementation with CoQ10 may confer a benefit in all of these conditions.205
Selenium
Selenium is a micronutrient involved in regulation of many enzymes involved in the cellular response to oxidative stress and inflammation, including glutathione peroxidase.206 The role of selenium in obstructive sleep apnea remains unclear. In a 2013 study of the concentrations of trace minerals in 44 individuals newly diagnosed with mild-to-moderate obstructive sleep apnea compared with healthy controls, those with sleep apnea had lower levels of red blood cell selenium and glutathione peroxidase activity than people without apnea, and selenium was negatively associated with AHI values.207 However, in a more recent study in 146 subjects, serum selenium levels were significantly elevated in individuals with obstructive sleep apnea compared with healthy controls, and selenium levels were positively correlated with AHI. The authors of the study postulated that elevated selenium may represent a defense mechanism in patients with obstructive sleep apnea and that antioxidant supplementation may improve the response.208 Although the exact role of selenium remains unclear, it is likely important for progression of obstructive sleep apnea, and further investigation is warranted.206
Vitamin D
Low vitamin D levels are associated with a wide variety of systemic effects, including modulation of the immune system, myopathy, and inflammation.209 In recent years, there has been an increased appreciation for the role of vitamin D deficiency in the occurrence and progression of sleep apnea. In a meta-analysis of 14 studies including almost 5,000 subjects, serum vitamin D levels were lower among patients with obstructive sleep apnea, which was incrementally exacerbated with increasing disease severity.210 Studies have also found that low vitamin D levels are associated with increased risk for insulin resistance, diabetes, and metabolic syndrome in patients with obstructive sleep apnea.211-214 There is often an inverse relationship between levels of vitamin D and parathyroid hormone, and higher parathyroid levels have also been found to be associated with increased risk for hypertension in adults with obstructive sleep apnea.211 In a small randomized controlled trial in 19 white adults with obstructive sleep apnea, daily vitamin D supplementation (4,000 IU) was associated with significant reductions in metabolic markers; however, neuropsychological and quality of life measures remained unchanged.215 Larger and, importantly, more diverse clinical trials are needed to examine the effects of vitamin D supplementation on obstructive sleep apnea and associated comorbidities. Mutations in the vitamin D receptor have been found to at least partially account for some of the variability in vitamin D levels observed in patients with obstructive sleep apnea; thus, genetically diverse populations should be included in clinical trials.216
Omega-3 Fatty Acids
Sufficient levels of omega-3 fatty acids are associated with a host of cardiovascular and anti-inflammatory benefits, which may have important implications for individuals with sleep apnea.217 A study that examined the red blood cell membrane fatty acid profiles of individuals with obstructive sleep apnea found that lower membrane levels of the omega-3 fatty acid docosahexaenoic acid (DHA) were associated with increased likelihood of severe sleep apnea.218 Although there has been speculation that omega-3 fatty acids may represent a therapeutic option for treatment of immune or metabolic comorbidities of sleep apnea, formal studies in humans are still needed.219
B Vitamins
A number of studies have found that people with obstructive sleep apnea, particularly those with severe disease, have higher levels of homocysteine, an amino acid linked to increased risk of cardiovascular and neurodegenerative diseases.220 A 6-year analysis of 1,825 patients with obstructive sleep apnea found that elevated homocysteine levels were associated with an 86% increased risk for developing concurrent hypertension.221 The effects of CPAP therapy on homocysteine levels is unclear, with some studies suggesting a modest improvement after three months of use and others observing no effect after six months of use.222,223
Supplementation with B vitamins can reduce serum homocysteine levels.224,225 A randomized controlled trial demonstrated that a high-dose B-complex vitamin reduced measures of oxidative stress and inflammation, and studies have shown that B vitamin supplementation is associated with reduced risk for stroke.224,226 Although trials are needed which demonstrate a clinical benefit of B vitamin supplementation in obstructive sleep apnea, evidence suggests it represents a promising option for exploration.
More information about the importance of keeping homocysteine levels low and strategies to do so are available in the Homocysteine Reduction protocol.
Probiotics
Changes in the gut microbiome have been linked to a variety of systemic diseases. The prevailing theory is that gut dysbiosis causes damage to the intestinal wall, resulting in a “leaky gut,” which causes chronic low-grade inflammation. There has been increasing interest in the role of the microbiome and gut dysbiosis on the development of obstructive sleep apnea and associated comorbidities, but studies have so far been limited primarily to animal models.227 However, an analysis of fecal samples from 93 patients with obstructive sleep apnea reported varying degrees of microbial dysbiosis in patients, dominated primarily by decreased levels of bacteria that produce short-chain fatty acids and increased prevalence of disease-causing bacteria.228 Studies involving animal models of sleep apnea and intermittent hypoxia have found that probiotics can improve some of the side effects of systemic inflammation in the heart and brain, as well as hypertension.229,230 Studies in humans are needed to explore the effects of probiotics on obstructive sleep apnea occurrence, severity, and comorbidities.
Disclaimer and Safety Information
This information (and any accompanying material) is not intended to replace the attention or advice of a physician or other qualified health care professional. Anyone who wishes to embark on any dietary, drug, exercise, or other lifestyle change intended to prevent or treat a specific disease or condition should first consult with and seek clearance from a physician or other qualified health care professional. Pregnant women in particular should seek the advice of a physician before using any protocol listed on this website. The protocols described on this website are for adults only, unless otherwise specified. Product labels may contain important safety information and the most recent product information provided by the product manufacturers should be carefully reviewed prior to use to verify the dose, administration, and contraindications. National, state, and local laws may vary regarding the use and application of many of the therapies discussed. The reader assumes the risk of any injuries. The authors and publishers, their affiliates and assigns are not liable for any injury and/or damage to persons arising from this protocol and expressly disclaim responsibility for any adverse effects resulting from the use of the information contained herein.
The protocols raise many issues that are subject to change as new data emerge. None of our suggested protocol regimens can guarantee health benefits. Life Extension has not performed independent verification of the data contained in the referenced materials, and expressly disclaims responsibility for any error in the literature.
- Patel SR. Obstructive Sleep Apnea. Ann Intern Med. Dec 3 2019;171(11):ITC81-ITC96. doi:10.7326/AITC201912030
- Acharya R, Basnet S, Tharu B, et al. Obstructive Sleep Apnea: Risk Factor for Arrhythmias, Conduction Disorders, and Cardiac Arrest. Cureus. Aug 24 2020;12(8):e9992. doi:10.7759/cureus.9992
- Punjabi NM. The epidemiology of adult obstructive sleep apnea. Proceedings of the American Thoracic Society. Feb 15 2008;5(2):136-43. doi:10.1513/pats.200709-155MG
- American Academy of Sleep Medicine. Rising prevalence of sleep apnea in U.S. threatens public health. Updated September 29, 2014. Accessed October 19, 2020. https://aasm.org/rising-prevalence-of-sleep-apnea-in-u-s-threatens-public-health/
- Mayo Clinic Staff. Obstructive sleep apnea. Updated June 5, 2019. Accessed October 19, 2020. https://www.mayoclinic.org/diseases-conditions/obstructive-sleep-apnea/symptoms-causes/syc-20352090
- Veasey SC, Rosen IM. Obstructive Sleep Apnea in Adults. The New England journal of medicine. Apr 11 2019;380(15):1442-1449. doi:10.1056/NEJMcp1816152
- Patil SP, Ayappa IA, Caples SM, Kimoff RJ, Patel SR, Harrod CG. Treatment of Adult Obstructive Sleep Apnea with Positive Airway Pressure: An American Academy of Sleep Medicine Clinical Practice Guideline. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Feb 15 2019;15(2):335-343. doi:10.5664/jcsm.7640
- Iftikhar IH, Valentine CW, Bittencourt LR, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a meta-analysis. Journal of hypertension. Dec 2014;32(12):2341-50; discussion 2350. doi:10.1097/HJH.0000000000000372
- Bratton DJ, Gaisl T, Wons AM, Kohler M. CPAP vs Mandibular Advancement Devices and Blood Pressure in Patients With Obstructive Sleep Apnea: A Systematic Review and Meta-analysis. JAMA. Dec 1 2015;314(21):2280-93. doi:10.1001/jama.2015.16303
- Lei Q, Lv Y, Li K, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a systematic review and meta-analysis of six randomized controlled trials. J Bras Pneumol. Sep-Oct 2017;43(5):373-379. doi:10.1590/S1806-37562016000000190
- Richards KC, Gooneratne N, Dicicco B, et al. CPAP Adherence May Slow 1-Year Cognitive Decline in Older Adults with Mild Cognitive Impairment and Apnea. J Am Geriatr Soc. Mar 2019;67(3):558-564. doi:10.1111/jgs.15758
- Chirinos JA, Gurubhagavatula I, Teff K, et al. CPAP, weight loss, or both for obstructive sleep apnea. The New England journal of medicine. Jun 12 2014;370(24):2265-75. doi:10.1056/NEJMoa1306187
- OpenStax CNX. Organs and Structures of the Respiratory System. Pharynx: Divisions of the Pharynx. Accessed 11/5/2020, https://courses.lumenlearning.com/suny-contemporaryhealthissues/chapter/pharynx/
- Dempsey JA, Veasey SC, Morgan BJ, O'Donnell CP. Pathophysiology of sleep apnea. Physiol Rev. Jan 2010;90(1):47-112. doi:10.1152/physrev.00043.2008
- Torre C, Camacho M, Liu SY, Huon LK, Capasso R. Epiglottis collapse in adult obstructive sleep apnea: A systematic review. The Laryngoscope. Feb 2016;126(2):515-23. doi:10.1002/lary.25589
- Owens RL, Eckert DJ, Yeh SY, Malhotra A. Upper airway function in the pathogenesis of obstructive sleep apnea: a review of the current literature. Current opinion in pulmonary medicine. Nov 2008;14(6):519-24. doi:10.1097/MCP.0b013e3283130f66
- Subramani Y, Singh M, Wong J, Kushida CA, Malhotra A, Chung F. Understanding Phenotypes of Obstructive Sleep Apnea: Applications in Anesthesia, Surgery, and Perioperative Medicine. Anesthesia and analgesia. Jan 2017;124(1):179-191. doi:10.1213/ANE.0000000000001546
- Donovan LM, Kapur VK. Prevalence and Characteristics of Central Compared to Obstructive Sleep Apnea: Analyses from the Sleep Heart Health Study Cohort. Sleep. Jul 1 2016;39(7):1353-9. doi:10.5665/sleep.5962
- Lehman S, Antic NA, Thompson C, Catcheside PG, Mercer J, McEvoy RD. Central sleep apnea on commencement of continuous positive airway pressure in patients with a primary diagnosis of obstructive sleep apnea-hypopnea. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Aug 15 2007;3(5):462-6.
- Javaheri S, Smith J, Chung E. The prevalence and natural history of complex sleep apnea. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Jun 15 2009;5(3):205-11.
- Pham LV, Schwartz AR. The pathogenesis of obstructive sleep apnea. J Thorac Dis. Aug 2015;7(8):1358-72. doi:10.3978/j.issn.2072-1439.2015.07.28
- Lee CH, Kim DK, Kim SY, Rhee CS, Won TB. Changes in site of obstruction in obstructive sleep apnea patients according to sleep position: a DISE study. The Laryngoscope. Jan 2015;125(1):248-54. doi:10.1002/lary.24825
- Nattie E. CO2, brainstem chemoreceptors and breathing. Progress in neurobiology. Nov 1999;59(4):299-331. doi:10.1016/s0301-0082(99)00008-8
- Orr JE, Malhotra A, Sands SA. Pathogenesis of central and complex sleep apnoea. Respirology (Carlton, Vic). Jan 2017;22(1):43-52. doi:10.1111/resp.12927
- Brown RE, Basheer R, McKenna JT, Strecker RE, McCarley RW. Control of sleep and wakefulness. Physiol Rev. Jul 2012;92(3):1087-187. doi:10.1152/physrev.00032.2011
- Nieto FJ, Young TB, Lind BK, et al. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. Sleep Heart Health Study. JAMA. Apr 12 2000;283(14):1829-36. doi:10.1001/jama.283.14.1829
- O'Connor GT, Caffo B, Newman AB, et al. Prospective study of sleep-disordered breathing and hypertension: the Sleep Heart Health Study. American journal of respiratory and critical care medicine. Jun 15 2009;179(12):1159-64. doi:10.1164/rccm.200712-1809OC
- Turnbull F, Blood Pressure Lowering Treatment Trialists C. Effects of different blood-pressure-lowering regimens on major cardiovascular events: results of prospectively-designed overviews of randomised trials. Lancet. Nov 8 2003;362(9395):1527-35. doi:10.1016/s0140-6736(03)14739-3
- Van Ryswyk E, Mukherjee S, Chai-Coetzer CL, Vakulin A, McEvoy RD. Sleep Disorders, Including Sleep Apnea and Hypertension. Am J Hypertens. Jul 16 2018;31(8):857-864. doi:10.1093/ajh/hpy082
- Floras JS. Sleep Apnea and Cardiovascular Disease: An Enigmatic Risk Factor. Circ Res. Jun 8 2018;122(12):1741-1764. doi:10.1161/circresaha.118.310783
- Javaheri S, Barbe F, Campos-Rodriguez F, et al. Sleep Apnea: Types, Mechanisms, and Clinical Cardiovascular Consequences. Journal of the American College of Cardiology. Feb 21 2017;69(7):841-858. doi:10.1016/j.jacc.2016.11.069
- McEvoy RD, Antic NA, Heeley E, et al. CPAP for Prevention of Cardiovascular Events in Obstructive Sleep Apnea. The New England journal of medicine. Sep 8 2016;375(10):919-31. doi:10.1056/NEJMoa1606599
- Peker Y, Glantz H, Eulenburg C, Wegscheider K, Herlitz J, Thunstrom E. Effect of Positive Airway Pressure on Cardiovascular Outcomes in Coronary Artery Disease Patients with Nonsleepy Obstructive Sleep Apnea. The RICCADSA Randomized Controlled Trial. American journal of respiratory and critical care medicine. Sep 1 2016;194(5):613-20. doi:10.1164/rccm.201601-0088OC
- Qureshi WT, Nasir UB, Alqalyoobi S, et al. Meta-Analysis of Continuous Positive Airway Pressure as a Therapy of Atrial Fibrillation in Obstructive Sleep Apnea. The American journal of cardiology. Dec 1 2015;116(11):1767-73. doi:10.1016/j.amjcard.2015.08.046
- Selim BJ, Ramar K. Management of Sleep Apnea Syndromes in Heart Failure. Sleep medicine clinics. Mar 2017;12(1):107-121. doi:10.1016/j.jsmc.2016.10.004
- Muscogiuri G, Barrea L, Annunziata G, et al. Obesity and sleep disturbance: the chicken or the egg? Crit Rev Food Sci Nutr. 2019;59(13):2158-2165. doi:10.1080/10408398.2018.1506979
- Fallahi A, Jamil DI, Karimi EB, Baghi V, Gheshlagh RG. Prevalence of obstructive sleep apnea in patients with type 2 diabetes: A systematic review and meta-analysis. Diabetes Metab Syndr. Jul - Aug 2019;13(4):2463-2468. doi:10.1016/j.dsx.2019.06.030
- Siwasaranond N, Nimitphong H, Manodpitipong A, et al. The Relationship between Diabetes-Related Complications and Obstructive Sleep Apnea in Type 2 Diabetes. J Diabetes Res. 2018;2018:9269170. doi:10.1155/2018/9269170
- Subramanian A, Adderley NJ, Tracy A, et al. Risk of Incident Obstructive Sleep Apnea Among Patients With Type 2 Diabetes. Diabetes Care. May 2019;42(5):954-963. doi:10.2337/dc18-2004
- Ota H, Fujita Y, Yamauchi M, Muro S, Kimura H, Takasawa S. Relationship Between Intermittent Hypoxia and Type 2 Diabetes in Sleep Apnea Syndrome. International journal of molecular sciences. Sep 25 2019;20(19)doi:10.3390/ijms20194756
- Martinez-Ceron E, Barquiel B, Bezos AM, et al. Effect of Continuous Positive Airway Pressure on Glycemic Control in Patients with Obstructive Sleep Apnea and Type 2 Diabetes. A Randomized Clinical Trial. American journal of respiratory and critical care medicine. Aug 15 2016;194(4):476-85. doi:10.1164/rccm.201510-1942OC
- Shaw JE, Punjabi NM, Naughton MT, et al. The Effect of Treatment of Obstructive Sleep Apnea on Glycemic Control in Type 2 Diabetes. American journal of respiratory and critical care medicine. Aug 15 2016;194(4):486-92. doi:10.1164/rccm.201511-2260OC
- Labarca G, Reyes T, Jorquera J, Dreyse J, Drake L. CPAP in patients with obstructive sleep apnea and type 2 diabetes mellitus: Systematic review and meta-analysis. The clinical respiratory journal. Aug 2018;12(8):2361-2368. doi:10.1111/crj.12915
- Gharsalli H, Oueslati I, Sahnoun I, et al. Effect of Continuous Positive Airway Pressure on glycated hemoglobin in patients with type 2 diabetes and Obstructive Sleep Apnea. La Tunisie medicale. Dec 2019;97(12):1345-1352.
- Furukawa S, Miyake T, Senba H, et al. The effectiveness of dapagliflozin for sleep-disordered breathing among Japanese patients with obesity and type 2 diabetes mellitus. Endocrine journal. Sep 27 2018;65(9):953-961. doi:10.1507/endocrj.EJ17-0545
- Tang Y, Sun Q, Bai XY, Zhou YF, Zhou QL, Zhang M. Effect of dapagliflozin on obstructive sleep apnea in patients with type 2 diabetes: a preliminary study. Nutrition & diabetes. Nov 4 2019;9(1):32. doi:10.1038/s41387-019-0098-5
- Iftikhar IH, Roland J. Obesity Hypoventilation Syndrome. Clin Chest Med. Jun 2018;39(2):427-436. doi:10.1016/j.ccm.2018.01.006
- Olaithe M, Bucks RS, Hillman DR, Eastwood PR. Cognitive deficits in obstructive sleep apnea: Insights from a meta-review and comparison with deficits observed in COPD, insomnia, and sleep deprivation. Sleep medicine reviews. Apr 2018;38:39-49. doi:10.1016/j.smrv.2017.03.005
- Mansukhani MP, Kolla BP, Somers VK. Hypertension and Cognitive Decline: Implications of Obstructive Sleep Apnea. Front Cardiovasc Med. 2019;6:96. doi:10.3389/fcvm.2019.00096
- Mubashir T, Abrahamyan L, Niazi A, et al. The prevalence of obstructive sleep apnea in mild cognitive impairment: a systematic review. BMC Neurol. Aug 15 2019;19(1):195. doi:10.1186/s12883-019-1422-3
- Hobzova M, Prasko J, Vanek J, et al. Depression and obstructive sleep apnea. Neuro endocrinology letters. Oct 2017;38(5):343-352.
- Kerner NA, Roose SP. Obstructive Sleep Apnea is Linked to Depression and Cognitive Impairment: Evidence and Potential Mechanisms. Am J Geriatr Psychiatry. Jun 2016;24(6):496-508. doi:10.1016/j.jagp.2016.01.134
- Bilyukov RG, Nikolov MS, Pencheva VP, et al. Cognitive Impairment and Affective Disorders in Patients With Obstructive Sleep Apnea Syndrome. Frontiers in psychiatry. 2018;9:357. doi:10.3389/fpsyt.2018.00357
- Lavie L. Oxidative stress in obstructive sleep apnea and intermittent hypoxia--revisited--the bad ugly and good: implications to the heart and brain. Sleep medicine reviews. Apr 2015;20:27-45. doi:10.1016/j.smrv.2014.07.003
- Kheirandish-Gozal L, Gozal D. Obstructive Sleep Apnea and Inflammation: Proof of Concept Based on Two Illustrative Cytokines. International journal of molecular sciences. Jan 22 2019;20(3)doi:10.3390/ijms20030459
- Van der Touw T, Andronicos NM, Smart N. Is C-reactive protein elevated in obstructive sleep apnea? a systematic review and meta-analysis. Biomarkers. Jul 2019;24(5):429-435. doi:10.1080/1354750X.2019.1600025
- Douglas RM, Haddad GG. Can O2 dysregulation induce premature aging? Physiology (Bethesda, Md). Dec 2008;23:333-49. doi:10.1152/physiol.00023.2008
- Polonis K, Becari C, Chahal CAA, et al. Chronic Intermittent Hypoxia Triggers a Senescence-like Phenotype in Human White Preadipocytes. Sci Rep. Apr 22 2020;10(1):6846. doi:10.1038/s41598-020-63761-7
- Gaspar LS, Alvaro AR, Moita J, Cavadas C. Obstructive Sleep Apnea and Hallmarks of Aging. Trends Mol Med. Aug 2017;23(8):675-692. doi:10.1016/j.molmed.2017.06.006
- Prather AA, Gurfein B, Moran P, et al. Tired telomeres: Poor global sleep quality, perceived stress, and telomere length in immune cell subsets in obese men and women. Brain Behav Immun. Jul 2015;47:155-62. doi:10.1016/j.bbi.2014.12.011
- Andrews NP, Fujii H, Goronzy JJ, Weyand CM. Telomeres and immunological diseases of aging. Gerontology. 2010;56(4):390-403. doi:10.1159/000268620
- Irwin M, McClintick J, Costlow C, Fortner M, White J, Gillin JC. Partial night sleep deprivation reduces natural killer and cellular immune responses in humans. FASEB journal : official publication of the Federation of American Societies for Experimental Biology . Apr 1996;10(5):643-53. doi:10.1096/fasebj.10.5.8621064
- Bozkurt NC, Karbek B, Cakal E, Firat H, Ozbek M, Delibasi T. The association between severity of obstructive sleep apnea and prevalence of Hashimoto's thyroiditis. Endocrine journal. 2012;59(11):981-8. doi:10.1507/endocrj.ej12-0106
- Vakil M, Park S, Broder A. The complex associations between obstructive sleep apnea and auto-immune disorders: A review. Med Hypotheses. Jan 2018;110:138-143. doi:10.1016/j.mehy.2017.12.004
- Kanbay A, Kokturk O, Pihtili A, et al. Obstructive sleep apnea is a risk factor for osteoarthritis. Tuberk Toraks. Dec 2018;66(4):304-311. Obstruktif uyku apnesi osteoartrit icin risk faktorudur. doi:10.5578/tt.57403
- Brenner R, Kivity S, Peker M, et al. Increased Risk for Cancer in Young Patients with Severe Obstructive Sleep Apnea. Respiration. 2019;97(1):15-23. doi:10.1159/000486577
- Sillah A, Watson NF, Schwartz SM, Gozal D, Phipps AI. Sleep apnea and subsequent cancer incidence. Cancer Causes Control. Oct 2018;29(10):987-994. doi:10.1007/s10552-018-1073-5
- Marshall NS, Wong KK, Cullen SR, Knuiman MW, Grunstein RR. Sleep apnea and 20-year follow-up for all-cause mortality, stroke, and cancer incidence and mortality in the Busselton Health Study cohort. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Apr 15 2014;10(4):355-62. doi:10.5664/jcsm.3600
- Nieto FJ, Peppard PE, Young T, Finn L, Hla KM, Farre R. Sleep-disordered breathing and cancer mortality: results from the Wisconsin Sleep Cohort Study. American journal of respiratory and critical care medicine. Jul 15 2012;186(2):190-4. doi:10.1164/rccm.201201-0130OC
- Kukwa W, Migacz E, Druc K, Grzesiuk E, Czarnecka AM. Obstructive sleep apnea and cancer: effects of intermittent hypoxia? Future oncology (London, England). 2015;11(24):3285-98. doi:10.2217/fon.15.216
- US National Library of Medicine. Gout. Updated July 8, 2020. Accessed October 16, 2020. https://medlineplus.gov/gout.html
- Singh JA, Cleveland JD. Gout and the Risk of Incident Obstructive Sleep Apnea in Adults 65 Years or Older: An Observational Study. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Sep 15 2018;14(9):1521-1527. doi:10.5664/jcsm.7328
- Zhang Y, Peloquin CE, Dubreuil M, et al. Sleep Apnea and the Risk of Incident Gout: A Population-Based, Body Mass Index-Matched Cohort Study. Arthritis Rheumatol. Dec 2015;67(12):3298-302. doi:10.1002/art.39330
- Blagojevic-Bucknall M, Mallen C, Muller S, et al. The Risk of Gout Among Patients With Sleep Apnea: A Matched Cohort Study. Arthritis Rheumatol. Jan 2019;71(1):154-160. doi:10.1002/art.40662
- Wiener RC, Shankar A. Association between Serum Uric Acid Levels and Sleep Variables: Results from the National Health and Nutrition Survey 2005-2008. International journal of inflammation. 2012;2012:363054. doi:10.1155/2012/363054
- Shi T, Min M, Sun C, et al. A meta-analysis of the association between gout, serum uric acid level, and obstructive sleep apnea. Sleep & breathing = Schlaf & Atmung. Dec 2019;23(4):1047-1057. doi:10.1007/s11325-019-01827-1
- Hirotsu C, Tufik S, Guindalini C, Mazzotti DR, Bittencourt LR, Andersen ML. Association between uric acid levels and obstructive sleep apnea syndrome in a large epidemiological sample. PLoS One. 2013;8(6):e66891. doi:10.1371/journal.pone.0066891
- Kanbay A, Inonu H, Solak Y, et al. Uric acid as a potential mediator of cardiovascular morbidity in obstructive sleep apnea syndrome. European journal of internal medicine. Jun 2014;25(5):471-6. doi:10.1016/j.ejim.2014.04.005
- Chen Q, Lin G, Chen L, et al. Does Continuous Positive Airway Pressure Therapy in Patients with Obstructive Sleep Apnea Improves Uric Acid? A Meta-Analysis. Oxid Med Cell Longev. 2019;2019:4584936. doi:10.1155/2019/4584936
- Ayyar L, Shaib F, Guntupalli K. Sleep-Disordered Breathing in Pregnancy. Sleep medicine clinics. Sep 2018;13(3):349-357. doi:10.1016/j.jsmc.2018.04.005
- Warland J, Dorrian J, Morrison JL, O'Brien LM. Maternal sleep during pregnancy and poor fetal outcomes: A scoping review of the literature with meta-analysis. Sleep medicine reviews. Oct 2018;41:197-219. doi:10.1016/j.smrv.2018.03.004
- Dominguez JE, Krystal AD, Habib AS. Obstructive Sleep Apnea in Pregnant Women: A Review of Pregnancy Outcomes and an Approach to Management. Anesthesia and analgesia. Nov 2018;127(5):1167-1177. doi:10.1213/ANE.0000000000003335
- Facco FL, Parker CB, Reddy UM, et al. Association Between Sleep-Disordered Breathing and Hypertensive Disorders of Pregnancy and Gestational Diabetes Mellitus. Obstet Gynecol. Jan 2017;129(1):31-41. doi:10.1097/AOG.0000000000001805
- Kim SD, Cho KS. Obstructive Sleep Apnea and Testosterone Deficiency. World J Mens Health. Jan 2019;37(1):12-18. doi:10.5534/wjmh.180017
- Bercea RM, Mihaescu T, Cojocaru C, Bjorvatn B. Fatigue and serum testosterone in obstructive sleep apnea patients. The clinical respiratory journal. Jul 2015;9(3):342-9. doi:10.1111/crj.12150
- Canguven O, Salepci B, Albayrak S, Selimoglu A, Balaban M, Bulbul M. Is there a correlation between testosterone levels and the severity of the disease in male patients with obstructive sleep apnea? Arch Ital Urol Androl. Dec 2010;82(4):143-7.
- Kellesarian SV, Malignaggi VR, Feng C, Javed F. Association between obstructive sleep apnea and erectile dysfunction: a systematic review and meta-analysis. Int J Impot Res. Jun 2018;30(3):129-140. doi:10.1038/s41443-018-0017-7
- Li Z, Fang Z, Xing N, Zhu S, Fan Y. The effect of CPAP and PDE5i on erectile function in men with obstructive sleep apnea and erectile dysfunction: A systematic review and meta-analysis. Sleep medicine reviews. Dec 2019;48:101217. doi:10.1016/j.smrv.2019.101217
- Cignarelli A, Castellana M, Castellana G, et al. Effects of CPAP on Testosterone Levels in Patients With Obstructive Sleep Apnea: A Meta-Analysis Study. Frontiers in endocrinology. 2019;10:551. doi:10.3389/fendo.2019.00551
- Bagabas N, Ghazali W, Mukhtar M, et al. Prevalence of Glaucoma in Patients with Obstructive Sleep Apnea. J Epidemiol Glob Health. Sep 2019;9(3):198-203. doi:10.2991/jegh.k.190816.001
- Lajoie AC, Lafontaine AL, Kimoff RJ, Kaminska M. Obstructive Sleep Apnea in Neurodegenerative Disorders: Current Evidence in Support of Benefit from Sleep Apnea Treatment. J Clin Med. Jan 21 2020;9(2)doi:10.3390/jcm9020297
- Ehrmann DA. Metabolic dysfunction in pcos: Relationship to obstructive sleep apnea. Steroids. Mar 10 2012;77(4):290-4. doi:10.1016/j.steroids.2011.12.001
- Thavaraputta S, Dennis JA, Laoveeravat P, Nugent K, Rivas AM. Hypothyroidism and Its Association With Sleep Apnea Among Adults in the United States: NHANES 2007-2008. J Clin Endocrinol Metab. Nov 1 2019;104(11):4990-4997. doi:10.1210/jc.2019-01132
- Tsai SH, Stoller ML, Sherer BA, Chao ZH, Tung TH. Risk of Nephrolithiasis in Patients With Sleep Apnea: A Population-Based Cohort Study. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . May 15 2018;14(5):767-773. doi:10.5664/jcsm.7102
- Lin CH, Lurie RC, Lyons OD. Sleep Apnea and Chronic Kidney Disease: A State-of-the-Art Review. Chest. Mar 2020;157(3):673-685. doi:10.1016/j.chest.2019.09.004
- Au CT, Zhang J, Cheung JYF, Chan KCC, Wing YK, Li AM. Familial Aggregation and Heritability of Obstructive Sleep Apnea Using Children Probands. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Nov 15 2019;15(11):1561-1570. doi:10.5664/jcsm.8012
- Perger E, Mattaliano P, Lombardi C. Menopause and Sleep Apnea. Maturitas. Jun 2019;124:35-38. doi:10.1016/j.maturitas.2019.02.011
- Zhang L, Ou X, Zhu T, Lv X. Beneficial effects of estrogens in obstructive sleep apnea hypopnea syndrome. Sleep & breathing = Schlaf & Atmung. Mar 2020;24(1):7-13. doi:10.1007/s11325-019-01896-2
- Lindberg E, Bonsignore MR, Polo-Kantola P. Role of menopause and hormone replacement therapy in sleep-disordered breathing. Sleep medicine reviews. Feb 2020;49:101225. doi:10.1016/j.smrv.2019.101225
- Chen X, Wang R, Zee P, et al. Racial/Ethnic Differences in Sleep Disturbances: The Multi-Ethnic Study of Atherosclerosis (MESA). Sleep. Jun 1 2015;38(6):877-88. doi:10.5665/sleep.4732
- Jordan AS, McSharry DG, Malhotra A. Adult obstructive sleep apnoea. Lancet. Feb 22 2014;383(9918):736-47. doi:10.1016/S0140-6736(13)60734-5
- Braido F, Baiardini I, Lacedonia D, et al. Sleep apnea risk in subjects with asthma with or without comorbid rhinitis. Respir Care. Dec 2014;59(12):1851-6. doi:10.4187/respcare.03084
- Chirakalwasan N, Ruxrungtham K. The linkage of allergic rhinitis and obstructive sleep apnea. Asian Pacific journal of allergy and immunology / launched by the Allergy and Immunology Society of Thailand . Dec 2014;32(4):276-86.
- Reid M, Maras JE, Shea S, et al. Association between diet quality and sleep apnea in the Multi-Ethnic Study of Atherosclerosis. Sleep. Jan 1 2019;42(1)doi:10.1093/sleep/zsy194
- de Melo CM, Del Re MP, Dos Santos Quaresma MVL, et al. Relationship of evening meal with sleep quality in obese individuals with obstructive sleep apnea. Clinical nutrition ESPEN. Feb 2019;29:231-236. doi:10.1016/j.clnesp.2018.09.077
- Lopes T, Borba ME, Lopes R, et al. Eating Late Negatively Affects Sleep Pattern and Apnea Severity in Individuals With Sleep Apnea. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Mar 15 2019;15(3):383-392. doi:10.5664/jcsm.7658
- Badr MS, Dingell JD, Javaheri S. Central Sleep Apnea: a Brief Review. Curr Pulmonol Rep. Mar 2019;8(1):14-21. doi:10.1007/s13665-019-0221-z
- Mold JW, Quattlebaum C, Schinnerer E, Boeckman L, Orr W, Hollabaugh K. Identification by primary care clinicians of patients with obstructive sleep apnea: a practice-based research network (PBRN) study. J Am Board Fam Med. Mar-Apr 2011;24(2):138-45. doi:10.3122/jabfm.2011.02.100095
- Lindberg E, Benediktsdottir B, Franklin KA, et al. Women with symptoms of sleep-disordered breathing are less likely to be diagnosed and treated for sleep apnea than men. Sleep Med. Jul 2017;35:17-22. doi:10.1016/j.sleep.2017.02.032
- Chung F, Subramanyam R, Liao P, Sasaki E, Shapiro C, Sun Y. High STOP-Bang score indicates a high probability of obstructive sleep apnoea. British journal of anaesthesia. May 2012;108(5):768-75. doi:10.1093/bja/aes022
- Jaspal Singh MD, Nicole M. Screening Tools for the Obstructive Sleep Apnea for the Cardiovascular Clinician - American College of Cardiology. Updated 07/14/2015. Accessed 12/01/2020, http://www.acc.org/latest-in-cardiology/articles/2015/07/14/11/04/screeing-tools-for-the-obstructive-sleep-apnea-for-the-cardiovascular-clinician
- Kline LR. Clinical presentation and diagnosis of obstructive sleep apnea in adults. UpToDate. Updated August 9, 2019. Accessed September 10, 2020. https://www.uptodate.com/contents/clinical-presentation-and-diagnosis-of-obstructive-sleep-apnea-in-adults?search=sleep%20apnea&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Mayo Clinic Staff. Polysomnography (sleep study). Updated November 17, 2018. Accessed October 17, 2020. https://www.mayoclinic.org/tests-procedures/polysomnography/about/pac-20394877
- American Academy of Sleep Medicine. Obstructive Sleep Apnea. Accessed October 17, 2020. https://aasm.org/resources/factsheets/sleepapnea.pdf
- Malhotra A, Mesarwi O, Pepin JL, Owens RL. Endotypes and phenotypes in obstructive sleep apnea. Current opinion in pulmonary medicine. Nov 2020;26(6):609-614. doi:10.1097/MCP.0000000000000724
- Patil SP, Ayappa IA, Caples SM, Kimoff RJ, Patel SR, Harrod CG. Treatment of Adult Obstructive Sleep Apnea With Positive Airway Pressure: An American Academy of Sleep Medicine Systematic Review, Meta-Analysis, and GRADE Assessment. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Feb 15 2019;15(2):301-334. doi:10.5664/jcsm.7638
- Batool-Anwar S, Goodwin JL, Kushida CA, et al. Impact of continuous positive airway pressure (CPAP) on quality of life in patients with obstructive sleep apnea (OSA). J Sleep Res. Dec 2016;25(6):731-738. doi:10.1111/jsr.12430
- Wickwire EM, Lettieri CJ, Cairns AA, Collop NA. Maximizing positive airway pressure adherence in adults: a common-sense approach. Chest. Aug 2013;144(2):680-693. doi:10.1378/chest.12-2681
- Rotenberg BW, Murariu D, Pang KP. Trends in CPAP adherence over twenty years of data collection: a flattened curve. J Otolaryngol Head Neck Surg. Aug 19 2016;45(1):43. doi:10.1186/s40463-016-0156-0
- Wang X, Zhang Y, Dong Z, Fan J, Nie S, Wei Y. Effect of continuous positive airway pressure on long-term cardiovascular outcomes in patients with coronary artery disease and obstructive sleep apnea: a systematic review and meta-analysis. Respiratory research. Apr 10 2018;19(1):61. doi:10.1186/s12931-018-0761-8
- Myllyla M, Hammais A, Stepanov M, Anttalainen U, Saaresranta T, Laitinen T. Nonfatal and fatal cardiovascular disease events in CPAP compliant obstructive sleep apnea patients. Sleep & breathing = Schlaf & Atmung. Dec 2019;23(4):1209-1217. doi:10.1007/s11325-019-01808-4
- da Silva Paulitsch F, Zhang L. Continuous positive airway pressure for adults with obstructive sleep apnea and cardiovascular disease: a meta-analysis of randomized trials. Sleep Med. Feb 2019;54:28-34. doi:10.1016/j.sleep.2018.09.030
- Baratta F, Pastori D, Fabiani M, et al. Severity of OSAS, CPAP and cardiovascular events: A follow-up study. European journal of clinical investigation. May 2018;48(5):e12908. doi:10.1111/eci.12908
- Lam JC, Lam B, Yao TJ, et al. A randomised controlled trial of nasal continuous positive airway pressure on insulin sensitivity in obstructive sleep apnoea. The European respiratory journal. Jan 2010;35(1):138-45. doi:10.1183/09031936.00047709
- Weinstock TG, Wang X, Rueschman M, et al. A controlled trial of CPAP therapy on metabolic control in individuals with impaired glucose tolerance and sleep apnea. Sleep. May 1 2012;35(5):617-625B. doi:10.5665/sleep.1816
- Marklund M, Braem MJA, Verbraecken J. Update on oral appliance therapy. European respiratory review : an official journal of the European Respiratory Society . Sep 30 2019;28(153)doi:10.1183/16000617.0083-2019
- Ramar K, Dort LC, Katz SG, et al. Clinical Practice Guideline for the Treatment of Obstructive Sleep Apnea and Snoring with Oral Appliance Therapy: An Update for 2015. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Jul 15 2015;11(7):773-827. doi:10.5664/jcsm.4858
- Zaghi S, Holty JE, Certal V, et al. Maxillomandibular Advancement for Treatment of Obstructive Sleep Apnea: A Meta-analysis. JAMA Otolaryngol Head Neck Surg. Jan 2016;142(1):58-66. doi:10.1001/jamaoto.2015.2678
- Stuck BA, Ravesloot MJL, Eschenhagen T, de Vet HCW, Sommer JU. Uvulopalatopharyngoplasty with or without tonsillectomy in the treatment of adult obstructive sleep apnea - A systematic review. Sleep Med. Oct 2018;50:152-165. doi:10.1016/j.sleep.2018.05.004
- Pang KP, Plaza G, Baptista JP, et al. Palate surgery for obstructive sleep apnea: a 17-year meta-analysis. European archives of oto-rhino-laryngology : official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS) : affiliated with the German Society for Oto-Rhino-Laryngology - Head and Neck Surgery . Jul 2018;275(7):1697-1707. doi:10.1007/s00405-018-5015-3
- Aishah A, Eckert DJ. Phenotypic approach to pharmacotherapy in the management of obstructive sleep apnoea. Current opinion in pulmonary medicine. Nov 2019;25(6):594-601. doi:10.1097/MCP.0000000000000628
- Aurora RN, Chowdhuri S, Ramar K, et al. The treatment of central sleep apnea syndromes in adults: practice parameters with an evidence-based literature review and meta-analyses. Sleep. Jan 1 2012;35(1):17-40. doi:10.5665/sleep.1580
- Chapman JL, Vakulin A, Hedner J, Yee BJ, Marshall NS. Modafinil/armodafinil in obstructive sleep apnoea: a systematic review and meta-analysis. The European respiratory journal. May 2016;47(5):1420-8. doi:10.1183/13993003.01509-2015
- Abad VC, Guilleminault C. Solriamfetol for the treatment of daytime sleepiness in obstructive sleep apnea. Expert review of respiratory medicine. Dec 2018;12(12):1007-1019. doi:10.1080/17476348.2018.1541742
- Yu JL, Thaler ER. Hypoglossal Nerve (Cranial Nerve XII) Stimulation. Otolaryngol Clin North Am. Feb 2020;53(1):157-169. doi:10.1016/j.otc.2019.09.010
- Gupta RJ, Kademani D, Liu SY. Upper Airway (Hypoglossal Nerve) Stimulation for Treatment of Obstructive Sleep Apnea. Atlas Oral Maxillofac Surg Clin North Am. Mar 2019;27(1):53-58. doi:10.1016/j.cxom.2018.11.004
- Certal VF, Zaghi S, Riaz M, et al. Hypoglossal nerve stimulation in the treatment of obstructive sleep apnea: A systematic review and meta-analysis. The Laryngoscope. May 2015;125(5):1254-64. doi:10.1002/lary.25032
- Woodson BT, Strohl KP, Soose RJ, et al. Upper Airway Stimulation for Obstructive Sleep Apnea: 5-Year Outcomes. Otolaryngology--head and neck surgery : official journal of American Academy of Otolaryngology-Head and Neck Surgery . Jul 2018;159(1):194-202. doi:10.1177/0194599818762383
- Kent DT, Carden KA, Wang L, Lindsell CJ, Ishman SL. Evaluation of Hypoglossal Nerve Stimulation Treatment in Obstructive Sleep Apnea. JAMA Otolaryngol Head Neck Surg. Sep 26 2019;doi:10.1001/jamaoto.2019.2723
- Edwards BA, Sands SA, Eckert DJ, et al. Acetazolamide improves loop gain but not the other physiological traits causing obstructive sleep apnoea. J Physiol. Mar 1 2012;590(5):1199-211. doi:10.1113/jphysiol.2011.223925
- Schmickl CN, Landry SA, Orr JE, et al. Acetazolamide for OSA and Central Sleep Apnea: A Comprehensive Systematic Review and Meta-Analysis. Chest. Aug 5 2020;doi:10.1016/j.chest.2020.06.078
- Eskandari D, Zou D, Grote L, Hoff E, Hedner J. Acetazolamide Reduces Blood Pressure and Sleep-Disordered Breathing in Patients With Hypertension and Obstructive Sleep Apnea: A Randomized Controlled Trial. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Mar 15 2018;14(3):309-317. doi:10.5664/jcsm.6968
- Hoff E, Zou D, Schiza S, et al. Carbonic anhydrase, obstructive sleep apnea and hypertension: Effects of intervention. J Sleep Res. Apr 2020;29(2):e12956. doi:10.1111/jsr.12956
- Liu HM, Chiang IJ, Kuo KN, Liou CM, Chen C. The effect of acetazolamide on sleep apnea at high altitude: a systematic review and meta-analysis. Therapeutic advances in respiratory disease. Jan 2017;11(1):20-29. doi:10.1177/1753465816677006
- Jaen-Aguila F, Vargas-Hitos JA, Mediavilla-Garcia JD. Implications of Renal Denervation Therapy in Patients with Sleep Apnea. International journal of hypertension. 2015;2015:408574. doi:10.1155/2015/408574
- Shantha GP, Pancholy SB. Effect of renal sympathetic denervation on apnea-hypopnea index in patients with obstructive sleep apnea: a systematic review and meta-analysis. Sleep & breathing = Schlaf & Atmung. Mar 2015;19(1):29-34. doi:10.1007/s11325-014-0991-z
- Bakris GL, Townsend RR, Liu M, et al. Impact of renal denervation on 24-hour ambulatory blood pressure: results from SYMPLICITY HTN-3. Journal of the American College of Cardiology. Sep 16 2014;64(11):1071-8. doi:10.1016/j.jacc.2014.05.012
- Kario K, Bhatt DL, Kandzari DE, et al. Impact of Renal Denervation on Patients With Obstructive Sleep Apnea and Resistant Hypertension- Insights From the SYMPLICITY HTN-3 Trial. Circulation journal : official journal of the Japanese Circulation Society . May 25 2016;80(6):1404-12. doi:10.1253/circj.CJ-16-0035
- Warchol-Celinska E, Prejbisz A, Kadziela J, et al. Renal Denervation in Resistant Hypertension and Obstructive Sleep Apnea: Randomized Proof-of-Concept Phase II Trial. Hypertension. Aug 2018;72(2):381-390. doi:10.1161/HYPERTENSIONAHA.118.11180
- Thomopoulos C, Spanoudi F, Kyriazis I, Anastasopoulos I, Ioannidis I. Metabolic effects of renal denervation. Current clinical pharmacology. Aug 2013;8(3):206-11. doi:10.2174/1574884711308030006
- Witkowski A, Prejbisz A, Florczak E, et al. Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension . Oct 2011;58(4):559-65. doi:10.1161/HYPERTENSIONAHA.111.173799
- Denolle T, Heautot JF. [Renal artery stenosis long time after renal denervation for resistant hypertension]. Ann Cardiol Angeiol (Paris). Jun 2018;67(3):204-207. Stenose de l'artere renale a distance d'une denervation renale pour HTA resistante. doi:10.1016/j.ancard.2018.04.011
- Riaz M, Certal V, Nigam G, et al. Nasal Expiratory Positive Airway Pressure Devices (Provent) for OSA: A Systematic Review and Meta-Analysis. Sleep Disord. 2015;2015:734798. doi:10.1155/2015/734798
- Wu H, Yuan X, Zhan X, Li L, Wei Y. A review of EPAP nasal device therapy for obstructive sleep apnea syndrome. Sleep & breathing = Schlaf & Atmung. Sep 2015;19(3):769-74. doi:10.1007/s11325-014-1057-y
- Wheeler NC, Wing JJ, O'Brien LM, et al. Expiratory Positive Airway Pressure for Sleep Apnea after Stroke: A Randomized, Crossover Trial. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Sep 15 2016;12(9):1233-8. doi:10.5664/jcsm.6120
- Hung TC, Liu TJ, Hsieh WY, et al. A novel intermittent negative air pressure device ameliorates obstructive sleep apnea syndrome in adults. Sleep & breathing = Schlaf & Atmung. Sep 2019;23(3):849-856. doi:10.1007/s11325-018-01778-z
- Yamaguchi Y, Kato M. Pilot Study of Oral Negative Pressure Therapy for Obstructive Sleep Apnea-Hypopnea Syndrome. J Sleep Disord Ther. 2017;6(3):271. doi:10.4172/2167-0277.1000271
- Netzer NC, Eliasson AH, Strohl KP. Women with sleep apnea have lower levels of sex hormones. Sleep & breathing = Schlaf & Atmung. Mar 2003;7(1):25-9. doi:10.1007/s11325-003-0025-8
- Shahar E, Redline S, Young T, et al. Hormone replacement therapy and sleep-disordered breathing. American journal of respiratory and critical care medicine. May 1 2003;167(9):1186-92. doi:10.1164/rccm.200210-1238OC
- Hersh AL, Stefanick ML, Stafford RS. National use of postmenopausal hormone therapy: annual trends and response to recent evidence. JAMA. Jan 7 2004;291(1):47-53. doi:10.1001/jama.291.1.47
- Mirer AG, Peppard PE, Palta M, Benca RM, Rasmuson A, Young T. Menopausal hormone therapy and sleep-disordered breathing: evidence for a healthy user bias. Annals of epidemiology. Oct 2015;25(10):779-84 e1. doi:10.1016/j.annepidem.2015.07.004
- Polo-Kantola P, Rauhala E, Helenius H, Erkkola R, Irjala K, Polo O. Breathing during sleep in menopause: a randomized, controlled, crossover trial with estrogen therapy. Obstet Gynecol. Jul 2003;102(1):68-75. doi:10.1016/s0029-7844(03)00374-0
- American College of Obstetricians and Gynecologists. Committee Opinion No. 556: Postmenopausal Estrogen Therapy: Route of Administration and Risk of Venous Thromboembolism. Obstetrics & Gynecology. 2013;121(4):887-890. doi:10.1097/01.AOG.0000428645.90795.d9
- Tuomilehto H, Seppa J, Uusitupa M. Obesity and obstructive sleep apnea--clinical significance of weight loss. Sleep medicine reviews. Oct 2013;17(5):321-9. doi:10.1016/j.smrv.2012.08.002
- Peppard PE, Young T, Palta M, Dempsey J, Skatrud J. Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA. Dec 20 2000;284(23):3015-21. doi:10.1001/jama.284.23.3015
- Peromaa-Haavisto P, Tuomilehto H, Kossi J, et al. Obstructive sleep apnea: the effect of bariatric surgery after 12 months. A prospective multicenter trial. Sleep Med. Jul 2017;35:85-90. doi:10.1016/j.sleep.2016.12.017
- Papandreou C, Schiza SE, Bouloukaki I, et al. Effect of Mediterranean diet versus prudent diet combined with physical activity on OSAS: a randomised trial. The European respiratory journal. Jun 2012;39(6):1398-404. doi:10.1183/09031936.00103411
- Harvard Men’s Health Watch. The Mediterranean diet: A model for Americans. Updated February, 2008. Accessed October 19, 2020. https://www.health.harvard.edu/newsletter_article/the-mediterranean-diet-a-model-for-americans#:~:text=The%20other%20pattern%20was%20a,the%20American%20or%20prudent%20patterns.
- Fiori CZ, Martinez D, Montanari CC, et al. Diuretic or sodium-restricted diet for obstructive sleep apnea-a randomized trial. Sleep. Apr 1 2018;41(4)doi:10.1093/sleep/zsy016
- Iftikhar IH, Kline CE, Youngstedt SD. Effects of exercise training on sleep apnea: a meta-analysis. Lung. Feb 2014;192(1):175-84. doi:10.1007/s00408-013-9511-3
- Iftikhar IH, Bittencourt L, Youngstedt SD, et al. Comparative efficacy of CPAP, MADs, exercise-training, and dietary weight loss for sleep apnea: a network meta-analysis. Sleep Med. Feb 2017;30:7-14. doi:10.1016/j.sleep.2016.06.001
- Kline CE, Ewing GB, Burch JB, et al. Exercise training improves selected aspects of daytime functioning in adults with obstructive sleep apnea. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Aug 15 2012;8(4):357-65. doi:10.5664/jcsm.2022
- Jurado-Garcia A, Molina-Recio G, Feu-Collado N, et al. Effect of a Graduated Walking Program on the Severity of Obstructive Sleep Apnea Syndrome. A Randomized Clinical Trial. International journal of environmental research and public health. Aug 31 2020;17(17)doi:10.3390/ijerph17176334
- Taveira KVM, Kuntze MM, Berretta F, et al. Association between obstructive sleep apnea and alcohol, caffeine and tobacco: A meta-analysis. Journal of oral rehabilitation. Nov 2018;45(11):890-902. doi:10.1111/joor.12686
- Simou E, Britton J, Leonardi-Bee J. Alcohol and the risk of sleep apnoea: a systematic review and meta-analysis. Sleep Med. Feb 2018;42:38-46. doi:10.1016/j.sleep.2017.12.005
- Vakulin A, Baulk SD, Catcheside PG, et al. Effects of alcohol and sleep restriction on simulated driving performance in untreated patients with obstructive sleep apnea. Ann Intern Med. Oct 6 2009;151(7):447-55. doi:10.7326/0003-4819-151-7-200910060-00005
- Zhu K, Bradley TD, Patel M, Alshaer H. Influence of head position on obstructive sleep apnea severity. Sleep & breathing = Schlaf & Atmung. Dec 2017;21(4):821-828. doi:10.1007/s11325-017-1525-2
- Laub RR, Tonnesen P, Jennum PJ. A Sleep Position Trainer for positional sleep apnea: a randomized, controlled trial. J Sleep Res. Oct 2017;26(5):641-650. doi:10.1111/jsr.12530
- Haas D, Birk R, Maurer JT, Hormann K, Stuck BA, Sommer JU. [Treatment of supine position-related obstructive sleep apnea with smartphone applications]. Hno. Feb 2017;65(2):148-153. Therapie ruckenlagebezogener obstruktiver Schlafapnoe mittels Smartphone-App. doi:10.1007/s00106-016-0330-8
- Freire AO, Sugai GC, Chrispin FS, et al. Treatment of moderate obstructive sleep apnea syndrome with acupuncture: a randomised, placebo-controlled pilot trial. Sleep Med. Jan 2007;8(1):43-50. doi:10.1016/j.sleep.2006.04.009
- Freire AO, Sugai GC, Togeiro SM, Mello LE, Tufik S. Immediate effect of acupuncture on the sleep pattern of patients with obstructive sleep apnoea. Acupuncture in medicine : journal of the British Medical Acupuncture Society . Sep 2010;28(3):115-9. doi:10.1136/aim.2009.001867
- Silva M, Lustosa TC, Arai VJ, et al. Effects of acupuncture on obstructive sleep apnea severity, blood pressure control and quality of life in patients with hypertension: A randomized controlled trial. J Sleep Res. Apr 2020;29(2):e12954. doi:10.1111/jsr.12954
- Wang L, Xu J, Zhan Y, Pei J. Acupuncture for Obstructive Sleep Apnea (OSA) in Adults: A Systematic Review and Meta-Analysis. Biomed Res Int. 2020;2020:6972327. doi:10.1155/2020/6972327
- Deleanu OC, Pocora D, Mihalcuta S, Ulmeanu R, Zaharie AM, Mihaltan FD. Influence of smoking on sleep and obstructive sleep apnea syndrome. Pneumologia. Jan-Mar 2016;65(1):28-35.
- Ben Amar J, Ben Mansour A, Zaibi H, Ben Safta B, Dhahri B, Aouina H. Impact of smoking on the severity of Obstructive Sleep Apnea Hypopnea Syndrome. La Tunisie medicale. Aug - Sep 2018;96(8-9):477-482.
- Bielicki P, Trojnar A, Sobieraj P, Wasik M. Smoking status in relation to obstructive sleep apnea severity (OSA) and cardiovascular comorbidity in patients with newly diagnosed OSA. Adv Respir Med. 2019;87(2):103-109. doi:10.5603/ARM.a2019.0011
- Lin YN, Zhou LN, Zhang XJ, Li QY, Wang Q, Xu HJ. Combined effect of obstructive sleep apnea and chronic smoking on cognitive impairment. Sleep & breathing = Schlaf & Atmung. Mar 2016;20(1):51-9. doi:10.1007/s11325-015-1183-1
- Zhu H, Xu H, Chen R, et al. Smoking, obstructive sleep apnea syndrome and their combined effects on metabolic parameters: Evidence from a large cross-sectional study. Sci Rep. Aug 18 2017;7(1):8851. doi:10.1038/s41598-017-08930-x
- Jouett NP, Moralez G, White DW, et al. N-Acetylcysteine reduces hyperacute intermittent hypoxia-induced sympathoexcitation in human subjects. Experimental physiology. Mar 2016;101(3):387-96. doi:10.1113/EP085546
- da Rosa DP, Forgiarini LF, e Silva MB, et al. Antioxidants inhibit the inflammatory and apoptotic processes in an intermittent hypoxia model of sleep apnea. Inflammation research : official journal of the European Histamine Research Society [et al] . Jan 2015;64(1):21-9. doi:10.1007/s00011-014-0778-5
- Krause BJ, Casanello P, Dias AC, et al. Chronic Intermittent Hypoxia-Induced Vascular Dysfunction in Rats is Reverted by N-Acetylcysteine Supplementation and Arginase Inhibition. Front Physiol. 2018;9:901. doi:10.3389/fphys.2018.00901
- Sadasivam K, Patial K, Vijayan VK, Ravi K. Anti-oxidant treatment in obstructive sleep apnoea syndrome. The Indian journal of chest diseases & allied sciences. Jul-Sep 2011;53(3):153-62.
- Celec P, Jurkovicova I, Buchta R, et al. Antioxidant vitamins prevent oxidative and carbonyl stress in an animal model of obstructive sleep apnea. Sleep & breathing = Schlaf & Atmung. May 2013;17(2):867-71. doi:10.1007/s11325-012-0728-9
- Grebe M, Eisele HJ, Weissmann N, et al. Antioxidant vitamin C improves endothelial function in obstructive sleep apnea. American journal of respiratory and critical care medicine. Apr 15 2006;173(8):897-901. doi:10.1164/rccm.200508-1223OC
- Singh TD, Patial K, Vijayan VK, Ravi K. Oxidative stress and obstructive sleep apnoea syndrome. The Indian journal of chest diseases & allied sciences. Oct-Dec 2009;51(4):217-24.
- Lee DS, Badr MS, Mateika JH. Progressive augmentation and ventilatory long-term facilitation are enhanced in sleep apnoea patients and are mitigated by antioxidant administration. J Physiol. Nov 15 2009;587(Pt 22):5451-67. doi:10.1113/jphysiol.2009.178053
- Zozina VI, Covantev S, Goroshko OA, Krasnykh LM, Kukes VG. Coenzyme Q10 in Cardiovascular and Metabolic Diseases: Current State of the Problem. Current cardiology reviews. 2018;14(3):164-174. doi:10.2174/1573403X14666180416115428
- Albuquerque RG, Hirotsu C, Tufik S, Andersen ML. Why Should We Care About Selenium in Obstructive Sleep Apnea? Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Jul 15 2017;13(7):931-932. doi:10.5664/jcsm.6674
- Chen PC, Guo CH, Tseng CJ, Wang KC, Liu PJ. Blood trace minerals concentrations and oxidative stress in patients with obstructive sleep apnea. The journal of nutrition, health & aging. 2013;17(8):639-44. doi:10.1007/s12603-013-0023-x
- Saruhan E, Sertoglu E, Unal Y, Bek S, Kutlu G. The role of antioxidant vitamins and selenium in patients with obstructive sleep apnea. Sleep & breathing = Schlaf & Atmung. Oct 8 2020;doi:10.1007/s11325-020-02207-w
- Archontogeorgis K, Nena E, Papanas N, Steiropoulos P. The role of vitamin D in obstructive sleep apnoea syndrome. Breathe (Sheffield, England) . Sep 2018;14(3):206-215. doi:10.1183/20734735.000618
- Neighbors CLP, Noller MW, Song SA, et al. Vitamin D and obstructive sleep apnea: a systematic review and meta-analysis. Sleep Med. Mar 2018;43:100-108. doi:10.1016/j.sleep.2017.10.016
- Barcelo A, Esquinas C, Pierola J, et al. Vitamin D status and parathyroid hormone levels in patients with obstructive sleep apnea. Respiration. 2013;86(4):295-301. doi:10.1159/000342748
- Archontogeorgis K, Papanas N, Rizos EC, et al. Reduced Serum Vitamin D Levels Are Associated with Insulin Resistance in Patients with Obstructive Sleep Apnea Syndrome. Medicina (Kaunas, Lithuania). May 20 2019;55(5)doi:10.3390/medicina55050174
- Fan Z, Cao B, Long H, et al. Independent association of vitamin D and insulin resistance in obstructive sleep apnea. Annales d'endocrinologie. Nov 2019;80(5-6):319-323. doi:10.1016/j.ando.2019.09.004
- Archontogeorgis K, Nena E, Papanas N, et al. Metabolic Syndrome and Vitamin D Levels in Patients with Obstructive Sleep Apnea Syndrome. Metabolic syndrome and related disorders. May 2018;16(4):190-196. doi:10.1089/met.2017.0181
- Kerley CP, Hutchinson K, Bramham J, McGowan A, Faul J, Cormican L. Vitamin D Improves Selected Metabolic Parameters but Not Neuropsychological or Quality of Life Indices in OSA: A Pilot Study. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Jan 15 2017;13(1):19-26. doi:10.5664/jcsm.6378
- Ragia G, Archontogeorgis K, Simmaco M, et al. Genetics of Obstructive Sleep Apnea: Vitamin D Receptor Gene Variation Affects Both Vitamin D Serum Concentration and Disease Susceptibility. Omics : a journal of integrative biology. Jan 2019;23(1):45-53. doi:10.1089/omi.2018.0184
- Scorza FA, Cavalheiro EA, Scorza CA, Galduroz JC, Tufik S, Andersen ML. Sleep Apnea and Inflammation - Getting a Good Night's Sleep with Omega-3 Supplementation. Frontiers in neurology. Dec 3 2013;4:193. doi:10.3389/fneur.2013.00193
- Ladesich JB, Pottala JV, Romaker A, Harris WS. Membrane level of omega-3 docosahexaenoic acid is associated with severity of obstructive sleep apnea. Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Aug 15 2011;7(4):391-6. doi:10.5664/JCSM.1198
- Alzoubi MR, Aldomi Al-Domi H. Could omega-3 fatty acids a therapeutic treatment of the immune-metabolic consequence of intermittent hypoxia in obstructive sleep apnea? Diabetes Metab Syndr. Oct - Dec 2017;11(4):297-304. doi:10.1016/j.dsx.2016.06.024
- Li K, Zhang J, Qin Y, Wei YX. Association between Serum Homocysteine Level and Obstructive Sleep Apnea: A Meta-Analysis. Biomed Res Int. 2017;2017:7234528. doi:10.1155/2017/7234528
- Kim J, Lee SK, Yoon DW, Shin C. Concurrent Presence of Obstructive Sleep Apnea and Elevated Homocysteine Levels Exacerbate the Development of Hypertension: A KoGES Six-year Follow-up Study. Sci Rep. Feb 8 2018;8(1):2665. doi:10.1038/s41598-018-21033-5
- Chen X, Niu X, Xiao Y, et al. Effect of continuous positive airway pressure on homocysteine levels in patients with obstructive sleep apnea: a meta-analysis. Sleep & breathing = Schlaf & Atmung. Dec 2014;18(4):687-94. doi:10.1007/s11325-014-0940-x
- Li J, Yu LQ, Jiang M, Wang L, Fang Q. Homocysteine level in patients with obstructive sleep apnea/hypopnea syndrome and the impact of continuous positive airway pressure treatment. Adv Clin Exp Med. Nov 2018;27(11):1549-1554. doi:10.17219/acem/74178
- Huang T, Chen Y, Yang B, Yang J, Wahlqvist ML, Li D. Meta-analysis of B vitamin supplementation on plasma homocysteine, cardiovascular and all-cause mortality. Clin Nutr. Aug 2012;31(4):448-54. doi:10.1016/j.clnu.2011.01.003
- Zhang DM, Ye JX, Mu JS, Cui XP. Efficacy of Vitamin B Supplementation on Cognition in Elderly Patients With Cognitive-Related Diseases. Journal of geriatric psychiatry and neurology. Jan 2017;30(1):50-59. doi:10.1177/0891988716673466
- Ford TC, Downey LA, Simpson T, McPhee G, Oliver C, Stough C. The Effect of a High-Dose Vitamin B Multivitamin Supplement on the Relationship between Brain Metabolism and Blood Biomarkers of Oxidative Stress: A Randomized Control Trial. Nutrients. Dec 1 2018;10(12)doi:10.3390/nu10121860
- Mashaqi S, Gozal D. Obstructive Sleep Apnea and Systemic Hypertension: Gut Dysbiosis as the Mediator? Journal of clinical sleep medicine : JCSM : official publication of the American Academy of Sleep Medicine . Oct 15 2019;15(10):1517-1527. doi:10.5664/jcsm.7990
- Ko CY, Liu QQ, Su HZ, et al. Gut microbiota in obstructive sleep apnea-hypopnea syndrome: disease-related dysbiosis and metabolic comorbidities. Clin Sci (Lond). Apr 15 2019;133(7):905-917. doi:10.1042/CS20180891
- Ganesh BP, Nelson JW, Eskew JR, et al. Prebiotics, Probiotics, and Acetate Supplementation Prevent Hypertension in a Model of Obstructive Sleep Apnea. Hypertension. Nov 2018;72(5):1141-1150. doi:10.1161/HYPERTENSIONAHA.118.11695
- Xu H, Wang J, Cai J, et al. Protective Effect of Lactobacillus rhamnosus GG and its Supernatant against Myocardial Dysfunction in Obese Mice Exposed to Intermittent Hypoxia is Associated with the Activation of Nrf2 Pathway. International journal of biological sciences. 2019;15(11):2471-2483. doi:10.7150/ijbs.36465
- Semelka M, Wilson J, Floyd R. Diagnosis and Treatment of Obstructive Sleep Apnea in Adults. Am Fam Physician. Sep 1 2016;94(5):355-60. https://pubmed.ncbi.nlm.nih.gov/27583421/
- AASM. American Academy of Sleep Medicine. Novel devices and applications for diagnosing obstructive sleep apnea. Updated 9/20/2023. Accessed 6/23/2025, https://aasm.org/novel-devices-applications-diagnosing-obstructive-sleep-apnea/
- Park JH, Wang C, Shin H. FDA-cleared home sleep apnea testing devices. NPJ Digit Med. May 13 2024;7(1):123. doi:10.1038/s41746-024-01112-w. https://pubmed.ncbi.nlm.nih.gov/38740907/
- Edouard P, Campo D, Bartet P, et al. 0579 Non Intrusive and Unattended Sleep Analyzer Effectively Screens Patients Suspected of Sleep Apnea: A Comparison With Polysomnography. Sleep. 2020;43(Supplement_1):A222-A222. doi:10.1093/sleep/zsaa056.576. https://academic.oup.com/sleep/article-abstract/43/Supplement_1/A222/5846812
- Kapur VK, Auckley DH, Chowdhuri S, et al. Clinical Practice Guideline for Diagnostic Testing for Adult Obstructive Sleep Apnea: An American Academy of Sleep Medicine Clinical Practice Guideline. J Clin Sleep Med. Mar 15 2017;13(3):479-504. doi:10.5664/jcsm.6506. https://pubmed.ncbi.nlm.nih.gov/28162150/
- IEEE. Institute of Electrical and Electronics Engineers. Body Sensor Networks Conference. Towards a Less Intrusive Sleep Monitoring System. Updated 2016. Accessed 6/23/2025, https://bsn.embs.org/2017/workshops/towards-a-less-intrusive-sleep-monitoring-system/
- Wick AZ, Combertaldi SL, Rasch B. The first-night effect of sleep occurs over nonconsecutive nights in unfamiliar and familiar environments. Sleep. Oct 11 2024;47(10)doi:10.1093/sleep/zsae179. https://pubmed.ncbi.nlm.nih.gov/39126649/
- Hamilton J. Half Your Brain Stands Guard When Sleeping In A New Place. NPR. Updated 4/21/2016. Accessed 6/23/2025, https://www.npr.org/sections/health-shots/2016/04/21/474691141/half-your-brain-stands-guard-when-sleeping-in-a-new-place
- Byun JH, Kim KT, Moon HJ, Motamedi GK, Cho YW. The first night effect during polysomnography, and patients' estimates of sleep quality. Psychiatry Res. Apr 2019;274:27-29. doi:10.1016/j.psychres.2019.02.011. https://pubmed.ncbi.nlm.nih.gov/30776709/
- Hussein O, Alkhader A, Gohar A, Bhat A. Home Sleep Apnea Testing for Obstructive Sleep Apnea. Mo medicine. Jan-Feb 2024;121(1):60-65. https://pubmed.ncbi.nlm.nih.gov/38404435/
- Collop N. Home Sleep Apnea Testing FOr Obstructive Sleep Apnea In Adults. Updated 06/10/2025. Accessed 06/17/25, https://www.uptodate.com/contents/home-sleep-apnea-testing-for-obstructive-sleep-apnea-in-adults
- Vera J. What Is an At-Home Sleep Study? Sleep Foundation. Updated 6/11/2025. Accessed 6/23/2025, https://www.sleepfoundation.org/sleep-studies/at-home-sleep-study
- Ichikawa M, Akiyama T, Tsujimoto Y, Anan K, Yamakawa T, Terauchi Y. Diagnostic accuracy of home sleep apnea testing using peripheral arterial tonometry for sleep apnea: A systematic review and meta-analysis. J Sleep Res. Dec 2022;31(6):e13682. doi:10.1111/jsr.13682. https://pubmed.ncbi.nlm.nih.gov/35793907/
- Zhai H, Yan Y, He S, Zhao P, Zhang B. Evaluation of the Accuracy of Contactless Consumer Sleep-Tracking Devices Application in Human Experiment: A Systematic Review and Meta-Analysis. Sensors (Basel, Switzerland). May 17 2023;23(10)doi:10.3390/s23104842. https://pubmed.ncbi.nlm.nih.gov/37430756/
- WatchPat How To Use. Accessed 05/05/2025, https://www.itamar-medical.com/patients/watchpat-home-sleep-testing/
- AASM. American Academy of Sleep Medicine. FDA clears disposable home sleep apnea test. Updated 2/18/2020. Accessed 6/23/2025, https://aasm.org/fda-clears-disposable-home-sleep-apnea-test/
- Lee YJ, Lee JY, Cho JH, Kang YJ, Choi JH. Performance of consumer wrist-worn sleep tracking devices compared to polysomnography: a meta-analysis. J Clin Sleep Med. Mar 1 2025;21(3):573-582. doi:10.5664/jcsm.11460. https://pubmed.ncbi.nlm.nih.gov/39484805/
- Robbins R, Weaver MD, Sullivan JP, et al. Accuracy of Three Commercial Wearable Devices for Sleep Tracking in Healthy Adults. Sensors (Basel, Switzerland). Oct 10 2024;24(20)doi:10.3390/s24206532. https://pubmed.ncbi.nlm.nih.gov/39460013/
- AASM. Samsung Galaxy Watch sleep apnea feature receives FDA authorization. Updated 2/12/2024. Accessed 06/09/2025, https://aasm.org/samsung-galaxy-watch-sleep-apnea-feature-receives-fda-authorization/
- AASM. American Academy of Sleep Medicine. Apple Watch Series 10 features sleep apnea notifications. Updated 9/18/2024. Accessed 6/23/2025, https://aasm.org/apple-watch-series-10-features-sleep-apnea-notifications/
- Knop FK, Urva S, Rettiganti M, et al. A long-acting glucose-dependent insulinotropic polypeptide receptor agonist improves the gastrointestinal tolerability of glucagon-like peptide-1 receptor agonist therapy. Diabetes, Obesity and Metabolism. 2024;26(11):5474-5478. doi:https://doi.org/10.1111/dom.15875. https://pubmed.ncbi.nlm.nih.gov/39188238/
- U.S. FDA. FDA Approves First Medication for Obstructive Sleep Apnea. Updated Dec. 20, 2024. Accessed Jun. 12, 2025, https://www.fda.gov/news-events/press-announcements/fda-approves-first-medication-obstructive-sleep-apnea
- Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity. N Engl J Med. Oct 3 2024;391(13):1193-1205. doi:10.1056/NEJMoa2404881. https://pubmed.ncbi.nlm.nih.gov/38912654/

